- Corresponding Author:
- D. M. Morkhade
Department of Pharmaceutical Sciences, Rashtrasant Tukadoji Maharaj Nagpur University Campus, Nagpur - 440 033, India
E-mail: dinesh622@rediffmail.com
| Date of Submission | 22 July 2005 |
| Date of Revision | 16 October 2006 |
| Date of Acceptance | 2 April 2007 |
| Indian J Pharm Sci, 2007, 69 (2): 263-268 |
Abstract
A natural gum, damar was investigated as a novel microencapsulating material for sustained drug delivery. Microparticles were prepared by oil-in-oil emulsion solvent evaporation method. Ibuprofen and diltiazem hydrochloride were used as model drugs. Microparticles were evaluated for particle size, encapsulation efficiency and in vitro drug release kinetics. Images of the microparticles were obtained by bright field microscopy. The effect of different gum:drug ratios and solubility of drug on microparticle properties was principally investigated. Gum damar could produce discrete and spherical microparticles with both drugs. With a freely water soluble drug (diltiazem hydrochloride), gum damar produced bigger (45-50 µm) and fast drug releasing microparticles with low encapsulation efficiencies (44-57%). Contrary, with a slightly water soluble drug (ibuprofen), gum damar produced small (24-33 µm) microparticles with better drug encapsulation (85-91%) and sustained drug delivery. The increase in gum:drug ratio showed an increase in particle size, encapsulation efficiency and decrease in drug release rate in all cases. Drug release profiles of all microparticles followed zero order kinetics. In conclusion, gum damar can be used successfully to produce discrete and spherical microparticles of ibuprofen and diltiazem hydrochloride.
Microparticulate systems more often provide controlled drug release patterns and thereby improve the drug therapeutics response and patient compliance. Microparticles can be produced for the protection of core material [1], reduction of gastric irritation [2], conversion of liquid to pseudo-solid [3], cell microencapsulation [4], peptide/protein delivery [5] and for designing pulsatile drug delivery systems [6]. Poly (D,L-lactide) (PLA) and poly (D,L-lactide-co-glycolide) (PLGA) are the commonly studied microencapsulating materials because these are biodegradable and have a broad regulatory approval [7]. But their high cost eventually increases the cost of formulation. Also the copolyesters of acrylic and methacrylic acid [8] (Eudragit® RS and RL) and cellulose derivatives [9] (ethylcellulose, hydroxypropylmethylcellulose (HPMC) and cellulose acetate phthalate) are some of the most frequently investigated microencapsulating materials for therapeutic substances. However, copolyesters of acrylic and methacrylic acid carries positive charge and have propensity to react with the therapeutic substances like heparin8. Ethylcellulose has a dense structure and thus has more probabilities to produce non-spherical and less permeable microparticles. Additionally, the plasticizers or pore formers are often needed with ethyl cellulose to obtain desired film features or the drug release patterns [10]. Cellulose acetate phthalate principally is enteric coating material. Hydrophilic HPMC cannot be used as a wall forming material and is usually incorporated for the specific purposes like mucoadhesion. Although the above details scratch only the surface of information available on the properties of some of the various wall forming materials, it apparently suggests that a range of polymeric materials has certain advantages as well as the own limitations. It therefore seems prerequisite to investigate the novel polymeric materials for the encapsulation of therapeutic substances. Such investigations usually begins with the natural gums because these are readily available, cost effective, eco-friendly, capable of multitude of chemical modifications, potentially degradable and compatible due to their natural origin [11,12]. Perhaps thus the study of natural gums has became a sought after goal of pharmaceutical scientists. However, scarce information is available pertaining to the investigation of natural gums as microencapsulating materials for therapeutic sunstances [13].
Few natural gums like guar gum, gum arabic, tamarind gum, gellan gum and xanthan gum have been investigated for their applicability in designing the microparticulate system. However, these gums have been utilized for specific purpose; xanthan has been used in association with chitosan to control microsphere degradation profile [14], gellan gum along with polyvinyl alcohol has been used to develop novel interpenetrating polymeric network microspheres [15], guar gum has been used for colon targeting [16,17], gum arabic has been used for the covalent binding of primaquine in microspheres [18], tamarind gum has been used with chitosan to prepare the composite microspheres as a support for immobilization of ß-galactosidase [18].
