- *Corresponding Author:
- Anumolu P.D
Gokaraju Rangaraju College of Pharmacy, Nizampet Road, Bachupally, Hyderabad-500 090, India
E-mail: panindrapharma@yahoo.co.in
| Date of Received : | 24 March 2014 |
| Date of Revised : | 10 January 2015 |
| Date of Accepted : | 30 May 2015 |
| Indian J Pharm Sci 2015;77(3):312-320 |
This is an open access article distributed under the terms of the Creative Commons Attribution‑NonCommercial‑ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non‑commercially, as long as the author is credited and the new creations are licensed under the identical terms.
Abstract
The establishment of biorelevant and discriminating dissolution procedure for drug products with limited water solubility is a useful technique for qualitative forecasting of the in vivo behavior of formulations. It also characterizes the drug product performance in pharmaceutical development. Lornoxicam, a BCS class-II drug is a nonsteroidal antiinflammatory drug of the oxicam class, has no official dissolution media available in the literature. The objective of present work was to develop and validate a discriminating and biorelevant dissolution test for lornoxicam tablet dosage forms. To quantify the lornoxicam in dissolution samples, UV spectrophotometric method was developed using 0.01M sodium hydroxide solution as solvent at λma× 376 nm. After evaluation of saturation solubility, dissolution, sink conditions and stability of lornoxicam bulk drug in different pH solutions and biorelevant media, the dissolution method was optimized using USP paddle type apparatus at 50 rpm rotation speed and 500 ml simulated intestinal fluid as discriminating and biorelevant dissolution medium. The similarity factor (f 2 ) were investigated for formulations with changes in composition and manufacturing variations, values revealed that dissolution method having discriminating power and method was validated as per standard guidelines. The proposed dissolution method can be effectively applied for routine quality control in vitro dissolution studies of lornoxicam in tablets and helpful to pharmacopoeias.
Keywords
Lornoxicam, biorelevant, discriminating, validation.
Dissolution tests are used in the pharmaceutical industry in a wide variety of application: To identify which sort of formulation will produce the best results in the clinic, to release products to the market, to verify batch-to-batch reproducibility, and to help identify whether changes made in the formulations or their manufacturing procedure after marketing approval are likely to effect the performance in the clinic. Further, dissolution tests can sometimes be implemented to determine whether a generic version of the medicine can be approved or not [1-3]. If the developed dissolution method mimics the GI-tract environment with discriminating power, it can be useful to forecast the in vivo behavior of formulations and to characterize the in vitro dissolution profiles of drug products during pharmaceutical development [4-8]. Bioequivalence problems arise in class II and class IV categories of the biopharmaceutical classification of drugs (BCS). The class II category, low solubility and high permeability drugs are identified as potential drug candidates for investigation. The low solubility aspects can be handled in order to develop a biorelevant and discriminating method for dissolution [9-13]. The development of a meaningful dissolution procedure for drug products with limited water solubility has been a challenge to both the pharmaceutical industry and the agencies that regulate them [14]. Lornoxicam, 6-chloro-4-hydroxy-2-methyl-N-2-pyridinyl-2Hthieno[ 2,3-e]-1,2-thiazine-3-carboxamide-1,1-dioxide (fig. 1) is a novel nonsteroidal antiinflammatory agent with marked analgesic properties [15]. Literature survey revealed that there is no official dissolution test has been reported for lorinoxicam. Therefore, the objective of present investigation was to develop and validate a discriminating and biorelevant dissolution test for lornoxicam.
Materials and Methods
All analytical grade chemicals and reagents were used for this investigation. Lornoxicam, the main active pharmaceutical ingredient was a gift sample from Hetero Drugs Limited, Hyderabad. The marketed formulations, Lornoxi 4, Lornoxi 8, Lorsaid 4 and Lorsaid 8 were procured from a local pharmacy. Chemicals like glacial acetic acid, hydrochloric acid, lactose, magnesium stearate, microcrystalline cellulose, potassium dihydrogen phosphate, sodium acetate, sodium dihydrogen phosphate, sodium hydroxide, sodium chloride, starch were purchased from S. D. Fine Chem Ltd, Mumbai, India; sodium lauryl sulphate (SLS) and Triton X100 were purchased from Himedia Ltd, Mumbai, India.
