- *Corresponding Author:
- D. Puri
Department of Biochemistry, University College of Medical Sciences, Delhi-110 095, India
E-mail: dineshpuri21@yahoo.com
| Date of Submission | 05 February 2005 |
| Date of Revision | 29 December 2005 |
| Date of Acceptance | 25 August 2006 |
| Indian J Pharm Sci,2006, 68 (5): 579-583 |
Abstract
An obese animal model having mild glucose intolerance was developed by intravenous administration of sub-optimal dose of alloxan to British Angora rabbits. Mild hypoglycaemic activity of crude extract prepared from leaves of a Nepalese herb Biophytum sensitivum, which was missed by the conventional diabetic animal models, was demonstrable in these animals. Initial fasting hyperglycaemia observed immediately after treatment with alloxan in these animals was followed by reverting of the fasting blood glucose to normal or near normal levels. However, the blood glucose levels rose to higher than normal values following oral glucose load in the 1 h, 2 h and 3 h samples, reflecting glucose intolerance which was stable for up to 4 mo. Hypoglycaemic effect in these animals, designated obese glucose-intolerant (OGI) rabbits, was judged by improvement in glucose tolerance, tested by prevention of blood glucose elevation following oral glucose load. Comparing the corresponding pre-treatment and post-treatment values could even quantitate the hypoglycaemic effect.
Introduction
Hypoglycaemic activity of drugs, including the products derived from medicinal plants, is conventionally assessed indiabetic animal models by observing drug-induced fall in fasting blood glucose (FBG) or suppression of glucose tolerance curve. Diabetes is induced experimentally by partial or total pancreatectomy, exposure to antiislet cell antibodies, but most commonly by injecting chemical agents,such as alloxan or streptozotocin, that cause widespread destruction of insulin-secreting pancreatic beta cells [1]. However, beta cells being extensively eliminated/or destroyed in the diabetic animals, several drugs that require functional pancreas cannot be satisfactorily tested in them.
Moreover, the drug-induced fall in FBG as a criterion for hypoglycaemic effect has been reported to be a relatively insensitive method of assessment and sometimes gives inconsistent results [2]. Post-treatment suppression of glucose tolerance curve, though a sensitive method of assessment, suffers from a major drawback in that the test is associated with high mortality in diabetic animals [3]. Need for better animal model for screening hypoglycaemic activity of compounds has therefore being felt; hence the present study carried out in British Angora rabbits,instead of conventionally used albino rabbits, and their suitability for serving as experimental animal models for the above stated purpose was evaluated. Sub-optimal doses of alloxan were injected intravenously for inducing beta cell destruction partially. The course of hyperglycaemic state was carefully monitored and changes in body weight recorded. It was observed that the low-dose alloxanated Angora rabbits showed increased feeding behaviour, gained weight rapidly, leading to moderate obesity, and developed impairment of glucose tolerance, though FBG remained near normal. Utility of these animals – moderately obese with mild glucose these animals – moderately obese with mild glucose intolerance – for detecting the mild hypoglycaemic activity of leaf extract of Biophytum sensitivum, a Nepalese herb [4], has been evaluated and discussed in this paper. A standard hypoglycaemic drug, tolbutamide, was taken for reference.
Materials and Methods
Alloxan monohydrate was purchased from Sigma. Tolbutamide (Hoechst Pharmaceuticals) was obtained locally. Inbred male British Angora rabbits of body weight 700 to 1000 g and age 2.0-2.5 mo were purchased from Palkhibas farm, located in mountainous Dhankuta district in Eastern Nepal. Permission for conducting experiments on these animals was granted by the research committee of B. P. Koirala Institute of Health Sciences, where the study was carried out, as also by Nepal Health Research Council. Having been brought down from high altitude to foothills, the animals were acclimatized to laboratory conditions and handling for at least 2 w, during which period they were given pallet diet obtained from of Hindustan Lever Ltd., Mumbai. Oral glucose tolerance test was performed in them to rule out diabetes and other forms of carbohydrate intolerance. Blood samples were collected from the marginal ear vein.
