- Corresponding Author:
- M. Ning
Research Center of Biomaterial and Novel Drug Delivery Systems, National Research Institute for Family Planning, 12 Dahuisi Rd, Haidian District, Beijing−100 081, China
E-mail: mayning999@126.com
| Date of Submission | 17 March 2014 |
| Date of Revision | 19 January 2015 |
| Date of Acceptance | 04 June 2015 |
| Indian J Pharm Sci 2015;77(3):335-342 |
Abstract
Preparation and in vitro/in vivo evaluation of mifepristone intravaginal ring formulations were investigated. In the present study, it is reported that a mifepristone intravaginal ring of reservoir design comprising of a mifepristone silicone elastomer core enclosed in a silicone layer. During the preparation of intravaginal ring solid dispersion method was employed which improved the release rate of drug from the intravaginal ring. In vitro release studies performed under sink conditions and the released drug amounts were estimated using UV spectrometry at 310 nm. In addition, the in vivo release profile of in-house devices was evaluated in female New Zealand white rabbits. The rabbit plasma samples were processed and analyzed using a validated HPLC-MS method. Norgestrel was used as internal standard, and plasma samples contained mifepristone and internal standard were deproteinized, and then subjected to HPLC-MS analysis under condition of electrospray ionization in the selected ion monitoring mode. The drug release from intravaginal ring made in house was constant for 21 days in rabbits, which suggested the mifepristone intravaginal ring release system would be useful in clinical practice in the future. The result indicated the in vitro/in vivo correlation is perfect, which explained in vitro release analysis method developed was feasible.
Keywords
Mifepristone, intravaginal ring, silicone elastomer, in vitro/in vivo correlation.
Mifepristone (fig. 1) is a synthetic 19-norsteroid that has potent antiglucocorticoid and antigestagenic properties [1]. It acts by blocking the action of progesterone at the receptor level, and thus has multiple potential antifertility actions. In the early clinical practice, mifepristone has been proved useful in medical abortion in the first and second trimester [2]. Recently, mifepristone also could be used in the clinic for the treatment of endometriosis [3], uterine myomas [4-6] and for contraceptive purpose [7]. Low doses of mifepristone (2-5 mg/day) orally had been reported for contraception, uterine myomas and treatment of endometriosis and no evidence of endometrial hyperplasia or cellular atypia [3]. Mifepristone is an oral tablet, which has the characteristics of safety, effectiveness and good tolerance, meanwhile its side effects are extremely low. However, the inconvenience of mifepristone daily intake could be resulting in the low compliance during treatment of the above-mentioned chronic diseases. The fluctuation of plasma level would lead to some side-effects. Therefore, our research group tried to develop a nonoral sustained mifepristone delivery, which could offer better compliance during the long-time treatment period. Among advanced sustained drug delivery systems, intravaginal ring (IVR) has been attracting more and more scientists and researchers recently. The intravaginal ring is self-administered, requires less frequent dosing. A successful case, NuvaRing (Organon, Oss, The Netherlands), the contraceptive vaginal ring has been approved for use in the United States. Most women note a beneficial effect on less bleeding profiles and are satisfied with NuvaRing [8]. Serious adverse events are rare. In Chile and Peru, progesterone-only vaginal contraceptive rings are available for nursing women. Recently more studies are ongoing new formulations of intravaginal rings in the world [8-15].
In present study, preparation and in vitro/in vivo evaluation of mifepristone IVR formulations were investigated. A method for the preparation of mifepristone intravaginal ring was reported. A mifepristone intravaginal ring of reservoir design, comprising a mifepristone silicone elastomer core enclosed in another silicone layer, manufactured by molding at 130° for several minutes. in vitro release studies were performed under sink condition and the sustained drug release was determined by sensitive and rapid HPLC method. The in vivo profile of the device was investigated in female New Zealand White rabbits and detected by a rapid method HPLC-MS developed in house.
Materials and Methods
Mifepristone (99.8%) powder and mifepristone standard (99.8%) were provided by Zizhu Phapharmaceutical Co. Ltd., Beijing, China. The internal standard was norgestrel (99.8%, National Institute for the Control of Pharmaceutical and Biological Products, Beijing, China). EDTA, hydrochloric acid, dehydrated alcohol and sodium dihydrogen phosphate were obtained from Sinopharm Chemical Reagent Beijing Co. Ltd., Beijing, China. Class VI elastomers (Dow Corning® C6-165, Part A and Part B) and Class VI Liquid Silicone Rubber Elastomers (Dow Corning® C6-550 LSR, Part A and Part B) were purchased from Dow Corning Corporation (USA). PVPK30 was obtained from Fengli Jingqiu Commerce and Trade Co. Ltd., Beijing, China. HPLC grade acetonitrile was purchased from Fisher Scientific, Fair Lawn, NJ, USA. Ultrapure water was obtained from a Milli-Q Plus water purification system, Millipore, Bedford, MA, USA. All other chemicals and solvents were analytical reagents and obtained from local commercial sources. Female New Zealand White rabbits were supplied by Vital River Laboratory Animal Technology Co. Ltd.
