- *Corresponding Author:
- Vidhya V. Iyer
School of Biosciences and Technology, VIT University, Vellore-632 014, India
E-mail: vidhyaiyer.v@vit.ac.in
| Date of Submission | 28 September 2016 |
| Date of Revision | 27 December 2016 |
| Date of Acceptance | 05 February 2017 |
| Indian J Pharm Sci 2017;79(2):175-185 |
Abstract
This review is aimed at equipping the reader with fundamental knowledge pertaining to vaccines, their pharmacokinetics and pharmacodynamics, along with specific examples with respect to clinical trials and mechanism of action. Poliomyelitis, rotavirus and diphtheria-pertussis-tetanus vaccines have been discussed in this review. Progress made in vaccines has been discussed, from failed clinical trials to recent ground-breaking developments. Efforts made to combat the recent outbreak of Zika virus have also been included. The recent development of the Nanopatch™ technology aims at revolutionizing the vaccine industry.
Keywords
Vaccines, clinical trials, pharmacokinetics, pharmacodynamics, virus, attenuated vaccine, Nanopatch™
Vaccination is the process of administration of an antigenic material (a vaccine) to stimulate the immune system so that adaptive immunity is developed towards a specific pathogen [1]. Vaccines have the ability to prevent or ameliorate morbidity from an infection. Inactivated (non-infective) or attenuated (reduced infectivity) forms of disease pathogens, or purified components of the pathogens, are highly immunogenic. Immunogens can be administered in many ways during vaccination [2]. A few types of vaccines are administered after the patient has already contracted a disease. Most vaccines are administered by hypodermic injection as they are not completely absorbed through the intestine. Certain typhoid and cholera vaccines, as well as the live attenuated polio vaccine, are usually given orally to produce immunity [2]. While vaccination has a long lasting effect, it takes several weeks to develop active immunity [1]. On the other hand, passive immunity (the transfer of antibodies) has an immediate effect. Herd immunity has been observed even in unvaccinated individuals when a sufficiently large proportion of a population has been vaccinated.
Vaccines contain either inactivated or attenuated forms of the immunogen (bacteria, virus) that evoke an immune response by generating antibodies that bind to the surface of the non-infectious immunogen. Along with the immunogen, other components include adjuvant that enhances the immune response and is usually aluminium-based [3]. Adjuvants can reduce the amount of antigens and also the number of immunizations needed to build up immunity [4]. Preservatives prevent unwanted contamination of the vaccines, usually 2-phenoxyethanol. Stabilizers are buffers to resist pH change and surfactants as wetting agents. Various vaccines that a growing child must receive in India are summarized in Table 1 [5].

Polio Vaccine
Polio was described by Michael Underwood in 1789, the first poliomyelitis outbreak in USA was in 1843, following which more than 21 000 paralytic cases were reported in 1952 [6]. Being a member of the enterovirus subtype, it has three serotypes namely P1, P2 and P3. It is inactivated rapidly and almost effectively by heat, ultraviolet light, chlorine and formaldehyde [7]. The route of infection being through the mouth, primary multiplication of the virus occurs at the site of implantation in the pharynx and gastrointestinal tract and it continues to be excreted in the stool for several weeks, giving rise to two routes of pathogenesis namely faecal-oral (under unhygienic conditions) and oral-oral [6]. It spreads along the nerve fibres, destroying motor neurons, thus leading to flaccid paralysis and in some cases, just a sore throat and cough [6]. Polio was eradicated in the early 1990s in the USA and less than 3000 cases were reported worldwide in 2000 [8]. Immunization led to the interruption of wild poliovirus transmission, the concept of which was based on stopping the circulation of wild poliovirus by depriving its susceptible human host via immunization [8]. So the continued decrease of polio in many countries and the progressive disappearance of polio virus genetic lineages suggest that the interruption of human-tohuman transmission is achievable.
Several vaccines were introduced to cure the polio epidemic. However, each vaccine has its own specific use, depending on the stage of the disease and the serotype that has infected the person [6]. In the chronological order of licensing, we have inactivated vaccine in 1955, types 1 and 2 monovalent oral poliovirus vaccines (OPV) in 1961, type 3 monovalent OPV in 1962, trivalent OPV in 1963 and enhancedpotency inactivated polio vaccine (IPV) in 1987.
Features of polio vaccines
Two vaccines developed to immunize against the polio virus are IPV, originally developed by Jonas Salk and colleagues and the live-attenuated OPV developed by Albert Sabin [9]. OPV and IPV are live-attenuated vaccines that induce systemic immunity against paralytic poliomyelitis. A consequence of immunization through vaccination is decreased virus shedding due to an increase in poliovirus-specific immunoglobulin A (IgA) secreted in the intestine. OPV is said to be more effective than IPV as secretory antibodies are not found after immunization with IPV unless the individual is exposed to live polio virus prior to vaccination [10]. IPV, which is administered parenterally is safe and induces good serum immunity. However, the resistance to infection in mucosal surfaces is poor after IPV when compared to that following OPV or natural infections even in the case of modern enhanced-potency preparations (E-IPV).
IPV
In 2000, USA switched to IPV from OPV [6]. Using an inactive (dead) form of the virus that cannot cause polio, the IPV is given as a shot in the arm or leg (either as a subcutaneous or intramuscular injection) [6]. IPV can be given at the same time as other vaccines and contains all three serotypes of the polio vaccine virus. IPV has been reported to be highly effective in producing immunity to the poliovirus; 90% or more of the recipients become immune after two doses and at least 99% become immune after three doses. However, its duration of activity is uncertain [6].
Different ways of combining OPV and IPV strategies were considered in the post-eradication era [8]. An OPVIPV combination vaccine was initially used OPV was removed to leave an IPV protective layer alone. However, this strategy did not work as protection against the spread of OPV [8]. A major disadvantage of IPV, when compared to OPV, is the cost. Substantial technology as well as time would be required to prepare an adequate supply of IPV to meet global requirements. Another drawback is that some individuals get a red spot (inflammation) where the shot is given and some may get an allergic reaction. Hence, it is not advisable to administer the polio vaccine if there has been a severe allergic reaction to a previous dose of the polio vaccine or to the antibiotics streptomycin, polymyxin B or neomycin. No side effects have been reported on pregnant women [6].
