- *Corresponding Author:
- B. N. Suhagia
Department of Quality Assurance, L. M. College of Pharmacy, Navrangpura, Ahmedabad–380 009, India.
E-mail: patelhary@rediffmail.com
| Date of Submission | 29 December 2004 |
| Date of Revision | 13 June 2005 |
| Date of Acceptance | 25 March 2006 |
| Indian J Pharm Sci, 2006, 68 (1): 127-130 |
Abstract
A simple and sensitive spectrophotometric method has been developed for determination of betaxolol hydrochloride in bulk powder, and its pharmaceutical dosage forms. In the proposed method, betaxolol is oxidized with sodium periodate, to liberate formaldehyde, which is determined in situ, using acetyl acetone in the presence of ammonium acetate. A yellow coloured chromogen was obtained having absorption maxima at 405 nm. The method is found to be linear in the concentration range of 5-30 µg/ml, with a regression co-efficient of 0.9997. No significant difference was found between the proposed method and official method, when one tailed and two tailed t-test are applied. Various reaction parameters such as concentration of sodium hydroxide and sodium periodate, time required for oxidation, reagent concentration, and time for maximum colour intensity were optimized. The method was validated, and can be used successfully to assay betaxolol in bulk powder, and its pharmaceutical dosage forms viz. ophthalmic solutions.
Betaxolol, potent adrenergic blocker [1] is structurally related to metoprolol. Chemically [2-3] it is 1-(4-(cyclopropylmethoxy) ethyl) phenoxy)-3-isopropylaminopropan-2-ol hydrochloride. It is used in glaucoma and ocular hypertension, as one drop of 0.5% solution twice daily [4]. The recommended starting dosage for mild to moderate hypertension is 10 to 20 mg per day in a single oral dose [5]. A dose of 10 mg per day should be used in elderly and patients with severe renal diseases.
The ophthalmic solution of betaxolol hydrochloride is official in the United States Pharmacopoeia, and assayed by high performance liquid chromatographic method [3]. Literature survey reveals that betaxolol is estimated in pharmaceuticals and biological fluids by HPLC [6-12] and spectropotometric methods [13].
In the proposed investigation, an attempt has been made to develop a simple, economical, accurate, and reproducible spectrophotometric method for estimation of betaxolol in pharmaceutical formulation. In the proposed method, betaxolol hydrochloride is first neutralized with sodium hydroxide, and the librated base is oxidized with sodium periodate to produce formaldehyde [14]. The reaction mixture, on treatment with acetyl acetone in presence of ammonium acetate, gives yellow coloured product having maximum absorbance at 405 nm. The proposed method was successfully applied for determination of betaxolol in bulk drug and pharmaceutical formulation.
A Double beam Shimadzu 160A UV/vis spectrophotometer with two matched quartz cells of 1 cm light path was employed for spectral measurement. A thermostatically controlled water bath (Remi Instruments, Mumbai) was used to control the temperature of the reaction mixture. Betaxolol hydrochloride USP working standard was procured as a gift sample from Torrent Pharmaceuticals Ltd., Ahmedabad. Sodium periodate, acetyl acetone (freshly distillated), sodium hydroxide, and anhydrous dibasic sodium phosphate were purchased from S. D. Fine Chem. Pvt. Ltd., Mumbai. Ammonium acetate (ExcelaR, Glaxo), acetonitrile (HPLC grade, Merck, Mumbai) and double distilled water were used.
Ammonium acetate- acetyl acetone reagent was prepared by dissolving 30 g of ammonium acetate in 50 ml of distilled water, followed by the addition of 1.0 ml of freshly distillated acetyl acetone. The final volume of this solution was adjusted to 100 ml with water, and stored in a refrigerator.
Betaxolol hydrochloride (25.0 mg) was accurately weighed and transferred to a 25 ml volumetric flask. It was dissolved and diluted to 25 ml with water. An aliquot (5.0 ml) was further diluted with water in 10 ml volumetric flask, to obtain the final concentration of 500 μg/ml.