Preparation of mifepristone-loaded IVRs
Silicon elastomer C6-165 Part A and Part B were blended by opening mixing mills to produce the silicon elastomer mix. Then the required amount of mifepristone or mifepristone solid dispersion (SD) was added to produce active mixture of 3% (wdrug/welastomer), and the 3% blends were molded at 130° for 10 min to produce the active core. The active core was coated with 30% (g/l) solution of silicon slastomer C6-550 dichloromethane solution for 10 s, then removed and vulcanized for 20 min at 130° to control the initial burst and the release rate of mifepristone. The preparation process of mifepristone SD and optimal ratio selection of drug to polymer were described as following chapter. To investigate the key influence factor of mifepristone in vitro release profile in silicon elastomer ring, the following factors were described in the following chapter. The drug release was analyzed using a UV spectrophotometer at 310 nm.
Influence of coating solution and coating time
Based on the pilot study, the coating solution was formulated with 10, 18, 25, 30 and 50 (g/l) silicon elastomer C6-550 in dichloromethane, respectively. The inner rings containing SD (1:0.5, wdrug/wpvp) of mifepristone were prepared and dipped in the coating solution for several seconds, then removed and vulcanized for 20 min at 130°. The drug release profiles of the different coated intravaginal rings were compared. At the same time, the drug release profiles of the rings at different coating time (5, 10 and 15 s) were investigated. All rings loaded same quantity 3% (wdrug/welastomer).
Influence of drug loading and cross-sectional diameter
The required amount of SD (1:0.5, wdrug/wpvp) of mifepristone was added into the silicone elastomer mixture to produce active mixes of 0.5, 1.0 and 3.0% (wdrug/welastomer), respectively, and then IVRs containing 15, 35 and 90 mg of drug were prepared by the above described method. At the same time, IVRs containing SD (1:0.5, wdrug/wpvp) of mifepristone were prepared with different cross-sectional diameter (Φ1=3, Φ2=4 and Φ3 = 5 mm), each ring loaded 3% (wdrug/welastomer) mifepristone.
Preparation of solid dispersion
Solid dispersion (SD) of mifepristone was prepared by the solvent evaporation method with PVPK30 as carrier. In this system, the solid dispersions of 1:0.5, 1:1, 1:3 (wdrug/wpvp) w/w of drug to carrier were prepared. The mixture of drug and carrier was dissolved in ethanol. The solvent was evaporated under reduced pressure using a vacuum dryer at 70° until complete evaporation. To ensure the residual solvent was completely removed, the solid mass was further dried in a vacuum oven at room temperature for 24 to 48 h or until the weight constant was obtained. The resulting solid was pulverized and sieved, and then stored in desiccator at room temperature until further evaluation and processed.
The solubility of mifepristone/PVPK30 systems were determined by adding excess amounts of mifepristone in a beaker containing 100 ml of phosphate buffer solution (pH 4.0) and incubated with continuous stirring at 37±0.5° for 24 h to achieve equilibrium. The dissolution of pure mifepristone, physical mixture (PM) and SD of mifepristone were performed in 900 ml of pH4.0 phosphate buffer solution at 37º by the USP-II paddle apparatus at 50 rpm. The resultant solutions were analyzed using a UV spectrophotometer at 310 nm. At the same time, physical mixture (PM) of mifepristone and carrier were prepared in order to study the release profiles.
Mechanical testing of intravaginal rings
The hardness of the intravaginal rings was tested with LD-A Digital Shore Durometer (Wenzhou Sundoo Instruments Company, China). The ring containing SD (1:0.5, wdrug/wpvp) of mifepristone was placed vertically in a holder subjected to an aluminium compression probe and tested different parts of the intravaginal ring three times.
in vitro release testing
The phosphate buffer solution (pH 4.0) was adopted as release media to provide sink conditions for mifepristone release due to the human vaginal pH fluctuating between 3.8 to 4.5 levels, in which the solubility of mifepristone were determined to be 129.8±1.2 mg/l. The rings were placed into screw-top glass bottles containing either 200 ml release medium. The bottles were placed in an orbital shaking incubator (37°, 60 rpm), and the release medium was sampled (5 ml) regularly during the 21- day study period. The release medium was completely replaced with fresh warmed medium every 24 h. Samples were stored at 4° until detection.
