- *Corresponding Author:
- P. Srinivasan
Department of Bioinformatics, Science Block, Alagappa University, Karaikudi-630 004, India
E-mail: sri.bioinformatics@gmail.com
| Date of Submission | 03 June 2013 |
| Date of Revision | 17 February 2014 |
| Date of Acceptance | 23 February 2014 |
| Indian J Pharm Sci 2014;76(2):170-174 |
Abstract
The present study was designed to evaluate the antioxidant and antibacterial activity of methanol extract of Kyllinga nemoralis. Six different in vitro antioxidant assays including 2,2-diphenyl-1-picrylhydrazyl, hydroxyl radical, superoxide anion radical, hydrogen peroxide radical, ferric reducing antioxidant power assay and reducing power were carried out to ensure the scavenging effect of the plant on free radicals. In addition, total antioxidant capacity assay, total phenolic contents, tannins, flavonoids and flavonol contents of the plant were also analysed by the standard protocols. Kyllinga nemoralis exhibited high antioxidant activity on 2,2-diphenyl-1-picrylhydrazyl assay (IC 50 = 90 μg/ml), superoxide radical scavenging assay (IC 50 = 180 μg/ml) and hydrogen peroxide radical scavenging assay (IC 50 = 200 μg/ml), compared with standards. These observations provide comprehensible supporting evidence for the antioxidant potential of the plant extract. Reducing power (IC 50 = 213.16 μg/ml) and hydroxyl radical scavenging activity (IC 50 = 223 μg/ml) of the plant extract was remarkable. The methanol extract of K. nemoralis exhibited significant antimicrobial activity against Gram-positive human pathogenic bacteria. Standard in vitro antioxidant assays assessed the electron donating ability of the plant extract in scavenging free radicals. The inhibitory effect of the plant extract against bacterial pathogens may be due to the presence of phytochemicals. Thus, the results suggest that Kyllinga nemoralis is a potential source of antioxidants and could serve as the base for drug development.
Keywords
Antioxidant activity, antimicrobial activity, Kyllinga nemoralis, phenolics, flavonoids
Free radicals and reactive oxygen species (ROS) including superoxide radical, hydroxyl radical, singlet oxygen and hydrogen peroxide are the by-products arising from numerous physiological and biochemical processes. Overproduction of ROS results in oxidative stress, which is responsible for the development of cell injury, aging, cardiovascular diseases, neurodegenerative diseases, autoimmune disorders, rheumatoid arthritis and cancer [1,2]. Natural antioxidants from plant sources are a group of compounds with quite different chemical structures. They are able to neutralise free radicals by inhibiting and scavenging them, thus providing protection against the onset of many diseases [3]. The two most commonly used synthetic antioxidants such as butylated hydroxyanisole (BHA) and butylated hydroxytoluene (BHT) induce DNA damage due to their toxicity and may have harmful effects on the lungs and liver of human beings. Therefore, screening of plants on the basis of their antioxidant activity is a challenge for the scientists [4].
Kyllinga nemoralis (Hutch and Dalz) (Family; Cyperaceae) is a perennial herb, used in traditional folk medicine to treat many diseases and disorders. Leaves of the plant are used as antivenom, relief of malarial chills, pruritus of the skin, thirst attributable to fever and diabetes. The paste of rhizomes mixed with milk is used internally for worm infection and the rhizome alone is used to treat hepatopathy, splenopathy, fever, tumour and diabetes [5]. The objective of the present study was to evaluate the in vitro antioxidant and antibacterial activity of methanol extract of K. nemoralis. To the best of our knowledge, there is no literature documenting the in vitro antioxidant activity of K. nemoralis. This is the first study where we are reporting the antioxidant and antimicrobial activity of methanol extract of K. nemoralis.
Whole plant of K. nemoralis was collected from Pandalur, Nilgiris District, Tamil Nadu, India. About 100 g of powder was continuously extracted with 500 ml of methanol and the residue was filtered and concentrated in rotary evaporator at 40° under reduced pressure. The obtained crude extract was stored in airtight container at 4° for further use.
