- *Corresponding Author:
- K. P. R. Chowdary
Industrial Pharmacy Division, University College of Pharmaceutical Sciences, Andhra University, Visakhapatnam-530 003, India
E-mail: profkprc@rediffmail.com
| Date of Submission | 12 July 2005 |
| Date of Revision | 27 February 2006 |
| Date of Acceptance | 15 September 2006 |
| Indian J Pharm Sci, 2006, 68 (5): 631-634 |
Abstract
Complexation of celecoxib with β-cyclodextrin and hydroxypropyl-β-cyclodextrin in the presence and absence of polyvinylpyrrolidone and the effect of polyvinylpyrrolidone on the solubilizing efficiency of cyclodextrins and on the dissolution rate of celecoxib from the cyclodextrin complexes were investigated. The phase solubility studies indicated the formation of celecoxib-β-cyclodextrin and celecoxib-hydroxypropyl-β-cyclodextrin inclusion complexes at a 1:1 M ratio in solution, both in the presence and absence of polyvinylpyrrolidone. The complexes formed were quite stable. The solubility and dissolution rate of celecoxib were markedly enhanced by complexation with β-cyclodextrin and hydroxypropyl-β-cyclodextrin. Celecoxib-hydroxypropyl-β-cyclodextrin (1:2) inclusion complex gave a 36.57-fold increase in the dissolution rate of celecoxib. Addition of polyvinylpyrrolidone resulted in higher complexation efficiency and markedly enhanced solubilizing efficiency of β-cyclodextrin and hydroxypropyl-β-cyclodextrin. Solid inclusion complexes of cyclodextrins with polyvinylpyrrolidone gave several times higher rates of dissolution than those of celecoxib and its complexes with cyclodextrins alone.
Cyclodextrins (CDs) are cyclic torus-shaped molecules with a hydrophilic outer surface and a lipophilic central cavity, which can accommodate a variety of lipophilic drugs. As a consequence of the inclusion process, many physicochemical properties such as solubility, dissolution rate, stability and bioavailability can be favourably modulated [1-3]. Cyclodextrins have been receiving increasing application in pharmaceutical formulations in recent years due to their approval by various regulatory agencies [4,5]. However, the use of cyclodextrins in solid oral dosage forms is limited to low dose drugs with large stability constants due to the mass limitations of oral dosage units [3]. Therefore, in cases where the low complexation efficiency would require a larger amount of CD than that acceptable for solid or liquid dosage forms, the enhancement of the complexation capacity of the chosen CD is of practical importance. On this subject, earlier papers have reported [2,6,7] the positive effect of the addition of small amounts of a suitable water-soluble polymer to a drug-CD system in improving both the complexing and solubilizing efficiencies of the CDs.
Celecoxib is the most widely prescribed COX-2 inhibitor. The poor aqueous solubility and wettability of celecoxib give rise to difficulties in the design of pharmaceutical formulations and lead to variable oral bioavailability. We have been working on the enhancement of solubility, dissolution rate and bioavailability of celecoxib through complexation with cyclodextrins [8]. In the present work, the effect of polyvinylpyrrolidone (PVP) on the complexation of celecoxib with ß- and hydroxypropyl-ß-cyclodextrins (HPßCD) was investigated. The effect of PVP on the solubilizing efficiency of cyclodextrins and dissolution of celecoxib from the CD complexes was also investigated.
Materials and Methods
Celecoxib was a gift sample from M/s Dr. Reddy’s Laboratories, Hyderabad. ß-CD and HPßCD were gift samples from M/s Cerestar Inc., USA. PVP (K40), dichloromethane (Qualigens), methanol (Qualigens) and sodium lauryl sulphate (Qualigens) were procured from commercial sources. All other materials used were of pharmacopoeial grade.
Phase solubility studies
Solubility studies were performed according to the method reported by Higuchi and Connors [9]. Excess drug (25 mg) was added to 15 ml of double distilled water (pH 6.8) containing various concentrations of ßCD or HPßCD (3-15 mM) in a series of 50 ml stoppered conical flasks, and the mixtures were shaken for 72 h at room temperature (28°) on a rotary flask shaker. After equilibration, 2 ml aliquots were withdrawn at 1 h intervals and filtered immediately using 0.45 μ nylon disc filter. The filtered samples were diluted suitably and assayed for celecoxib at 254 nm against blanks prepared in the same concentration of ßCD or HPßCD in water so as to cancel any absorbance that may be exhibited by the cyclodextrin molecules. Shaking was continued until three consecutive estimations were the same. Phase solubility studies were conducted in each case with and without addition of PVP. PVP was added at a concentration of 0.5% w/v to the solution containing CDs. The solubility experiments were conducted in triplicate.
