- *Corresponding Author:
- Zhumei Sun
School of Clinical Medicine, North China University of Technology, China
E-mail: zhujichuo1870@163.com
| Date of Received | 24 August 2021 |
| Date of Revision | 30 October 2022 |
| Date of Acceptance | 02 February 2023 |
| Indian J Pharm Sci 2023;85(1):241-248 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
One of the leading causes of diabetes-related deaths is myocardial damage, which may be the cause of heart failure in people with type 2 diabetes mellitus. The objective of the research was to look at the impact of coronary failure with kind a pair of type 2 diabetes mellitus. This project aimed to explore the protective impact of oleanolic acid on myocardial damage in type 2 diabetes mellitus and to investigate the connected mechanism. Specific pathogen free grade db/db male mice were elected as model, while the Db/m mice were opted for control. Different doses of drug intervention were performed and the general condition, cardiac function, blood glucose, blood lipids, degree of myocardial injury, and degree of oxidative stress were examined by morphological examination of myocardial tissues, biochemical examination, and gene and protein amount detection. The results showed that lactate dehydrogenase, creatine kinase isoenzyme, total cholesterol, triglycerides and malondialdehyde levels in serum of Db/db mice were increased, while phosphatidylinositol 3 kinase, protein kinase B, glucose transporter 4 expression and superoxide dismutase level in myocardial tissue were decreased. After using oleanolic acid, the serum concentrations of lactate dehydrogenase, creatine kinase isoenzyme, total cholesterol, triglycerides and malondialdehyde were declined in Db/db mice, while phosphatidylinositol 3 kinase, protein kinase B, glucose transporter 4 expression and superoxide dismutase level were rised. The results recommend that oleanolic acid has protective impact on cardiac muscle injury in Db/db mice and conjointly the mechanism could also be correlated with promoting the activation of phosphatidylinositol 3 kinase/protein kinase B/glucose transporter 4 signal transduction.
Keywords
Oleanolic acid, type 2 diabetes mellitus, myocardial injury, phosphatidylinositol 3 kinase/protein kinase B/glucose transporter 4, insulin resistance, oxidative stress
The 2019 International Diabetes Federation report shows that China has the best variety of individuals with Diabetes (DM) within the world, with a complete of 114 million individuals with DM. Sort a pair of Type 2 Diabetes Mellitus (T2DM) accounts for 90 %-95 % of the overall variety of DM patients[1]. At the same time, people with T2DM have multiple cardiovascular complications and it is expected that 75 % of people with DM will die from cardiovascular disease by 2030. Heart failure is 20 %-40 % more common in people with DM and the risk of hospitalization is 33 % higher than normal[2], posing a serious threat to patient safety.
Insulin Resistance (IR) could be an ordinary pathogenic property of T2DM and is influenced by variety of factors. IR reduces the insulin response of target tissues, resulting in reduced glucose uptake and utilization by target tissues.
The Phosphatidylinositol 3 Kinase (PI3K)/Protein Enzyme B (Akt) signal transduction has been shown to be a really crucial signal pathway involved in IR and performs a crucial function among the event of T2DM[3]. Apart from reducing cellular inflammatory damage, promoting angiogenesis and regulating energy metabolism, this pathway can also exert hypoglycaemic effects by regulating glucose transport, lipid metabolism and protein synthesis[4]. Glucose Transporter 4 (Glut 4), a signaling macromolecule lower reaches the PI3K/ Akt signaling route, plays a significant part in glucose transport[5] (fig. 1). Meanwhile, recent studies suggest that the prevention and treatment of DM should be synchronized with lipid-lowering, glucose-lowering and antioxidants. Currently, the commonly used clinical hypoglycaemic drugs are biguanides, sulphonylureas and dipeptidyl peptidase VI inhibitors, the long-term use of these drugs can have negative effects on the body and can even aggravate heart failure[6]. Oleanolic Acid (OA), which is widely found in herbal medicine, has been discovered to have antibacterial, antitumor, antiinflammatory and analgesic efficacy, in addition to antioxidant, hepatoprotective and vascular effects[7]. However, the mechanism of the protecting impact of OA on cardiac muscle injury during a high glucose setting isn't clear. Therefore, during this study, the PI3K/Akt/Glut 4 signal pathway was used because the target to research the impact of OA on the viscous part of DM mice and to undertake out its mechanism of action to produce an experimental foundations for the interference and therapy of cardiac muscle injury in DM.