Plant material
Samples of the Biophytum sensitivum (family Oxalidaceae; Accession No. 199600026), a traditional antidiabetic herb,were obtained from the Botany Department, Mohender Morang Campus (Postgraduate Institute, Biratnagar), for this study. With the help of these samples, our field workers were trained so as to be able to study its morphology to avoid any mistake during collection from the thick Tarahara forests. The plant was then collected from these forests (Sunsari district), on the eastern bank of Koshi River, during the months of August to October 1997, the time when it grows in abundance. For reliable identification, the new plant samples were sent to the Botany Department of the above institute, where they were authenticated.
The plant leaves were separated, dried, soaked in water for about 16 h and homogenized. The homogenate was centrifuged at 3000 g for 15 min. The supernatant was decanted, filtered and the extract so obtained was separated and used for the present study. The above process yielded 13.2% w/w extract, as reported by us earlier [5]. It was orally administered to the experimental animals, using feeding tube (No. 7).
Biochemical estimations
Blood glucose level was estimated by glucose-oxidase/ peroxidase method [6], using kits from Ranbaxy, Delhi. Insulin was estimated in sera by ELISA technique using commercial kits from Immunosystems, Italy.
Induction of alloxan diabetes
Overnight fasted rabbits were rendered diabetic by single intravenous injection of alloxan monohydrate at a dose of 80 mg/kg body weight, which was found to be the optimum dose for the induction of diabetes in our earlier study [7]. Serum glucose level was checked after 72 h of alloxanization, and the animals with glucose level exceeding 200 mg% were considered diabetic. Lower doses of 70 mg/kg- and 60 mg/kg-body weight of alloxan were also given to the separate groups of animals for inducing diabetes of milder severity course of diabetes following alloxanization was monitored, as described in the next section. It may be mentioned presently that FBG initially rose in all alloxanated rabbits. In most, the hyperglycaemic state stabilized, resulting in overt diabetes; whereas in some others, initial high FBG levels returned to near normal after few days. The latter group of rabbits developed moderate obesity as well and showed impairment of glucose tolerance; they were designated as obese glucose-intolerant (OGI) rabbits.
Four rabbits, which did not show any increase in FBG levels even initially after alloxanization, were considered totally resistant and were excluded from the study.
Assessment of hypoglycaemic effect
Ability of a compound to lower the FBG or attenuate the glucose tolerance curve is conventionally taken to be an indicator of its hypoglycaemic effect, as mentioned earlier. In OGI rabbits, FBG being normal, suppression of glucose tolerance test (GTT) only was taken into consideration. Five to seven days before the commencement of the experiment, GTT was performed in these animals following an overnight fast. Fasting blood sample was taken, glucose was orally administered at a dose of 3 g/kg body weight and then plasma glucose was estimated in blood samples drawn 1 h, 2 h and 3 h later from marginal ear vein. The GTT curve was drawn and area under curve (AUC) calculated, which not only confirmed hyperglycaemic status of the animal but also showed the pre-treatment base-line glucose tolerance pattern. One week later, the test (GTT) was repeated following administration of the test compound orally and 90 min later, oral glucose load was given at the above dose. Blood samples were collected before the administration of glucose and 1 h, 2 h and 3 h later. The corresponding pre-treatment and post-treatment glucose levels at these three points of time and the AUCs were compared to assess post-treatment suppression of the curve, if any, which would reflect hypoglycaemic activity of the test compound.
In contrast to OGI, in overt diabetic rabbits both fall of FBG and attenuation of GTT were taken as criteria of hypoglycaemic effect. However, GTT was associated with high mortality in the diabetic rabbits and therefore generally avoided in those having severe hyperglycaemia (FBG > 250 mg/dl).
