- *Corresponding Author:
- Kinnari n. Mistry
Ashok and Rita Patel Institute of Integrated Study and Research in Biotechnology and Allied Sciences (ARIBAS), Affiliated to Sardar Patel University, Anand-388 121, India
E-mail: kinnarimistry@aribas.edu.in
| Date of Submission | 13 March 2016 |
| Date of Revision | 29 March 2017 |
| Date of Acceptance | 27 April 2017 |
| Indian J Pharm Sci 2017;79(3):431-437 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
In current study, the phytochemical, antioxidant and cytotoxic effects of methanol extracts of Vitex negundo, Lantana camara, Bauhinia variegata and Bauhinia racemosa were reported. Methanol extract of medicinal plants (V. negundo, L. camara, B. variegata and B. racemosa) were extracted using different extraction methods and then tested for their antioxidant activity by 2,2-diphenylpicrylhydrazyl, superoxide scavenging method, chelation of metal ions and cytotoxic potency using MTT assay on human cancer cell lines. In different extraction methods, highest yield was obtained with hot extraction method. Total phenolic and flavonoid contents were found higher in methanol extract of V. negundo having 173.3±1.54 mg gallic acid equivalent/g DW and 24.41±1.5 mg quercetin equivalent/g DW contents, respectively. Leaves of V. negundo have maximum IC50 value of 73±2.76 µg/ml and 350±1.23 µg/ml in 2,2-diphenylpicrylhydrazyl and iron chelating activity compared to all other methanol extracts. B. racemosa leaves have higher superoxide scavenging effect with IC50 value of 223±1.23 µg/ml. V. negundo and L. camara leaves showed pronounced cytotoxic effect against HELA and KB human cancer cell lines with LD50 value of 222±3.35 and 188.69±1.4 µg/ml, respectively. The present study shows that V. negundo and L. camara may be a probable source of natural antioxidant and as an anticancer drugs.
Keywords
Cancer, superoxide scavenging activity, metal chelating activity, MTT assay, DPPH assay
Cancer is largely an uncontrolled cell proliferative disease in humans and currently there is a significant scienti?c and commercial interest in discovering new antitumor drugs from natural sources[1]. The possibility of using natural products as anticancer agents was acknowledged in 1950s by the U.S. National Cancer Institute (NCI), which was involved in the discovery of new natural products as anticancer agents[2]. This is the second most common disease after cardiac disorders leading to high rates of mortality in the world[3]. It is believed that in the upcoming years the number of cancer patients would increase in the emerging and underdeveloped countries up to 70% to become an issue of serious concern. In India, lung, breast, colon, rectum, stomach and liver cancers are frequently detected[4-6].
Plants have been used as medicines from thousands of years and people relay on plant products for their primary health[7]. These medicines initially were taken in the form of crude drugs such as powders, teas, poultices, tinctures and other natural remediates[7,8]. Secondary metabolites present in plants among which phenolic compounds form the groups of secondary phytochemicals, which occur in high amounts and been found to be responsible for antiinflammatory, antiallergic and anticancer effects[9,10]. Use of herbal drug as an alternate form in health care is increasing and screening of plants for bioactive compounds became an essential source of cancer-related drugs[11].
Hydrogen peroxide (H2O2), superoxide anion (O2) and hydroxyl radical (HO) are some spontaneous oxygen species that are natural by-products of human metabolism. They are hazardous in high amount and interfere with proteins, enzymes, RNA and DNA, leading to tissue injury and degenerative diseases[12,13]. Mammalian body has certain protection mechanisms to combat and reduce oxidative injury. Eating of food products rich in antioxidant phytonutrients especially flavonoids and other polyphenolic compound is beneficial for human health[14,15]. Crude plant extracts with potentially useful biological properties have been used for further detailed pharmacological investigations through in vitro screening methods. The medicinal plants selected for the present investigation, which included leaves of Vitex negundo, Lantana camara, Bauhinia variegata and Bauhinia racemosa have been used in the traditional system because of their possible health endorsing and pharmacological attributes. The present study was carried out to study antioxidant and cytotoxic activity of crude methanol extracts.
Materials and Methods
Selected plant materials were collected from Pune in August 2013 under close supervision and all plant materials were authenticated in the Botany department, Pune.