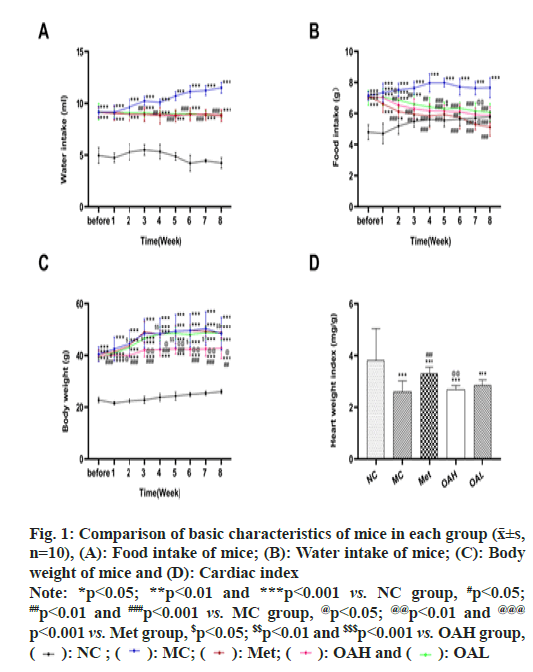
Fig. 1: Comparison of basic characteristics of mice in each group (x͞±s,
n=10), (A): Food intake of mice; (B): Water intake of mice; (C): Body
weight of mice and (D): Cardiac index
Note: *p<0.05; **p<0.01 and ***p<0.001 vs. NC group, #p<0.05; ##p<0.01 and ###p<0.001 vs. MC group, @p<0.05; @@p<0.01 and @@@ p<0.001 vs. Met group, $p<0.05; $$p<0.01 and $$$p<0.001 vs. OAH group,  OAL
OAL
Materials and Methods
Grouping and dosing:
Forty Specific Pathogen Free (SPF) grade male Db/ db mice (28-34) g and ten SPF grade male Db/m mice (18-24) g were bought from Changzhou Cavins Laboratory Animal Co., Ltd (License No.: SCXK (Su) 2018-0002), all animals were ate and drank freely under the identical conditions. Experiments were launched in conformity to the rules of China Animal Analysis Committee.
Grouping: Db/m mice were utilized as the Normal Control (NC) group (n=10) following a week of acclimation. Using a random number table, the Db/ db mice were separated into four groups; the Model Control (MC) group (n=10), the Oleanolic Acid High dosage (OAH) group (n=10), the Oleanolic Acid Low dose (OAL) group (n=10) and the Metformin (Met) group (n=10). Saline was administered at the dose of 10 ml/kg in the NC and MC groups, 200 mg of Met (Sino-US Shanghai Squibb 20190427, specification: 0.5), 200 mg of OA (O110087, Abcam, purity: >98 %) and 100 mg of OA in the OAL group. For 8 w, the medication was given once daily by gavage.
Feeding and water intake measurements:
Each group of mice was placed in a cage and an equal amount of feed was weighed on an electronic balance and placed in the cage of each group. A graduated water dispenser was used to measure the amount of water drunk by the mice.
Blood glucose and glucose tolerance testing:
Blood was taken from the tail vein of every group of mice when 4 h of fasting, and glucose was measured and recorded employing a Roche glucose meter once per week. After 8 w, the glucose tolerance of each group of mice was assessed; fasting for 12 h, fasting blood glucose (0 point) was measured first and glucose (2 g/kg) was administered to each group 30 min after administration, followed by the level of blood sugar measurement utilizing a blood sugar meter at 30, 60, 90 and 120 min respectively.
Blood was gathered from the eye and the supernatant was centrifuged and the serum levels of Total Cholesterol (TC), Triglycerides (TG), Lactate Dehydrogenase (LDH), Creatine Kinase Isoenzyme (CK-MB) were measured using a fully automatic biochemical instrument (Hitachi Automatic Immunochemical Instrument, 7600-010) according to the operating instructions. The levels of Malondialdehyde (MDA), Hydroxyproline (Hyp), Superoxide Dismutase (SOD) in serum were examined by Enzyme Linked Immunosorbent Assay.
Calculation of the heart mass index:
The myocardial tissue was quickly removed, washed with pre-cooled saline and then blotted dry with absorbent paper. Fat, blood vessels and other non- myocardial tissue were removed and the heart weight (HW) was weighed (g).
Heart Index=Cardiac weight (mg)/weight (g)
Protein immunoblot analysis:
For each group, equal amounts of myocardial tissue were weighed, cut up and placed in centrifuge tubes and lysis solution (Radioimmunoprecipitation Assay (RIPA):protease inhibitor:phosphatase inhibitor=50:1:1) was added to extract total myocardial tissue protein and determine the protein concentration by Bicinchoninic Acid (BCA). The macromolecule specimen were isolated by polyacrylamide gel cataphoresis and shifted to polyvinylidene halide membrane, closed with 5 % skimmed milk powder; primary antibodies PI3K, Akt, Glut 4 and Glyceraldehyde 3-Phosphate Dehydrogenase (GAPDH) (all diluted 1:2000) were added and incubated for one night at 4°; Goat Anti- Rabbit IgG H&L/HRP antibody (Abcam, 1:2000) was covered and cultivated for 1 h at 26°-28°, Image J 11.0 software was utilized to dissect the grayscale value of the band. GAPDH was regard as the internal reference.