Doses
Poliovirus vaccine can be given to both adults and children in varying doses. Although children are at a greater risk of suffering from the disease, even the adult immune system can fail to tackle the virus [6]. Routine vaccination of adults aged 18 y and above is not necessary, but laboratory workers and travellers to polio-endemic countries should be vaccinated [6].
Efficacy of polio vaccines
Polio virus vaccines are essentially produced from a mixture of three types of attenuated polioviruses that are usually grown in human diploid cell cultures or are produced from cultures propagated in monkey kidney cells. The live OPV usually contains attenuated, live polioviruses. OPV controls the wild-type poliovirus circulation in the human body by inducing intestinal immunity against poliovirus reinfection [11]. It persists in the pharynx for one to 2 w and is then excreted through faeces for several weeks or longer, after the administration of the vaccine [11]. The vaccine virus can be transmitted through physical contact, thus resulting in the immunization of individuals.
The effectiveness of OPV has been amply demonstrated by its success in preventing poliomyelitis and in inhibiting the circulation of wild-type polioviruses in almost all countries. The OPV is essentially a three-dose series against any of three poliovirus serotypes in more than 95% of recipients worldwide [11]. Data available on antibody persistence suggest that immunity is usually prolonged and mostly lifelong.
Rotavirus Vaccine
Rotaviruses (RV) belong to the family of Reoviridae [12]. Their ability to be maintained in animal models has helped us to study the mechanisms of pathogenesis and immunity for the development of vaccines [12]. RV is the major cause for acute gastroenteritis in infants and young children worldwide. They are transmitted faecal-orally and are extremely infectious, leading to serious dehydration and death if left untreated [12]. Treatment is mainly by oral or intravenous. Diarrhoea caused by RV is the cause for over half a million deaths and 2.4 million hospitalizations worldwide every year [13]. Symptoms can range from mild watery diarrhoea to severe diarrhoea with forceful vomiting, abdominal distress and fever and can lead to serious complications, including dehydration, electrolyte imbalance, seizures and death [14].
The strains of the virus are categorized on the basis of serotypes that are defined by two surface proteins: VP7 (glycoprotein; the G type) and VP4 (proteasesensitive protein; the P type) [15]. There are five major combinations of G and P types worldwide: G1P [8], G2P [4], G3P [8], G4P [8] and G9P [8] [15]. The two main constituents of the rotavirus are the two shells the inner core containing the antigenic protein VP6 and the outer core containing VP4 and VP7. Virus neutralization mainly involves neutralizing VP7 [15].
Mechanisms of action
Two mechanisms of protection against RV have been proposed [16]. The first one is based on the antibodies that recognize serotype-specific antigens within outer capsid VP4 or VP7 proteins of the virus particle [18]. The second one is based on effectors other than neutralizing antibodies following natural infections. These theories form the basis of development of vaccines [16].
Types of RV vaccines
Diarrhoea caused by RV strains G1, G2, G3 and G4 is prevented by RotaShield vaccine, the mechanism of which is not completely understood [17]. The vaccine, consisting of four live viruses-a rhesus rotavirus (serotype 3) and three rhesus-human reassortant viruses (serotype 1, 2 and 4), stimulates the production of IgG and IgA antibodies, initiating a local immune response [17]. Initially, live attenuated vaccines were developed as they were efficient in mimicking the recurring natural infections, resulting in protection against the virus. However, due to a vast number of intussusception cases in patients who received this vaccine, this quadrivalent vaccine was withdrawn from the market. Intussusception occurs when part of the intestine invaginates into the other part of the intestine causing bowel blockage [17]. Rotarix, a single strain vaccine, underwent large, extensive safety trials and was proven safe for use in Singapore, Finland and Latin America [18]. It was not associated with fever and was highly immunogenic. The vaccine also did not interfere with other concomitantly used vaccines, even polio vaccines [18].
Rotarix single strain vaccine has given the best correlation between rotavirus antibody after vaccination and protection against the infection when compared to other vaccines, such as RotaTeq and RotaShield [17]. In three placebo-controlled trials, 61-91% of vaccinated infants developed rotavirus-specific IgA antibodies after two doses [19]. RotaTeq (RV5) is a pentavalent human bovine reassortant vaccine, developed from the attenuated bovine rotavirus strain WC3, a G6P [5] [19]. WC3 was reassorted with human strains to yield the five viruses that comprise the licensed vaccine: each of the parental bovine virus with one gene segment substitution from a human strain, leading to expression of either G1, G2, G3 or G4 [20].
Diphtheria-Pertussis-Tetanus (Dpt) Vaccine
DPT stands for diphtheria, pertussis (whooping cough) and tetanus, three serious bacterial diseases caused by Corynebacterium diphtheriae, Bordetella pertussis and Clostridium tetani, respectively. Diphtheria and pertussis are spread from one person to another, while tetanus enters the body through cuts and wounds, which may not appear dangerous and are often unnoticed or neglected [21].
Symptoms and harmful effects of DPT
Diphtheria causes a thick covering in the back of the throat. It can lead to breathing problems, paralysis, heart failure and even death in severe cases [21]. Pertussis causes severe coughing spells in infants, making it difficult for them to eat, drink or even breathe. These spells can last for weeks. Pertussis can lead to pneumonia, convulsions, brain damage and death [21]. Tetanus causes painful tightening of the muscles all over the body and can lead to locking of the jaw, because of which, patients cannot open their mouths or swallow [22].
