- *Corresponding Author:
- S. M. Jain
Department of Pharmacology, L. M. College of Pharmacy, Gujarat University, Ahmedabad-380 009, India
E-mail: rx_mukesh@rediffmail.com
| Date of Submission | 04 April 2011 |
| Date of Revision | 08 November 2011 |
| Date of Acceptance | 22 November 2011 |
| Indian J Pharm Sci, 2011, 73 (6): 682-687 |
Abstract
Triphala is categorized as rejuvenator and traditionally been used in various gastric disorders including intestinal inflammation. The aim of present study was to examine the comparative gastroprotective effects of Triphala formulations against experimental gastric ulcer in rats to substantiate its traditional claim. Gastric ulcer was induced by water immersion plus stress-induced ulcers in rats. The drug effects were assessed by studying macroscopic gross injury and stomach tissue biochemical parameters. Triphala unequal formulation and Chinnodbhavadi kwath showed significant antiulcer activity and this is evident from reduction of ulcer index, lipid peroxidation and hydroxyl radical levels and concomitantly raised levels of catalase and superoxide dismutase. Though similar kind of activity was observed in Triphala equal formulation the magnitude was much less. Further, Chinnodbhavadi kwath significantly increased the glutathione and ATPase level but Triphala equal formulation significantly increased glutathione level only. Based on the data generated, it is suggested that among the three formulations studied, Chinnodbhavadi kwath and Triphala unequal formulations provides significant protection in gastric ulcer as compared to Triphala equal formulation.
Keywords
Chinnodbhavadi kwath, gastroprotection, rejuvenator, Triphala
Peptic ulcer is the most common gastrointestinal disorder in clinical practice. There is evidence concerning the participation of reactive oxygen species in the etiology and pathophysiology of human diseases, such as neurodegenerative disorders, inflammation, viral infections, autoimmune pathologies and digestive system disorders such as gastrointestinal inflammation and gastric ulcer [1]. Study have shown that alteration in the antioxidant status following stress-induced ulcer indicate free radicals are directly implicated in the mechanism of stressinduced ulceration in rats [2]. Considering the several side effects (arrhythmias, impotence, gynaecomastia and hematopoietic changes) of modern medicine, indigenous drugs possessing fewer side effects should be looked for as a better alternative for the treatment of peptic ulcer [3].
The edifice of the drug science of Ayurveda stands on a strong foundation of the basic fundamentals of pancha-mahabhutas and tridosa. The three doshas, namely vata, pitta and kapha are biological representatives for physiological functions in the state of homeostasis and for pathological disorders in the state of imbalance [4]. The vitiation of pitta dosha lead to impairment of agni resulted in to amlapitta (hyperacidity), grahani roga (malabsorption syndrome) and other gastrointestinal disorders. Triphala formulation is one of the renowned Ayurvedic formulation used alone or along with other ingredients in Ayurvedic therapeutics for the treatment of gastrointestinal problems. It is categorized as tridoshik rejuvenator and reported to be an antioxidant rich herbal formulation [5,6]. As per Ancient text, one of the Triphala formulations called as Chinnodbhavadi kwath (decoction) is used for chronic hyperacidity and gastric problems [7]. Pharmacological studies have shown that Triphala extract possess antioxidant activity and reduce the damage due to oxidative stress [8]. It has been reported to be cytotoxic to breast cancer cells and prostate cancer cells [9], radio protective [6] and displays antidiabetic and free radicals scavenging activities [10].
The present study was thus aimed to investigate the comparative gastroprotective effects of Triphala formulations in stress-induced gastric ulcer in rats to determine which of the two formulation- Triphala equal or Triphala unequal is better for the above property and to ascertain whether this property is retained when Triphala formulation used as an ingredient of Chinnodbhavadi kwath (decoction) to substantiate its traditional claim.