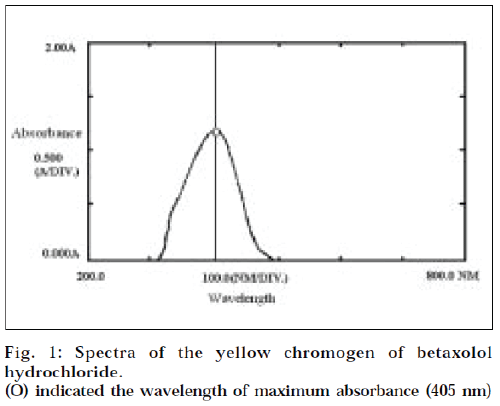
In a 25 ml volumetric flask, standard betaxolol solution (1.0 ml), sodium hydroxide solution (1.0 ml, 0.015M), and sodium periodate solution (2.0 ml, 0.01M) were pipetted successively. The reaction flasks were allowed to stand at 37° for 30 min, with occasional shaking. Ammonium acetate- acetyl acetone reagent (5.0 ml) was added and mixed thoroughly. The reaction mixture was heated on water bath at 60 for 5 min, and cooled to room temperature. The volume was adjusted upto the mark with distilled water. Absorbance of the coloured solution was scanned on Shimadzu UV/vis spectrophotometer from 800 nm to 200 nm, against reagent blank. Maximum absorbance was obtained at 405 nm (Fig. 1).
The standard solution of betaxolol hydrochloride (0.25-1.5 ml, 500 μg/ml) was pipetted out in a series of 25 ml volumetric flasks, and analyzed as above. The Beer’s law is obeyed in the concentration range of 5-30 μg/ml of betaxolol hydrochloride (Table 1).
| Concentration (µg/ml) | Absorbance at 405 nm ± SDa | % C.V. |
|---|---|---|
| 5 | 0.123 ± 0.0055 | 4.48 |
| 10 | 0.243 ± 0.0066 | 2.72 |
| 15 | 0.387 ± 0.0064 | 1.66 |
| 20 | 0.519 ± 0.0047 | 0.91 |
| 25 | 0.649 ± 0.0075 | 1.15 |
| 30 | 0.783 ± 0.0056 | 0.72 |
Accurately measured volume of ophthalmic solution, equivalent to 5 mg of betaxolol, was transfered into a 10 ml volumetric flask, and the volume was made up to mark with distilled water. The solution (1.0 ml) was pipetted in to a 25 ml volumetric flask, and analyzed as above. Amount of betaxolol hydrochloride was computed from the calibration curve (Table 2).
| Formulation | Label claim(%w/v) | % Amount found by | |
|---|---|---|---|
| Proposed method Mean ± S.D.b | Official method3Mean ± SDb | ||
| Bulk powder | - | 99.88 ± 0.98 | 99.29 ± 2.16 |
| Ophthalmic solution-1 | 0.5 | 100.09 ± 4.01 | 101.81 ± 5.22 |
| Ophthalmic solution-2 | 0.5 | 96.20 ± 0.64 | 96.40 ± 3.40 |
| t-test (one tailed)c | - | Tcal =0.212 | Ttab =6.313 |
| t-test (two tailed)c | - | Tcal =0.424 | Ttab =12.706 |
Table 2: Analysis Of Pharmaceutical Formulations
It is known that alpha-amino alcohol, in which the amine group is primary or secondary, liberates formaldehyde on periodate oxidation. [14] The librated formaldehyde produced yellow coloured chromogen, 3,5- diacetyl-1,4dihydrolutidine, on reaction with acetyl acetoneammonium acetate reagent solution. The coloured solution exhibits a wavelength of maximum absorption at 405 nm. Betaxolol has also alpha-amino alcohol moiety. Therefore, the above principle was used to analyze betaxolol in bulk, and in its dosage forms.