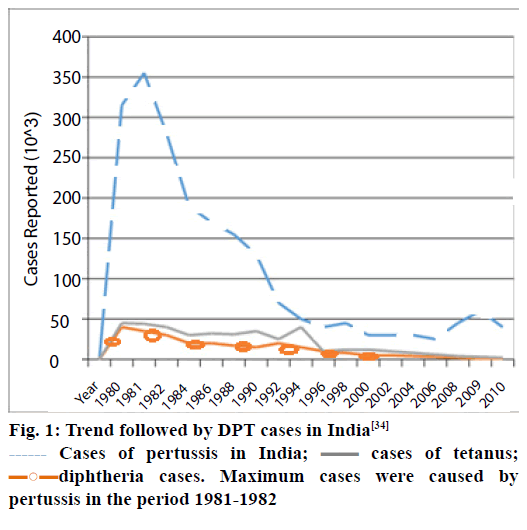
The three diseases are prevalent in infants and children, as compared to adolescents. DPT can be prevented by vaccinating children [22]. The vaccine is usually given as an intramuscular injection. The trend followed by DPT cases in India is depicted in Figure 1 [23].
Figure 1: Trend followed by DPT cases in India[34]
 Cases of pertussis in India;
Cases of pertussis in India;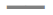 cases of tetanus;
cases of tetanus;
 diphtheria cases. Maximum cases were caused by
pertussis in the period 1981-1982
diphtheria cases. Maximum cases were caused by
pertussis in the period 1981-1982
Types of DPT vaccines
There are three types of DPT vaccines namely diphtheria pertussis whole-cell toxoids (DPwT), diphtheria toxoids acellular pertussis (DPaT) and toxoids diphtheria acellular pertussis (Tdap). DPwT contains the entire killed form of the pertussis bacterium, whereas DPaT contains only few proteins of the pertussis bacterium (acellular pertussis). Both vaccines are very effective, but DPaT has fewer side effects. Tdap contains lower potency of diphtheria toxoid and pertussis protein [24]. The DT vaccine affords protection against diphtheria and tetanus, whereas the TT vaccine protects only against tetanus. The DT vaccine has 1/10th the dose of diphtheria toxoid present in DT/DPT and is used in patients in whom the pertussis component is causes a severe allergic reaction [24].
TT is routinely used for children older than 10 y of age as they do not need both diphtheria and pertussis components. It is given as a booster dose at the ages of 10 and 16 y to children who have already received their primary doses/boosters of DPT/DT [24]. All children should get five doses of DPT vaccine at the end of 6 w, 10 w, 14 w, 1st booster after 16 to 18 mon and 2nd booster after 5 y. Even after three doses of DPT vaccine, the protection level is only 80%. Hence, it is prudent to administer two boosters for better protection against DPT [21].
Features of DPT vaccines
DTwP vaccines essentially consist of a mixture of the detoxified toxins (toxoids) of tetanus and diphtheria and inactivated B. pertussis, all of which have been adsorbed onto an aluminum salt [25]. Tetanus, DTaP vaccines contain pertussis toxin and one or more of the following components of B. pertussis namely fimbrial antigens (FIM), which are found on fimbriae that assist the bacteria to adhere to the epithelia while pertactin (PRN, also known as 69 kDa protein) is an autotransporter, which helps the microbe to get to the cell surface [25].
Even though serological correlates of pertussis immunity are not well defined, it has been determined through various experiments that DTaP vaccines have the potential to stimulate immune responses that far exceed those to DTwP vaccines with respect to the antibodies that are measured [25]. As far as PT is concerned, the immunogenicity seems to depend on antigen concentration, antigen derivation and formulation. In case of diphtheria, after intramuscular injection, the diphtheria toxoid induces the formation of antibodies against the diphtheria toxin. Similarly, in tetanus, after intramuscular injection, tetanus toxoid induces the formation of antibodies against the tetanus toxin. In pertussis, after intramuscular injection, whole-cell or acellular pertussis vaccine induces the formation of several clinically protective antibodies. However, the levels of antibodies that are necessary for protection have not been calculated till date [25].
The Multicenter Acellular Pertussis Trial was a safety and immunogenicity study that was conducted in six centers across USA and was sponsored by the National Institutes of Health (NIH). It compared the antibody response of infants who were vaccinated at 2, 4 and 6 mon of age with DTwP or with any of the 13 different DTaP vaccines [26]. Antibodies to pertussis antigens were measured in serum samples taken before administration of the first dose and 1 mon after the administration of the third dose of any one of the 13 different DTaP vaccines [26]. The following results were observed: 86% and 99% of children had fourfold or greater increases in titers of antibody to filamentous hemagglutinin adhesin (FHA) and PT, respectively. More than 90% of children administered with one of the 13 different DTaP vaccines and more than 90% of those administered DTwP developed diphtheria and tetanus antibody levels, which were indicative of immunity to these diseases. In case of diphtheria, primary immunization with DPT protects more than 95% of the population for a minimum of 10 y. While in tetanus, primary immunization with DPT protects 95% of individuals for at least 10 y [26]. In the case of pertussis, after primary immunization with DPT vaccine, immunity to pertussis usually persists through childhood, but is thought to decrease over time.
Lifelong immunity is attained through subsequent mild pertussis infection [26].