The free radical scavenging activity by antioxidants in the methanol extract of K. nemoralis was measured using 2,2-diphenyl-1-picrylhydrazyl (DPPH) [6], reducing power [7], ferric reducing antioxidant power assay [8], superoxide radical scavenging capacity [9], hydroxyl radical scavenging capacity [10] and hydrogen peroxide radical scavenging assay [11]. All the scavenging activities except reducing power and ferric reducing antioxidant power assay were calculated by the following formula: Radical scavenging activity (%) = (Ac−As/As)×100, where as is the absorbance of the control without extract and As is the absorbance of the tested sample with the presence of plant extract. Graph was plotted with the mean values of plant extract and standard. Inhibitory concentration (IC50) of the extract that caused 50% inhibition was calculated from the graph (Table 1).
| Concentration (µg/ml) | Inhibitory activity (%) | |||
|---|---|---|---|---|
| DPPH | Superoxide radical | Hydroxyl radical | Hydrogen peroxide | |
| 50 | 36.48±0.012 | 9.93±0.010 | 22.48±0.030 | 16.98±0.009 |
| 100 | 53.23±0.011 | 20.69±0.010 | 30.64±0.095 | 29.61±0.001 |
| 150 | 67.16±0.019 | 38.31±0.012 | 37.80±0.010 | 32.89±0.013 |
| 200 | 71.47±0.010 | 56.14±0.005 | 44.74±0.011 | 48.58±0.006 |
| 250 | 88.88±0.011 | 61.27±0.010 | 57.24±0.009 | 62.60±0.010 |
| IC50 | 90.94 | 182.10 | 220.49 | 200.23 |
| µg/ml | µg/ml | µg/ml | µg/ml | |
All the values are represented as means±SD (n=3), DPPH=diphenyl-1- picrylhydrazyl
Table 1: Radical scavenging activity of extracts at different concentrations
Total antioxidant capacity of the methanol extract of K. nemoralis was evaluated by the method of Prieto et al. [12] and it was expressed as mg BHT equivalent per gram dry weight. Total flavonoid content was measured based on the method described by Jia et al. [13] and it was expressed as mg/g quercetin equivalent using the following equation based on the calibration curve: Y = 0.002x, R2 = 0.97, where x was the absorbance and y was the quercetin equivalent (mg/g).
The flavonol content of the plant was determined according to the modified method of Kumaran and Karunakaran [14]. Total flavonol content was calculated by using the same equation as for flavonoids. The total phenols were determined by the method of Singleton and Rossi [15] and it was calculated as gallic acid (mg/g) equivalent using the following equation based on the calibration curve: Y=0.005x, R2=0.989, where x was the absorbance and y was the gallic acid equivalent. Total tannin was estimated by the method of Prince and Butler [16] and it was calculated as tannic acid (mg/g) equivalent using the following equation based on the calibration curve: Y=0.037x, R2=0.993, where x was the absorbance and y was the tannic acid equivalent.
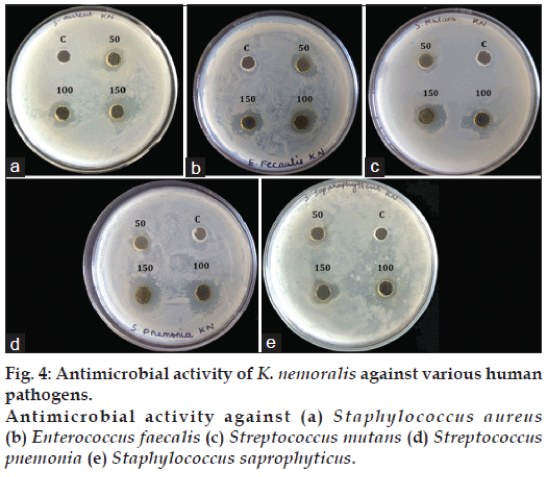
Antimicrobial activity of methanol extract was tested using agar well diffusion method against five different Gram-positive human pathogenic bacteria such as Staphylococcus aureus, S. saparophyticus, Streptococcus mutans, S. pneumoniae and Enterococcus faecalis. All results were expressed as means±SD (standard deviation) of three replicates. Statistical Package for Social Science (SPSS 10.0) was used to analyse the variance (ANOVA). P<0.05 were regarded as significant.
The inhibitory activity of different concentrations of K. nemoralis (50-250 μg) on DPPH ranged from 36.48 to 88.88%, whereas BHT ranged from 29 to 87%. The results showed that the DPPH radical scavenging activity was more efficient than BHT. The IC50 value of K. nemoralis on DPPH radical was found to be 90.94 μg/ml, which was compared with BHT (IC50=109.07 μg/ml). Several studies reported that the DPPH radical is reduced by the hydrogen donating ability of phenolics and flavonoids.