The drug release was evaluated using a UV/Vis spectrophotometric method (PerkinElmer′s Lambda 35 UV/Vis systems, USA). Absorbances of the standard and sample solutions were measured at the wavelength of 310 nm, daily release and cumulative release versus time profiles were plotted respectively during the 21-day study period.
in vivo study in rabbits
Six female New Zealand White rabbits (SPF, 2.50±0.10 kg) were housed in the laboratory animal center of National Research Institute for Family Planning (IACUC Issue No. NRIFP13031601) in accordance with recommendations in the Guide for the Care and Use of Laboratory Animals of the NIH. At the onset of treatment, the rabbits were approximately 5 to 8 months old and their mean body weight was 2.5±0.10 kg. The vaginal opening was infused with 2% liquid lidocaine, a midline laparotomy incision was made, and the vagina was isolated from surrounding soft tissues. The 1/2 segment of vaginal ring containing SD (1:0.5, wdrug/wpvp) of mifepristone was inserted through vaginal opening. Insertion was performed as a clean procedure by trained technical staff. The implant was anchored with a 5-0 Prolene suture to the vagina from the outer ventral wall. The laparotomy incision was closed with absorbable suture and skin staples. The IVRs were inserted on day 0 into the vaginal vault and retained for a period of 21- day. Blood samples (approximately 0.5 ml each) were collected from marginal ear vein every day during 21-day. Blood samples were treated with EDTA before plasma was separated by centrifugation (3000 rpm, 10 min) and stored (-80°) prior to analysis.
The internal standard norgestrel was mixed with 0.1 ml plasma sample. Then the plasma samples were treated with 100 µl acetonitrile. After the plasma samples were mixed for 1 min and centrifuged for 10 min at 16 000 rpm, the upper solution transferred into the inner glass sample tube and 10 µl was injected into the LC/ MS. Peak area ratios of mifepristone/IS were calculated and calibration curves were developed. The equations of the calibration curves were used to interpolate the concentrations of mifepristone in the samples using their peak area ratios. The rabbit plasma samples were processed and analyzed using accuracy, precision, sensitivity, specificity and stability validated method.
HPLC-MS
Norgestrel was used as internal standard (IS), and plasma samples contained mifepristone and IS were deproteinized, and then subjected to LC-MS analysis under condition of electrospray ionization (ESI) in the selected ion monitoring (SIM) mode. The separation was performed on a Waters Symmetry C18 (2.1×150 mm, 5µm) column with isocratic elution at a flow rate of 0.3 ml/min. The mobile phase consisted of acetonitrile and water (82:18, v/v) containing 0.5% formic acid. Target ions were monitored at [M+H]+ m/z 430.3 and 313.5 in positive ESI mode for mifepristone and IS, respectively. The calibration curve of mifepristone has acceptable linearity in the range of 5 to 500 ng/ml. The lowest limit of quantification (LLOQ) was 5 ng/ml.
Results and Discussion
The drug loaded ring was prepared by moulding process with the following characteristics: 4.0 mm ring cross-sectional diameter, 50.0 mm overall ring diameter and 3.0±0.2 g mean weight of rings. The preparation process had a good reproducibility. The solid dispersion method and coating solution were two key factors influencing the drug release profile. The drug loaded rings daily released mifepristone with a little burst effect over the 21 day. The coated rings should be one of core-type rings that comprise drug-loaded core(s) encapsulated by a non-medicated polymer membrane. The mechanical properties of an IVR must ensure optimal intravaginal compatibility and user acceptability. If the mechanical strength is too weak, the ring could either be expulsed from the vagina or be prone to rupture. If the mechanical strength is too strong, the inflexibility of the device could cause irritation/ulceration of the intravaginal tissue. The hardness of the intravaginal ring containing SD (1:0.5, wdrug/wpvp) of mifepristone was in the range of 60 to 70 HA, which was suitable for human.
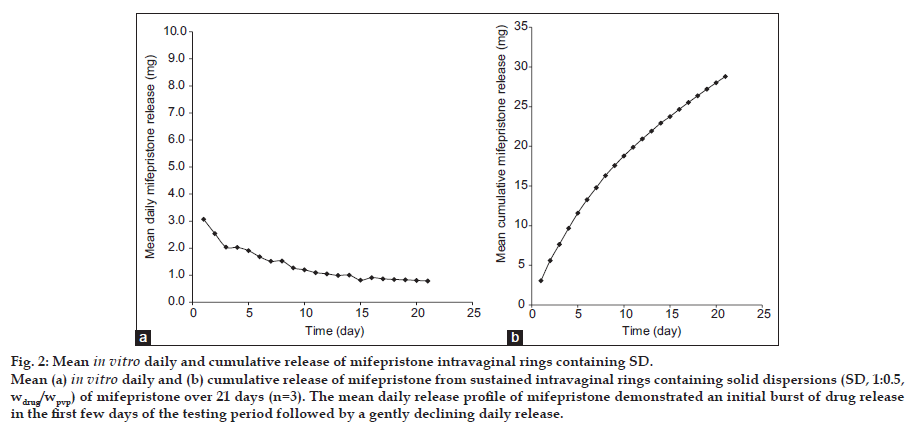
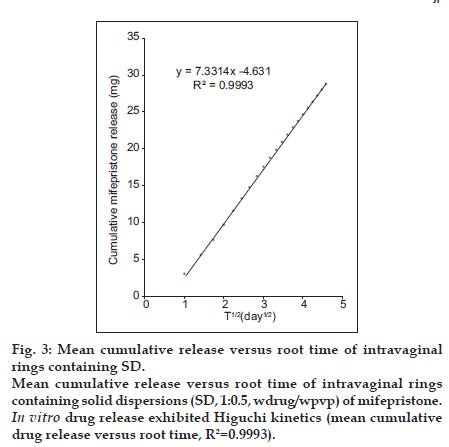
Profiles of daily and cumulative in vitro drug release from IVR devices containing SD (1:0.5, wdrug/wpvp) of mifepristone, shown in fig. 2, exhibit Higuchi kinetics (mean cumulative drug release versus root time; R2=0.9993, fig. 3), i.e. a matrix diffusion controlled mechanism, and sustained release of mifepristone over the 21-day. The daily release of mifepristone from silastic ring for the 21-day period ranges from 1.00 mg/day to 3.00 mg/day (fig. 2a), and the cumulative release reaches to 28.81 mg (30%) on the day 21 (fig. 2b). The average rate of the drug release was 1.37 mg/day over the study period, and the average rate was 1.22 mg/day from day 3 to 21, which might meet the clinic treatment requirements [16,17]. The mean daily release profile of mifepristone was a little burst of drug release with the initial days of the testing period followed by a gently declining daily release over the 21-day study period.