Efficacy of DPT vaccines
Diphtheria and tetanus toxoids induce antibodies against toxins made by C. diphtheriae and C. tetani. Pertussis vaccine protects against B. pertussis. The following are common adverse reactions caused by DPT vaccines in certain cases. There are two types of adverse reactions seen in the body-local and systemic side effects. Local side effects, such as pain, swelling and redness, occur in a specific site where the vaccine is administered. Besides these, difficulty in walking is seen in 30-40% of DPT vaccines. These symptoms persist for 24-72 h and respond to paracetamol. Sometimes a nodule forms at the injection site, which may persist for several days to weeks. It may soften and form a sterile abscess, but if it shows fluctuation, it should be drained [24]. Systemic side effects occur due to absorption of vaccine parts into the bloodstream and acts at multiple levels of the body. They include fever, lassitude, anorexia, vomiting, irritability, excessive crying and is seen in 30-40% of patients. Fever is usually mild to moderate, lasts for 2-3 d and responds to paracetamol [24]. Sometimes, the pertussis component of DPT is responsible for fever of more than 105°F, excessive crying and screaming spells lasting for more than 4 h and convulsions [27]. If a patient develops any of those adverse reactions, it is a contraindication to further use of DPT, only DT should be used in such a case. Such reactions should be immediately reported to the physician [28]. The DPT vaccine was found to be associated with an increased risk of febrile seizures only on the day of vaccination [24]. Also, the number of febrile seizures attributable to the administration of DPT was estimated to be 6-9 per 1 00 000 children. When compared with other children with febrile seizures that were not associated with vaccination, the children who had febrile seizures after vaccination were not found to be at higher risk for subsequent seizures or neurodevelopmental disabilities [28]. While DTaP is a relatively new vaccine first approved for use in USA in 1991, its history spans over half a century. Year-wise incident occurrence of DPT is summarized in Table 2 [22,29-31]
| Year | Incident |
|---|---|
| Early 1940s | DT whole-cell P-incidence of three life-threatening illnesses plummeted. Drawback: whole-cell pertussis component was associated with a range of adverse side effects. |
| Mid 1940s | After a series of deaths and adverse reactions, safety of whole-cell pertussis was of huge concern, especially in USA and Europe. |
| 1970-1985 | Excessive media coverage led to several product liability issues and exiting of manufacturers from the market[29]. In 1984, the sole supplier of DPT vaccine was in USA |
| 1981 | Meanwhile, a safer version of DTwP called DTaP, containing an acellular pertussis component, was produced by Japanese scientists and licensed[29,31]. |
| 1986 | Against this backdrop, Congress in USA responded by passing the National Childhood Vaccine Injury Act that was immediately effective in controlling the DPT lawsuit[22]. |
Table 2: Year-Wise incident occurrence for dpt vaccines
Paracetamol or ibuprofen can be used to treat pain, swelling, redness, difficulty in walking, and fever. It takes 2-3 d for the symptoms to disappear. In case of abscesses, antibiotic treatment and drainage are to be adopted [27].
Current challenges of DTaP vaccination
Diphtheria and tetanus are quite uncommon today. However, the main issue is the deteriorating immunity to pertussis following administration of five doses of DTaP. Despite the higher efficacy shown by this vaccine, incidence of pertussis has increased substantially in 7 to 10 y olds in recent years [32]. Thus, the duration of protection may wane earlier than expected and is currently not well established [32].
Failed Clinical Trials
Cutter incident
Approximately 2 00 000 children received a polio vaccine in April 1955 in USA [33]. The process of inactivating the live virus was defective. Several reports of paralysis appeared and the vaccination campaign was abandoned in the first few months [33]. The attenuated vaccine used contained Sabin’s strain and was manufactured by Cutter Laboratories [33]. Sabin’s attenuated strain replaced Salk’s inactivated strain, wherein the latter involved inactivating the virus by using formaldehyde. Although Sabin’s attenuated strain proved advantageous due to its oral administration and boosting contact immunity, the strain could be re-activated in the gut. The vaccine caused 40 000 cases of polio, causing varying degrees of paralysis and even death in 200 children. Lack of experience and inefficient expertise led to the outbreak of this disaster [33].
Status in India
In 2005, OPV failed to eradicate polio in India due to certain misconceptions, the main one being the concept of herd immunity [34]. The virus replicates in the gut and is shed in the faeces. It was thought that the oral form of the vaccine when excreted through the faeces of an immunized person would immunize even unvaccinated persons coming in contact with it. However, this concept of herd immunity failed because the attenuated strain in OPV had low infectivity and the vaccine load was low in faeces [34]. Also, while replicating in the gut, the attenuated virus reverted into its pathogenic form to cause paralytic poliomyelitis [34]. In 2007, there was a sudden increase in the number of polio cases [35]. This was due to “vaccine pressure” that increased after the failure of the primary vaccine, which gave rise to mutant stains that were resistant to the antibodies produced [36]. One of the main reasons for failure was the low efficacy of oral polio vaccine against the wild type polio virus. The number of cases declined steadily after the peak of the 2007 outbreak [35]. Towards the end of 2009, wild type polio virus 1 (WPV1) had almost disappeared, but WPV3 was still prevalent. Hence, it was decided to use the monovalent form of the oral polio virus to eradicate WPV3 [35]. In 2010, the bivalent form of the oral polio virus was introduced to simultaneously reduce the number of cases due to WPV1 and WPV3. Currently, India is declared as polio-free after struggling for years to eradicate polio with the last case reported in 2011.
Rotavirus vaccine failure
RotaShield is a tetravalent, live attenuated rhesus vaccine (RRV-TV) that was released by American Home Products in August 1998 [36]. RotaShield was nearly 100% effective to prevent rotaviral diarrhoea [36]. The path to vaccine development and implementation had been tortuous with a major setback in 1999 due to an unexpected association of the vaccine with intussusceptions because of which, it was withdrawn from the market [36]. Researchers had shown no significant difference in the rates of intussusception between vaccinated and the control populations because it failed to consider the cumulative risk of intussusception or had not considered the onset of wild-type rotaviral infection in control populations [37]. Currently a combination of Rotaeq and Rotarix is given to prevent infant diarrheal deaths due to RV infections. In spite of a spike in intussusception in a span of 7 d after administering the vaccine, the drawback is outweighed by its advantage of preventing RV-associated gastroenteritis [37].