Biorelevent media like simulated gastric fluid (SGF) without enzyme, SGF media with 0.25% w/v SLS, SGF media with 0.1% v/v Triton Χ 100, simulated intestinal fluid (SIF) without enzyme, fasted state simulated intestinal fluid (FaSSIF) pH 6.5, fed state simulated intestinal fluid (FeSSIF) pH 5.0 were prepared according to the USP procedures [16,17].
Electrolab-TDT-08L dissolution test apparatus, double beam 1800 UV/Vis spectrophotometer (Shimadzu, Japan), analytical balance (Shimadzu AUX 220, Japan), orbital shaker bath (Bio-Technics, India), pH meter (Elico, Hyderabad), ultrasonic cleaner (Sonica), tablet compression machine (Lab press- CIP machineries, Ahmedabad, India) and hardness tester (Secor, Hyderabad, India) were used in this study.
Analytical method
Standard solution of lornoxicam was diluted appropriately with 0.01M sodium hydroxide (NaOH) to obtain a solution containing 10 μg/ml lornoxicam. This solution was scanned between 200-400 nm [18].
Saturation solubility studies
The solubility of lornoxicam was determined by preparing saturated solutions in different media such as double distilled water, 0.1M hydrochloric acid, acetate buffers (pH 2.7, 3.7 and 4.7), phosphate buffers (pH 5.8, 6.8 and 7.4), simulated gastric fluid without enzyme and with surfactants (SGF, SGF+0.25% w/v SLS and SGF+0.1% v/v Triton X-100), and blank fasted and fed state intestinal fluids (FaSSIF and FeSSIF). An excess of lornoxicam was taken in a 50 ml volumetric flask containing 10 ml of media. An orbital shaker bath was used for continuous shaking of the sample solutions in the volumetric flasks for a period of 24 h at temperature 37±1°. The equilibrated solutions were removed, filtered (Whattman filter paper No. 41) to separate the saturated solutions from excess undissolved drug. The saturated solutions of different systems were suitably diluted with 0.01 M sodium hydroxide solution and the concentration of lornoxicam was determined spectrophotometrically by measuring the absorbance at 376 nm, as reported in the analytical method.
Dissolution test conditions
Dissolution testing of lornoxicam (8 mg) bulk drug filled capsule (n=3) was performed in a paddle type USP tablet dissolution apparatus, in 500 ml of various buffers, such as 0.1M HCl, acetate buffers (pH 4.7), phosphate buffers (pH 6.8 and 7.4) and media such as SGF without enzyme, blank FaSSIF, FeSSIF and SIF without enzyme at 50 rpm and 37±1° for 45 min. Aliquots of 5.0 ml were withdrawn at each time (5, 10, 15, 20, 30 and 45 min) interval up to 45 min, and replaced with an equal volume of fresh medium to maintain sink conditions. At the end of the test, the withdrawn samples were filtered, diluted with 0.01M sodium hydroxide and quantified by the spectrophotometric method (n=3). The amount of dissolved drugs was computed from the respective calibration curves and then plotted against time. The medium in which highest drug release occurred for lornoxicam bulk drug, that medium was chosen for in vitro dissolution studies of tablet dosage form.
Stability determination
Samples of lornoxicam were collected from the optimized dissolution medium and were diluted with 0.01M sodium hydroxide to get final concentrations of samples within the linearity range of analytical method to measure the absorbance at 376 nm. The drug concentration calculated by using regression equation in samples of 0, 24 and 48 h were compared.
Discriminating power of the dissolution media
Discriminatory power of the dissolution method was determined by manufacturing the tablets under different conditions like hardness variation and disintegrated effect (with and without disintegrant). The dissolution behavior of punched tablets was investigated in the presence of proposed dissolution test conditions [19]. The dissolution profile data were also compared mathematically using the similarity fit factor (f2), 
If f2 is less than 50, then two dissolution profiles are considered as dissimilar. The similarity increases as the f2 value increases above 50 and approaches 100.