Preparation of solid complexes
Solid inclusion complexes of celecoxib-ßCD and celecoxib-HPßCD were prepared in 1:1 and 1:2 ratios by kneading method with and without the addition of PVP. PVP was added at a concentration of 10% w/w of the a mortar with a small volume of a solvent blend of water :methanol : dichloromethane (4:6:1). The thick slurry formed was kneaded for 45 min and then dried at 55°. The dried mass was powdered and sieved through mesh prepared to study the effect of PVP on the solubility and dissolution rate of celecoxib.
Estimation of celecoxib
A UV spectrophotometric method based on the measurement of absorbance at 254 nm in water containing 1% sodium lauryl sulphate (SLS) was developed and used for the estimation of celecoxib. The method obeyed 2006 Beer’s law in the concentration range of 2-10 μg/ml. When a standard drug solution was assayed repeatedly (n = 6), the relative error (accuracy) and relative standard deviation (precision) were found to be 0.8% and 1.2% respectively.
Dissolution rate study
The dissolution rate of pure celecoxib and the celecoxib from all CD inclusion complexes prepared was studied using DISSO 2000, Lab India 8–station dissolution rate test apparatus with a paddle stirrer. The dissolution rate was studied in 900 ml of water containing 1% SLS. Celecoxib (50 mg) or its inclusion complex equivalent to 50 mg of celecoxib, a speed of 50 rpm and a temperature of 37 ± 1° were used in each test. Samples of dissolution medium (5 ml) were withdrawn through a filter (0.45 μ) at different time intervals, suitably diluted and assayed for celecoxib by measuring absorbance at 254 nm. The volume withdrawn at each time was replaced with dissolution medium (5 ml). The dissolution experiments were conducted in triplicate.
Results and Discussion
The complexation of celecoxib with ßCD and HPßCD was investigated by phase solubility studies. The phase solubility diagrams for the complex formation between celecoxib and CDs in the presence and absence of PVP are shown in fig. 1. The aqueous solubility of celecoxib was found to increase linearly as a function of the concentration of CD. The phase solubility diagrams of celecoxib-CD complexes can be classified as type AL according to Higuchi and Connors [9]. Because the straight line had a slope < 1 in each case, the increase in solubility was due to the formation of a 1:1 M complex in solution with both ßCD and HPßCD in the presence and absence of PVP. The apparent stability constant (Kc) was calculated from the slope of the linear plot of the phase solubility diagram according to the equation Kc = Slope/ So(1-Slope), where So is the solubility of the drug in the absence of CD. The estimated Kc values of various complexes are given in Table 1. The values of Kc indicated that the complexes formed between celecoxib and CDs are quite stable.
| Sample | Kc (M-1) | Solubilizing efficiency* |
|---|---|---|
| C-βCD | 310.54 | 5.74 |
| C-HPβCD | 695.56 | 11.07 |
| C-βCD-PVP | 798.87 | 11.04 |
| C-HPβCD-PVP | 912.19 | 15.28 |
*Ratio between drug solubility in aqueous solution (15 mM) of CDs (with or without PVP) and drug solubility in water
Table 1: Effect of pvp on stability constant (Kc) and solubilizing efficiency of Cyclodextrins
To evaluate the effect of PVP, the solubilizing efficiency of CDs in the presence and absence of PVP was calculated as a ratio between drug solubility in aqueous solution of CD (15 mM), with and without PVP, and drug solubility in water. The solubilizing efficiency values are given in Table 1. ßCD alone gave a 5.74-fold increase in the solubility of celecoxib, whereas in the presence of PVP, it gave an 11.04-fold increase. Similarly HPßCD gave a 15.28- and 11.07-fold increase in the solubility of celecoxib respectively in the presence and absence of PVP. Thus, the addition of PVP markedly enhanced the solubilizing efficiency of ßCD and HPßCD. Among the two CDs, HPßCD exhibited higher solubilizing efficiency both in the absence and presence of PVP. The values of stability constant (Kc) were found to be higher in the presence of PVP, with both ßCD and HPßCD indicating higher complexation efficiency. A 2.57- and 1.31-fold increase in the Kc value was observed respectively with ßCD and HPßCD in the presence of PVP.