Results and Discussion
Course of diabetes was carefully monitored in all alloxanated rabbits by estimating FBG after every 5 d. With 80 mg/kg body weight dose of alloxan, FBG showed progressive rise and reached 260 ± 16 mg/dl by the 15th day (Table 1). Relatively smaller variation in FBG was seen after that, and the glucose level recorded on the 35th day was 296 ± 20 mg/dl. Similar pattern was observed with 70 mg/kg of alloxan, though the final value stabilized at a lower value (216 ± 12 mg/dl) on the 35th day. With 60 mg/kg alloxan, the FBG predictably rose initially till about the 2nd week but showed a spontaneous reversal after that, reaching normal value by 30-35 d. Moreover, the latter animals gained weight rapidly to develop moderate obesity (body weight reaching 1718 ± 88 g) and showed impairment of glucose tolerance, as discussed later in this article. Their mean body weight was significantly higher than the animals of the control group, but fasting blood glucose and plasma insulin levels were not significantly different from controls. Comparative features of these rabbits, designated OGI, versus the diabetic rabbits and controls are outlined in Table 2. It may be observed that unlike diabetic animals, they did not exhibit any significant hyperglycaemia or hypoinsulinaemia.
| Doseofalloxan: mg/kgbodyweight | 0 | 5 | 10 | 15 | 20 | 25 | 30 | 35 |
|---|---|---|---|---|---|---|---|---|
| 80 | 84±6 | 213±9 | 218±12 | 260±16 | 288±16 | 292±15 | 282±14 | 296±20 |
| 70 | 86±7 | 126±8 | 212±10 | 210±11 | 252±11 | 232±11 | 208±11 | 216±12 |
| 60 | 84±6 | 110±9 | 168±9 | 206±10 | 180±9 | 102±6 | 88±7 | 96±6 |
Blood glucose values expressed as mg/dl and dose of alloxan as mg/kg body weight. The fasting blood glucose levels were recorded at 5 days’ interval up to 35 days after alloxan treatment for following course of diabetes
Table1: Fastingbloodglucoselevels(Mg/Dl)Ofalloxanatedangorarabbits
| Control | OGI | Diabetic | |
|---|---|---|---|
| (n=10) | (n=10) | (n=20) | |
| Body weight (g) | 1008±77 | 1718±88 | 860±68 |
| FBG (mg/dl) | 86.1±6 | 92±7 | 272.4±16 |
| Plasma insulin (µU/ml) | 11.4±1.2 | 9.8±1.4 | 0.12±0.03 |
| Fluid intake (ml/day) | 166±9 | 206±10 | 218±10 |
The parameters were measured 35 days after alloxanization in OGI and diabetic rabbits. Values of FBG and insulin are not statistically different in controls and OGI but are significantly different in diabetic rabbits versus controls and OGI (P < 0.01; student’s t-test), OGI - Obese glucoseintolerant, FBG - Fasting blood glucose
Table 2: General Parameters Of The Control, Ogi And Diabetic Rabbits
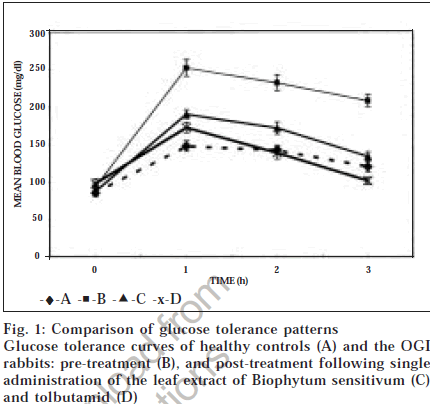
Reversal of experimental diabetic state, as seen in OGI animals in our study, has been reported in rats, mice and rabbits in earlier studies2,8. Abnormalities of glucose metabolism and dyslipidaemias were also observed in those animals. In view of this, the alloxanated animals showing apparent reversal of diabetes in the present study (i.e., the OGI rabbits) were tested for any abnormality of glucose metabolism by glucose tolerance test. Greater elevations of blood glucose at 1, 2 and 3 h were observed in OGI rabbits compared to corresponding values in the control rabbits (fig. 1). The area under curve in the OGI rabbits, 635.8 mg/dl/h, was also higher than that for the control rabbits (394.2 mg/dl/ h), suggesting that the OGI rabbits are not able to prevent elevation of blood glucose levels following glucose load. Serum insulin levels were lower in these animals in fasted state (Table 2) as also in 1 h, 2 h and 3 h samples (13.2±1.1, 14.6±1.1 and 10.3±1.2 μU/ml respectively) compared to the corresponding values in controls (28.8±2.3, 28.4±2.4 and 13.6±1.2 μU/ml). Thus, mild hypoinsulinaemia and impaired glucose-induced insulin response is seen in the OGI animals, which suggests that the pancreatic beta cells were partially damaged by sub-optimal dose of alloxan. This primarily accounts for the impaired glucose tolerance pattern observed, though obesity may accentuate glucose observed, though obesity may accentuate glucose cells to insulin. Further studies, including histopathological examination and receptor analysis, are necessary to validate the latter point. Abnormality of the glucose tolerance pattern as described above was reproducible even after 4 mo of alloxanization, suggesting development of a stable subdiabetic state.