Fruits of Terminalia chebula Retz. (Combretaceae), Terminalia belerica (Gaertn) Roxb. (Combretaceae) and Emblica officinalis Gaertn. (Euphorbiaceae) were collected from forest of Dang and Valsad (Gujarat, India) in the month of December (2005). Stem of Tinospora cordifolia (Willd.) Miers. (Menispermaceae), stem bark of Azadirachta indica A. Juss. (Meliaceae) and leaves of Trichosanthes dioica Roxb. (Cucurbitaceae) were collected from forest of Barda hills, Jamnagar (Gujarat, India) in the month of September and October 2005. The plant materials were authenticated and voucher specimens of each submitted to phamacognosy laboratory of Institute of Postgraduate Teaching and Research, Gujarat Ayurved University, Jamnagar, India. Triphala unequal formulation was prepared by mixing one part of T. chebula, two parts of T. belerica and four parts of E. officinalis [5,11] and Triphala equal formulation was prepared by mixing these three ingredients in equal proportion (1:1:1) [12]. Chinnodbhavadi kwath (decoction) was prepared by mixing equal proportion of T. chebula, T. belerica, E. officinalis, T. cordifolia, A. indica and T. dioica. Coarse powder (48 g) of mixture and 768 g water was added; boiled on low to medium heat till the liquid portion was reduced to 1/8th of the original volume (96 g) and filtered [11]. All chemicals used in the study and for biochemical assay were of analytical grade.
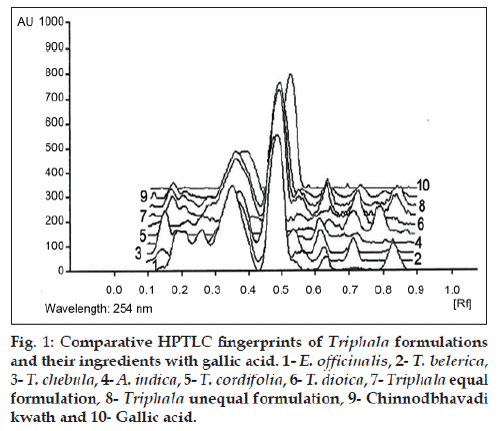
Triphala formulations and their ingredients were standardized using gallic acid as a marker compound by HPTLC finger print. The plate was developed in toluene:ethyl acetate:formic acid (5:5:1) solvent system. Gallic acid was observed at 0.52 Rf value, when scanned at 254 nm. E. officinalis, T. belerica, T. chebula and all three Triphala formulations shows almost the same Rf values as observed for gallic acid (fig. 1). The concentration of trace heavy metals such as lead, cadmium, arsenic and mercury present in formulations were analyzed by Atomic Absorption Spectrophotometer. The data obtained indicated that trace metals do not seem to be present in significant quantities in Triphala equal, Triphala unequal formulations and Chinnodbhavadi kwath.
Figure 1: Comparative HPTLC fingerprints of Triphala formulations and their ingredients with gallic acid. 1- E. officinalis, 2- T. belerica, 3- T. chebula, 4- A. indica, 5- T. cordifolia, 6- T. dioica, 7- Triphala equal formulation, 8- Triphala unequal formulation, 9- Chinnodbhavadi kwath and 10- Gallic acid.
Adult Wistar rats (180 to 220 g) of either sex were used in the experiment. The animals were maintained under standard conditions of temperature, humidity and exposed to 12 h light and dark cycles. All animals were exposed to the same environmental conditions and were maintained on standard diet and water ad libitum. The experimental protocol was approved by the Institutional Animal Ethical Committee as per guidelines of Committee for the Purpose of Control and Supervision on Experiments on Animals, India.
Acute oral toxicity of Triphala formulations were carried out in female rats as per the 423 guideline of OECD (Organization of Economic Co-operation and Development) [13]. The formulations were tested at limit dose of 2000 mg/kg body weight of female rats. The result showed that Triphala formulations did not produce any changes in observed parameters and there was no mortality at the dose level tested. Hence, animal dose of Triphala formulations were fixed on the basis of human therapeutic dose mentioned in literatures [11,12].
Rats were randomized in to six groups, each consisting of six animals. Group (I) normal control group and Group (II) stress control group, received vehicle as an aqueous suspension of 1% carboxymethyl cellulose (CMC) in the dose of 10 ml/kg body weight. Rats of group (III) and (IV) received Triphala equal and Triphala unequal formulations, respectively in a dose of 540 mg/ kg. Triphala formulations were suspended in 1% CMC and administered orally once daily for seven consecutive days to respective groups. Rats of group (V) were treated with Chinnodbhavadi kwath in a dose of 8.7 ml/kg for seven days. Group (VI) was treated with omeprazole 48, 24 and 1 h prior to induction of ulcers in a dose of 20 mg/kg and used as a positive control group.