Preparation of extracts and stock solutions:
The selected plants were washed, shade-dried and chopped in to fine pieces. Extractions were performed using cold maceration and Soxhlet methods. Soxhlet extractions of all selected plant powders were carried out using methanol and all methanol extracts were concentrated in a rotary evaporator at 45º and desiccated under low pressure. The plant powders were extracted thrice with methanol to obtain 3 different extracts, which were found to have no major variation in yield and content of phytoconstituents.
Using cold maceration method, the extracts of all plants were prepared in methanol at room temperature. Dried plant powder of 10 g was mixed with 100 ml solvent in 250 ml flasks and kept on shaker for 24 h and then it was allowed to stand for 4 h to settle down the plant materials. Thereafter, supernatant was filtered and collected. The solvent was evaporated at 45º in rota evaporator (Heildolph, Germany). Yield was calculated for both extraction methods by Eqn. 1: extraction yield (%) = (weight of the dried extract)/(weight of original plant sample)×100. In airtight bottles dried extracts were stored at –20° for further studies. Dimethyl sulfoxide (DMSO) was used to dissolve each extracts and sterilized using 0.22 μm syringe filters (Axiva, Scichem Biotech) for further use.
Total phenolic content:
Total phenolic content was determined using a method reported by Savitree et al.[16] with certain modifications. Standard curve of gallic acid was plotted by mixing Folin-Ciocalteu reagent with methanol solution of gallic acid (r2=0.914). About 0.5 ml of plant extracts dissolved in respective solvent was mixed with 0.1 ml Folin-Ciocalteu reagents (0.5 N) and the solution was kept for 15 min prior to the addition of 2.5 ml of saturated sodium carbonate. Absorbance was measured at 760 nm after 30 min of incubation. Gallic acid equivalents (GAE) mg/g of the dry extract was used to measure total phenolic content in the extract.
Determination of total flavonoid content:
For determination of total flavonoid content, a method described by of Pourmorad et al. was used with minor changes[17]. Briefly, 1 ml of plant fraction and quercetin standard solution was mixed with 0.5 ml of potassium acetate (120 mM), followed by addition of 0.5 ml of aluminium chloride (1.2%) solution. The mixture was kept for an half an hour. At 415 nm the absorbance was measured instantly against prepared blank. Quercetin equivalents (QE) mg/g of the dry extract was used to measure total flavonoids content. A calibration curve was plotted using quercetin (r2= 0.955).
2,2-diphenylpicrylhydrazyl (DPPH) activity:
Scavenging activity on DPPH free radicals by the extract was assessed according to the method reported by Blois[18] with slight modifications. Briefly, 1.0 ml solution of the extract at different concentrations in ethanol was mixed with 1.0 ml of 0.2 mM DPPH in ethanol. The mixture was kept at room temperature for 30 min and the absorbance was measured at 517 nm. Ascorbic acid was used as the standard to compare the result. The DPPH activity of the sample extract was presented as IC50 value and also represented that the concentration of sample essential in reduction of DPPH radicals by 50%. DPPH radical scavenging activity was calculated using the following Eqn., percent scavenging = ((Ac−As)/Ac)×100, where, Ac is the absorption of control, As is the absorption of tested extract.
Superoxide radicals scavenging assay:
The superoxide radical scavenging activity was measured by the reduction of nitro blue tetrazolium (NBT) according to a previously reported method with slight modification[19]. Superoxide radical formed the non-enzymatic PMS/NADH system by the oxidation of NBT to purple formazan[20]. Superoxide radical formed in 3 ml reaction mixture containing sodium phosphate buffer (20 mM pH 7.8) NADH (73 mM), NBT (50 mM), PMS (15 mM) and different concentrations of sample solution were mixed. The solution mixture was kept at 25o for 5 min. The formation of blue colour formazan was followed by observing the raise in absorbance at 560 nm against an appropriate blank. Reaction mixture without plant sample served as blank.
Metal chelating activity:
The metal ion chelating potential of plant extracts was measured by a standard method with slight modification[21]. Briefly, in 1 ml of FeSO4 solution (12.5 μM) was mixed with plant extracts of different concentration and ferrozine (75 μM) was added in this mixture to start the reaction. The mixture was kept for 20 min at room temperature and then the absorbance was determined at 562 nm. Citric acid was used as standard to compare the result. Following equation used to calculate metal chelating activity was calculated using following Eqn., chelating effect (%)=(Ac–As)/ Ac×100, where, Ac is the absorbance of control and As is the absorbance of sample.