Gum damar (GD) is a natural gum of plant Shorea Wiesneri (family Dipterocarpaceae). It contains about 40% alpha-resin, 22% beta resin, 23% dammarol acid and 2.5% water [19]. It has been used as emulsifier and stabilizer for the production of colour, paints, inks and aromatic emulsions [20]. It has been used in food and cosmetic industries and in the manufacture of paper, wood, varnishes, lacquers, polishes and additive for beverages [20]. It has also been used for the water-resistant coating and in pharmaceutical and dental industries for its strong binding properties [21]. In India, Sal damar is widely utilized as an indigenous system of medicine [20]. These applications of GD project its substantial binding and film forming propensity and fair compatibility with the physiologic environment. In view of the above along with the merits of drug encapsulation, an attempt has been made in the present study to investigate GD as a novel microencapsulating material for therapeutic substances. With respect to the short biological half-life, ibuprofen [22] (half-life: around 2 h) and diltiazem hydrochloride (DLTZ) [23] (half-life: 3-4 h), were selected as model drugs for encapsulation in this study.
Materials and Methods
Gum damar (GD) was received as a gift sample from Innovative Marketing Services, Mumbai. Ibuprofen and diltiazem hydrochloride (DLTZ) were the gift samples from M/s. H-Joules and Co. Ltd., Nagpur. Magnesium stearate was obtained from M/s. S. D. Fine Chemicals Ltd., Mumbai. Acetone and potassium dihydrogen phosphate were procured from M/s. Ranbaxy Fine Chemicals Ltd., New Delhi. Hydrochloric acid, liquid paraffin (heavy) and petroleum ether (60-80) were purchased from M/s. Central Scientific India Ltd., Nagpur. All other materials were of analytical or reagent grade and used as received.
Preparation of GD microparticles
GD microparticles were prepared by oil-in-oil emulsion solvent evaporation technique [24]. In brief, GD (1 or 2 g) was dissolved in 10 ml acetone and drug powder (1 g) was added followed by magnesium stearate (9% w/w of total gum wt.). The above mixture was stirred for 10 minutes on a magnetic stirrer and poured gradually into 160 ml of liquid paraffin (viscosity -165 cps) rotating at a speed of 1100 rpm at 31±1.5° in a 250 ml glass beaker. The above dispersion was maintained at the same temperature and stirring was continued till 9 h. Microparticles were collected by vacuum filtration, washed two times with 12 ml of petroleum ether (60-80) and air dried. The dried microparticles were again washed rapidly with phosphate buffer pH 7.4 to remove the free or loosely bound drug. The microparticles were air dried and stored at 0% relative humidity (RH) before study. The gum:drug ratios used throughout the study were 1:1 and 2:1. The effect of gum:drug ratio on particle size, encapsulation efficiency (%) and in vitro drug release profile of microparticles was principally investigated.
Encapsulation efficiency
About 50 mg of microparticles were dissolved in acetone (0.5 ml) and drug materials were extracted several times in phosphate buffer pH 7.4. The samples after suitable dilutions were analysed by UV Spectrophotometer (Shimadzu UV-1601, Kyoto, Japan) at 221 and 236.4 nm for ibuprofen and DLTZ content respectively. Encapsulation efficiency was determined by the formula, Encapsulation efficiency (%)= (calculated drug content)/(theoretical drug content)×100.