Water immersion stress-induced gastric ulcer was induced by following the method described earlier [14]. On seventh day, the individually over night fasted rats were exposed to the water immersion stress inside specially arranged containers, which were made up of plexi-glass with holed lids. The rats were sacrificed at the end of 14 h period after water immersion stress. Abdominal cavity was opened carefully and the stomach was excised for assessment of ulcer index and tissue biochemical studies.
The stomach was excised, cleaned and opened along its greater curvature and the inner surface gently washed with cold saline solution and examined for ulceration with a magnifying lens. Severity and total number of ulcers in each rat were recorded for calculating ulcer index [15]. The scoring was done as, 0- no visible ulcer, 1- maximum diameter of 1 mm, 2- maximum diameter of 1-2 mm, 3- maximum diameter of 2-3 mm, 4- maximum diameter of 3-4 mm, 5- maximum diameter of 4-5 mm, 10- An ulcer over 5 mm in diameter, 25- A perforated ulcer.
The stomach was subsequently divided in to longitudinal pieces and stored immediately at -20º for estimation of tissue biochemical parameters. Protein content was quantitated using bovine serum albumin as a standard and was expressed as mg/g wet tissue [16]. Lipid peroxidation (LPO) was measured as thiobarbituric acid reactive substances (TBARS) formation [17]. TBARS concentrations were calculated by the use of malondialdehyde (MDA) as a standard and results were expressed as nmol MDA/g tissue. Hydroxyl radical ion react with thiobarbituric acid resulted in to pink colour which was estimated and the results were expressed as Unit/mg protein [18].
Superoxide dismutase (SOD) activity was determined by the nitro blue tetrazolium reduction method [19]. One unit of enzyme activity that inhibit rate of reaction by 50% in one minute under the defined assay conditions and the results have been expressed as Unit/mg protein. Catalase in stomach tissue was measured and content was expressed as μmoles H2O2 consumed/mg protein/min [20]. Glutathione present was estimated by the method of Grunert and Phillips [21] and the results were expressed as nmol/g wet tissue.
The estimation of adenosine triphosphatases includes Na+_K+-ATPase [22], Ca2+-ATPase [23] and Mg2+-ATPase [24]. The concentrations of ATPase were expressed as μmoles of phosphorus liberated /mg protein/min at 37º. Deoxyribonucleic acid and ribonucleic acid was estimated in stomach mucosa, homogenized in trichloro acetic acid and extracted with alcohol diethyl ether reagent as per the method described previously [25].
The data are expressed as mean±standard error of mean for six rats per experimental group. One way analysis of variance (ANOVA) was used to compare the mean values of quantitative variables among the groups followed by Dunnet’s multiple t-test for unpaired data to determine significant difference between groups at P<0.05.
Triphala is considered as one of the most important formulation in Ayurvedic therapeutics for its multiple organ protective effects including gastroprotection. Various physical and psychological stresses cause gastric ulceration in humans and experimental animals. Oxidative damage is considered to be common factor in the pathogenesis of ulcers by different experimental and clinical models. The increases in free radicals generation during stress are held responsible for induction of ulcers [26]. In the present study, rats subjected to water immersion stress showed significant increased in ulcer index as shown in (Table 1). Rats pretreated with Triphala unequal formulation (P<0.05) and Chinnodbhavadi kwath (P<0.05) showed significant reduction in ulcer index when compared with the stress control group. Triphala equal formulation exhibited a moderate non-significant decrease in ulcer index.
It is observed that stress procedure significantly elevated the lipid peroxidation and hydroxyl radicals (P<0.05) and concomitantly reduced the activity of SOD, catalase and total glutathione (P<0.05) content in gastric mucosa of rats which is indicative of oxidative stress (Table 1). Cell membrane lipid is very susceptible to hydroxyl radical attack and initiates the formation of LPO. Triphala unequal formulation and Chinnodbhavadi kwath treatment improve stomach oxidative balance in rats because they were able to reduce the level of hydroxyl radical and malondialdehyde, a good indicator of lipid peroxidation [17]. Triphala unequal formulation and Chinnodbhavadi kwath significantly reversed the stress-induced free radical generation. This may be due to restoration of free radicals scavenging enzymes viz; SOD, catalase and glutathione in gastric mucosa Table 1, effectively counter acting the free radicals generated during the stress condition. Earlier study also revealed that biological activities of Triphala due to its ability to scavenge free radicals [7]. The presence of the in vitro antioxidant activity of Triphala by scavenging oxygen radicals together with the inhibition of lipid peroxidation due to presence of phenolic compound particularly gallic acid [9]. Further, the flavanoid isolated from E. officinalis is reported to lower the LPO level in rats [27].