Fig. 2:Mean in vitro daily and cumulative release of mifepristone intravaginal rings containing SD. Mean (a) in vitro daily and (b) cumulative release of mifepristone from sustained intravaginal rings containing solid dispersions (SD, 1:0.5, wdrug/wpvp) of mifepristone over 21 days (n=3). The mean daily release profile of mifepristone demonstrated an initial burst of drug release in the first few days of the testing period followed by a gently declining daily release.
Fig. 3:Mean cumulative release versus root time of intravaginal rings containing SD. Mean cumulative release versus root time of intravaginal rings containing solid dispersions (SD, 1:0.5, wdrug/wpvp) of mifepristone. In vitro drug release exhibited Higuchi kinetics (mean cumulative drug release versus root time, R2=0.9993).
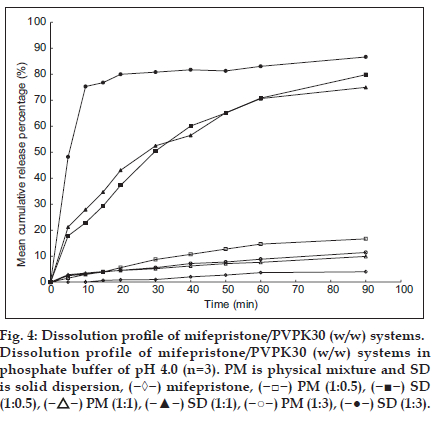
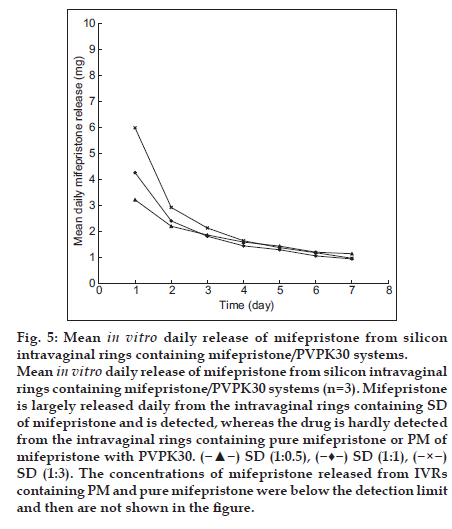
Mifepristone is insoluble in water. It was found that mifepristone could sparsely be released from the silastic intravaginal rings. The silicone elastomer solubility of mifepristone was detected to be 16.80 mg/g by the differential scanning calorimetry (DSC) method according to the reference method [18] and mifepristone could not be released from IVR devices, which is different from what was found in the patents [16,17]. The SD of mifepristone was therefore prepared in order to improve the solubility and dissolution of mifepristone in vitro, and it could be used in the preparation process of mifepristone-loaded IVR. The influence of PVPK30 upon the solubility of mifepristone is presented in Table 1. The increase in solubility was linear with respect to the weight fraction of the carrier, and the solubility of SD was 4-5 folds compared with pure drug. It can be observed that the release kinetics of SD of mifepristone is higher (60%-80%), when compared with pure mifepristone and physical mixture (PM) of mifepristone with polyvinylpyrrolidone (PVPK30, fig. 4). Then SD of mifepristone, PM of mifepristone with PVPK30 and pure mifepristone were respectively incorporated into silicone elastomer to prepare IVRs. It can be seen from fig. 5 that mifepristone is largely released daily from the intravaginal rings containing SD of mifepristone and is detected, whereas the drug is hardly detected from the intravaginal rings containing pure mifepristone or PM of mifepristone with PVPK30, presumably due to the fact that the solubility of mifepristone decreased relatively by solid dispersion method in silicon elastomer and the increase of solubility and dissolubility in vitro which were agreed with document reported [15]. The result also showed that the high ratio of PVPK30 could increase the initial burst effect of intravaginal ring, and the optimized ratio of mifepristone and PVPK30 was 1:0.5 with higher solubility and wide range of drug loading. SD method was the first application successfully in the preparation of mifepristone-loaded IVR, and this method might apply for other similar insoluble drugs.