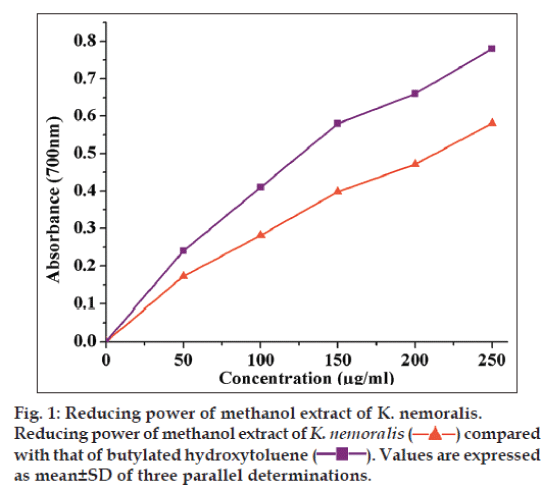
In reducing power assay, the dose-dependent curve of the plant extract at different concentrations (50, 100, 150, 200 and 250 μg/ml) was compared with that of BHT (fig. 1) and the inhibitory activity of BHT was higher than that of K. nemoralis. The absorbance of the plant extract was varied from 0.172±0.005 (50 μg) to 0.581±0.092 (250 μg), whereas BHT was varied from 0.28±0.111 (50 μg) to 0.76±0.091 (250 μg).
The IC50 value of the plant extract was 213.16 μg/ml and that of BHT was 122 μg/ml (Table 2). Reducing power may be due to the presence of polyphenols, which can donate electrons and scavenge free radicals by converting them into more stable products and can terminate the radical chain reaction [17].
| Total flavonoidsa | Total flavonolsa | Total phenolsb | Total tanninsc |
|---|---|---|---|
| 86.36±0.020 | 70.45±0.003 | 51.6±0.007 | 38.10±0.050 |
All the values are represented as means±SD (n=3), DPPH=diphenyl-1- picrylhydrazyl
Table 2: Total flavonoids, flavonols, phenols and tannin contents of plant extract
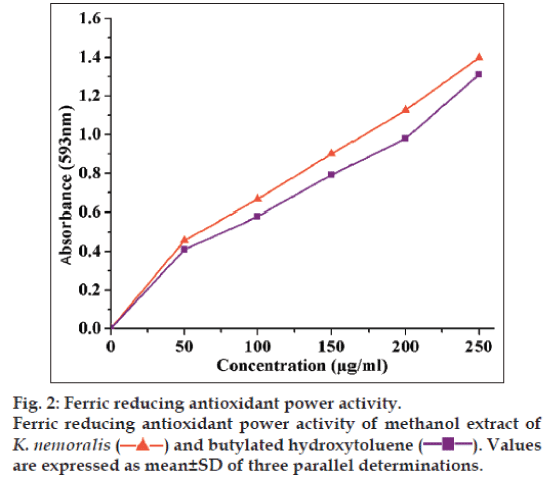
The reducing ability of the plant extract is associated with the antioxidant activity. Linear dose-response curve of the plant extract was compared with the standard BHT (fig. 2). At 250 μg/ml, the highest ferric reducing antioxidant activity was 1.46±0.086, significantly higher when compared with BHT (1.32±0.111). The IC50 value of K. nemoralis was found to be 51.23 μg/ml and BHT was found to be 60.56 μg/ml. Therefore, ferric reducing antioxidant activities of methanol extract of K. nemoralis indicating the ability of plant extract to reduce Fe3+ to Fe2+.
When the concentration of plant extract is from 50 to 250 μg, the superoxide radical scavenging activity was ranged from 9.83 to 61.27% and ascorbic acid ranged from 7.73 to 59.42%. The plant extract exhibited superoxide radical scavenging activity (IC50=182.10 μg/ml), higher than that of ascorbic acid (IC50=207.67 μg/ml). Several studies reported that the presence of free hydroxyl group of phenolic compounds in plants is responsible for the superoxide radical scavenging activity.