Antiproliferative assay:
Hela (cervical cancer) and KB (oral cancer) cell lines were procured from National Centre for Cell Science (NCCS) Pune, used to study antiproliferative activity. Cells were grown in DMEM-F12 (Sigma, Germany), having 10% fetal bovine serum (FBS), 100 U/ml penicillin and 100 μg/ml streptomycin solution. Cells were incubated in CO2 incubator containing 5% CO2 at 37° temperature.
Cytotoxic activity:
In MTT assay, cytotoxicity of methanol extracts of all selected plant extracts were determined on Hela (cervical cancer) and KB (oral cancer) cells depended on the reduction of MTT dye (Sigma, Germany) by calculating the quantity of insoluble formazan produced in live cells. Briefly, in 96-well plate the cells were inoculated in 100 μl at number varying from 10 000- 20 000 cells/well based on cell growth characteristics. Then the 96-well plates were kept at 37° and 5% CO2 humidified incubator for 24 h before addition of plant extracts. Furthermore, methanol extracts dissolved in 2% DMSO were added into a 96-well plate at different concentration (50, 100, 200, 400, 500 μg/ml) by diluting it in medium. After 24 h, in each well 20 μl of MTT reagent (5 mg/ml) was added. After 4 h of incubation 100 μl DMSO was added, which was followed by 1 h of incubation at 37° to dissolve formazan crystals. Using an ELISA plate reader (Promega, USA) absorbance was taken at 570 nm and background wavelength at 750 nm. Following equation was used to calculate the percentage of cell viability: cell viability (%) = (OD sample (mean))/(OD control (mean))×100, where, OD is the optical density; OD of sample: mean absorbance of treated cells and OD of control: mean absorbance of control cells. The dose-response graph was plotted and from the graph (LD50) value was calculated[22,23]. Concentration that required inhibiting 50% of cell growth was used as a stricture for cytotoxicity.
Results and Discussion
In the present study, methanol extracts of four selected plants were evaluated for their antioxidant and anticancer activities using DPPH, superoxide scavenging, metal chelating activity and MTT assay. The antioxidant and anticancer activity of medicinal plants were not similar due to the different array of bioactive components present in the plants that might be responsible for their activity. Table 1 showed the yield of crude methanol extract of different plants by hot and cold extraction method. Table 1 also summarized that maximum yield was obtained by hot extraction method compare to cold extraction method in all plants. The maximum yield was obtained in L. camara followed by V. negundo, B. racemosa and B. variegata. Phenolic compounds are commonly found in both edible and non-edible plants and plant parts. Flavonoids represented a largely well-known group of polyphenolic plant secondary metabolites. These compounds were reported to play a significant role in many activities including free radical scavenging properties[24]. Total phenolic and flavonoid contents in methanol extracts of different plants were exhibited in Table 2. Table 2 also summarized that methanol extract of V. negundo leaves have the maximum total phenolic (173.3±1.54 mg GAE/g dry extract) and flavonoid content (24.41±1.54 mg QE/g dry extract). Phenols and flavonoids are very important constituents of plants and play a significant role in scavenging activity. These components contain hydroxyl group, which do not directly take part in plants antioxidant activity[25]. In protection against oxidative stress, phenolic compounds like flavonoids play important roles. Due to functional hydroxyl groups present in these groups, they showed antioxidant activity by chelating metal ions or scavenging free radicals[26,27]. Metals ion chelation could be essential in the avoidance of free ion generation, which harms target biomolecules[28].