The dissolution method was validated by analyzing specificity, linearity, accuracy, precision and robustness as per standard guidelines [20].
Specificity
The specificity of the proposed method was evaluated through the analysis of a placebo solution, which was prepared with the common excipients (lactose, starch, microcrystalline cellulose, magnesium stearate, titanium dioxide and talc) of the pharmaceutical formulation. Thus, the mixture of component inert was prepared in their usual concentration employed in tablets (concentrations were determined based on the Handbook of Pharmaceutical Excipients and calculated for medium weight of content) [21]. The developed method was applied in order to check if any component of the formulation and dissolution medium could interfere with the absorbance of lornoxicam at selected wavelength.
Linearity
Aliquots of lornoxicam stock solution (1000 μg/ml) were diluted with 0.01M sodium hydroxide solution to give concentration of 2-22 μg/ml. Each solution was prepared in triplicate and linearity was determined by constructing the plot between analyte intensity vs. concentration to calculate the regression line by linear least squares methodology.
Precision
The precision of the method was determined by intraday precision and interday precision variations as per standard guidelines. The intraday precision and interday precision were assessed after subjecting six tablets to the dissolution test conditions, on the same day and on three different days respectively. The % RSD was calculated.
Accuracy
The accuracy was carried out by adding known amount of standard drug at 80, 100 and 120% of the nominal assay of lornoxicam to the placebo sample in the dissolution medium and then subjected to the proposed dissolution method. The experiment was conducted in triplicate. The percentage recovery and percentage relative standard deviation (% RSD) were calculated for each concentration.
Robustness
The robustness was studied by evaluating the effect of small but deliberate variations in the optimized dissolution test conditions. Different parameters that were altered to evaluate the robustness of the method such as bath temperature (±2.0°), pH of the dissolution medium (±2.0) and paddle rotation speed (±2 rpm)
Results and Discussion
The standard solution of lornoxicam (10 μg/ml) was scanned between 200-400 nm and found maximum absorbance at 376 nm. Hence, this wavelength has been selected for the development of standard plot and for the analysis of dissolution samples. The solubility profile of lornoxicam revealed that the solubility is pH-dependent. Lornoxicam exhibited significant solubility in water, phosphate buffers pH 6.8 and 7.4; this may be due to hydrogen bond formation and electrostatic interactions between –OH group and sodium hydroxide which is present in phosphate buffers. The pKa value of lornoxicam is 6.8, indicates high solubility of lornoxicam at above pH 7.0 (Table 1). The ratio of saturation solubility and drug concentration (dose) signify the closeness to the sink conditions. The term sink condition is defined as a volume of medium at least three times the volume required to form a saturated solution of a drug substance. The sink conditions of lornoxicam was calculated based on the solubility in different medias revealed that only phosphate buffer pH 7.4 and water exhibit the sink condition, but water is not ideal dissolution medium for in vitro dissolution test of immediate release dosage forms and it does not mimic the GI tract environment. The solubility behavior of lornoxicam was studied in different biorelevant media and the results of which have been shown in Table 2. These results revealed that sink conditions were achieved in SIF.
| Type of media | Solubility (mg/ml) | Sink conditions |
|---|---|---|
| *mean ± SD | (CS/CD) | |
| Water | 0.163 ± 0.006 | 10.187 |
| 0.1N HCl | 0.003 ± 0.0001 | 0.187 |
| Acetate buffer (pH? 2.8) | 0.014 ± 0.0002 | 0.875 |
| Acetate buffer (pH? 3.7) | 0.005 ± 0.0001 | 0.312 |
| Acetate buffer (pH? 4.7) | 0.002 ± 0.0006 | 0.125 |
| Phosphate buffer (pH? 5.8) | 0.005 ± 0.0008 | 0.312 |
| Phosphate buffer (pH? 6.8) | 0.047 ± 0.0002 | 2.937 |
| Phosphate buffer (pH? 7.4) | 0.172 ± 0.002 | 10.75 |
*Mean of three replications; CS indicates saturation solubility of lornoxicam in 500 ml of dissolution media; CD maximum (8 mg) dose of lornoxicam in tablet formulations
Table 1: Saturation solubility and sink Conditions of lornoxicam in different Dissolution media.