The antioxidants in the plant extract can scavenge hydroxyl radical by donating hydrogen atoms and accelerating the conversion of H2O2 to H2. It can act as Fe3+ ion chelators of the system, thereby preventing them from complexing with the deoxyribose [18]. The methanol extract showed scavenging activity on •OH radicals ranged from 22.48 to 57.24% (50-250 μg), whereas ascorbic acid showed scavenging activity that ranged from 20.5 to 84.28% (50-250 μg). The IC50 value of plant extract was found to be 220.49 μg/ml, which was compared with ascorbic acid (IC50=143.72 μg/ml), attributed to their antioxidant activity.
Hydrogen peroxide radical scavenging activity of plant extract at various concentrations of 50, 100, 150, 200 and 250 μg/ml was found to be 16.98, 29.61, 32.89, 48.58 and 62.60%, respectively. The results were comparable to the scavenging activity of ascorbic acid, which was ranged from 30.42 to 55.69% (50-250 μg/ml). The methanol extract possessed the IC50=200.23 μg/ml and ascorbic acid possessed the IC50=200.23 μg/ml.
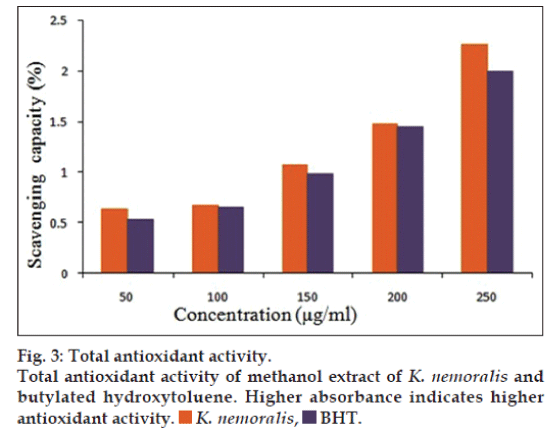
Antioxidant activity of the plant is related to the presence of phenolic compounds such as flavonoids, phenolic acids and tannins. The total antioxidant activity of K. nemoralis at 250 μg/ml was 2.265±0.01, while BHT at the same concentration was found to be 1.995±0.14 (fig. 3). Here, K. nemoralis possessed higher antioxidant activity than the standard, which may be due to the presence of phenolic content in the plant extract.
Polyphenols play an important role in the stabilisation of lipid oxidation and may contribute directly to antioxidative action. It is suggested that polyphenolic compounds have inhibitory effects on mutagenesis and carcinogenesis [19]. Many plant species were tested for their total phenolic content to correlate with their antioxidant activity. Therefore, it is essential to find the total phenolic content of K. nemoralis. In the present study, the plant possessed total phenolic content of 51.6±0.007 mg of gallic acid equivalents/g dry weight of plant extract (Table 2).
Flavonoids are widely distributed group of plant phenolic compounds responsible for the antioxidant activity of the plants. It has been proven to display a wide range of pharmacological and biochemical activities including radical scavenging properties [20]. Methanol extract of K. nemoralis exhibited the highest flavonoid and flavonol contents (86.36±0.020 and 70.45±0.003 mg of quercetin equivalents/g dry weight of plant extract, respectively).
Several studies reported that tannins possess high antioxidant activity than low molecular weight phenolic compounds. The total tannin content of plant extract was found to be 38.10±0.050 mg tannic acid equivalents/g dry weight of plant extract (Table 2). Thus, antioxidant activity of our plant extract may also due to the presence of this phenolic compound.
Natural antimicrobial compounds from plants act as therapeutics that can inhibit the growth of pathogens and have been used to overcome the side effects associated with the synthetic antimicrobial agents. The results of the present study revealed that the methanol extract showed significant inhibitory activity against all the five different human pathogenic bacteria. Among the five different bacteria, the highest antimicrobial activity was observed on S. pneumonia and the least antibacterial activity was observed on S. saprophyticus. The plant extract showed almost similar zone of inhibition against other three organisms (S. aureus, S. mutans and E. faecalis). Methanol control was not found to inhibit any pathogenic bacteria (fig. 4). The inhibitory activity of the plant extract could be attributed to the presence of phenols since it controls the growth and multiplication of bacteria by disturbing the function of bacterial cell membranes.