Activity of the Biophytum sensitivum leaf extract was tested by oral administration of the extract at optimum dose of 200 mg/kg following an overnight fast7, recording the glucose tolerance pattern and comparing it with the pattern observed before the treatment. Consistent reductions in blood glucose levels were observed at all points (fig. 1), which were significantly lower than the corresponding pre-treatment levels. The AUC in the treated animals (473.6 mg/dl/h) showed a significant fall of 25.6% compared to the pre-treatment value of 635.8 mg/dl/h. Activity of tolbutamide, a standard oral hypoglycaemic agent, was also tested in the OGI model. At a dose of 500 mg/kg, tolbutamide caused improvement in the glucose tolerance pattern and significantly reduced the AUC to 411.8 mg/dl/h (P < 0.01) compared to the corresponding pre-treatment AUC, thereby ensuring that the OGI model responds to a known standard drug as well.
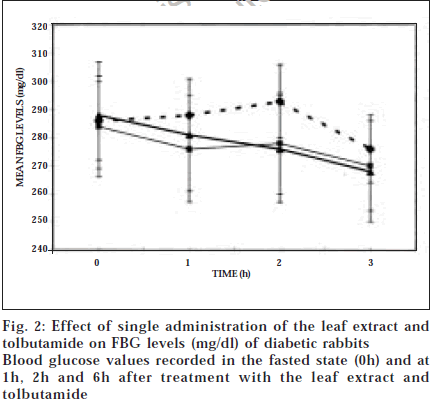
In diabetic rabbits, elevations of both FBG and glucose tolerance curve occur; therefore, the hypoglycaemic activity was judged by taking into consideration the post-treatment fall of FBG as well as suppression of glucose tolerance curve. Following administration of the leaf extract, FBG showed marginal alterations at 1 h, 2 h and 6 h (fig. 2). Compared to the corresponding values in the untreated diabetic rabbits, these values were slightly lower but not significantly. Similar trend was observed in the glucose tolerance pattern when the pre-treatment
pattern was compared to the post-treatment pattern to look for evidence of suppression, if any (Table 3). Decreases in the 1 h, 2 h and 3 h blood glucose values were small, insignificant and rather inconsistent – the inconsistency and variability of response of these animals being reflected by large standard deviations. Fall in AUC in the treated animals compared to the pre-treated animals (865 to 849 mg/dl/h) was also marginal (1.9%) and insignificant. It is noteworthy that single administration of the same dose of plant extract had produced significant fall of the glucose tolerance curve in OGI rabbits (fig. 1) but failed to elicit such effect in diabetic rabbits. Likewise, tolbutamide treatment led to inconclusive results in FBG (fig. 2) and GTT pattern of the diabetic rabbits (results not shown), though it had elicited pronounced hypoglycaemic effect in OGI rabbits. Thus the diabetic animals as animal models are not as sensitive for assessing hypoglycaemic activity of test compounds as the OGI rabbits. This is explained by the fact that extensive damage of pancreatic beta cells in diabetic animals limits their utility for assessing hypoglycaemic potential of the test compounds that require functional pancreas, such as the plant leaf extract, which has earlier been shown to act by inducing secretion of insulin from the pancreatic beta cells [7]. An additional limiting factor in use of diabetic animals is that GTT resulted in high mortality in them, whereas no such problem was encountered in OGI rabbits.