| Groups | Ulcer index | LPO (n mol MDA/g tissue |
Total protein (mg/g tissue) |
Hydroxyl Radical (U/mg protein) |
SOD (U/mg protein) | Catalase (µmoles H2O2 consumed/mg protein/min) |
Total glutathione (n moles/g tissue) |
|---|---|---|---|---|---|---|---|
| Normal control | 00 | 5.04 ± 0.57 | 92.66 ± 4.12 | 4.52 ± 0.49 | 1.26 ± 0.04 | 7.80 ± 0.31 | 1418.36 ± 48.17 |
| Stress control | 17.66 ± 1.82a | 8.38 ± 0.53a | 86.90 ± 4.83 | 9.36 ± 0.58a | 1.06 ± 0.05a | 6.39 ± 0.26a | 0618.79 ± 58.42a |
| Triphala equal | 12.66 ± 1.64 | 7.36 ± 0.29 | 89.77 ± 3.56 | 7.84 ± 0.24 | 1.07 ± 0.05 | 6.97 ± 0.21 | 0813.47 ± 47.87* |
| Triphala unequal | 11.50 ± 1.33* | 6.68 ± 0.25 * | 85.75 ± 3.82 | 7.59 ± 0.32* | 1.24 ± 0.04* | 7.83 ± 0.31* | 0681.37 ± 45.27 |
| Chinnod. kwath | 9.33 ± 2.80* | 6.68 ± 0.39 * | 82.87 ± 7.87 | 7.37 ± 0.31* | 1.24 ± 0.06* | 7.91 ± 0.55* | 0889.93 ± 55.61* |
| Omeprazole | 4.33 ± 1.11* | 6.13 ± 0.55 * | 87.47 ± 4.93 | 5.53 ± 0.53* | 1.19 ± 0.03 | 7.28 ± 0.44 | 1035.94 ± 48.66* |
Table 1: Effect of triphala formulations on ulcer index and gastric mucosal biochemical Parameters in stress-induced ulcer in rats
In rats subjected to stress, there was significant decrease in the level of membrane bound Na+-ATPase, Ca2+-ATPase and Mg2+-ATPase (P<0.05)in comparison to normal control group (Table 2). Chinnodbhavadi kwath significantly reversed decreased level of membrane bound ATPase near to normal control group in stress condition. The effect might be due to decrease in LPO and restoration of the level of GSH significantly and thereby protecting the protein thiol and sulfahydryl groups which are essential for structural integrity and function of ATPase. It is pertinent here to refer to a previous report which suggested that Emblica officinalis extract [28] and Azadirachta indica [29] significantly prevented depletion of nonprotein sulfahydryl groups, that might have contributed to the protective effect of Chinnodbhavadi kwath.
| Groups | Na-ATPase (μmoles of Pi liberated /mg protein/min) | Ca-ATPase (μmoles of Pi liberated /mg protein/min) | Mg-ATPase (μmoles of Pi liberated /mg protein/min) | DNA (mg/g tissue) | RNA (mg/g tissue) |
|---|---|---|---|---|---|
| Normal control | 3.53 ± 0.19 | 2.62 ± 0.14 | 2.49 ± 0.16 | 641.23 ± 39.21 | 1199.24 ± 82.70 |
| Stress control | 2.16 ± 0.08a | 1.28 ± 0.15a | 1.35 ± 0.21a | 228.26 ± 49.81a | 858.08 ± 72.37a |
| Triphala equal | 2.38 ± 0.23 | 0.98 ± 0.10 | 1.09 ± 0.07 | 293.47 ± 60.12 | 847.74 ± 65.38 |
| Triphala unequal | 2.67 ± 0.18 | 1.84 ± 0.15 | 1.60 ± 0.13 | 369.56 ± 57.51 | 951.12 ± 52.31 |
| Chinnod. kwath | 3.81 ± 0.53* | 2.34 ± 0.23* | 2.01 ± 0.16* | 369.56 ± 43.48 | 909.77 ± 54.70 |
| Omeprazole | 3.38 ± 0.47* | 2.14 ± 0.12* | 1.94 ± 0.06* | 358.69 ± 53.25 | 951.12 ± 63.39 |
Table 2: Effect of triphala formulations on gastric mucosal biochemical parameters in Stress-induced ulcer in rats
Increase or decrease in life span of mucosal cells can be expressed as amount of DNA and RNA in the gastric wall mucosa. The increased DNA and RNA content of gastric wall mucosa to some extent in Triphala unequal formulation and Chinnodbhavadi kwath treated groups indicate decreased cell shedding and increased life span of cells [30]. A recent study suggested that phenolic compound, ascorbic acid and flavanoids in Triphala formulations are responsible for the protection of DNA [6].