| Plants name | Parts | Solvent | Hot extraction yield (%) | Cold extraction yield (%) |
|---|---|---|---|---|
| Vitexnegundo | Leaves | Methanol | 23.7% | 11.2% |
| Lantana camara | Leaves | Methanol | 45.8% | 8% |
| Bauhinia variegate | Leaves | Methanol | 18% | 16% |
| Bauhinia racemosa | Leaves | Methanol | 18.4% | 12.3% |
Table 1: Extraction Yield (%) of Different Plants Through Hot and Cold Extraction Methods
| Plants | Total phenolic content (mg GAE/g dry extract) |
Total flavonoids content (mg QE/g dry extract) |
|---|---|---|
| Vitexnegundo | 173.3±1.54 | 24.41±1.54 |
| Lantana camara | 144.7±1.34 | 12.44±2.85 |
| Bauhinia variegate | 112.0±2.43 | 20.73±1.43 |
| Bauhinia racemosa | 61.5±1.29 | 21.59±1.61 |
Table 2: The Total Phenolics and Flavonoids Content In Different Methanol Extracts
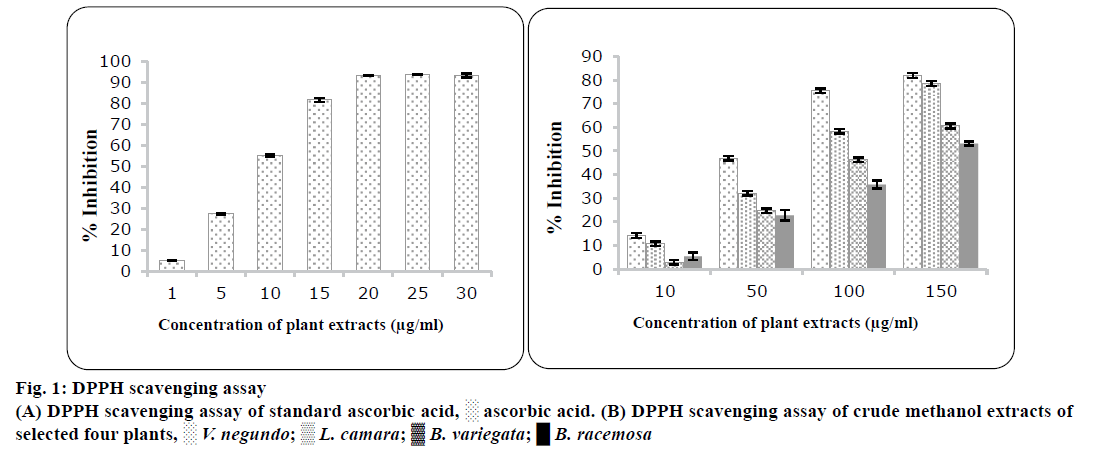
DPPH free radical scavenging activity was carried out with the methanol extracts of all selected plants and the results were presented in figure 1. Different plants for free radicals of DPPH showed significant scavenging activities mentioned in Table 3. The DPPH scavenging activity was increased as concentration was increased. These results provide a comparison of the antioxidant activity directly with ascorbic acid. V. negundo exhibited the highest scavenging activity (IC50; 73±2.76 μg/ml) followed by L. camara, B. variegata and B. racemosa. The influence of antioxidants on scavenging of DPPH radical was supposed to be owed to their hydrogendonating capacity. In the presence of oxygen radicals formed (ROO., HO.,O2•-) are extremely reactive species vary in their lifespan and organic properties.
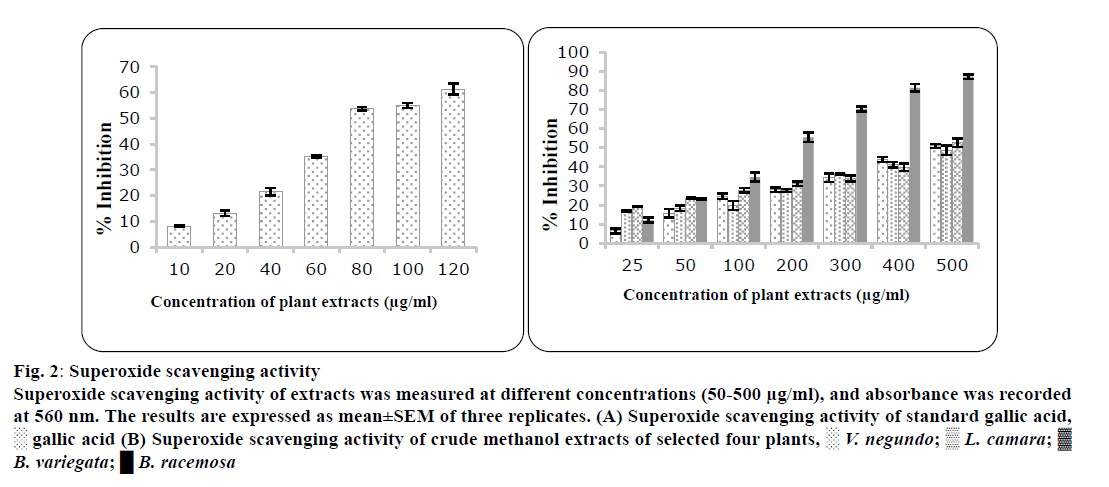
| Plants | DPPH radical scavenging assay activity [IC50(µg/ml)] | Super oxide radical scavenging assay [IC50(µg/ml)] |
Iron chelating activity assay [IC50(µg/ml)] |
|---|---|---|---|
| Vitex negundo | 73±2.76 | 465±1.02 | 350±1.23 |
| Lantana camara | 89.24±2.21 | 487±3.12 | 355±2.32 |
| Bauhinia variegate | 117±3.33 | 485±2.72 | 430±2.44 |
| Bauhinia racemosa | 139±2.66 | 223±1.23 | 512±3.34 |
| Standard | Ascorbic acid 11.40±0.33 |
Gallic acid 93.115±1.23 |
EDTA 288±3.23 |
Table 3: Ic50Value of Different Plants for Various Antioxidant Systems