| Composition | Mifepristone | SD1 (1:0.5) | SD2 (1:1) | SD3 (1:3) |
| Solubility (mg/l) | 28.1±0.2 | 129.8±1.2 | 137.2±0.9 | 143.5±0.6 |
| Solubility in phosphate buffer of pH 4.0, n=3 | ||||
Table 1: Solubitlity of mifepristone/PVPK30 systems in phosphate buffer
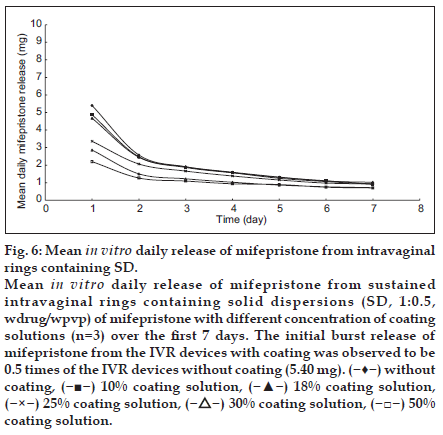
The mean daily release profiles of mifepristone from sustained IVRs with or without coating are shown in fig. 6. The initial burst release of mifepristone from the IVR devices with coating was observed to be 0.5 times of the IVR devices without coating (5.40 mg). The concentration of coating solution had effects on both the burst effect and the release rate. It could be concluded that the release rate and the amount of drug in initial burst release were decreased by increasing the concentration of coating solution. The results showed that 50 and 30% (g/l) coating solution could prevent significantly the initial burst release effect and provide a fairly stable and efficacious release of mifepristone from the ring. The optimal concentration of coating solution is 30% (g/l), which can reduce the burst release of ring and provide good appearance of ring. It is well known that a large amount of mifepristone trapped on the surface of the silicon elastomer during the manufacture process of the active core, and was released immediately to the release medium (burst effect). Surface coating is an effective and simple way to control the drug release of rings. In lower concentration (10 and 18%) of coating solution, the content of C6-550 liquid silicon rubber (LSR) was too low to form a continuous film on the surface of ring, which had no effect on the drug burst effect. When the concentration was greater than or equal to 25%, a continuous film was formed and encapsulated the active core, which reduced the initial burst of mifepristone. C6- 550 LSR has similar properties to that of C6-165 silicon elastomer, so they have similar permeation characteristics for mifepristone. Mifepristone on the surface of the active core had to be dissolved by dissolution medium, diffused through the coating layer, and finally partitioned into the dissolution medium, thereby increasing the diffusional pathway for drug release. At the same time, the dissolution of the active within the core of the ring was enhanced under the high temperature conditions associated with curing, and its subsequent diffusion into and deposition within the coating layer on cooling to ambient temperature. Based on the mechanisms above mentioned, the initial amount of drug released within the first 24 h was significantly decreased to 2.2 mg, followed a fairly stable release of mifepristone from rings.
Fig. 4:Dissolution profile of mifepristone/PVPK30 (w/w) systems. Dissolution profile of mifepristone/PVPK30 (w/w) systems in phosphate buffer of pH 4.0 (n=3). PM is physical mixture and SD is solid dispersion, ( ) mifepristone, (
) mifepristone, ( ) PM (1:0.5), (
) PM (1:0.5), ( ) SD (1:0.5), (
) SD (1:0.5), ( ) PM (1:1), (
) PM (1:1), ( ) SD (1:1), (
) SD (1:1), ( ) PM (1:3), (
) PM (1:3), ( ) SD (1:3).
) SD (1:3).
Fig. 5:Mean in vitro daily release of mifepristone from silicon intravaginal rings containing mifepristone/PVPK30 systems. Mean in vitro daily release of mifepristone from silicon intravaginal rings containing mifepristone/PVPK30 systems (n=3). Mifepristone is largely released daily from the intravaginal rings containing SD of mifepristone and is detected, whereas the drug is hardly detected from the intravaginal rings containing pure mifepristone or PM of mifepristone with PVPK30. ( ) SD (1:0.5), (
) SD (1:0.5), ( ) SD (1:1), (
) SD (1:1), ( ) SD (1:3). The concentrations of mifepristone released from IVRs containing PM and pure mifepristone were below the detection limit and then are not shown in the figure.