DPT vaccine failure
A paediatric patient developed recurrent seizures and acute encephalopathy on administering the first dose of DPT. A computed tomographic (CT) scan of the brain revealed that the pertussis fraction of the vaccine was the reason for the acute encephalopathy [38]. When whole cell vaccines were initially prepared, the cells elicited an immune complex-mediated type III hypersensitivity reaction called Arthus reaction, which involved high concentrations of circulating antibodies [38]. According to a study in 1978, neurological illness, encephalitis and convulsions were reported after the administration of the whole cell vaccine [39]. The study also reported that local reactions were generally more frequent with increasing dosage. On the other hand, systemic reactions (with the exception of fever) were less frequent with increasing dosage [39]. According to Centers of Disease Control (CDC) report of 1991, infants who had convulsions or had immediate family members with such history, were more likely to have seizures following DPT vaccination. Currently, the acellular form of DPT (DTaP) is used as it is associated with a lower incidence of neurological complications.
Recent Developments
Zika virus (ZKV) and Zika virus vaccine development
ZKV is a Flavivirus that is borne by arthropods, such as mosquitoes [40]. It was first isolated from monkeys in Uganda in 1947 and was associated only with mild illness from the 1960s to the 1980s [40]. The first large outbreak of disease due to ZKV was reported only in 2007 from the Island of Yap [41]. Severe congenital microcephaly associated with ZKV has been reported in Brazil in 2015 [40]. Besides horizontal transmission through arthropod bites, transmission can also occur from mother to child, nosocomially, by transfusion, via bone marrow or organ transplantation and sexually [40]. Being a Flavivirus, ZKV is a single-stranded, positive sense RNA virus with a 10 794 kb genome with two flanking, non-coding regions [40]. Human skin fibrobasts, epidermal keratinocytes and immature dendritic cells were found to permit entry of ZKV [42]. ZKV-infectioninduced formation of autophagosomes, expression of antiviral antigen clusters (RIG-1, MDA-5 and TLR3) and activation of T cells (Th1, Th2, Th9 and Th17) [43].
The vaccine produced by Inovio Pharmaceuticals has shown promising results in mice by inducing robust and durable response. Researchers are currently working on a vaccine, named SynCon, as a means to protect people from the mosquito-transmitted Zika virus [44]. Vaxart began preclinical testing of an oral Zika virus vaccine. Vaxart’s vaccine platform enables delivery of recombinant vaccines using a convenient room temperature-stable tablet that can be shipped and stored without refrigeration and is ideally suited for viruses such as Zika [45]. National Institute of Allergy and Infectious Diseases (NIAID, Bethesda, USA), Hawaii Biotech (Honolulu, USA) and Bharat Biotech International Limited (Hyderabad, India) have developed candidate vaccines that are undergoing preclinical testing [46]. A Zika virus vaccine has been approved by US-Food and Drug Administration (USFDA) for phase I human trial [46]. The vaccine, GLS- 5700, is a synthetic DNA plasmid vaccine that encodes for the premembrane-membrane and envelope regions of ZIKV.
A prototype subunit and adenoviral-based Zika vaccine was developed by Kim et al., which encodes the T4 fibritin foldon trimerization domain (Efl) with a fused extracellular portion of the ZIKV envelope gene (E) [47]. Carboxymethyl cellulose microneedle array (MNA) was used to intradermally deliver this subunit vaccine [47]. The two vaccines were named, Ad5.ZIKVEfl and ZIKV-rEfl and were subsequently tested in C5BL/6 mice [47]. The study concluded with E-ZIKV antigen vaccination showing preliminary efficacy in controlling ZIKV infectivity [47].
Plants as bioreactors
When it comes to recent developments in vaccine development, transgenic plants are being heavily researched as promising expression systems for producing vaccine antigens [48]. Higher plants can be transformed into bioreactors by a stably integrated gene within the plant’s nuclear and/or chloroplast genome by the process of recombinant plant viruses infecting plants and acting as transient expression vectors [49]. These bioreactors can yield subunit vaccines for oral or parenteral administration [49].
In phase I clinical trials several plant-derived vaccine antigens have been found to be safe with the ability to induce sufficiently high immune response [48].
Estimations indicate that one acre of crop can provide for antigenic production to vaccinate millions of individuals, provided the antigen is expressed in the seeds of an edible legume [48].
Vaccine delivery using nanoparticles
Immune stimulating complexes (ISCOMs), liposomes and virus-like particles (VLPs) are nanoscale size (<1000 nm) materials that have the ability to behave as adjuvants and stabilize the vaccine antigens at the same time [50]. These nanoparticle-based vaccine delivery systems are able to modulate the immune response to an antigen by utilizing different pathways to enter antigen-presenting cells [50]. Their nanoscale nature and properties make them specifically suited for intradermal antigen delivery antigens or at mucosal surfaces [50].
VLPs
VLPs have a self-assembled viral envelope [51]. This envelope is generated from a single protein and forms a multimeric complex displaying a high density of epitopes [51]. VLPs have the ability to stimulate strong immune responses and also boast an ease of production [51-54].
VLPs are non-infectious and non-replicating because they assemble without encapsulating any viral RNA [50]. Integration of the packed genome into the host cell is prevented by deleting the genes coding for viral integrase prior to expression [55]. This procedure also safeguards against recombination with defective or live virus in an infected individual [55].
Liposomes
Liposomes are a phospholipid bilayer shell encompassing an aqueous core [56,57]. These liposomes can have one of two different constructions [50]. One of them is unilameller vesicles, which are made up of a single phospholipid bilayer [50]. The other one, is multilameller vesicles, which are made up of several concentric phospholipid shells separated by layers of water [50]. The presence of two distinctly differentially natured layers allows us to tailor the incorporation of molecules such as vaccines to either one of these layers, that is hydrophobic molecules within the phospholipid bilayers and hydrophilic molecules into the aqueous core [50].
ISCOMs nanoparticles
ISCOMs are colloidal saponin containing 40 nm micelles and are largely employed as self-adjuvanting vaccine delivery systems [50]. ISCOMs have immune stimulating properties and are hence chiefly used as a vaccine adjuvant in order to induce longer protection and a stronger immune response.
ISCOMs are largely composed of phospholipid, cholesterol and saponin [58-60]. These form when the above three constituents are mixed together under a specific stoichiometry, generally 1:1:5:0.1/1 (cholesterol:phospholipid:saponin) for classical ISCOMs [59,61].