Real-Time Polymerase Chain Reaction (PCR):
Using RNAiso Plus (TaKaRa, Dalian, China) to withdraw total RNA from myocardial tissue and the concentration was measured spectrophotometrically by NanoDropTM. Using SuperScriptTM IV, the complementary DNA (cDNA) was reverse transcribed into cDNA for qPCR. SYBR® Green was used in the amplification processes with real-time fluorescence quantitative PCR equipment (Bio-rad, USA) (TaKaRa, Dalian, China). There were 35 cycles of the following reaction conditions; 95° for 10 min, 95° for 15 s, 60° for 30 s and 72° for 30 s. The over 2-CT approach was used for relative quantification. Refer to the Oe Biotech designed and GenBank published PI3K, Akt and Glut 4 sequence (Table 1).
| Protein | Sequences |
|---|---|
| PI3K | 5-TTAAACGCTACGGCAACGA-3 5-AGTCTCCTACTGCTGTAGAT-3 |
| AKT | 5-TGAGGTTGCCCACACGCTTA-3 5-GAATCAGCTTCGCATAGGTG-3 |
| Glut 4 | 5-GGGGGCTGACAGGGACCATAAC-3 5-AAGACCCTACGGTAACGAGTAC-3 |
| GAPDH | 5-AGGGCGAGACCCCACTAACA-3 5-AGGGGGGCTAAGCAGTTGGT-3 |
Table 1: PI3K, AKT, GLUT 4 and GAPDH Sequences of Real Time PCR Primers
Hematoxylin and Eosin (H&E) Staining:
Coronal sections of 0.5 cm of myocardial tissue were taken from each group of mice. All specimens required to 4 % paraformaldehyde fixed, paraffin embedded and finally stored at 4°. The tissue was cut into ultra-thin sections of 4.5 μm and the sections were subjected to two 100 %, 50 % xylene transparency for 5 min, two gradients of 100 %, 95 %, 85 %, 75 % ethanol dehydration for 3 min each, distilled water rinsing for 3 min, hematoxylin (Shanghai Biotech, lot no. E607318) staining for 5 min, 70 % ethanol maceration for 30 min The tissues were then stained with eosin (Shanghai Biotech, lot E607318) for 60 s, dehydrated again with gradient ethanol, washed twice with xylene solution and sealed with neutral resin size. Finally, the morphological structure of the tissue was examined under light microscope.
Masson Staining:
The aforesaid paraffin-embedded tissues were stained with hematoxylin, washed and placed in Lincoln red acidic magenta solution for 10 min. They were then dipped in and douched with 2 % acetic acid glacial solution, decomposed for 4 min with 1 % phosphomolybdic acid aqueous solution, stained for 5 min with toluidine blue solution, sealed for 5 min with 0.2 % acetic acid glacial.
Electron microscopic observation:
For each group of mice, myocardial tissue <1 mm in volume was fixed in 2.5 % glutaraldehyde and phosphate buffer for 3 h, rinsed three times in 0.1 M sodium phosphate buffer solution (pH 7.35), fixed for 3 h in 1 % uric acid, rinsed and dehydrated each time by continuous immersion in a low to high concentration of ethanol and pure acetone for 15 min. After dehydration, the tissue was immersed in pure embedding solution for 3 h, baked overnight at 37°, cut into 50-60 nm slices, double dyed with 3 % uranyl acetate and lead citrate, discovered with transmission electron microscopy.
Statistical Analysis:
Data results are available at x͞ ±s. Data were statistically analysed using Statistical Package for the Social Sciences (SPSS) 19.0. Least Significant Difference (LSD) multiple comparisons and one way Analysis of Variance (ANOVA) were utilized to determine statistical significance. Difference was regarded significant while p<0.05.
Results and Discussion
Water intake and food intake were monitored during the experiment. We discovered that the other mouse groups consumed considerably more food and water than the NC group (p<0.05) (fig. 1A and fig. 1B). In comparison with MC group, food and fluid intake were lower dramatically in the Met and OA groups (OAH and OAL groups) (p<0.01). There was none discernible distinction between Met and OA groups. There was no discernible distinction between OAH and OAL groups. From 3rd w of administration, the food consumption of the mice in the Met and OA groups was considerably cut down (p<0.05). The body weight of mice in OAH group was considerably inferior to the MC group from 1st w and lower than that of the Met group from 2nd w onwards (p<0.05) (fig. 1C).