| Fasting | 1h | 2h | 3h | |
|---|---|---|---|---|
| Pre-treated | 232±14 | 306±16 | 300±15 | 286±16 |
| Post-treated | 236±16 | 288±21 | 302±22 | 282±21 |
| % change | +1.72 | -5.88 | +0.67 | -1.39 |
| Significance | NS | NS | NS | NS |
Percent change was calculated by comparing blood glucose values before and after treatment in the same group of animals (self-controls). NS = Not significant (student’s t-test). (N = 16)
Table 3: Effect Of The Leaf Extract On Glucose Tolerance Pattern Of The Diabetic Rabbits
Taken together, these observations in the OGI and the overt diabetic rabbits suggest that the OGI model is more sensitive in detecting mild hypoglycaemic activity of a crude extract. The crude extracts generally interfere less with the regulatory mechanisms involved in carbohydrate metabolism, and their use causes significantly less hypoglycaemic effect compared to more potent synthetic drugs [9]. Such mild hypoglycaemic effect was missed by conventional diabetic model but effectively detected by the OGI rabbits. Further advantage of using the OGI model over the conventional way of testing is that the same animal serves as its own control because the blood glucose values within the same group of animals were compared before and after treatment. This results in minimizing the biological variations, which provides additional advantage of enhancing reliability of the results. In view of these results, it is hoped that OGI rabbits would serve as a useful experimental tool for screening crude extracts with slight activity, thereby successfully putting to test hypoglycaemic potential of plant products.Though recently, sophisticated animal models [e.g., Bio-Breeding (BB) rat, nonobese diabetic (NOD) mouse,cross of BB and Zucker fatty (BBZ) rats, Long Evans Tokushima lean rat] are being used for detecting mild hypoglycaemic activity of plant products with varying degree of success, they are not readily available in this part of the world and moreover may not be able to adopt to the harsh climate in this region. Development of locally available breeds is therefore of special importance, at least during initial screening of mild hypoglycaemic activities of compounds. The OGI Angora rabbits have been found to be useful for this purpose and may prove worthwhile for unravelling hypoglycaemic potential of many more unexplored plants growing in these regions.
Finally, it is fitting to mention that the purpose of any research on herbal products is to isolate active principle from it for development of the drug. Having established hypoglycaemic potential of the new medicinal plant Biophytum sensitivum, with the help of the OGI model, further steps for purification were initiated in our laboratory, and a partially purified preparation has been obtained from the plant extract, which is active at lower dose.
Acknowledgements
The author is grateful to Dr. S. N. Jha and Mr. Sunil Dhungel, Postgraduate Institute, Biratnagar, Nepal, for their collaboration in providing the plant samples, training our field workers and authenticating the plants subsequently collected. Grateful acknowledgements are due to Dr. Kalyani, Department of Botany, University of Delhi, for providing the required information about the plant from scientific literature.
References
- Nakamura. M.. Nagafuchi. S.z Yamaguchi, K. and Ryosaburs, T., Diabetes. 1984. 33. 894.
- Babu, B.V.. Raclin Moortrpugazhenthi, S., Prabhu, K.M. and Mouthy, P.S.. Indian J. Biochem. Biophys, 1988, 25, 714.
- Puri. D.. Prabhu. K.M., Murthy, P.S., Indian J. Physiol. Pharmacol., 2002. 46, 457.
- Grover. J.K; Yaclav, S., and Vats, V., J. Ethnopharmacol.. 2002, 81, 81.
- J Puri. D. Phrm.-24o1., 2003, 41, 253.
- Btwtis. C.A.. Ashwod, E.R. Eds. Tietz Text book of clinical Chemistry 3rd Edn.. W. B. Saunders and Co.,Philadelphia, 1976, 1.
- Puri. D.. J. Ethnopharmacol., 2001, 78, 89.
- Dixit P.K.. Lowe, I. and Lazarow, A., Nature, 1962, 196, 388.
- Vats, V.. Grover, J.K and Rathi, S.S., J. Ethnopharmacol., 2001, 79, 95.