| Type of Bio ?relevant media |
Solubility (mg/ml) | Sink conditions |
|---|---|---|
| *mean±SD | (CS/CD) | |
| SGF | 0.005±0.0002 | 0.312 |
| SGF+0.25% SLS | 0.085±0.002 | 5.312 |
| SGF+Triton X?100 (0.1%v/v) | 0.008±0.004 | 0.500 |
| SIF | 0.376±0.020 | 23.500 |
| FaSSIF | 0.057±0.0002 | 3.562 |
| FeSSIF | 0.006±0.0005 | 0.375 |
*Mean of three replications; CS indicates saturation solubility of lornoxicam in 500 ml of biorelevant dissolution media; CD maximum (8 mg) dose of lornoxicam in tablet formulations. SGF is simulated gastric fluid. SLS is sodium lauryl sulphate, SIF is simulated intestinal fluid, FaSSIF is fasted state simulated intestinal fluid and FeSSIF is fed state simulated intestinal fluid
Table 2: Saturation solubility and sink Conditions of lornoxicam in different Biorelevant dissolution media.
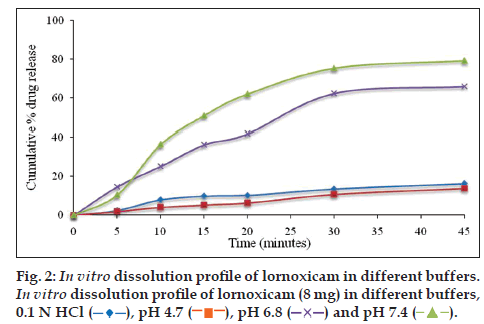
Dissolution testing of lornoxicam bulk drug was carried out in 500 ml of different dissolution media at 50 rpm, temperature 37±1° and samples were collected at 5, 10, 15, 20, 30 and 45 min time intervals. The amount of drug dissolved was analyzed by the developed UV method. The cumulative percent drug released was plotted against time. The dissolution medium with satisfactory drug release of lornoxicam was selected as optimized dissolution medium. The dissolution study results in different dissolution media for lornoxicam bulk drug are given in figs. 2 and 3, which revealed that the % cumulative drug release was as expected in phosphate buffer pH 7.4 and in SIF. Based on the results of saturation solubility studies and sink conditions of lornoxicam in different dissolution media, final optimized dissolution conditions were arrived at, which included USP apparatus 2 at a paddle speed 50 rpm and temperature 37±1° in 500 ml of SIF (pH 7.5) or phosphate buffer pH 7.4 as dissolution media with sampling time intervals of 5, 10, 15, 20, 30 upto 45 min. The optimized dissolution media mimics the gastrointestinal environment conditions (biorelevant) and pH is 7.5, within the range of GI tract pH levels. The optimized dissolution conditions were applied to test the in vitro dissolution of lornoxicam tablets, Lornoxi 4, Lornoxi 8, Lorsaid 4 and Lorsaid 8. The results as shown in fig. 4 revealed that more than 90% of drug was released from all tablet formulations within 45 min.
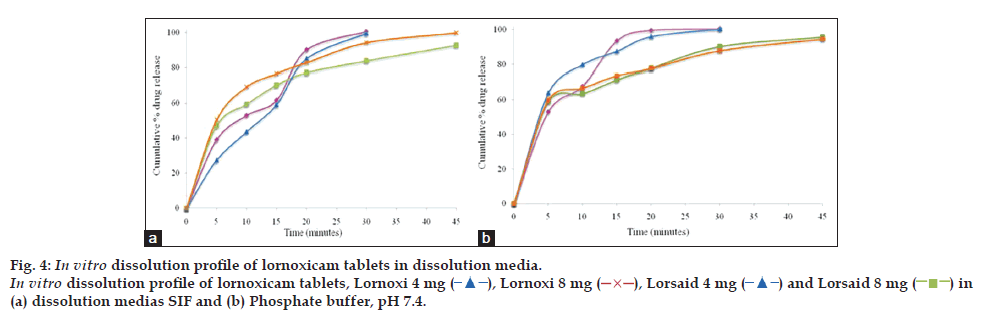
Discriminatory power of the dissolution method was determined by manufacturing the tablets (n=12) under different conditions and checking the dissolution behavior in the presence of proposed dissolution test conditions. The dissolution study results for the effect of tablet hardness (4 kg/cm2 vs. 8 kg/cm2) and disintegrant (with disintegrant vs. without disintegrant) are shown in figs. 5 and 6, which revealed that dissolution rate of both drugs was slightly faster for tablets with lesser hardness and with disintegrant.