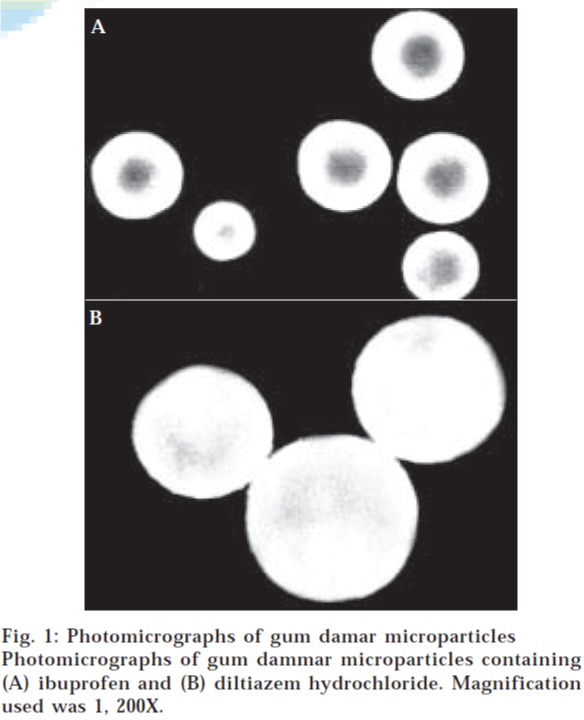
Bright field microscopy and particle size
To examine the sphericity and the discreteness of microparticles, images were taken by the automated Leica LaborLux Leitz S bright field microscope (Germany). The microparticles were examined by the optical microscopy and the mean particle diameter was determined by measuring about 100 particles using 1-mm stage micrometer. The magnification used was 1200X.
In vitro drug release study
The USP XXV type II (paddle type) dissolution test apparatus was used to study the drug release profile of microparticles using the buffer change method. About 50 mg of microparticles were placed in a muslin cloth (mesh 400), which was tied to the paddle, rotating at a speed of 100 rpm. A hydrochloric acid buffer pH 1.2 for initial 2 h and phosphate buffer pH 7.4 for the remaining 8 h was used as a dissolution medium. A 900 ml of dissolution medium maintained at 37±0.5° was employed for each batch of 50 mg microparticles throughout the study. At a periodic interval, 5 ml of the sample was withdrawn and analysed at 221 nm and 236.4 nm for ibuprofen and DLTZ content, respectively.
Drug release kinetics
To study the underlying mechanism of drug release, dissolution data was computed in the light of different kinetic equations as follows25, Qt=Kot--(1), In Qt= In Qo-K1t--(2), Qt=KH.√t--(3), 3√Qo-3√Qt=KHC.t--(4), 3/2[1-(1-Qt)2/3]-Qt=KBC.t-(5), where, Qt is the amount of drug released in time t. Qo is the initial amount of drug released. Ko, K1, KH, KHC and KBC are the release rate constants for Zero order, First order, Higuchi, Hixson-Crowell and Baker-Lonsdale rate equations, respectively. In the equations above, the zero order rate Eqn. 1 describes the systems where the drug release rate is independent of its concentration. The first order Eqn. 2 describes the systems where drug release rate depends on its concentration. Higuchi Eqn. 3 describes the drug release by diffusion from an insoluble matrix. The Hixson-Crowell Eqn. 4 describes systems through which drug releases with the changes in surface dimensions and Baker-Lonsdale Eqn. 5 describes drug release from the spherical matrix.
Results and Discussion
Numerous techniques like the multiorifice-centrifugal [26], wurster coating [27], emulsion congealing [28,29], spray drying [30,31], interfacial polymerization [32], coacervation-phase separation [33] and emulsion solvent evaporation [34-37] have been extensively investigated by the researchers to produce microparticulate systems. Amongst these, emulsion solvent evaporation technique seems more useful because it offers several advantages; it requires only mild conditions such as ambient temperature and constant stirring [35-38]. Thus, GD microparticles were produced by the emulsion solvent evaporation technique. The microparticles prepared without droplet stabilizer showed strong aggregation. Therefore, magnesium stearate in view of its droplet stabilizing potential [39] was incorporated as droplet stabilizer in all formulations. The pilot experimentation suggested; magnesium stearate at 9% w/w of the total gum wt. was efficient in producing the discrete and free flowing GD microparticles. GD microparticles were prepared with slightly water soluble (ibuprofen) as well as freely water soluble (DLTZ) drug material to examine the actual drug release retarding ability of GD. Variations in the stirring speed, concentration of droplet stabilizer, height of stirrer’s shaft in external medium, diameter of the container (containing dispersion medium) and the volumes of internal and external phases were tried and optimized during the initial set up of the formulation. Higher ibuprofen contents in the formulation markedly plasticized GD and thus facilitated aggregation of microparticles, which was not preventable even at the higher concentrations of magnesium stearate (data not shown). Therefore gum:drug ratios of 1:1 and 2:1 were employed in the present study. The effect of gum:drug ratio and drug solubility was investigated in terms of microparticle size, encapsulation efficiency and in vitro drug release patterns.