Previous report suggested that E. officinalis, T. cordifolia and A. indica produced significant antiulcer activity [31]. E. officinalis produced antiulcer activity due to presence of saponin and tannin [28]. The major antiulcer compound isolated from the aqueous extract of A. indica characterized to be phenolic glycoside in nature [32]. Triphala formulation is rich source of tannins, which is known to affect the integrity of mucus membrane. Tannins with their protein precipitating and vasoconstriction effects could be advantageous in preventing ulcer development [28]. Further, Triphala formulations contains Terminalia belerica and Emblica officinalis which are well established major antioxidants and their free radical scavenging effect is due to the presence of ellagic acid and gallic acid compared to Terminalia chebula [10].
From the present study, it is concluded that Triphala formulations possess significant anti-ulcer activity. This activity depends mainly on inhibition of free radical generation due to restoration of free radicals scavenging enzymes, enhancing the stability of gastric mucosal barrier and gastric cytoprotection against stress-induced gastric ulceration in rats. Among the three formulations studied Chinnodbhavadi kwath and Triphala unequal formulation provides significant protection against gastric ulcer as compared to Triphala equal formulation. The reason for the observed efficacy of this preparation is the increased quantity of two of its ingredients namely Terminalia belerica and Emblica officinalis in Triphala unequal formulation. Of the two, Emblica officinalis is well-known for its cytoprotective effect. Chinnodbhavadi kwath produced antiulcer effect due to the presence of Triphala formulation along with T. cordifolia and A. indica.
Acknowledgments
The authors wish to thank Dr. Mukesh C. Gohel, Principal, L. M. College of Pharmacy, Prof. M. S. Baghel, Director, Institute of PG Teaching and Research in Ayurveda and Dr. G. S. Lavekar, Director, CCRAS for their constant support.
References
- Repetto MG, Llesuy SF. Antioxidant properties of natural compounds used in popular medicine for gastric ulcers. Braz J Med Biol Res 2002;35:523-34.
- Sood S, Muthuraman A, Gill NS, Bali M, Sharma PD. Effect of Citrus karna peel extract on stress-induced peptic ulcer in rat. J Biol Sci 2010;10:231-6.
- Bafna PA, Balaraman R. Antiulcer and antioxidant activity of Pepticare, a herbomineral formulation. Phytomedicine 2005;12:264-70.
- Karnick CR. Pharmacology of Ayurvedic medicinal plants. Delhi: Sri Satguru Publications; 1996. p. 1-3.
- Vaidya BG. Nighantu Adarsa. 2nd ed. Varanasi: Chowkhamba Bharati Academy; 1998.
- Jagetia GC, Baliga MS, Malagi KJ, Sethukumar Kamath M. The evaluation of the radioprotective effect of Triphala (an ayurvedic rejuvenating drug) in the mice exposed to γ-radiation. Phytomedicine 2002;9:99-108.
- Tripathi I. Amlapitta Chikitsa. In: Dwivedi R, Deo S, editors. Chakradatta of Shri Chakrapanidatta. 3rd ed. Varanasi: Chowkhamba Sanskrit Sansthan; 1997. p. 45-54.
- Naik GH, Priyadarsini KI, Bhagirathi RG, Mishra B, Mishra KP, Banavalikar MM, et al. In vitro antioxidant studies and free radical reactions of Triphala, an Ayurvedic formulation and its constituents. Phytother Res 2005;19:582-6.