Therefore, this dissolution method has the ability to discriminate certain changes in composition and manufacturing process. The dissolution profile data was also compared mathematically using the similarity fit factor (f2). Table 3, listed out the f2 values used for comparison of the dissolution profiles for each of the process parameters evaluated. Based on the results of similarity factor values, SIF media has more discriminating power for variation in composition difference and manufacturing process of lornoxicam self punched tablets than phosphate buffer, pH 7.4. So, finally SIF media was considered as discriminating and biorelevant dissolution medium for lornoxicam tablets and the dissolution method was validated in SIF media.
| Media | Process parameter | |
|---|---|---|
| Tablets withhardness of 4 kg/cm2vs 8 kg/cm2 |
Tablets containing disintegrantvs no disintegrant |
|
| Phosphate buffer pH 7.4 | 69.431 | 78.17 |
| SIF | 46.10 | 51.35 |
Table 3: Similarity factor (f2) for dissolution Profiles of tablets with different parameters.
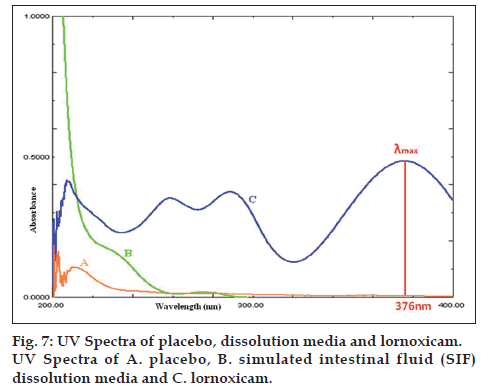
The UV spectrum of placebo and lornoxicam in the dissolution medium is shown in fig. 7. A perusal to figure indicated that there is no interference from the excipients in the tablets and dissolution medium with UV absorbance of lornoxicam at their respective analytical wavelengths. Therefore, the proposed method is specific.
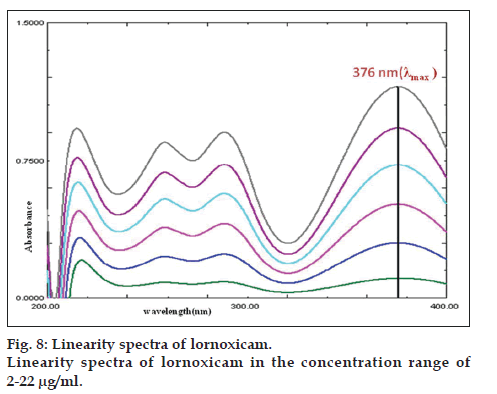
The linearity was evaluated by the least square regression method. The responses for lornoxicam at 376 nm were found to be linear in the concentration range of 2-22 μg/ml, with a correlation co-efficient (r) value of 0.997. The results indicate a good linear relationship between UV absorbance and concentration. Linearity spectra are shown in fig. 8.
The % relative standard deviation values for intra- and inter-day precision studies were found to be less than 2 (Table 4) and no significant difference was observed between intraday and interday values, which indicate that the proposed dissolution method was reproducible and precise. The accuracy states the agreement between the standard value and observed value. The accuracy results of proposed dissolution method were reported in Table 5, % recovery was found from 93.75-95.83% and % relative standard values were less than 2; signify that the dissolution method is accurate for its intended use.