) SD (1:3). The concentrations of mifepristone released from IVRs containing PM and pure mifepristone were below the detection limit and then are not shown in the figure.
Fig. 6:Mean in vitro daily release of mifepristone from intravaginal rings containing SD. Mean in vitro daily release of mifepristone from sustained intravaginal rings containing solid dispersions (SD, 1:0.5, wdrug/wpvp) of mifepristone with different concentration of coating solutions (n=3) over the first 7 days. The initial burst release of mifepristone from the IVR devices with coating was observed to be 0.5 times of the IVR devices without coating (5.40 mg). ( ) without coating, (
) without coating, ( ) 10% coating solution, (
) 10% coating solution, ( ) 18% coating solution, (
) 18% coating solution, ( ) 25% coating solution, (
) 25% coating solution, ( ) 30% coating solution, (
) 30% coating solution, ( ) 50% coating solution.
) 50% coating solution.
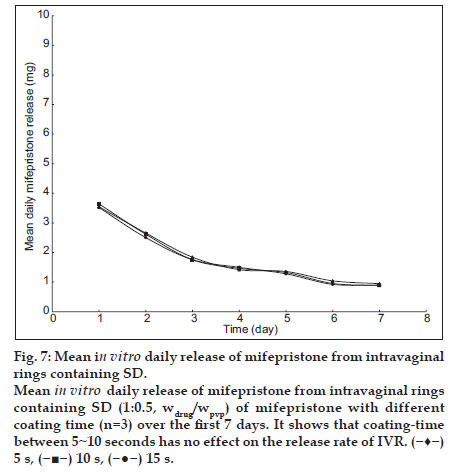
Fig. 7 depicts the daily release profiles of mifepristone from IVRs with three different coating times. The result shows that coating-time has no effect on the release rate of IVR. When the coating-time is more than 15 s, the rings are difficult to dry. The optimum time for coating is 5 s. The release profile was similar to the document reported, and the drug burst decreased after coating with liquid silicone elastomer [19].
Fig. 7:Mean in vitro daily release of mifepristone from intravaginal rings containing SD.
Mean in vitro daily release of mifepristone from intravaginal rings containing SD (1:0.5, wdrug/wpvp) of mifepristone with different coating time (n=3) over the first 7 days. It shows that coating-time between 5˜10 seconds has no effect on the release rate of IVR. ( ) 5 s, (
) 5 s, ( ) 10 s, (
) 10 s, ( ) 15 s.
) 15 s.
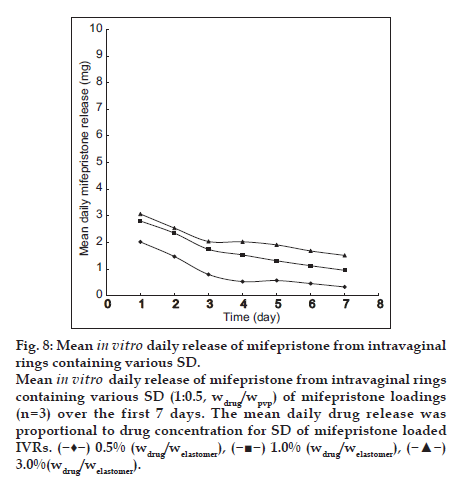
Diffusion-controlled release characteristic is that mean daily drug release was proportional to drug concentration [18,20]. The rings made in house displayed diffusion-controlled release characteristics. The mean daily drug release was proportional to drug concentration for SD of mifepristone loaded IVRs, as exemplified in fig. 8. Therefore, the release rate of drug could be modified by changing the initial drug loading for different clinical applications to need the duration of desired released rate of mifepristone.
Fig. 8:Mean in vitro daily release of mifepristone from intravaginal rings containing various SD.
Mean in vitro daily release of mifepristone from intravaginal rings containing various SD (1:0.5, wdrug/wpvp) of mifepristone loadings (n=3) over the first 7 days. The mean daily drug release was proportional to drug concentration for SD of mifepristone loaded IVRs. ( ) 0.5% (wdrug/welastomer), (
) 0.5% (wdrug/welastomer), ( ) 1.0% (wdrug/welastomer), (
) 1.0% (wdrug/welastomer), ( ) 3.0%(wdrug/welastomer).
) 3.0%(wdrug/welastomer).
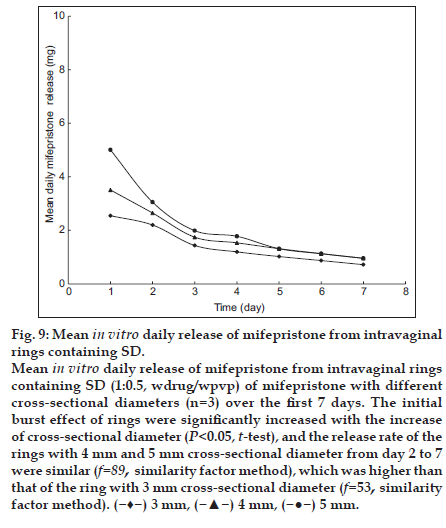
The initial burst effect of rings were significantly increased with the increase of cross-sectional diameter (P<0.05, t-test, fig. 9), and the release rate of the rings with 4 mm and 5 mm cross-sectional diameter from day 2 to 7 were similar (f=89, similarity factor method), which was higher than that of the ring with 3 mm cross-sectional diameter (f=53, similarity factor method). Because the surface area of ring was increased with increasing cross-sectional diameter, the release rate and the magnitude of burst were significantly increased [20]. Therefore the optimum cross-sectional diameter of ring was 4 mm, which had a low initial burst and high drug release rate.