After 8 w of administration, the heart mass index of the Met and OA groups rebounded (p<0.001), with a more significant increase in the Met group (p<0.01) and no prominent distinguish between the OAH and OAL groups. Besides, in comparison with the NC group, the heart mass index of mice in all other groups declined dramatically (p<0.001) (fig. 1D).
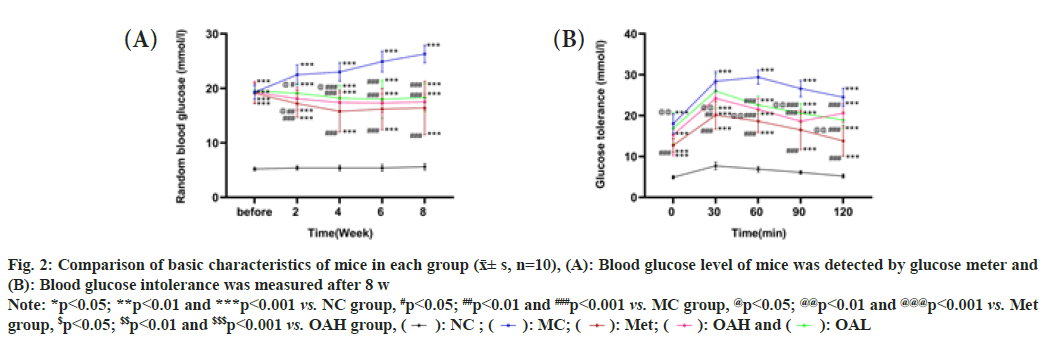
The glucose level of the mice were randomly measured during feeding and we found that the blood glucose levels in all other groups were increased when comparing with NC group (p<0.001). The sugar content of blood in Met and OA groups were lower than MC group after treatment (p<0.001) and there were no remarkable distinguishes between OAH and OAL groups. What’s more, glucose levels in each group dropped within 2 w (fig. 2A), with no significant difference seen after 2 w between Met group and the OA group (p>0.05). The findings of the glucose tolerance test revealed that the mice in the other groups had considerably worse glucose tolerance than the NC group (p<0.001). The glucose tolerance of Met and OA groups was inferior to the MC group 60 min later (p<0.001), and the Met group's glucose tolerance was even worse than the OAL group's (p<0.01). The OAH and OAL groups did not significantly differ from one another (fig. 2B).
Fig. 2: Comparison of basic characteristics of mice in each group (x͞± s, n=10), (A): Blood glucose level of mice was detected by glucose meter and (B): Blood glucose intolerance was measured after 8 w
Note: *p<0.05; **p<0.01 and ***p<0.001 vs. NC group, #p<0.05; ##p<0.01 and ###p<0.001 vs. MC group, @p<0.05; @@p<0.01 and @@@p<0.001 vs. Met
group, $p<0.05; $$p<0.01 and $$$p<0.001 vs. OAH group,  OAL
OAL
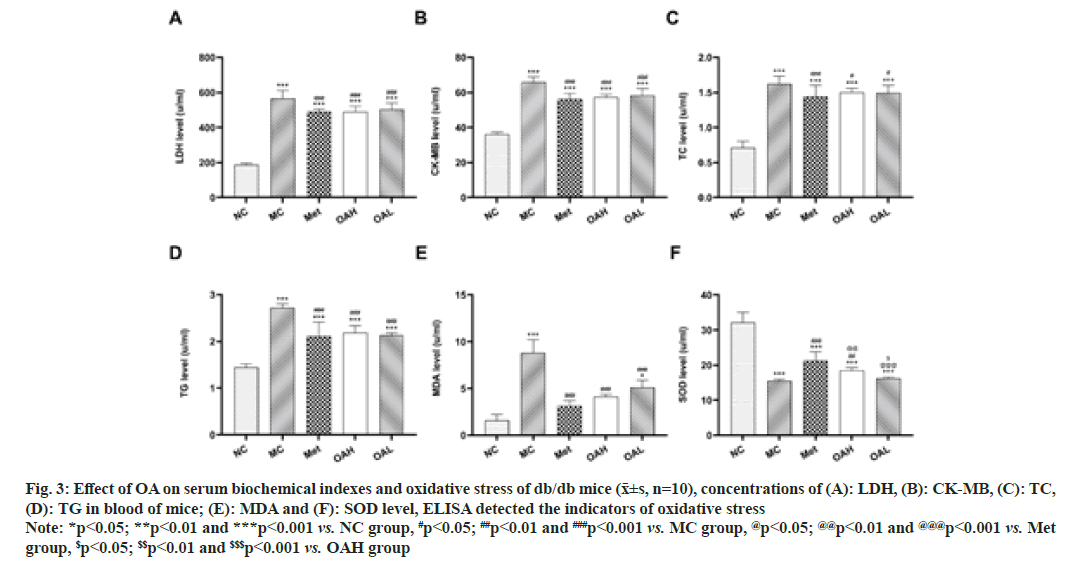
The findings demonstrated that all other groups had observably greater levels of LDH, CK-MB, TC, TG and MDA than NC group (p<0.05). In comparison to MC group, LDH, CK-MB, TC, TG and MDA levels were considerably reduced in the Met and OA groups (p<0.05). Between the Met and OA groups, as well as the OAL and OAH groups, there were no appreciable differences (fig. 3A-fig. 3E). As opposed to the NC group, SOD levels were markedly decreased in the other groups (p<0.001). When comparing to MC group, SOD standards were significantly higher in Met and OAH groups (p<0.01) and much higher in Met group (p<0.01). The OAH and OAL groups did not exhibit any appreciable differences in the findings (fig. 3F).
