- *Corresponding Author:
- A. Husain
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Jamia Hamdard, New Delhi-110 062
E-mail: drasifhusain@yahoo.com
| Date of Submission | 11 April 2005 |
| Date of Revision | 29 September 2005 |
| Date of Acceptance | 10 August 2006 |
| Indian J Pharm Sci, 2006, 68 (4): 536-538 |
Abstract
Starting from 3-3-(4-methyl-benzoyl)propionic acid and using appropriate reagents, some new 2-arylidene-4-(4-methylphenyl)but-3-en-4-olides (IIIa-g) and their corresponding pyrrolone derivatives (IVa-d) have been synthesized. These compounds were evaluated for their antibacterial activity against Staphylococcus aureus and Escherichia coli as well as antifungal activity against Candida albicans . The compounds have considerable antifungal activity and moderate antibacterial activity.
γ-Lactones are well-known heterocycles of biological interest. Butenolides consist of unsaturated γ-lactone rings, which are also known as 2, 3 and 2, 5dihydrofuran-2-ones. Some well-known lactones of natural origin are santonin, cardiac glycosides, sesquiterpene lactones and patulin (an antibiotic) [1-3]. A wide range of biological activities are exhibited by this class of compounds, such as anthelmintic, ascaricidal, antifungal, antiinflammatory, antitumor and antiviral [1-7]. The γ-lactone ring present in butenolide derivatives is significantly reactive and can be utilized for the synthesis of nitrogen heterocycles, i.e., pyrrolones of potential biological activities [8-12] like antifungal, antibacterial and antiinflammatory. In view of potential biological activity of members of γ-lactone ring system, it was of interest to us to prepare some new derivatives in this class.
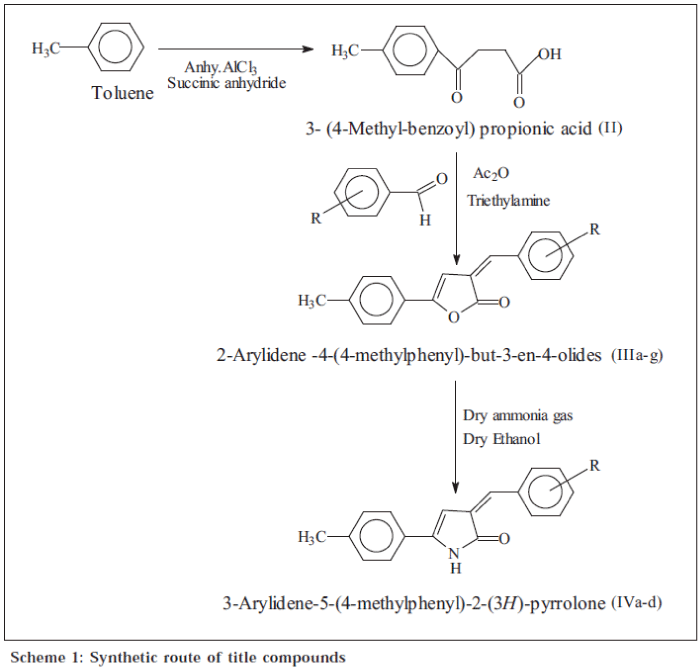
In the present communication, a series of 2-arylidene-4(4-methylphenyl)but-3-en-4-olides (IIIa-g) and 3-arylidene5-(4-methylphenyl)-2(3H)-pyrrolones (IVa-d) were screened for antibacterial activity against Staphylococcus aureus and Escherichia coli as well as antifungal activity against Candida albicans, the synthesis of which has been reported in our previous communication [13].
The starting compound, i.e., 3-(4-methyl-benzoyl) propionic acid (II), was obtained by the condensation of succinic anhydride with dry toluene in presence of anhydrous aluminium chloride, following Friedel-Craft’s acylation reaction conditions. 2-Arylidene-4-(4-methyl* phenyl)but-3-en-4-olides (IIIa-g) were synthesized from 3(4-methyl-benzoyl)propionic acid (II) by treating it with different aromatic aldehydes in the presence of triethylamine in acetic anhydride. Some of the selected butenolides (IIIa-d) were treated with dry ammonia gas at room temperature to obtain 3-arylidene-5-(4-methyl phenyl)-2(3H)-pyrrolones (IVa-d). The synthesized compounds were characterized by elemental analysis, IR, 1H NMR and mass spectral data. The compounds were synthesized according to Scheme 1.