Solid inclusion complexes of celecoxib-ßCD and celecoxib-HPßCD in 1:1 and 1:2 ratios were prepared with and without PVP by kneading method. All the solid complexes prepared were found to be fine and free flowing powders. Low C.V. (<1.0%) values in percen drug content indicated uniformity of drug content in each batch of solid inclusion complex prepared. The dissolution rate of pure celecoxib and celecoxib from various solid inclusion complexes including celecoxib-PVP (9:1) system was studied in water containing 1% SLS. SLS was included in the dissolution medium to maintain sink condition. The dissolution profiles of various complexes are shown in fig. 2. The dissolution of celecoxib from all the solid inclusion complexes was more rapid and higher when compared to celecoxib pure drug. The dissolution of pure celecoxib and celecoxib from various complexes followed first order kinetics (r > 0.980). Dissolution rate constants (K1) were calculated from the slopes of the first order linear plots of the dissolution data. Dissolution efficiency (DE30) values based on the dissolution data were calculated as per Khan [10]. T50 (time taken for 50% dissolution) values were recorded from the dissolution profiles. The dissolution parameters are summarized in Table 2.
| Product | Percent dissolved in 10 min | T50 (min) | DE50(%) | K1 (min-1) | Increase in K1 (folds)* |
|---|---|---|---|---|---|
| Celecoxib | 29.63 ± 1.06 | >60 | 28.91 | 0.0075 | - |
| C-βCD (1:1) | 55.12 ± 1.71 | 8.0 | 55.49 | 0.0523 | 6.97 |
| C-βCD (1:2) | 73.86 ± 2.06 | 3.0 | 72.24 | 0.0996 | 13.28 |
| C-HPβCD (1:1) | 75.29 ± 1.37 | 3.5 | 74.94 | 0.1573 | 20.97 |
| C-HPβCD (1:2) | 85.04 ±1.05 | 1.5 | 83.75 | 0.2743 | 36.57 |
| C-PVP (9:1) | 36.13 ± 1.57 | 28.0 | 37.84 | 0.0136 | 1.81 |
| C-βCD-PVP (1:1:0.2) | 71.91 ± 1.70 | 4.0 | 68.84 | 0.0913 | 12.17 |
| C-βCD-PVP (1:2:0.3) | 88.86 ± 2.10 | 2.0 | 83.14 | 0.1339 | 17.85 |
| C-HPβCD-PVP (1:1:0.2) | 82.97 ± 1.21 | 2.5 | 82.47 | 0.2959 | 39.45 |
| C-HPβCD-PVP (1:2:0.3) | 99.57 ±1.71 | 1.0 | 89.23 | 0.5445 | 72.60 |
*Ratio between K1 of CD complex and K1 of celecoxib. T 50 is time taken for 50% dissolution; DE30 is dissolution efficiency up to 30 min; and K1 is first order dissolution rate constant
Table 2: Dissolution parameters of various celecoxib solid complexes
All CD complexes exhibited higher rates of dissolution and dissolution efficiency values than celecoxib and celecoxib-PVP (9:1) system, indicating more rapid and higher dissolution of celecoxib from its CD complexes. The K1 and DE30 values increased as the proportion of CD in the complex was increased. The increase in K1 (folds) with various CD systems is shown in Table 2.