ISCOMs have been used to entrap viral envelope proteins while recent applications include proteins from a range of parasites and bacteria [62-64]. The complexes that do not incorporate viral proteins are referred to as ISCOM matrices [60].
Vaxxas Nanopatch™
The NanopatchTM approach works by using a grid of approximately thousands of microprojections that are coated with vaccines. Their sharp tipped nature allows them to pierce through the outer layers of the skin when applied using a customized applicator device [65]. This vaccine coating incorporates a formulation containing the vaccine, which has been fixed onto the grid’s projections by using a gas-jet method to powder-coat the spikes. The dimensions of the projections are such that it allows the release of this coated material directly to the large numbers of key immune cells immediately below the skin surface [65].
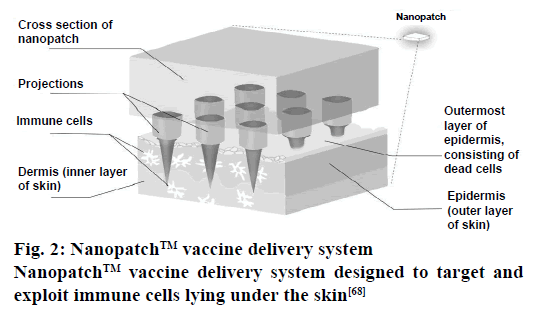
The Nanopatch infused with the vaccine is designed to be heat-stable, which allows it to transported without refrigeration [66]. Since, the process does not draw blood, it offers another key advantage, which is the lower risk of infection [67]. Nanopatch vaccine delivery system is illustrated in Figure 2 [68]. The projections are manufactured while keeping precise geometry as the most essential factor. Add to this the fact that the highest standards are used to ensure a uniform and consistent coating and the Nanopatch ends up delivering an effective amount of the vaccine that too to the desired target or targets [65].
Figure 2: Nanopatch™ vaccine delivery system
Nanopatch™ vaccine delivery system designed to target and
exploit immune cells lying under the skin[68]
The outer layer of the skin differs considerably in its properties mostly because of factors such as age, health, gender and even environmental humidity [65]. By utilizing the mechanical properties of our skin under different conditions, the applicator was designed to yield uniform delivery and penetration in almost all patient population variations [65].
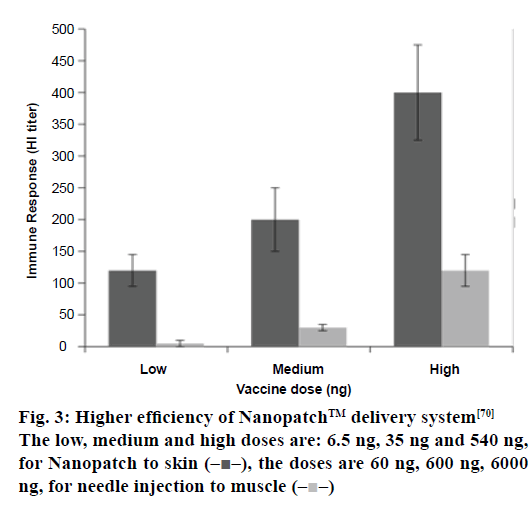
The features of the NanopatchTM delivery technology provide some important benefits that are improved immunogenicity: delivery of the vaccine directly to key immune cells, allows the enhancement of the immune response generated by the vaccine on a Nanopatch system or allow the generation of an effective immune response with a fraction of a full vaccine dose [69]. A display of higher efficiency of the Nanopatch delivery system is shown in Figure 3 [70]. No cold chain: the formulations used to coat the patches can ensure ambient temperature stability of the vaccine [71]. Temperature stability also introduces the option of distributing vaccines to parts of the world where cold chain infrastructure is unreliable or non-existent [71]. Needle-free: the use of an applicator allows the arrays of Nanopatch microprojections to penetrate through the protective outer layer of the skin to deliver a vaccine. Pain-free: the design of the microprojection array of the Nanopatch is such that it allows the targeting of vaccine delivery directly towards key immune cells, which are generally present just below the skin surface [72]. Since these projections do not penetrate up to a depth wherein they might interact with nerve endings, hence they don’t produce a pain response [72].
Figure 3:Higher efficiency of Nanopatch™ delivery system[70]
The low, medium and high doses are: 6.5 ng, 35 ng and 540 ng,
for Nanopatch to skin  the doses are 60 ng, 600 ng, 6000
ng, for needle injection to muscle
the doses are 60 ng, 600 ng, 6000
ng, for needle injection to muscle 
Cost-effective: the development process of the patch itself has renowned manufacturing techniques incorporated into it to allow it to undergo low cost and large volume manufacture.
In general, most of the contributions in the field of vaccines go unnoticed because a vaccination is inherently a preventive measure. As such, it gets clubbed along with all other prophylaxis entities that seldom get their due credit. With the recent omnipresence of vaccines and government vaccination programs, vaccines have been shown to impart safety, security and stability to health and biological status. Vaccines can save the lives of many children, a very important benefit in any society. The ingredients in vaccines are tested and proved to be safe in the amounts used; any adverse effect reported is usually due to bad manufacturing practice or quality control. Major medical organizations and healthcare departments classify vaccines as safe, which can be a useful guideline in vaccination programs. Adverse reactions to vaccines are extremely rare and in most cases, can be avoided, by a simple allergy patch test to save the individual from a more extensive reaction. By having a significant population vaccinated, the rampant spread of diseases, disease-causing agents and potential pandemics can be curbed. Vaccines have contributed to the eradication of smallpox and the suppression of nearly eradicated diseases such as polio.
Conflict of interest
The authors report no conflict of interest.
Acknowledgements
All the authors have contributed equally to this paper. The authors thank Natasha Pathil for her assistance in Figure 2.
Financial support and sponsorship
Nil.