Fig. 3: Effect of OA on serum biochemical indexes and oxidative stress of db/db mice (x͞±s, n=10), concentrations of (A): LDH, (B): CK-MB, (C): TC,
(D): TG in blood of mice; (E): MDA and (F): SOD level, ELISA detected the indicators of oxidative stress
Note: *p<0.05; **p<0.01 and ***p<0.001 vs. NC group, #p<0.05; ##p<0.01 and ###p<0.001 vs. MC group, @p<0.05; @@p<0.01 and @@@p<0.001 vs. Met
group, $p<0.05; $$p<0.01 and $$$p<0.001 vs. OAH group
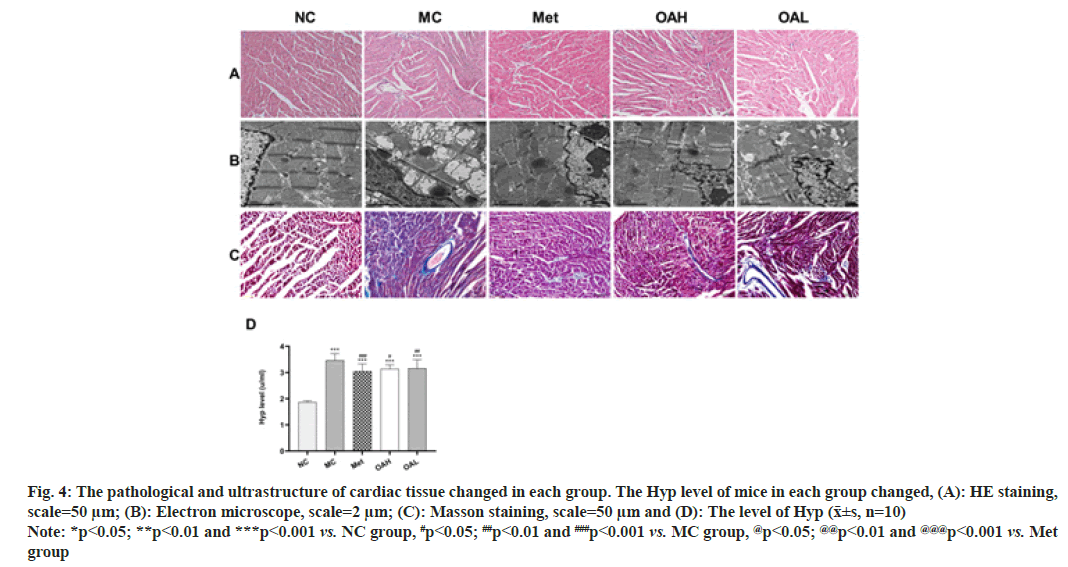
HE staining revealed that the cardiomyocytes in group of NC were neatly and compactly arrayed with a clear hierarchy, while the other groups had obvious damage and a large number of inflammatory cells infiltrating the cardiomyocytes, with the Met and OA groups showing reduced damage and the Met group showing more obvious damage. the OAH group showed reduced damage compared to the OAL group (fig. 4A).
Fig. 4: The pathological and ultrastructure of cardiac tissue changed in each group. The Hyp level of mice in each group changed, (A): HE staining,
scale=50 μm; (B): Electron microscope, scale=2 μm; (C): Masson staining, scale=50 μm and (D): The level of Hyp (x͞±s, n=10)
Note: *p<0.05; **p<0.01 and ***p<0.001 vs. NC group, #p<0.05; ##p<0.01 and ###p<0.001 vs. MC group, @p<0.05; @@p<0.01 and @@@p<0.001 vs. Met
group
Transmission electron microscopy showed that the nuclei of cardiomyocytes in the NC group were oval in shape, with a uniform distribution of material in the nuclei and neatly arranged mitochondrial cristae. In the MC group, a series of morphological changes occurred in the ultrastructure of the cardiomyocytes, mitochondria were swollen and deformed, cristae were absent, and a large number of vacuoles were produced. The myofilaments in the Met and OAH groups were significantly less damaged, with neatly arranged myofilaments, intact mitochondrial structure, a small amount of vacuolation and clear and intact cristae. The Met and OAH groups showed the most pronounced signs (fig. 4B).