Fig. 9:Mean in vitro daily release of mifepristone from intravaginal rings containing SD. Mean in vitro daily release of mifepristone from intravaginal rings containing SD (1:0.5, wdrug/wpvp) of mifepristone with different cross-sectional diameters (n=3) over the first 7 days. The initial burst effect of rings were significantly increased with the increase of cross-sectional diameter (P<0.05, t-test), and the release rate of the rings with 4 mm and 5 mm cross-sectional diameter from day 2 to 7 were similar (f=89, similarity factor method), which was higher than that of the ring with 3 mm cross-sectional diameter (f=53, similarity factor method). ( ) 3 mm, (
) 3 mm, ( ) 4 mm, (
) 4 mm, ( ) 5 mm.
) 5 mm.
Available in vivo analytical methods for mifepristone include radioimmunoassay (RIA) [21], radioreceptor assay (RRA) [22], HPLC [23,24], LC-MS/MS [25,26]. The disadvantage of RIA and RRA could not distinguish drug from metabolic materials, which might result in the deviation of analysis. The reported HPLC method had relative high detection limit which was not suitable for the sample of plasma due to the low drug concentration in blood. HPLC-MS/MS was relative high cost and the reported LC-MS/MS method for determination of mifepristone in human plasma [25,26] might not be applied to rabbit. Therefore, it was to be developed a simple, specific, rapid and sensitive HPLC-MS analytical method for the quantification of mifepristone in rabbit plasma in house. The specificity, precision, accuracy, matrix effect and stability of method had been validated and the results met the related requirements.
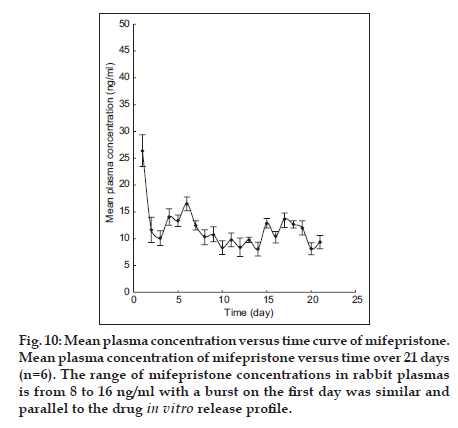
The plasma concentration−time profiles of IVR containing SD (1:0.5, wdrug/wpvp) of mifepristone are shown in fig. 10. That the range of mifepristone concentrations in rabbit plasmas is from 8 to 16 ng/ml was observed except a burst on the first day. The result of sustained-release mifepristone with a burst on the first day was similar and parallel to the drug in vitro release profile, which indicated the in vitro release determination method feasible. In addition, no serious adverse effects on the rabbits were observed during in vivo study period.
Fig. 10:Mean plasma concentration versus time curve of mifepristone. Mean plasma concentration of mifepristone versus time over 21 days (n=6). The range of mifepristone concentrations in rabbit plasmas is from 8 to 16 ng/ml with a burst on the first day was similar and parallel to the drug in vitro release profile.
The present investigation clearly demonstrated the advantages of the silastic intravaginal rings of mifepristone, including preparation process, high drug release rate and low drug concentration in rabbit plasma. The solid dispersion method was one of important factors improving the drug release rate of rings in vitro. Moreover, coating solution was found to significantly reduce the magnitude of the observed burst effect, and obtained a steady in vivo drug release rate. The IVR device showed preliminary safety and exhibited sustained release of mifepristone over 21− day with a little burst on the initial day in vitro, the plasma drug concentration of mifepristone-loaded IVR was keeping constant in rabbits which was consistent with the in vitro release profile. Therefore, IVR device containing mifepristone might have potential in the treatment of uterine myoma and uterine endometriosis, and even for the contraceptive purpose.
Financial support and sponsorship
Nil.
Conflict of interest
There are no conflicts of interest.
References
- Weems YS, Bridges PJ, Sasser RG, LeMaster BR, ChingL,Vincent DL, et al. Effect of mifepristone on pregnancy specific protein B (PSPB), progesterone, estradiol 17-â, prostaglandin F2a (PGF2a) and prostaglandin E (PGE) inovariectomized90-day pregnant ewes. Prostaglandins Other Lipid Mediat 2002;70:195-208.