Thus, the present study concluded that the methanol extract of K. nemoralis is a potential source of natural antioxidants to react against free radicals such as DPPH, superoxide radical, hydrogen peroxide radical and hydroxyl radical. Our findings revealed that the K. nemoralis was found to contain a noticeable amount of total phenols, flavonoids, flavonols and tannins. In addition, it was observed that methanol extract showed a dose-dependent inhibition on the growth of human pathogenic bacteria. Radical scavenging capacity on different free radicals and antimicrobial activity of the plant extract are, may be, due to the presence of phenols and flavonoids. At present, work is in progress for the isolation and characterisation of bioactive compounds from K. nemoralis.
References
- Wu JH, Tung YT, Chyu CF, Chien SC, Wang SY, Chang ST, et al. Antioxidant activity and constituents of extracts from theroot of Garciniamultiflora. J Wood Sci 2008;54:383-9.
- Shodehinde SA, Oboh G. Assessment of antioxidant capacity, proximate composition and inhibitory activity of unripe plantain (Musa paradisiaca) products on Fe2+ and sodium nitroprusside-induced oxidative stress in vitro. J Toxicol Environ Health Sci 2012;4:46-56.
- Arranz S, Perez-Jimenez J, Saura-Calixto F. Antioxidant capacity of walnut (Juglansregia L.): Contribution of oil and defatted matter. Eur Food Res Technol 2008;227:425-31.
- Roy N, Laskar RA, Ismail SK, Kumari D, Ghosh T, Begum NA. A detailed study on the antioxidant activity of the stem bark of DalbergiasissooRoxb., an Indian medicinal plant. Food Chem2011;126:1115-21.
- Raju S, Kavimani S, Rao VU, Reddy SK. Kyllinganemoralis (Hutch and Dalz) (Cyperacea): Ethnobotany, Phytochemistry and Pharmacology. Pharmacogn J 2011;3:7-10.
- Blois MS. Antioxidant determinations by the use of a stable free radical. Nature 1958;181:1199-200.
- Yen GC, Duh PD. Antioxidative properties of methanolic extracts from peanut hulls. J Am Oil ChemSoc 1993;70:383-6
- Benzie IF, Strain JJ. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal Biochem 1996;239:70-6.
- Liu F, Ooi VE, Chang ST. Free radical scavenging activities of mushroom polysaccharide extracts. Life Sci 1997;60:763-71.
- Halliwell B, Gutteridge JM, Aruoma OI. The deoxyribosemethod: A simple “test tube” assay for determination of rate constants for reactions of hydroxyl radicals. Anal Biochem1987;165:215-9.
- Ruch RJ, Cheng SJ, Klaunig JE. Prevention of cytotoxicity and inhibition of intracellular communication by antioxidant catechins isolated from Chinese green tea. Carcinogenesis 1989;10:1003-8.
- Prieto P, Pineda M, Aguilar M. Spectrophotometric quantification of antioxidant capacity through the formation of a phosphomolybdenum complex: Specific application to the determination of vitamin E. Anal Biochem 1999;269:337-41.
- Jia Z, Tang M, Wu J. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem 1999;64:555-9.
- Kumaran A, Karunakaran J. In vitro antioxidant activities of methanol extracts of five Phyllanthusspecies from India. LWTFood SciTechnol 2006;40:344-52.
- Singleton VL, Rossi JA. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J EnolVitic 1965;16:144-58.
- Prince ML, Butler LG. Rapid visual estimation and spectrophotometric determination of tannin content of sorghum grain. J Agric Food Chem 1977;25:1268-73.
- Rajamanikandan S, Sindhu T, Durgapriya D, Sophia D, Ragavendran P, Gopalakrishnan VK. Radical scavenging activity of ethanolic extract of Mollugonudicaulis by in vitro assays. Indian J Pharm Educ Res 2011;45:310-6.
- Awah FM, Uzoegwu PN, Ifeonu P, Oyugi JO, Rutherford J, Yao X, et al. Free radical scavenging activity, phenolic contents and cytotoxicity of selected Nigerian medicinal plants. Food Chem 2012;131:1279-86.
- Gursoy N, Sarikurkcu C, Cengiz M, Solak MH. Antioxidant activities, metal contents, total phenolics and flavonoids of seven Morchellaspecies. Food ChemToxicol 2009;47:2381-8.
- Prasad KN, Yang B, Dong X, Jiang G, Zhang H, Xie H, et al.Flavonoid contents and antioxidant activities from Cinnamomum species. Innov Food SciEmergTechnol 2009;10:627-32.