All the compounds were screened for their antimicrobial activity against the microorganisms, viz, Staphylococcus aureus (NCTC-6571), Escherichia coli (ATCC-25922) and Candida albicans (ATCC-10231), in meat peptone agar medium at a concentration of 100 μg/ml by cup plate method [14]. Compounds inhibiting growth of one or more of the above microorganisms were further tested for minimum inhibitory concentration (MIC). A solution of the compounds was prepared in dimethylformamide (DMF) and a series of doubling dilutions prepared with sterile pipettes. To each of a series of sterile stoppered test tubes, a standard volume of nutrient broth medium was added. A control tube containing no antimicrobial agent was included. The inoculum consisting of an overnight broth culture of microorganisms was added to separate tubes. The tubes were incubated at 37° for 24 h and examined for turbidity. The tube with highest dilution showing no turbidity was the one containing compound with MIC. Results are presented in Table 1.
| Compound | Antibacterial activity | Antifungal activity Candida albicans | ||
|---|---|---|---|---|
| No. | R | S. aureus | E. coli | |
| I IIa | H | - | - | >100 |
| I IIb | 2,4-(Cl)2 | >100 | - | >100 |
| I II c | 3,4-(OCH3)2 | - | - | - |
| I I Id | 2-OCOCH3 | - | - | - |
| I IIe | 2,4-(OCOCH3)2 | - | - | - |
| I II f | 4-Br | >100 | >100 | >100 |
| I IIg | 4-N-(CH3)2 H | -50 | - | -50 |
| IVa | >100 | |||
| IVb | 2,4-(Cl)2 | 50 | >100 | 25 |
| Ivc | 3,4-(OCH3)2 | >100 | - | - |
| IVd | 2-OH | - | - | >100 |
*= in μg/ml; -= insignificant activity
Table 1: Antimicrobial activity (mic*) of the compounds iiia-g and iva-d.
3-(2,4-Dichloro-benzylidene)-5-(4-methylphenyl)-2(3H) pyrrolone (compound IVb) showed excellent activity against Candida albicans with MIC-25 μg/ml and good activity against Staphylococcus aureus with MIC-50 μg/ml. 3Benzylidene-5-(4-methylphenyl)-2(3H) pyrrolone (compound IVa) exhibited good activity against Staphylococcus aureus and Candida albicans with MIC-50 μg/ml. Both the compounds were active against Escherichia coli with MIC100 μg/ml. Majority of the compounds were insignificant in their action against Escherichia coli.
An analysis of results showed that introduction of chloro groups (electron withdrawing) in arylidene ring increases antimicrobial action, while introduction of hydroxyl and methoxy group (electron donating) decreases the antimicrobial activity. The replacement of oxygen atom in the butenolide ring by nitrogen atom (pyrrolones) enhances antimicrobial action.
Acknowledgements
The authors are thankful to (late) Hakim Abdul Hameed Sahib (Founder, Chancellor of Jamia Hamdard) and Mr. A. Mueed (President, Hamdard National Foundation) for providing facilities to carry out this work. Two of the authors (MMA and SL) are grateful to the University Grant Commission (UGC), New Delhi, for providing research fellowships.
References
- Lee, K.H., Kim, S.H. and Piantadosi, C., J. Pharm. Sci., 1976, 63, 1163.
- Gringuaz, A., In: Introduction to Medicinal Chemistry, Wiley-VCH, New York 1997, 305.
- Avetisyan, A.A. and Tokmadzhyan, G.G., Chem. Heterocycl.Compd., 1987, 23, 595.
- Khan, M.S.Y. and Husain, A., Pharmazie, 2002, 57, 448.
- Khan, M.S.Y., Husain, A. and Sharma, S., Indian J. Chem., 2002,41B, 2160.
- Michiyoshi, M., MasahKliro, K. and Mikio, M., Jpn. Kokai TokkyoKoho JP, 2001, 278, 708, through Chem. Abstr., 2001, 135,268773c.
- Yi-Chen, C., Fang-Rong, C. and Yang-Chang, W. TetrahedronLett., 1999, 40, 7513, through Chem. Abstr., 2000, 132, 2037k.
- Awad, W.I., Hashem, A.I. and El-Badry, K., Indian J. Chem.,1975, 13, 1139.
- Khattab, S.A. and Hosny, M., Indian J. Chem., 1980, 19B, 1038.
- Cuiper, A.D. and Brzostowska, M., J. Org. Chem., 1999, 64, 2567.
- Griffart-Brunet, D. and Langois, N., Tetrahedron Lett., 1994, 35, 119.
- Klaver, W.J., Hiemstra, H. and Speckamp, W.N., J. Amer. Chem.Soc., 1989, 11, 2588.
- Husain, A., Hasan, S.M., Lal, S. and Mumtaz Alam M., Indian J.Heterocycl. Chem., 2004, 16, 163.
- R. Cruickshank, J.P. Dugid, D.P. and Marmion, R.H.A. Swain, Eds., In: Medical Microbiology, Vol.II, Churchill, London, 1975.