- Fiala C, Gemzell-Danielsson K. Review of medical abortion using mifepristone in combination with a prostaglandin analogue. Contraception 2006;74:66-86.
- Chang YF, Sean M, Kevin F. Improved quality of life is partlyexplained by fewer symptoms after treatment of fibroids with mifepristone. Int J GynaecolObstet 2010;109:121-4.
- Carbonell JL, Acosta R, Heredia B, Pérez Y, Yero MC, Valle A. Evolution of uterine leiomyoma after treatment with mifepristone: A randomizedclinical trial. Progresos de Obstetricia y Ginecologia 2010;53:231-6.
- Carbonell JL, Acosta R, Heredia B, Pérez Y, Yero MC, Valle A. Mifepristone for treatment of uterine fibroids. Rev Cuba GynecologiaSaludReprod 2010;36:236-50.
- CarbonellEsteve JL, Riverón AM, Cano M, Ortiz AI, Valle A, Texidó CS, et al. Mifepristone 2.5 mg versus 5 mg daily in the treatment of leiomyoma before surgery. Int J Womens Health 2012;4:75-84.
- Pei KY, Xiao BL, Jing XP, Lu SQ, Wei L, Zhao H. Weekly contraception with mifepristone. Contraception 2007;75:40-4.
- Edwardson J, Jamshidi R. The contraceptive vaginal ring.SeminReprod Med 2010;28:133-9.
- Kerns J, Darney P. Vaginal ring contraception. Contraception 2011;83:107-15.
- Malcolm RK, Edwards KL, Kiser P, Romano J, Smith TJ. Advances in microbicide vaginal rings. Antiviral Res 2010;88:S30-9.
- Duan XY, Chen K, Wang JD,Ning MY.Review of Drug Loaded Vaginal Ring. Chin Pharm J 2012;47:841-6.
- Whaley KJ, Hanes J, Shattock R, Cone RA, Friend DR. Novelapproaches to vaginal delivery and safety of microbicides:Biopharmaceuticals, nanoparticles, and vaccines. Antiviral Res2010;88:S55-66.
- Friend DR, Doncel GF.Combining prevention of HIV-1, other sexually transmitted infections and unintended pregnancies: Development of dual-protection technologies. Antiviral Res 2010;88:S47-54.
- Han YA, Singh M, Saxena BB. Development of vaginal rings forsustained release of nonhormonal contraceptives and anti-HIV agents. Contraception 2007;76:132-8.
- Zhang Y, Li CX, Ning MY, Duan XY, Liu Y. Preparation and evaluation of intravaginal ring containing drospirenone. AdvPharmacolSci 2013;192408.
- Chen HL, Shao HH, Chen JX, Chen LK. Vaginal ring preparation and its application.2002, US2002/0161352 A1.
- Chen HL, Shao HH, Chen JX, Chen LK. Vaginal ring preparation and its application.2005, US2005/0197651 A1.
- Woolfson AD, Malcolm RK, Gallagher RJ. Design of a silicone reservoir intravaginal ring for the delivery of oxybutynin. J Control Release 2003;91:465-76.
- Chen HL, Shao HH, Chen JX, Chen LK. Vaginal ring preparation and its application.2002, CN1382500A.
- Malcolm K, Woolfson D, Russell J, Tallon P, Mcauley L, Craig D. Influence of silicone elastomer solubility and diffusivity on the in vitro release of drugs from intravaginal rings. J Control Release2003;90:217-25.
- Leminer R,Ranta S, Hertzen HV, Oehler J, Heikinheimo O. Pharmacokinetics of 10 mg of mifepristone. Contraception 2003;68:427-9.
- Földesi I, Falkay G, Kovács L. Determination of RU486 (Mifepristone) in blood by radioreceptorassay: A pharmacokinetic study. Contraception 1996;54:27-32.
- Guo ZY, Chu CD, Yin GX, He ML, Fu KQ, Wu JL. An HPLC method for the determination of ng mifepristone in human plasma.JChromatogr B AnalytTechnol Biomed Life Sci 2006;832:181-4.
- Guo ZY, Wang S, Wei DY, Zhai JX. Development of a highperformanceliquid chromatographic method for the determination of mifepristone in human plasma using norethisterone as an internal standard: Application to pharmacokinetic study. Contraception 2007;76:228-32.
- Tang C, Bi HC, Zhong GP, Chen X, Huang ZY, Huang M. Simultaneous determination of mifepristone and monodemethylmifepristonein human plasma by liquid chromatography−tandem mass spectrometry method using levonorgestrel as an internal standard: Application to a pharmacokinetic study. Biomed Chromatogr2009;23:71-80.
- Jiang B, Ruan ZR, Lou HG, Yuan H. Determination of cymipristone in human plasma by a liquid chromatography-electrospray ionizationtandemmass spectrometry. J Chromatogr B AnalytTechnol Biomed Life Sci 2010;878:719-23.