| Formulation | Intra?day precision | Inter?day precision | ||
|---|---|---|---|---|
| mean ± SD (n=6)* | % RSD | mean ± SD (n=6) | % RSD | |
| Lornoxi 4 | 100.60 ± 0.421 | 0.418 | 100.02 ± 0.625 | 0.624 |
| Lornoxi 8 | 95.78 ± 1.273 | 1.273 | 94.54 ± 0.542 | 0.573 |
| Lorsaid 4 | 100.47 ± 1.973 | 1.973 | 100.48 ± 1.256 | 1.252 |
| Lorsaid 8 | 94.62 ± 1.185 | 1.252 | 95.45 ± 1.092 | 1.144 |
*Number of six replications; RSD is relative standard deviation
Table 4: Precision data for the proposed Dissolution Method.
| Analyte | % Level of | Amount (mg) | % Recovery | % RSD | |
|---|---|---|---|---|---|
| recovery | Added | Recovered | |||
| Lornoxicam | 80 | 3.2 | 3.0 | 93.75 | 0.94 |
| 100 | 4 | 3.8 | 95.00 | 1.25 | |
| 120 | 4.8 | 4.6 | 95.83 | 1.02 | |
Table 5: Accuracy Data For The Proposed Dissolution Method.
The robustness was examined against small but deliberate variations of critical parameters, including bath temperature, pH of the dissolution medium and paddle rotation speed. The robustness results of proposed dissolution method were reported in figs. 9 and 10, revealed that no significant differences were found among the amounts of drug dissolved at the predetermined time points under the different temperatures, rpm and pH with respect to those corresponding to the optimal dissolution conditions, confirming the robustness of test at ±2 level. The optimized conditions proved to be robust with regards to temperature, paddle rotation speed and pH of the dissolution medium.
Figure 9: In vitro dissolution profile of Lornoxi 8 and Lorsaid 8 tablets.
In vitro dissolution profile of Lornoxi 8 (above panel) and Lorsaid 8 (below panel) tablets under varying conditions such as (a) bath temperature 35º ( ), 37º (
), 37º ( ), 39º (
), 39º ( ), (b) paddle rotation speed, 48 (
), (b) paddle rotation speed, 48 ( ), 50 (
), 50 ( ), 52 (
), 52 (  ) and (c) pH 7.3 (
) and (c) pH 7.3 (  ), 7.5 (
), 7.5 ( ), 7.7 (
), 7.7 ( ) demonstrating robustness of the method.
) demonstrating robustness of the method.
Figure 10: In vitro dissolution profile of Lornoxi 4 and Lorsaid 4 tablets.
In vitro dissolution profile of Lornoxi 4 (above panel) and Lorsaid 4 (below panel) tablets under varying conditions such as (a) bath temperature 35º (  ), 37º (
), 37º (  ), 39º (
), 39º ( ); (b) paddle rotation speed 48 (
); (b) paddle rotation speed 48 ( ), 50 (
), 50 ( ), 52 (
), 52 (  ) ; (c) pH 7.3 (
) ; (c) pH 7.3 ( ), 7.5 (
), 7.5 ( ), 7.7 (
), 7.7 ( ) demonstrating robustness of the method.
) demonstrating robustness of the method.
Lornoxicam was found to be stable under dissolution test conditions and the measured absorbance of the initial time, 24 and 48 h were similar. There was no evidence of degradation of the drugs under dissolution test conditions, revealed that solutions were stable for more than 48 h (Table 6).
| Analyte | % Recovery | ||
|---|---|---|---|
| Initial time | After 24 h | After 48 h | |
| Lornixicam | 98.00 | 98.46 | 99.05 |
Table 6: Data For Stability Of Dissolution Samples.
The discriminating and biorelevant dissolution test for quantification of lornoxicam tablets has been developed and validated. Based on strategy of saturation solubility studies, sink conditions, stability evidence and discriminating power of dissolution media; the best conditions optimized for dissolution testing for lornoxicam tablets were: 500 ml of SIF without enzyme, paddle type apparatus, stirring speed is 50 rpm, at a temperature of 37±10 and collection time of 45 min. The proposed dissolution test was applied to study the in vitro dissolution profiles of lornoxicam tablet formulations, demonstrated that the drug release was more than 90% from all formulations within 45 min. The proposed dissolution method was validated and results proved that the method is scientifically sound with selectivity, linearity, precise, accurate and robustness. The dissolution studies of lornoxicam tablets in proposed dissolution medium was found to better, because of its discriminating power and surrogate the GIT environment (biorelevant). These advantages promote that; the developed dissolution method can be routinely employed in quality control analysis of lornoxicam in tablet dosage forms since a dissolution test not indicated in any official monograph and it may help to pharmaceutical researchers.