In the proposed method, betaxolol hydrochloride is first neutralized with sodium hydroxide and oxidized with sodium periodate, to produce formaldehyde. The librated formaldehyde was treated with acetyl acetone-ammonium acetate reagent solution. A yellow coloured product was obtained, having maximum absorbance at 405 nm (Fig. 1). The colour is found to be stable for at least 2 h.
In the proposed method, various parameters such as concentration of sodium hydroxide, concentration of sodium periodate, time required for oxidation, reagent concentration, and time for maximum colour development were studied and optimized, to obtain maximum colour intensity. The optical characteristics of Betaxolol hydrochloride such as Beer’s law limit, Sandell’s sensitivity, and molar extinction coefficient were determined. The linear regression equation for determination of betaxolol hydrochloride is y = 0.0266x 0.0143, with co-relation co-efficient being 0.9997. The RSD was found to be 0.72-4.48% (Table 3).
| Parameters | Values |
|---|---|
| Wavelength for measurement (nm) | 405 |
| Beer’s Law limit (µg/ml) | 5-30 |
| Molar absoptivity (lit/mole/cm) | 7.64 × 103 |
| Sandell’s sensitivity (µg/ml/cm2/0.001 abs. unit) | 4.06 × 10-2 |
| Regression equation (Yd) | |
| Slope (b) | 0.0266 |
| Intercept (a) | 0.0143 |
| Correlation coefficient (r)e | 0.9997 |
| Relative standard deviation (%)f | 0.72-4.48 |
| % Recovery (%) | 99.10-100.90 |
Table 3: Optical Characteristics Of The Proposed Method
For recovery study, known amounts of pure drug was added to the previously analyzed pharmaceutical preparations, the mixtures were analyzed by the proposed method, and the percent recovery was calculated, which was found to be 99.10-100.90% for betaxolol. The analysis was carried out in triplicate for two ophthalmic solutions. The results of analysis of ophthalmic solutions are shown in Table 2. The good recovery confirmed the accuracy and the specificity of the proposed method, and the lack of interference from the usual diluents and preservatives used in the manufacture of ophthalmic solutions. The developed method was also compared with the official method, [3] and no significant difference was observed. This method is particularly useful for routine in-process quality control for bulk drug, and its pharmaceutical preparations i.e. ophthalmic solutions.
Acknowledgements
The authors thank Torrent Pharmaceuticals Limited, Ahmedabad for supplying gift sample of working standard of betaxolol hydrochloride.
References
- Reynolds, J.E.F., Eds., In; Martindale: The Extra Pharmacopoeia, 30th Edn., The Pharmaceutical Press, London, 1994, 630.
- Budavari, S. Eds., In; The Merck Index, 12h Edn., Merck and Co. Inc., Whitehouse Station, N.J.,1996, 198.
- United State Pharmacopoeia, XXIII, NF XVIII, The USP convention, Inc, Rockville, MD, 1995, 197.
- Beresfold, R. and Heel, R.C., Drugs, 1996, 31, 6.
- Micaela, M.T., Buckley, K.L. and Goa, S.P., Drugs, 1990, 40, 75.
- Erram, S.V. and Tipnis, H.P., Indian Drugs, 1993, 30, 61.
- Erram, S.V. and Tipnis, H.P., Indian Drugs, 1992, 29, 553.
- Erram, S.V. and Tipnis, H.P., Indian Drugs, 1993, 30, 195.
- Santoro, M.I., Choi, H.S., Kedor, H. and Erika, R.M., Anal. Lett., 1995, 28, 71.
- Caqueret, H. and Bianchetti, G., J. Chromatogr. Biomed. Appl., 1984, 311, 199.
- Wong, Y.W.J. and Ludden, T.M., J. Chromatogr. Biomed. Appl., 1990, 534, 161.
- Bianchetti, G., Ganasia, J. and Morselli, P.L., J. Chromatogr., 1979, 176, 134.
- Rao, K.V., Kumar B.V., Rao, M.E. and Rao, S.S., Indian J.Pharm. Sci., 2003, 65, 516.
- Micolet, B.H. and Shinn, L.A., J. Amer. Chem. Soc., 1939, 61, 1614.