- Kaur S, Michael H, Arora S, Harkonen PL, Kumar S. The in vitro cytotoxic and apoptotic activity of Triphala- an Indian herbal drug. J Ethnopharmacol 2005;97:15-20.
- Sabu MC, Kuttan R. Antidiabetic activity of medicinal plants and its relationship with their antioxidant property. J Ethnopharmacol 2002;81:155-60.
- Tripathi B. Kwathadi kalpana and Churna kalpana. In: Sarngadhara Sanhita of Sri Sarngadharacharya. 4th ed. Varanasi: Chowkhamba Surbharati Prakashan; 2008. p. 133 and 174.
- Anonymous. The Ayurvedic Formulary of India, Part-1. 2nd ed. New Delhi: Department of ISM, Ministry of Health and Family Welfare, Government of India; 2003. p. 103-10.
- Ecobichon DJ. The basis of Toxicology testing. New York: CRC Press; 1997. p. 43-86.
- Parmar NS, Jagruti KD. A review of the current methodology for the gastric and duodenal antiulcer agents. Indian J Pharmacol 1993;25: 120-35.
- Bafna PA, Balaraman R. Antiulcer and antioxidant activity of Normacid, a herbomineral formulation. Indian J Exp Biol 2004;42: 674-80.
- Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem 1951;193:265-75.
- Ohkawa H, Ohishi, N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 1979;95:351-8.
- Ohkawa H, Ohishi N, Yagi K. Reaction of linoleic acid hydroperoxide with thiobarbituric acid. J Lipid Res 1978;19:1053-7.
- McCord JM, Fridovich I. Superoxide dismutase: An enzymatic function of erythrocuprein. (hemocuprein). J Biol Chem 1969;244:6049-55.
- Sinha AK. Colorimetric assay of catalase. Anal Biochem 1972;47:389-94.
- Grunert RR, Phillips PH. A modification of nitroprusside method of analysis for glutathione. Arch Biochem 1951;30:217-25.
- Bonting SL. Sodium-potassium activated adenosine triphosphatase and cation transport. In: Bittar EE editor. Membranes and ion transport. London: Wiley-Inter Science; 1970. p. 257-63.
- Hjerten S, Pan H. Purification and characterization of two forms of a low-affinity Ca2+-ATPase from erythrocyte membranes. Biochim Biophys Acta 1983;728:281-8.
- Ohnishi T, Suzuki T, Suzuki Y, Ozawa K. A comparative study of plasma membrane Mg2+-ATPase activities in normal, regenerating and malignant cells. Biochim Biophys Acta 1982;684:67-74.
- Chinoy NJ. Essential techniques in reproductive physiology and endocrinology. Ahmedabad: Anand Book Depot; 1993. p. 59-61.
- Sairam K, Rao ChV, Babu MD, Kumar KV, Agrawal VK, K Goel RK. Antiulcerogenic effect of methanolic extract of Emblica officinalis: an experimental study. J Ethnopharmacol 2002;82:1-9.
- Anila L, Vijayalakshmi NR. Antioxidant action of flavonoids from Mangifera indica and Emblica officinalis in hypercholesterolemic rats. Food Chem 2003;83:569-74.
- Al-Rehaily AJ, Al-Howiriny TA, Al-Sohaibani MO, Rafatullah S. Gastroprotective effects of ‘Amla’ Emblica officinalis on in vivo test models in rats. Phytomedicine 2002;9:515-22.
- Bandyopadhyaya U, Biswas K, Chatterjee R, Bandyopadhyay D, Chattopadhyay I, Ganguly CK, et al. Gastroprotective effect of Neem (Azadirachta indica) bark extract: Possible involvement of H+- K+-ATPase inhibition and scavenging of hydroxyl radical. Life Sci 2002;71:2845-65.
- Prabha T, Dora Babu M, Priyambada S, Agrawal VK, Goel RK. Evaluation of Pongamia pinnata root extract on gastric ulcers and mucosal offensive and defensive factors in rats. Indian J Exp Biol 2003;41:304-10.
- Dharmani P, Palit G. Exploring Indian medicinal plants for antiulcer activity. Indian J Pharmacol 2006;38:95-9.
- Bandyopadhyay U, Chatterjee R, Bandyopadhyay RK. Process for the isolation of an active principle from Azadirachta indica useful for controlling gastric hyperacidity and gastric ulceration. 1998. US patent 5730986.