Acknowledgements
The authors thank the management of Gokaraju Rangaraju College of Pharmacy for providing necessary facilities and infrastructure to carry out this research work.
Financial support and sponsorship
Nil.
Conflict of interest
There are no conflicts of interest.
References
- Brahmankar DM, Jaiswal SB. Biopharmaceutics and Pharmacokinetics. 1st ed. New Delhi: VallabhPrakashan; 2007. p. 19-25.
- Subrahmanyam CV. Physical Pharmaceutics. 2nd ed. New Delhi: VallabhPrakashan; 2005. p. 85-96.
- Wang J, Flanagan DR. Fundamentals of Dissolution. 1st ed. Academic Press; 2009. p. 309-18.
- Lagace M, Gravelle L, Di Maso M, McClintock S. Developing a discriminating dissolution procedure for a dual active pharmaceutical product with unique solubility characteristics. DissolutTechnol2004;2:13-7.
- Soni T, Nagda C, Gandhi T, Chotai NP. Development of discriminating method for dissolution of aceclofenac marketed formulations. Dissolut Technol 2008;5:31-5.
- Anumolu PD, Sunitha G, Raju YV, Babu PRS, Subrahmanyam CVS. Development of biorelevant and discriminating dissolution media for efavirenz and its formulations. Asian J Pharm Clin Res 2012;5:220-3.
- Qingxi W, Nikoletta F, Yun M. Biorelevant dissolution methodology and application in drug development. DissolutTechnol 2009;8:6-12.
- Zoeller T, Klein S. Simplified biorelevant media for screening dissolution performance of poorly soluble drugs. DissolutTechnol 2007;11:8-13.
- Dressman J, Kramer J. Pharmaceutical dissolution Testing. 1st ed. London: Taylor and Francies group; 2007. p. 92-8.
- Rohrs BR. Dissolution method development of poorly soluble compounds. DissolutTechnol 2001;8:1-5.
- Tenhoor NC, Bakatselou V, Dressman J. Solubility of mefenamic acid under simulated fed and fasted state conditions. J Pharm Res 1991;8:1203-5.
- Jogia H, Mehta T, Patel M. Evaluation of dissolution media containing a novel synthetic surfactant by in vitrotesting of BCS class II drugs.DissolutTechnol 2009;8:14-9.
- Anumolu PD, Gurrala S, Yeradesi VR, Puvvadi SB, Chavali SVS. Development of dissolution test method for drotaverine hydrochloride/ mefenamic acid combination using derivative spectrophotometry. Trop J Pharm Res 2013;12:227-32.
- Huang Z, Lozano R, Francis R, Aubry A-F, Steckbeck A, Sciascia DO. Development of a single in vitro dissolution method for a combination trilayer tablet formulation of clopidogrel and pravastatin. DissolutTechnol 2011;2:12-9.
- O?Neil MJ. The Merck index, An Encyclopedia of Chemicals, Drugs and Biologicals. 14th ed, Whitehouse Station NJ, USA: Division of Merck and Co., Inc; 2006. p. 5582.
- Boni JE, Brickl RS, Dressman J, Pfefferle ML. Instant FaSSIF and FeSSIFbiorelevance meets practicality. DissolutTechnol 2009;8:41-5.
- United States Pharmacoepia and National Formulary USP 29-NF 24, Rockville, MD, USA: The United States Pharmacoepial Convention, Inc; 2006. p. 2673.
- Ramyakrishna N, Ramanjaneyulu KV, Deepti MK, Sudhakar AM, Rao PV, Pramod N. Method development and validation of lornoxicam in pharmaceutical dosage form by using UV-Visible spectrophotometry. Int J Pharm Biol Archives 2012;3:1012-6.