Superoxide (O2•-) radical is known to be very dangerous to cellular components as a precursor of the extra reactive oxygen species, cause damage to tissue and because of that various diseases occur. B. racemosa leaves (223±3.21 μg/ml) possessed the highest superoxide activity while L. camara showed the lowest superoxide radical scavenging activity (487±3.12 μg/ml). Results are presented in figure 2 and Table 3.
Figure 2: Superoxide scavenging activity
Superoxide scavenging activity of e xtracts was measured at different concentrations (50-500 μg/ml), and absorbance was recorded at 560 nm. The results are expressed as mean±SEM of three replicates. (A) Superoxide scavenging activity of standard gallic acid, ? gallic acid (B) Superoxide scavenging activity of crude methanol extracts of selected four plants, ? V. negundo; ? L. camara;  B. variegata; ? B. racemosa
B. variegata; ? B. racemosa
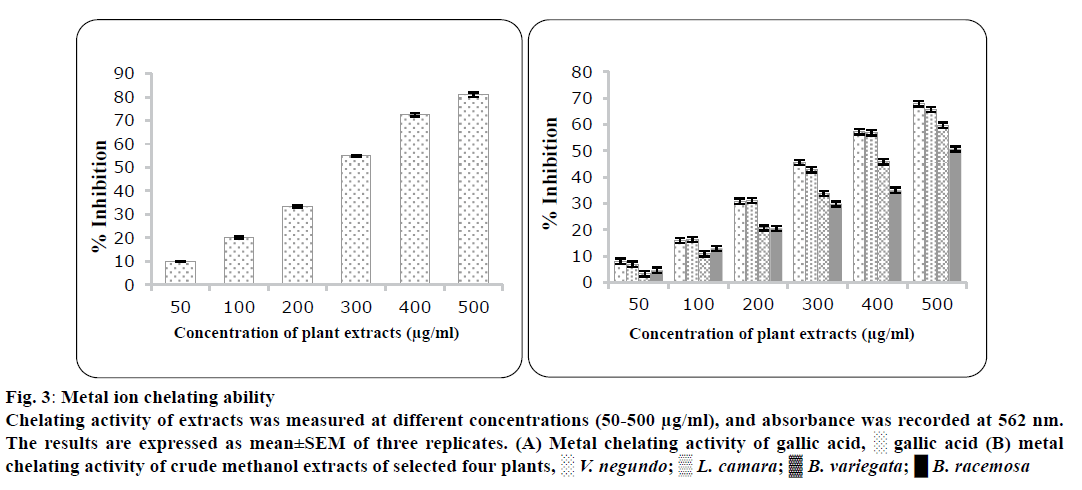
The IC50 values for metal chelating ability of the V. negundo, L. camara, B. variegata and B. racemosa are 350±1.23, 355±2.32, 430±2.44 and 512±3.34 μg/ml, respectively, while that of EDTA was 288±3.23 μg/ml as shown in Table 3. Fe2+- ferrozine complex formation was inhibited by leaf extracts of all four plants as indicated by the formation of colour decrease in dose-dependent manner (figure 3). A main mechanism behind antioxidant action is reduction of the Fe2+ ion, which indicate electron-donating activity of extract. Ferriccyanide complex is converted to ferrous form in the presence of antioxidants in the extracts. Oxidative stress mediated because formation of ROS via Fenton chemistry, which may lead to human diseases. Free radicals are generated due to metal ions leads to lipid peroxidation and DNA damage[29]. These harmful effects of metal ions may be stuck by metal deactivation and chelation. A Ferrozine form complex with Fe2+, but the complex formation is not occurs in the presence of metal chelating agents. Many reports on metal chelation by other plant extracts also authenticate these findings[29,30]. Previous studies reported that oxidized form of metal ion stabilized by chelating agents because they reduce the redox potential therefore they were considered as efficient as antioxidants[31].