Masson staining showed essentially no collagen fibril expression in myocardial tissue in the NC group and a dramatic augment in both interstitial and perivascular fibrosis in the MC group. However, this fibrotic response was less pronounced in the Met and OA groups, with the most significant improvement in the OAH group and a slightly more pronounced improvement in the Met group comparing with the OAL group (fig. 4C). We looked at the Hyp levels in mouse serum, which were considerably greater in the other groups comparing with the NC group (p<0.001), to further investigate this; the Hyp levels were declined in Met and OA groups comparing with MC group, but there were no discernible diversities between Met and OA groups or between OAH and OAL groups (fig. 4D).
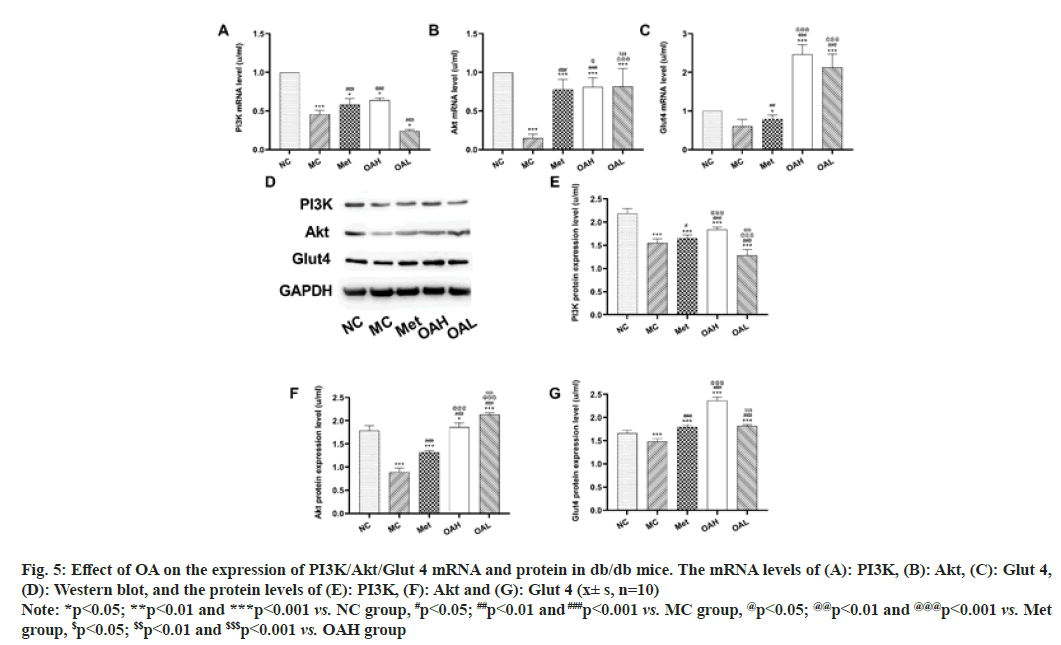
The outcomes revealed that OA could improve the microenvironment of the body and inhibit myocardial tissue damage. Reverse Transcription–Polymerase Chain Reaction (RT-PCR) and Western Blot to observe the mRNA standard and protein expression of PI3K, Akt and Glut 4 in mouse myocardial tissues. We discovered that PI3K, Akt and Glut 4 all showed similar trends. When comparing with mice in NC group, the content of PI3K, Akt and Glut 4 mRNA, and protein were considerably lower in the MC group (p<0.05). The Met group considerably outperformed MC group in terms of PI3K, Akt and Glut 4 protein, and mRNA standards (p<0.05), and OAH group outperformed the Met group even more. Between the Met and OAL groups, the expression levels of Glut 4 did not significantly change (fig. 5). These findings imply that by stimulating the PI3K/Akt signal pathway and encouraging the large expression of Glut 4 downstream, OA may regulate glycolipid metabolism and promote glycogen production.