The increase in gum:drug ratio showed an increase in microparticle size in all cases (Table 1). This can be explained by the fact that increase in gum concentration produces a significant increase in the viscosity of internal phase, which increases the microemulsion droplet size and eventually the microparticle size [40-42]. Also, it has been revealed that increase in polymer content of sample increases the frequency of collision, which may result in fusion of semiformed or visco-elastic particles producing large microparticles [36,43]. Importantly, the solubility property of drug showed more significant effect as compared to the gum:drug ratio on particle size of GD microparticles (Table 1 and fig. 1). Ibuprofen was completely soluble while DLTZ was insoluble in GD solution of acetone. Due to this, ibuprofen could form microspheres with GD, whereas, DLTZ particles since were not soluble acquired GD coatings to form microcapsules. Therefore, the size of ibuprofen microspheres was dependent perhaps on the viscosity of internal phase and the solvent removal time. On the other hand, the particle size of DLTZ microparticles was dependent on the particle size of DLTZ in internal phase. GD however could form the spherical and discrete microparticles with both drugs (fig. 1).
| Gum:drug ratio | Particle size (μm) | Encapsulation efficiency (%) | ||
|---|---|---|---|---|
| Ibuprofen* | Diltiazem HCla | Ibuprofen* | Diltiazem HCla | |
| 1:1 | 24 ± 3 | 45 ± 2 | 85.23 ± 2.01 | 43.72 ± 3.24 |
| 2:1 | 33 ± 2 | 50 ± 2 | 91.07 ± 2.19 | 57.19 ± 3.11 |
*Gum damar microparticles containing ibuprofen, aGum damar microparticles containing diltiazem hydrochloride.
Table 1: The effect of gum:drug ratio and drug type on particle size and encapsulation efficiency of gum damar microparticles
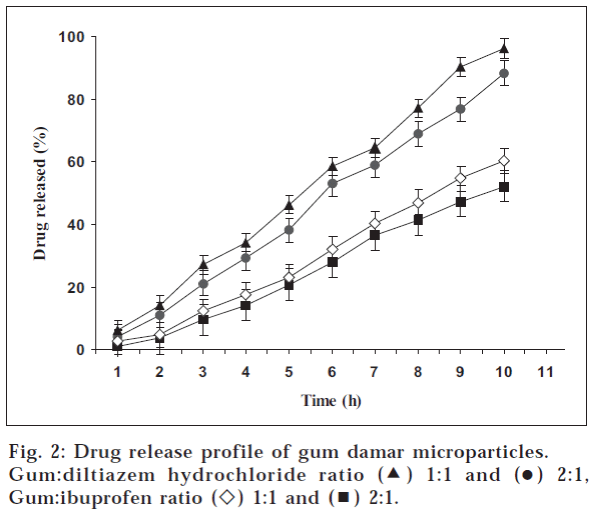
The solubility of drug significantly affected the in vitro drug release profile of GD microparticles. GD microparticles containing ibuprofen showed much slow drug release profiles as compared to that of DLTZ (fig. 2). This may be attributed to the marked difference in the solubility of these drugs at pH 7.4. The saturation solubility of ibuprofen and DLTZ in pH 7.4 is 7.43±0.51 mg/ml and 593.20±3.67 mg/ml, respectively [13,23]. This suggests that DLTZ has greater solubility than ibuprofen in dissolution media. Furthermore, ibuprofen has propensity to plasticize the polymeric materials, thereby making their film more flexible and less permeable [45]. So, the plasticization of GD by ibuprofen also might have contributed to the slow drug release profile of GD microparticles containing ibuprofen. On the other hand, DLTZ is a freely water soluble drug; which might have facilitate DLTZ release from GD microparticles. Notably, the microparticles containing both the drugs showed slow drug release for initial 2 h, which may be due to the low solubility of GD in acidic media. The increase in GD concentrations in the formulation has shown the decrease in drug release rates (fig. 2). GD has low solubility at pH 1.2 and 7.4 and in higher concentrations might have produced the thick wall or dense matrix around the drug particles. For dissolution, drug particles apparently have to travel more distance through such thick wall or matrix as compared to thin ones, which eventually slows down the drug release rate.