Figure 3: Metal ion chelating ability
Chelating activity of extracts was measured at different concentrations (50-500 μg/ml), and absorbance was recorded at 562 nm.
The results are expressed as mean±SEM of three replicates. (A) Metal chelating activity of gallic acid, ? gallic acid (B) metal chelating activity of crude methanol extracts of selected four plants, ? V. negundo; ? L. camara;  B. variegata; ? B. racemosa
B. variegata; ? B. racemosa
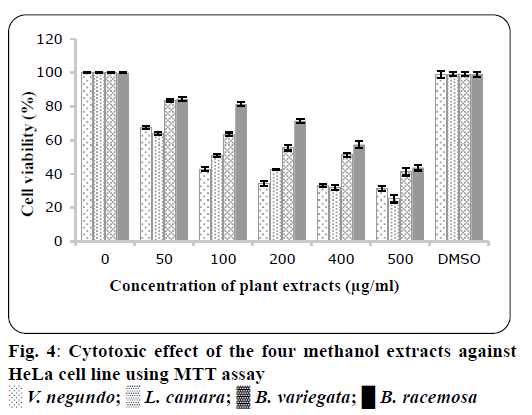
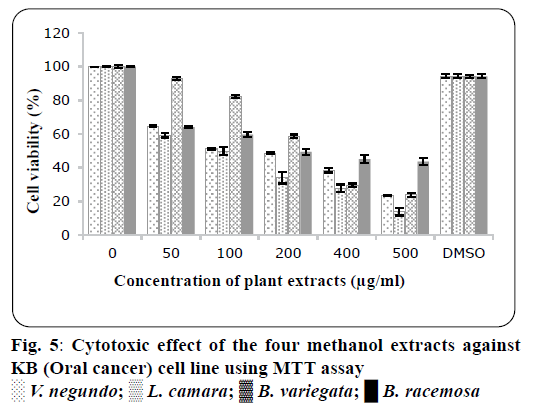
The crude methanol extracts of four plants were tested for cytotoxicity against Hela (cervical cancer cell line) and KB cells (oral cancer cell line). Results showed that crude methanol extracts inhibited the viability of Hela and KB cells with their LD50 values, where shown in Table 4. Crude methanol extracts were tested at concentration range (500, 400, 200, 100 and 50 μg/ml). As shown in figure`s 4 and 5, these extracts show significant concentration-dependent inhibition on propagation and viability of the Hela cells and KB cells. The results exhibited that cytotoxic activity of V. negundo extract on Hela cells was more potent than all other extracts, similarly L. camara was more effective on KB cells. LD50 values for all plants in both cell lines were summarized in Table 4. Many anticancer drugs derived from plants were useful clinically include vinblastine irinotecan, topotecan and paclitaxel[31,32]. Several biological processes and biological interactions occur between flavonoids, phenolic compounds or polyphenols, with enzymes, proteins that make them toxic to the cell or inhibit the growth of cells[33]. Several reports indicated that the cytotoxicity and anticancer properties of natural plants were due to the existence of flavonoid compound and it play a significant function in chemoprevention through showing its effects on transduction of signal and angiogenesis of cells[34,35]. In 1998, Bravo reported that polyphenolic compounds, including flavonoids are important for cancer prevention[35]. It was reported in previous study that methanol extracts from V. negundo had high concentrations of polyphenolic compounds[36]. This could have contributed to the cytotoxicity of the cells to methanol V. negundo and L. camara extract.