- Moore J, Flanner H. Mathematical comparison of curves with an emphasis on in vitrodissolution profiles. Pharm Technol 1996;20:64-74.
- FDA Guidance for industry: Immediate release solid dosage forms: In vitro dissolution testing and in-vivo bioequivalence documentation. Rockville MD, USA: Food and Drug Administration; 1995.
- Raymond CR, Paul JS, Sian CO. Hand Book of Pharmaceutical Excipients. 2nd ed. London: Pharmaceutical Press and American Pharmacists Association; 2006. p. 132, 385,725.
 ), pH 6.8 (
), pH 6.8 (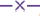 ) and pH 7.4 (
) and pH 7.4 ( 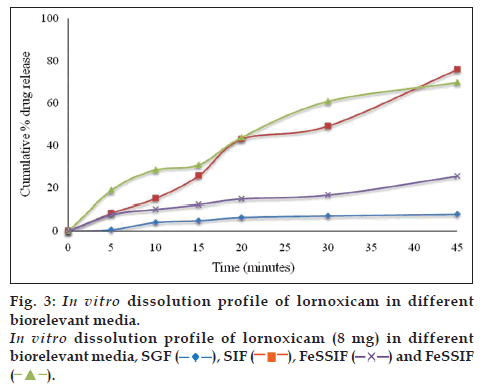
 ), Lorsaid 4 mg (
), Lorsaid 4 mg ( ) and Lorsaid 8 mg (
) and Lorsaid 8 mg (  ) in(a) dissolution medias SIF and (b) Phosphate buffer, pH 7.4.
) in(a) dissolution medias SIF and (b) Phosphate buffer, pH 7.4.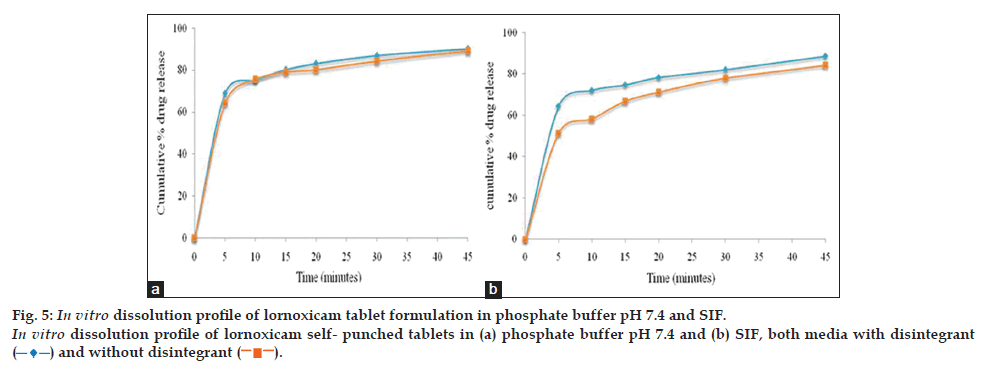
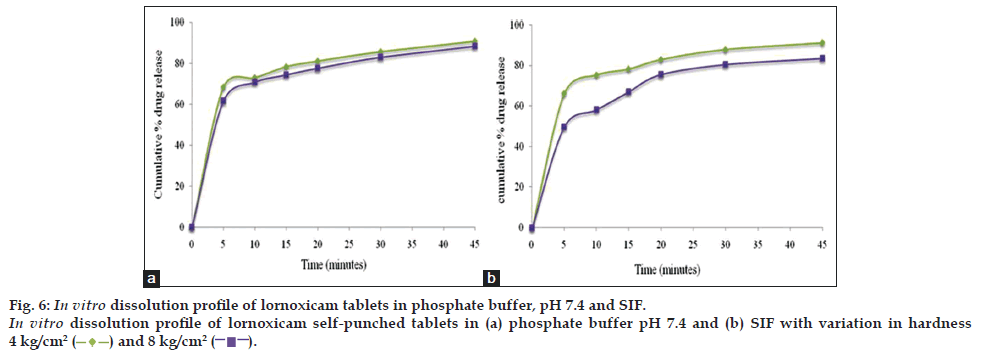
 ) and 8 kg/cm2 (
) and 8 kg/cm2 (  ).
).