The linearity of percentage drug released versus time plots (fig. 2) and correlation coefficient values (Table 2) of all produced microparticles indicate that the drug release from all the formulations follows zero order kinetics. In case of ibuprofen microparticles, drug diffusion as well as polymer erosion might have controlled the drug release pattern whereas in case of DLTZ, solubility characteristics of GD and DLTZ together might have governed and controlled the drug release profiles. In both cases, however, it may be stated that the GD has potential to produce controlled release ibuprofen and DLTZ microparticles.
| Kinetic models | Ibuprofen microparticles Gum:drug ratio | Diltiazem HCL microparticles Gum:drug ratio | ||
|---|---|---|---|---|
| 1:1 | 2:1 | 1:1 | 2:1 | |
| First order | 0.947 | 0.926 | 0.936 | 0.930 |
| Baker-Lonsdale | 0.937 | 0.948 | 0.911 | 0.919 |
| Hixson-Crowell | 0.981 | 0.974 | 0.973 | 0.972 |
| Zero order | 0.996 | 0.996 | 0.998 | 0.998 |
| Higuchi | 0.975 | 0.977 | 0.986 | 0.985 |
Table values represent the correlation coefficients for linearity according to different kinetic equations used to describe the drug release from gum damar microparticles containing ibuprofen and diltiazem hydrochloride.
Table 2: Correlation coefficient values for gum damar microparticles
In the present study, GD was investigated as a novel microencapsulating material for ibuprofen and DLTZ. A GD could develop spherical and discrete microparticles with both drugs. Increase in GD content of internal phase showed an increase in particle size, encapsulation efficiency and decrease in drug release rates of the microparticles. GD in microparticulate system effectively sustained the drug release rates for the longer period of time. Freely water soluble and slightly water soluble drugs exhibited fast and slow release profiles, respectively. The drug release of GD microparticles followed zero order kinetics indicating that GD can be successfully used to prepare controlled release ibuprofen and DLTZ microparticles.
Acknowledgements
Authors are grateful to Dr. N. K. Subhedar, A. J. Sakharkar and D. M. Kokare for their help in the bright field microscopy. Authors kindly acknowledge the Innovative Marketing Services, Mumbai for providing the gift sample of GD. The authors also greatly acknowledge M/s H-Joules & Co. Ltd., Nagpur for providing the gift samples of ibuprofen and DLTZ.
References
- Quong, D. and Neufeld., J. Microencapsul., 1999, 16, 573.
- Calis, S., Bozdag, S., Kas, S. and Hinchal, A.A., J. Control.Release, 2000, 64, 269.
- Kulkarni, A.R., Soppimath, K.S., Aminabhavi, T.M., Deve, A.M. and Mehta, M.H., J. Control. Release, 2000, 63, 97.
- Peirone, M., Ross, C.J., Hortelano, G., Brash, J.L. and Chang, P.L., J.Biomed. Res., 1998, 42, 587.
- Cohen, S., Yoshioka, T., Lucarrelli, M., Hwang, L.H. and Langer, R.,Pharm. Res., 1991, 8, 713.
- Demenueie, A. and Dtnarvana, R., Int. J. Pharm., 1995, 118, 237.
- Tuncay, M., Calis, S., Kas, H.S., Ercan, M.T., Peksoy, I. and Hincal, A.A., Int. J. Pharm., 2000, 195, 179.
- Jiao, Y.Y., Ubrich, N., Hoffart, V., Marchandarvier, M., Vigneron, C.Hoffman, M. and Maincent, P., Drug Develop. Ind. Pharm., 2002, 28, 1033.
- Chowdary, K.P.R. and Ratna, V., Indian J. Pharm Sci., 1990, 10, 279.
- Narisawa, S., Yoshino, H., Hirakawa, Y. and Noda, K., Chem. Pharm. Bull., 1994, 42, 1485.
- Bharadwaj, T.R., Kanwar, M., Lal, R. and Gupta, A.,Develop. Ind. Pharm., 2000, 26, 1025.