| Plants | LD50value on HELA cells (µg/ml) | LD50 value on KB cells (µg/ml) |
|---|---|---|
| Vitex negundo | 222±3.35 µg/ml | 247±2.33 µg/ml |
| Lantana camara | 229±4.23 µg/ml | 188.6±1.4 µg/ml |
| Bauhinia variegate | 371.8±3.12 µg/ml | 299.23±3.55 µg/ml |
| Bauhinia racemosa | 442.61±2.1 µg/ml | 334.65±2.34 µg/ml |
Table 4: Ld50value of Different Plants on Hela And Kb Cancerous Cell Lines
The current study showed that the presence of phytochemicals in selected plant leaf extracts has effective cytotoxic activity against cervical and oral cancer cells. The free radical scavenging ability probably is one of the mechanisms by which herbal medicines exhibit higher antioxidant activity. In addition crude methanol extracts have potential to fight against oxidative damage because of its DPPH activity, superoxide scavenging activity and iron binding ability. Knowing the exact compounds responsible for the plant’s anticancer activity will be useful in finding new anticancer agents. Lastly, in vivo and clinical evaluations need to be performed for the successful commercialization of this plant to benefit both the food and pharmaceutical industries.
Acknowledgements
Authors are grateful to Charotar Vidya Mandal (CVM) and ARIBAS Vallabh Vidyanagar, Gujarat for providing platform for this research work. We are also thankful to P.D. Patel Institute of Applied Sciences, Charusat University for providing us facility for our research work. Authors acknowledge the support of Dr. Subhash Sadhu Deokule, Head of Botany department, Pune in authentication of plant materials..
Conflict of interest:
All authors declare no conflict of interests.
Financial support:
Nil.
References
- Kinghorn A, Farnsworth N, Soejarto D, Cordell G, Swanson S, Pezzuto J, et al. Novel strategies for the discovery of plant-derived anticancer agents. Pharm Biol 2003;41:53-67.
- Cragg GM, Newman DJ. A tale of two tumor targets: topoisomerase I and tubulin. The Wall and Wani contribution to cancer chemotherapy. J Nat Prod 2004;67:232-44.
- Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun M. Cancer statistics, 2007. CA Cancer J Clin 2007;57:43-66.
- Nandakumar A, Kishor RG, Chandra KA, Poonamalle BP, Gupta PC, Gangadharan P, et al. Concurrent chemoradiation for cancer of the cervix: Results of a multi-institutional study from the setting of a developing country (India). J Glob Onco 2015;1:11-22.
- Rao D, Ganesh B. Estimate of cancer incidence in India in 1991. Indian J Cancer 1998;35:10-18.
- Murthy N, Mathew A. Cancer epidemiology, prevention and control. CurrSci 2004;86:518-27.
- Samuelson G. Drugs of Natural Origin: A Textbook of Pharmacognosy. 4th ed. Stockholm: Apotekarsocieteten; 1999.
- Balick MJ. Plants, people, and culture: the science of ethnobotany. New York: Scientific American Library; 1996.
- Afolayan A, Jimoh F. Nutritional quality of some wild leafy vegetables in South Africa. Int J Food SciNutr 2009;60:424-31.
- Middleton E, Kandaswami C, Theoharides TC. The effects of plant flavonoids on mammalian cells: implications for inflammation, heart disease, and cancer. Pharmacol Rev 2000;52:673-751.
- Alpinar K, Oezyuerek M, Kolak U, Guclu K, Aras C, Altun M, et al. Antioxidant capacities of some food plants wildly grown in Ayvalik of Turkey. Food Sci Tech Res 2009;15:59-64.
- Jung HA, Park JC, Chung HY, Kim J, Choi JS. Antioxidant flavonoids and chlorogenic acid from the leaves of Eriobotrya japonica. Arch Pharm Res 1999;22:213-18.
- Pietta P, Simonetti P, Mauri P. Antioxidant activity of selected medicinal plants. J Agric Food Chem 1998;46:4487-90.
- Cao G, Booth SL, Sadowski JA, Prior R. Increases in human plasma antioxidant capacity after consumption of controlled diets high in fruit and vegetables. Am J ClinNutr 1998;68:1081-87.