HPßCD gave higher enhancement in the dissolution rate and efficiency of celecoxib when compared to ßCD. A 36.57-fold increase in the dissolution rate of celecoxib was observed with celecoxib-HPßCD (1:2) complex. PVP alone gave only a marginal increase in the dissolution rate of celecoxib. A 1.81-fold increase in the dissolution rate was observed with celecoxib-PVP (9:1) system, whereas addition of PVP has markedly enhanced the dissolution rate and efficiency of celecoxib from CD complexes. Celecoxib-ßCD (1:2) complex gave a 13.28-fold increase in the dissolution rate of celecoxib, whereas celecoxib ßCD-PVP (1:2:0.3) complex gave a 17.85-fold increase. Similarly celecoxib-HPßCD (1:2) and celecoxib-HPßCD- PVP (1:2:0.3) complexes gave 36.57- and 72.60-fold increase in the dissolution rate of celecoxib respectively. Thus inclusion of PVP in the CD complexes has markedly enhanced both complexation and solubilizing efficiencies of the CDs, and the solid inclusion complexes of CDs with PVP gave higher rates of dissolution - several times higher than those of celecoxib and its complexes with CDs alone.
Because of the enhancement in CD complexation and solubilizing efficiencies by the presence of PVP, celecoxib-ßCD-PVP (1:1:0.2) complex gave a dissolution rate equal to that of celecoxib-ßCD (1:2) complex, and celecoxib-HPßCD-PVP (1:1:0.2) complex gave a dissolution rate equal to that of celecoxib-HPßCD (1:2) complex. These observations indicate that in the presence of PVP, a low amount of CD can be used to get the desired dissolution rate and efficiency.
Thus, the results of the study indicated the formation of celecoxib-ßCD and celecoxib-HPßCD inclusion complexes at a 1:1 M ratio in solution both in the presence and absence of PVP. The complexes formed are quite stable. The solubility and dissolution rate of celecoxib were markedly enhanced by complexation with ßCD and HPßCD, and addition of PVP resulted in higher complexation efficiency and markedly enhanced the solubilizing efficiency of ßCD and HPßCD. Thus, addition of PVP, a water-soluble polymer, could be a strategy that can be employed to improve the usefulness of cyclodextrins.
Acknowledgements
One of the authors (SVS) is grateful to CSIR, New Delhi, for the award of CSIR-SRF to work on cyclodextrin complexation for enhancing dissolution and bioavailability.
References
- Uekama, K., Hirayama, F. and Irie, T., Chem. Rev., 1998, 98, 2045.
- Loftsson, T. and Brewster, M.E., J. Pharm. Sci., 1996,85,1017.
- Rajewski, R.A. and Stella, V.J., J. Pharm. Sci., 1996, 85,1142.
- Thompson, D.O., Crit. Rev. Ther. Drug Carrier Syst., 1997,14, 1.
- 5. Hedges, A.R., Chem. Rev., 1998, 98, 2035.
- J. Pharm., 1994, 110, 169.
- Mura, P., Faucci, M.T. and Bettinetti, G.P., Eur. J. Pharm. Sci.,2001, 13, 187.
- Chowdary, K.P.R., Reddy, M.N. and Diwan, P.V., AAPS Pharm.Sci., 2004, 6, 7.
- Higuchi, T. and Connors, K.A., Adv. Anal. Chem. Instr., 1965, 4, 117.
- Khan, K.A., J. Pharm. Pharmacol., 1975, 27, 48.
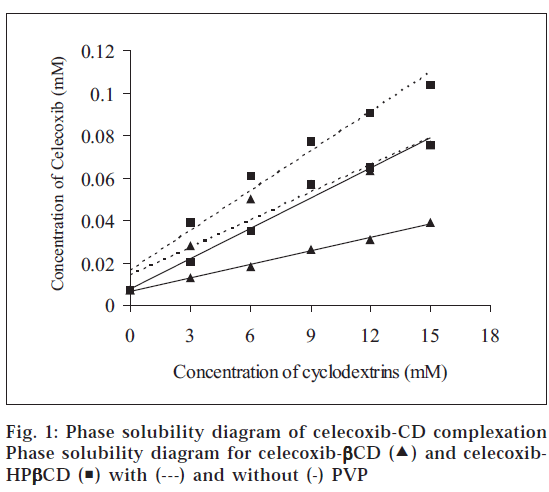
 ) and celecoxib-HPβCD (
) and celecoxib-HPβCD (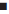 ) with (---) and without (-) PVP
) with (---) and without (-) PVP
 ), C-HPßCD-PVP (
), C-HPßCD-PVP ( );Drug:CD ratios 1:1 (-), 1:2 (---) and C-PVP (9:1) (+)
);Drug:CD ratios 1:1 (-), 1:2 (---) and C-PVP (9:1) (+)