Fig. 5: Effect of OA on the expression of PI3K/Akt/Glut 4 mRNA and protein in db/db mice. The mRNA levels of (A): PI3K, (B): Akt, (C): Glut 4,
(D): Western blot, and the protein levels of (E): PI3K, (F): Akt and (G): Glut 4 (x± s, n=10)
Note: *p<0.05; **p<0.01 and ***p<0.001 vs. NC group, #p<0.05; ##p<0.01 and ###p<0.001 vs. MC group, @p<0.05; @@p<0.01 and @@@p<0.001 vs. Met
group, $p<0.05; $$p<0.01 and $$$p<0.001 vs. OAH group
One of the most dangerous complications of DM is cardiovascular injury, which has a 2-4 times greater incidence than that of non-diabetic individuals and can cause cardiac failure and even sudden coronary death in patients. The etiology of IR is defined by a high glucose environment in the body due to impaired insulin target organ response and impaired glucose absorption and utilization. Long-term hyperglycemia can harm the heart, liver, kidneys and other tissues and organs. Therefore, early intervention and treatment are essential to the management of T2DM[8]. The tissues of the liver, kidney, cardiovascular system and other organs have been demonstrated to be affected by OA, which has been proven to have hypoglycemic, hypolipidemic, antioxidant and antiatherosclerotic effects[9]. Additionally, Su et al. discovered that OA could reduce the inflammatory response and increase glycogen production in DM mice to ameliorate symptoms like IR. However, it is unclear how OA helped diabetic mice preserve their hearts from damage. For the purpose of preliminarily research the protective role and mechanism of OA on the myocardium of diabetic mice, we used the PI3K/Akt/Glut 4 signaling pathway as our target in this research. We then observed the morphological changes and ultrastructural alterations in the myocardium of Db/db mice to confirm its regulation of PI3K/Akt/Glut 4 signaling pathway.
According to the results of Wang et al.[10], the current study revealed that the blood glucose and glucose tolerance of Db/db mice were distinctly lower than the MC group after the application of OA. This result provided more evidence that OA could successfully cut blood sugar and enhance IR. Additionally, electron microscopic analysis revealed that, compare to the model group, cardiomyocytes in the OA group were relatively neatly arranged and structurally intact, and myocyte myofibrillar lysis was improved. In the meantime, myocardial enzyme profiles in OA mice were restored to normal; myocardial fiber breaks and collagen fibers were decreased, suggesting that OA could delay the onset and progression of myocardial injury in Db/db mice. These findings imply that in Db/ db mice, OA can effectively maintain heart function, lessen the severity of myocardial injury, and increase IR.
One of the primary causes of IR production is oxidative stress, and raising this reaction also increases the risk of islet cell malfunction, decreased glucose tolerance, and aberrant glucolipid metabolism[11]. The OA and Met groups showed significantly lower MDA levels and significantly higher SOD levels, demonstrating that the degree of oxidative partial pressure in Db/db mice was improved after the drug was administered, whereas the MC group showed dramatically higher MDA levels and lower SOD levels compared to the NC group, indicating elevated levels of oxidative stress.
PI3K/Akt signal transduction pathway is a crucial signaling system concerned with IR and in connection with glucose transport, lipid metabolism and protein synthesis regulation. Insulin can act as a metabolic administrative role in physiological processes with the activation of PI3K/Akt pathway[12], which mediates glucose uptake and utilization and exerts hypoglycemic effects. Glut 4 is one of the many signal proteins lower reach of the PI3K/Akt signal transduction route and acts a crucial role in the adjust and control of systemic sugar homeostasis. In addition, Glut 4 stimulates glucose uptake and intracellular gluconeogenesis in adipose tissue, activates the PI3K/Akt signal route in HEPG2 cells and greatly reduces high glucose-induced IR[13]. Endorphins can improve skeletal muscle glucolipid metabolism and IR in KKAy mice by activating PI3K/ Akt and its downstream pathway[14]. In the present study, we found higher standards of PI3K, Akt, Glut 4 protein and mRNA content in OA and Met groups, suggesting that OA may cause insulin to bind to IR and phosphorylate its Substrate (IRS), which indirectly or directly activates the PI3K/Akt signal transduction in myocardial tissue of db/db mice, causing p-Akt to activate a large amount of the downstream signaling molecule Glut 4 expression, accelerating the diffusion of glucose into the cell, thus exerting a role in regulating glucolipid metabolism and promoting glycogen synthesis[15].
In this study, we discovered that the use of OA can effectively motivate the PI3K/Akt/Glut 4 signaling pathway to decline oxidative stress and improve IR. OA increased the expression of PI3K, Akt, Glut 4 proteins and genes involved in the insulin signal transduction route in the myocardial tissue of Db/db mice. Therefore, we propose that PI3K/Akt/Glut 4 pathway inhibition results in aberrant glucose metabolism, oxidative stress and IR, which are all induced by PI3K/Akt/Glut 4 pathway inhibition.
This study showed that OA can lower blood glucose levels in db/db mice's internal environment, improve the myocardial enzyme profile in DM mice, reduce myocardial fibrosis and protect against myocardial injury. Its mechanism involves activating the PI3K/ Akt signaling pathway, increasing Glut 4 protein translocation to speed up glucose transport and glycogen synthesis, lowering oxidative stress, and reducing IR. We established the close connection between the PI3K/Akt/Glut 4 pathway and myocardial injury in this study using only this pathway as the starting point for tests and analysis, but we cannot yet rule out the impact of other pathways on the myocardium of diabetic mice. While db/db mice are the study's experimental subjects, the impact on clinical patients must be deeply tested.