- Pulido R, Bravo L, Saura-Calixto F. Antioxidant activity of dietary polyphenols as determined by a modified ferric reducing/antioxidant power assay. J Agric Food Chem 2000;48:3396-402.
- Mongkolsilp S, Pongbupakit I, Sae-Lee N, Sitthithaworn W. Radical scavenging activity and total phenolic content of medicinal plants used in primary health care. SWU J Pharm Sci 2004;9:32-35.
- Pourmorad F, Hosseinimehr S, Shahabimajd N. Antioxidant activity, phenol and flavonoid contents of some selected Iranian medicinal plants. Afr J Biotechnol 2006;5:1142-45.
- Blois MS. Antioxidant determinations by the use of a stable free radical. Nature 1958;181:1199-200.
- Beauchamp C, Fridovich I. Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 1971;44:276-87.
- Martinez CA, Loureiro ME, Oliva MA, Maestri M. Differential responses of superoxide dismutase in freezing resistant Solanumcurtilobumand freezing sensitive Solanumtuberosumsubjected to oxidative and water stress. Plant Sci 2001;160:505-15.
- Soler?Rivas C, Espín JC, Wichers HJ. An easy and fast test to compare total free radical scavenger capacity of foodstuffs. Phytochem Anal 2000;11:330-38.
- Freshney RI. Culture of Animal Cells: A Manual of Basic Technique and Specialized Applications. 6th ed. New Jersey: John Wiley & Sons, Inc.; 2000. p. 383-432.
- Park JG, Lee SK, Hong IG, Kim HS, Lim KH, Choe KJ, et al. MDR1 gene expression: its effect on drug resistance to doxorubicin in human hepatocellular carcinoma cell lines. J Nat Cancer Inst 1994;86:700-5.
- Kumar S, Mishra A, Pandey AK. Antioxidant mediated protective effect of Partheniumhysterophorusagainst oxidative damage using in vitromodels. BMC Complement Altern Med 2013;13:120.
- Chuanphongpanich S, Suttajit M, Phanichphant S, Buddhasukh D, Sirithunyalug B. Antioxidant capacity of broccoli seeds grown in Thailand. Chiang Mai J Sci 2006;33:117-22.
- Kumar S, Pandey AK. Phenolic content, reducing power and membrane protective activities of Solanumxanthocarpumroot extracts. Vegetos 2013;26:301-7.
- Leopoldini M, Russo N, Chiodo S, Toscano M. Iron chelation by the powerful antioxidant flavonoid quercetin. J Agric Food Chem 2006;54:6343-51.
- Hider R, Hall A. Clinically Useful Chelators of Tripositive Elements. Prog Med Chem 1991;28:41-173.
- Singh R, Arora S. Attenuation of free radicals by acetone extract/fractions of Acacia niloti-ca (l.) Willd. Ex del. J Chin Clin Med 2007;2:196-204.
- Gordon M. The mechanism of antioxidant action in vitro. In: Hudson BJF, editor. Food Antioxidants. Netherlands: Springer; 1990. p. 1-18.
- Cragg GM, Newman DJ. Plants as a source of anticancer agents. J Ethnopharmacol 2005;100:72-9.
- Murray B, Carter R, Imrie C, Evans S, O’Suilleabhain C. Diclofenac reduces the incidence of acute pancreatitis after endoscopic retrograde cholangiopancreatography. Gastroenterology 2003;124:1786-91.
- Weber G, Shen F, Prajda N, Yeh YA, Yang H, Herenyiova M, et al. Increased signal transduction activity and down-regulation in human cancer cells. Anticancer Res 1995;16:3271-82.
- Fotsis T, Pepper MS, Aktas E, Breit S, Rasku S, Adlercreutz H, et al. Flavonoids, dietary-derived inhibitors of cell proliferation and in vitroangiogenesis. Cancer Res 1997;57:2916-21.
- Bravo L. Polyphenols: chemistry, dietary sources, metabolism, and nutritional significance. Nutr Rev. 1998;56:317-33.
- Guha G, Rajkumar V, Kumar RA. Polyphenolic constituents of methanolic and aqueous extracts of Vitexnegundo render protection to Hep3B cells against oxidative cytotoxicity. Food ChemToxicol 2010;48:2133-38.