References
- Clem AS. Fundamentals of Vaccine Immunology. J Glob Infect Dis 2011;3:73-8.
- Hamborsky J, Kroger A, Wolfe S. Epidemiology and Prevention of Vaccine-Preventable Diseases. 13th ed. Washington (DC): Public Health Foundation; 2015.
- http://www.cdc.gov/vaccines/vac-gen/additives.htm.
- Petrovsky N, Aguilar JC. Vaccine adjuvants: Current state and future trends. Immunol Cell Biol 2004;82:488-96.
- https://www.drugs.com/cg/inactivated-polio-vaccine-ambulatory-care.html.
- Murdin AD, Barreto L, Plotkin S. Inactivated poliovirus vaccine: Past and present experience. Vaccine 1996;14:735-46.
- apps.who.int/iris/bitstream/10665/68205/1/WHO_V-B_03.11_eng.pdf.
- Knobler S, Lederberg J, Pray LA. Considerations for Viral Disease Eradication. Washington (DC): National Academies Press (US); 2002.
- Zurbriggen S, Tobler K, Abril C, Diedrich S, Ackermann M, Pallansch MA, et al. Isolation of sabin-like polioviruses from wastewater in a country using inactivated polio vaccine. Appl Environ Microbiol 2008;74:5608-14.
- Valtanen S, Roivainen M, Piirainen L, Stenvik M, Hovi T. Poliovirus‐specific intestinal antibody responses coincide with decline of poliovirus excretion. J Infect Dis 2000;182:1-5.
- https://www.cdc.gov/mmwr/preview/mmwrhtml/00046568.htm.
- Westerman LE, Jiang B, McClure HM, Snipes-Magaldi LJ, Griffin DD, Shin G, et al. Isolation and characterization of a new simian rotavirus, YK-1. Virol J 2006;3:40.
- Correia JB, Patel MM, Nakagomi O, Montenegro FMU, Germano EM, Correia NB, et al. Effectiveness of monovalent rotavirus vaccine (Rotarix) against severe diarrhea caused by serotypically unrelated G2P[4] strains in Brazil. J Infect Dis 2010;201:363-9.
- Plosker GL. Pharmacoeconomic spotlight on rotavirus vaccine RIX4414 (RotarixTM) in developed countries. Drugs R D 2012;12:239-44.
- Gerald TK, Fontaine O, Bhargava A, Cynthia BP, Zulfiqar A, et al. Diarrheal Diseases. In: Jamison DT, Breman JG, Measham AR, Alleyne G, Claeson M, Evans DB, editors. Disease Control Priorities in Developing Countries. 2nd ed. Washington (DC): Oxford University Press; 2006. p. 371-88
- Ward R. Mechanisms of protection against rotavirus infection and disease. Pediatr Infect Dis J 2009;28:S57-9.
- Desselberger U. Rotaviruses. Virus Res 2014;190:75-96.
- Ward RL, McNeal MM, Steele DA. Why does the world need another rotavirus vaccine? TherClin Risk Manag 2008;4:49-63.
- Bucardo F, Rippinger CM, Svensson L, Patton JT. Vaccine-derived NSP2 segment in rotaviruses from vaccinated children with gastroenteritis in Nicaragua. Infect Genet Evol 2012;12:1282-94.
- Dennehy PH. Rotavirus vaccines: An overview. ClinMicrobiol Rev 2008;21:198-208.
- http://www.seruminstitute.com/content/faq_dtp.htm.
- Galanis E. Changing epidemiology and emerging risk groups for pertussis. Can Med Assoc J 2006;174:451-2.
- Lahariya C. A brief history of vaccines and vaccination in India. Indian J Med Res 2014;139:491-511.
- http://www.pediatriconcall.com/fordoctor/Diseases_a_z/article.aspx?artid=387.
- Bass JW. Pertussis: current status of prevention and treatment. Pediatr Infect Dis 1985;4:614-9.
- Michael D. Decker, Kathryn M. Edwards. The multicenteracellular pertussis trial: An overview. J Infect Dis 1996;174:270-5.
- https://www.cdc.gov/mmwr/preview/mmwrhtml/00048610.htm.
- Barlow WE, Davis RL, Glasser JW, Rhodes PH, Thompson RS, Mullooly JP, et al. The risk of seizures after receipt of whole-cell pertussis or measles, mumps and rubella vaccine. N Engl J Med 2001;345:656-61.
- https://www.cdc.gov/mmwr/preview/mmwrhtml/00000452.htm.
- Noble GR, Bernier NH, Esber EC, Hardegree MC, Hinman AR, Klein D, et al. Acellular and whole-cell pertussis vaccines in Japan. Report of a visit by US scientists. J Am Med Assn 1987;257:1351-6.
- Mowery DC, Mitchell V. Improving the reliability of the U.S. vaccine supply: an evaluation of alternatives. J Health Polit Policy Law 1995;20:973-1000.
- Tartof SY, Lewis M, Kenyon C, White K, Osborn A, Liko J, et al. Waning immunity to pertussis following 5 doses of DTaP. Pediatrics 2013;131:e1047-52.
- Fitzpatrick M. The cutter incident: How America’s first polio vaccine led to a growing vaccine crisis. J R Soc Med 2006;99:156.
- Paul Y. Why has polio eradication program failed in India? Indian Pediatr 2008;45:381-8.
- John JT, Vashishtha VM. Eradicating poliomyelitis: India’s journey from hyperendemic to polio-free status. Indian J Med Res 2013;137:881-94.
- https://web.stanford.edu/~siegelr/ShadmanRotashieldPaper.pdf.
- Jiang V, Jiang B, Tate J, Parashar UD, Patel MM. Performance of rotavirus vaccines in developed and developing countries. Hum Vaccin 2010;6:532-42.
- Patel MK, Patel TK, Tripathi CB. Diphtheria, pertussis (whooping cough) and tetanus vaccine induced recurrent seizures and acute encephalopathy in a pediatric patient: Possibly due to pertussis fraction. J PharmacolPharmacother2012;3:71-3.