Diabetes mellitus is a chronic condition that requires ongoing therapy. Numerous studies have demonstrated that OA has an anti-diabetic effect and has inherent advantages over many medications used to treat DM. It is also more affordable and safe than insulin injections and frequently prescribed medications, and it can reduce the long-term medical costs of patients. The positive utility and mechanism of OA on myocardial injury in Db/db mice were elucidated in this study using PI3K/Akt/Glut 4 as the target. This study can serve as a solid experimental basis for the introduction of more potent, affordable and safe Chinese medicine ingredients into the field of treating DM.
Funding:
This study was supported by a Grant from the Medical Science Research Project of Hebei Province (No.20221519), Undergraduate and Middle School Students Training Program for Science and Technology Innovation Ability (No:22E50103D).
Author contributions’:
Zhumei Sun and Shuying Han designed this study. Xinyu Shang, Jiashuo Li and Wenji Liang performed the experiments. Fengmei Xing, Jinkun Xi analysed and interpreted the data. Xiaohan Yu and Shuang Zhao checked and proved the authenticity of the raw data. Zhumei Sun and Xinyu Shang wrote the manuscript. All authors read and approved the final manuscript.
Conflict of interests:
The authors declared no conflict of interests.
References
- IDF Diabetes Atlas. 9th edition. International Diabetes Federation; 2019.
- Petersen MC, Shulman GI. Mechanisms of insulin action and insulin resistance. Physiol Rev 2018;98(4):2133-223.
[Crossref] [Google Scholar] [PubMed]
- Sun GP, Yuan Li, Fang XL. Effects of Perilla leaf polysaccharides on oxidative stress and PI3K/AKT/Glut 4 signaling pathway in pancreatic tissue of diabetic mice. Chin Pharm J 2020;31(15):1874-9.
- Wang Y, Chen Y, Nie QQ. Extraction technology and antioxidant activity of oleanolic acid from Ligustrum lucidum. Jiangsu Agric Sci 2017;45 (16):174-6.
- Huang Y, Zhou T, Zhang Y, Huang H, Ma Y, Wu C, et al. Antidiabetic activity of a flavonoid-rich extract from flowers of Wisteria sinensis in type 2 diabetic mice viaactivation of the IRS-1/PI3K/Akt/GLUT4 pathway. J Funct Foods 2021;77:104338.
- Jin JQ. Analysis of 359 cases of adverse reactions caused by hypoglycemic drugs. Chin Pharm J 2014;25(10):920-2.
- Hao BB, Rao JH, Dai JM. Oleanolic acid alleviates hepatic ischemia-reperfusion injury in mice by activating Nrf2/HO-1 signaling pathway. Jiangsu Pharm J 2016;42(12):1317-9.
- Bharadwaja S, Issac PK, Cleta J, Jeganathan R, Chandrakumar SS, Sundaresan S. An in vitro mechanistic approach towards understanding the distinct pathways regulating insulin resistance and adipogenesis by apocynin. J Biosci 2021;46:1-6.
[Google Scholar] [PubMed]
- Wang X, Liu R, Zhang W, Zhang X, Liao N, Wang Z, et al. Oleanolic acid improves hepatic insulin resistance via antioxidant, hypolipidemic and anti-inflammatory effects. Mol Cell Endocrinol 2013;376(1-2):70-80.
[Crossref] [Google Scholar] [PubMed]
- Wang ZL. Protective effect of oleanolic acid on type 2 diabetic rats. Qingdao Univ J; 2019.
- Asmat U, Abad K, Ismail K. Diabetes mellitus and oxidative stress-A concise review. Saudi Pharm J 2016;24(5):547-53.
[Crossref] [Google Scholar] [PubMed]
- Chen J, Liu YR. Effects of curcumin on glucose transport and PI3K/Akt signaling pathway in adipocytes of type 2 diabetic rats. Chin J Comp Med 2019;29(5):90-7.
- Wang J, Wang Y, Liu M, MU Y, Zhang B, Han S. Effects of extract from fermented buckwheat flower and leaf (EFBFL) on myocardial injury in type 2 diabetic db/db mice. Chin Pharmacol Bull 2017;33(7):1026-31.
- Yang D, Yao Q, Zhang H. Effects of visfatin on glucose and lipid metabolism and insulin resistance in skeletal muscle of diabetic KKAy mice through PI3K/Akt/FoxO1 signaling pathway. Chin J Comp Med 2021;31(9):16-23.
- Tong JL, Wang YM, Deng DT. Effects of metformin and aerobic exercise on adiponectin and glucose transporter 4 in skeletal muscle of obese rats. Anhui Med J 2018;22(10):1873-6.