- Satturwar, P.M., Fulzele, S.V. and Dorle, A.K., AAPS Pharm. Sci. Tech.,.2003, 4, 55.
- Sipahigil, O. and Dortunc, B., Int J. Pharm., 2001, 228, 119.
- Chellat, F., Tabrizian, M., Dumitriu, S., Chornet, E., Rivard, C.H. and Yahia, L., J. Biomed. Mat. Res., 2000, 53, 592.
- Agnihotri, S.A. and Aminabhavi, T.M., Drug Dev. Ind. Pharm.,2005, 31, 491.
- Chourasia, M.K. and Jain, S.K., J. Drug Target., 2004, 12, 435.
- Chaurasia, M., Chourasia, M.K., Jain, N.K., Jain, A., Soni, V., Gupta, Y. and Jain, S.K., AAPS PharmSciTech., 2006, 7, article 74.
- Zhang, J. and Zeng, J., J. Bioact. Compat.Polym., 2006, 21, 415.
- Jafarsidik, J., Duta Rimba, 1987, 13, 7.
- Conelly, W.T., Economic Botany, 1985, 39, 39.
- Billing, H.J., Oil Colour Trades J., 1994, 3, 666.
- Kristmundsdottir, T. and Ingvarsdottir, K., Drug Develop. Ind.Pharm., 1994, 20, 769.
- Singh, B. and Ahuja, N., Drug Develop. Ind. Pharm., 2002, 28, 431.
- Haznedar, S. and Dortunc, B. Int. J. Pharm., 2004, 131-140.
- Costa, P. and Lobo, J.M.S., Eur. J. Pharm. Sci., 2001, 13, 123.
- Bakan, J.A. Microencapsulation. In: Lachman, L., Lieberman, H.A. and Kanig, J.L., Eds., The Theory and Practice of Industrial Pharmacy, 3rd Edn., Varghese Publishing House, Mumbai, 1990, 412.
- Ichikawa, H., Fukumori, Y. and Adeyeye, C.M., Int. J. Pharm.,1997, 156, 39.
- Adeyeye, C.M. and Price, J.C., Pharm. Res., 1991, 8, 1377.
- Yüksel, N., Aydmli, A., Ozalp, Y. and Ozdemir, N., Drug Res., 2000,57, 187.
- He, P., Davis, S.S. and Illum, L., Int. J. Pharm., 1999, 187, 53.
- Billon, A., Bataille, B., Cassanas, G. and Jacob, M., Int. J. Pharm., 2000, 203, 159.
- Matsunami, Y. and Ichikawa, K., Int. J. Pharm., 2002, 242, 147.
- Sajeev, C., Vinay, G., Archana, R. and Saha, R.N., J.Microencapsul., 2002, 19, 753.
- Bodmeier, R. and McGinity, J.W., J. Microencapsul., 1987, 4, 279.
- Das, S.K., Drug Develop. Ind. Pharm., 1991, 17, 2521.
- Jeffery, H., Davis, S.S. and Ohagan, D.T., Int. J. Pharm., 1991, 77, 169.
- Satturwar, P.M., Mandaogade, P.M. and Dorle, A.K., J. Microencapsul., 2002, 19, 407.
- Kim, B.K., Hwang, S.J., Park, J.B. and Park, H.J., J. Microencapsul., 2002, 19, 811.
- Haznedar, S. and Dortunc, B., Int. J. Pharm., 2004, 269, 131.
- Rodriguez, M., Jose, L., Vilajato, J.C. and Torres, D., J. Control . Release, 1998, 55, 67.
- Lee, J.H., Park, T.G. and Choi, H.K., Int. J. Pharm., 2000, 196, 75.
- Dinarvand, R., Mirfattahi, S. and Atyabi, F., J. Microencapsul., 2002, 19, 73.
- Tamilvanan, S.S.B., J. Microencapsul., 2000, 17, 57.
- Alex, R. and Bodmeier, R., J. Microencapsul., 1990, 7, 347.
- Wu, C. and Mcginity, J.W., Int. J. Pharm., 1999, 177, 15.
 1:1 and (◾) 2:1.
1:1 and (◾) 2:1.