- Cherry JD. Historical review of pertussis and the classical vaccine. J Infect Dis 1996;174:S259-63.
- Musso D, Gubler DJ. Zika Virus. ClinMicrobiol Rev 2016;29:487-524.
- http://www.who.int/mediacentre/factsheets/zika/en/.
- Hamel R, Dejarnac O, Wichit S, Ekchariyawat P, Neyret A, Luplertlop N, et al. Biology of Zika virus infection in human skin cells. J Virol 2015;89:8880-96.
- Tappe D, Perez-Giron JV, Zammarchi L, Rissland J, Ferreira DF, Jaenisch T, et al. Cytokine kinetics of Zika virus-infected patients from acute to reconvalescent phase. Med MicrobiolImmunol 2016;205:269-73.
- http://ir.inovio.com/news/news-releases/news-releases-details/2016/Inovio-Pharmaceuticals-DNA-Vaccine-for-Zika-Virus-Induces-Robust-Immune-Responses-in-Preclinical Study/default.aspx.
- http://www.businesswire.com/news/home/20160303005451/en/Vaxart-Begins-Preclinical-Testing-Oral-Zika-Virus.
- http://www.the-scientist.com/?articles.view/articleNo/46370/title/Zika-Vaccine-Ready-for-Human-Trials/.
- Kim E, Erdos G, Huang S, Kenniston T, Falo LD Jr., Gambotto A. Preventative Vaccines for Zika Virus Outbreak: Preliminary Evaluation.EBioMedicine2016;13:315-20.
- Tiwari S, Verma PC, Singh PK, Tuli R. Plants as bioreactors for the production of vaccine antigens. BiotechnolAdv 2009;27:449-67.
- Sala F. Vaccine antigen production in transgenic plants: Strategies, gene constructs and perspectives. Vaccine 2003;30;21:803-8.
- Gregory AE, Titball R, Williamson D. Vaccine delivery using nanoparticles. Front Cell Infect Microbiol2013;3:13.
- Grgacic EVL, Anderson DA. Virus-like particles: Passport to immune recognition. Methods 2006;40:60-5.
- Kingsman SM, Kingsman AJ. Polyvalent recombinant antigens: A new vaccine strategy. Vaccine 1988;6:304-6.
- Roldão A, Mellado MCM, Castilho LR, Carrondo MJ, Alves PM. Virus-like particles in vaccine development. Expert Rev Vaccines 2010;9:1149-76.
- Zeltins A. Construction and characterization of virus-like particles: A review. MolBiotechnol 2012;53:92-107.
- Young KR, McBurney SP, Karkhanis LU, Ross TM. Virus-like particles: Designing an effective AIDS vaccine. Methods 2006;40:98-117.
- Heurtault B, Frisch B, Pons F. Liposomes as delivery systems for nasal vaccination: Strategies and outcomes. Expert Opin Drug Deliv 2010;7:829-44.
- Henriksen-Lacey M, Korsholm KS, Andersen P, Perrie Y, Christensen D. Liposomal vaccine delivery systems. Expert Opin Drug Deliv2011;8:505-19.
- Kersten G, Teerlink T, Derks H, Verkleij A, Wezel van, Crommelin DJ, et al. Incorporation of the major outer membrane protein of Neisseria gonorrhoeae in saponin-lipid complexes (iscoms): Chemical analysis, some structural features and comparison of their immunogenicity with three other antigen delivery systems. Infection and immunity. Infect Immun 1988;56:432-8.
- Lövgren K, Morein B. The requirement of lipids for the formation of immunostimulating complexes (iscoms). BiotechnolApplBiochem 1988;10:161-72.
- Barr IG, Sjölander A, Cox JC. ISCOMs and other saponin based adjuvants. Adv Drug Deliv Rev 1998;32:247-71.
- Kersten GFA, Spiekstra A, CoenBeuvery E, Crommelin DJA. On the structure of immune-stimulating saponin-lipid complexes (iscoms). BiochimBiophysActa 1991;1062:165-71.
- Morein B, Simons K. Subunit vaccines against enveloped viruses: Virosomes, micelles and other protein complexes. Vaccine 1985;3:83-93.
- Claassen I, Osterhaus A. The iscom structure as an immune-enhancing moiety: Experience with viral systems. Res Immunol 1992;143:531-41.
- Morein B, Lövgren K, Rönnberg B, Sjölander A, Villacrés-Eriksson M. Immunostimulating complexes. ClinImmunother 1995;3:461-75.
- http://www.vaxxas.com/nanopatch-technology.
- Williams SC. Under the skin of intradermal vaccines. ProcNatlAcadSci USA 2013;110:10049-51.
- Xiong Z, Dong X, Sun P, Zhang Y. [Dose-effect research using nanopatch to deliver siRNA in the inhibition of HPV gene expression]. Sheng Wu Yi Xue Gong Cheng XueZaZhi 2013;30:1283-9.
- Pallavi B, Thofeeq MD, Venkat Reddy B Ch. An Advanced Approach of NFID - Nanopatch Technology. Austin J NanomedNanotechnol 2015;3:1039.
- Muller DA, Pearson FE, Fernando GJP, Agyei-Yeboah C, Owens NS, Corrie SR, et al. Inactivated poliovirus type 2 vaccine delivered to rat skin via high density microprojection array elicits potent neutralising antibody responses. Sci Rep 2016;6:22094.
- http://antyweb.pl/nanopatch-szczepienia-bez-igly/
- Pearson FE, McNeilly CL, Crichton ML, Primiero CA, Yukiko SR, Fernando GJP, et al. Dry-coated live viral vector vaccines delivered by nanopatchmicroprojections retain long-term thermostability and induce transgene-specific T cell responses in mice. PLoSOne 2013;8:e67888.
- Suh H, Shin J, Kim YC. Microneedle patches for vaccine delivery. ClinExp Vaccine Res 2014;3:42.