- Corresponding Author:
- K. R. S. Sambasiva Rao
Achrya Nagarjuna University, Nagarjunanagar-522 510, Guntur
E-mail: krssrao@yahoo.com
| Date of Submission | 10 December 2010 |
| Date of Revision | 28 February 2011 |
| Date of Acceptance | 24 March 2011 |
| Indian J Pharm Sci, 2011, 73 (1): 243-247 |
Abstract
Solid dispersions of mefanamic acid with a water-soluble polymer polyvinyl pyrrolidine and a super disintegrant, primojel were prepared by common solvent and solvent evaporation methods employing methanol as the solvent. The dissolution rate and dissolution efficiency of the prepared solid dispersions were evaluated in comparison to the corresponding pure drug. Solid dispersions of mefenamic acid showed a marked enhancement in dissolution rate and dissolution efficiency. At 1:4 ratio of mefenamic acid-primojel a 2.61 fold increase in the dissolution rate of mefenamic acid was observed with solid dispersion. The solid dispersions in combined carriers gave much higher rates of dissolution than super disintegrants alone. Mefanamic acid-primojel-polyvinyl pyrrolidine (1:3.2:0.8) solid dispersion gave a 4.11 fold increase in the dissolution rate of mefenamic acid. Super disintegrants alone or in combination with polyvinyl pyrrolidine could be used to enhance the dissolution rate of mefenamic acid.
Keywords
Dissolution rate, mefenamic acid, polyvinyl pyrrolidone, solid dispersions
Mefenamic acid (MA), an anthranilic acid derivative, is a non-steroidal antiinflammatory drig (NSAID) [1] and short (2 h) plasma half-life drug. It is used as antipyretic analgesic [2], and antirheumatic [3] for the treatment of headache, dental pain, postoperative and postpartum pain, dysmenorrhea and osteoarthritis. The usual dose orally is 500 mg three times daily. Mefenamic acid is absorbed from the gastrointestinal tract. Peak plasma concentrations occur about 2 to 4 h after ingestion. Most of the NSAIDs belong to class II category under biopharmaceutical classification system, since they are inherently highly permeable through biological membranes, but exhibit low aqueous solubility. Rate of absorption and/or extent of bioavailability for such hydrophobic drugs are controlled by rate of dissolution in gastrointestinal fluids [4]. Solid dispersions are one of the most promising strategies to improve the oral bioavailability of poorly water-soluble drugs. By reducing drug particle size to the absolute minimum, and hence improving drug wettability, bioavailability may be significantly improved [5]. The present study aims at enhancing the dissolution rate and bioavailability of MA with solid dispersions [6] by employing common solvent and solvent evaporation methods. Water dispersible super disintegrants, a new class of tablet excipients were evaluated as carriers, alone and in combination with polyvinyl pyrrolidine (PVP), for enhancing the dissolution rate and bioavailability of MA.
MA was a gift sample from M/s. Sigma Laboratories, Mumbai, India, methanol (Qualigens) and polyvinyl pyrrolidone (PVP K30) was a gift sample from M/s. Sun Pharma Ind. Ltd., Mumbai, India. All other materials used were of pharmacopoeial grade and were procured from commercial sources.
Solid Dispersions of mefenamic Acid were prepared by common solvent method employing methanol as solvent. The required quantities of MA and PVP were weighed and dissolved in the corresponding solvent in a round bottom flask to get a clear solution. The solvent was then removed by evaporation under reduced pressure (vacuum) at 60° with constant mixing. The mass obtained was crushed, pulverized and shifted through mesh no. 100. In each case solid dispersions were prepared in the ratio of drug carrier namely 8:2.
Solid dispersions of MA in superdisintegrate primojel were prepared by solvent evaporation method. The required quantities of MA were dissolved in methanol to get a clear solution in a dry mortar. The superdisintegrate primojel (PJ, passed through 120 No. mesh) was then added to clear drug solution and dispersed. The solvent was removed by continuous titration until a dry mass was obtained. The mass obtained was further dried at 50° for 4 h in an oven. The product was crushed, pulverized and shifted through mesh no. 100. In each case solid dispersions in the superdisintegrate primojel were prepared at three different ratios of MA, PJ namely 1:1, 1:2 and 1:4, respectively.
The required quantities of MA and water-soluble carrier (PVP) were dissolved in the solvent to get a clear solution in a dry mortar. The superdisintegrate (PJ) was then added to the drug solution and dispersed. The solvent was then evaporated by continuous titration until a dry mass was obtained. The mass obtained was further dried at 50° for 4 h in an oven. The product was crushed, pulverized and shifted through mesh no. 100. Various solid dispersions prepared with their composition are listed in Table 1.
| Composition | |||
|---|---|---|---|
| Drug | Carriers | SD code | Drug:carriers |
| Mefenamic acid (8) | PVP (2) | MA-PVP | 8:2 |
| Mefenamic acid (1) | PJ (1) | MA-PJ | 1:1 |
| Mefenamic acid (1) | PJ (2) | MA-PJ | 1:2 |
| Mefenamic acid (1) | PJ (4) | MA-PJ | 1:4 |
| Mefenamic acid (1) | PJ (3.2) PVP (0.8) | MA-PJ-PVP | 1:3.2:0.8 |
PVP: Polymer poly vinyl pyrrolidine, PJ: Primojel, SD Code: Solid dispersions code (Dissolution rate or ratio of the prepared formulation of drug and carrier).
Table 1: Composition of various solid dispersions prepared
A spectrophotmetric method based on the measurement of absorbance at 279 nm in phosphate buffer pH 7.4 was used in the present study for the estimation of mefenamic acid. The method was validated for reproducibility, accuracy, precision and linearity by analyzing six individually weighed samples of MA. The stock solution of MA was subsequently diluted to a series of dilution containing 5, 10, 15 and 20 μg/ml of solution, using phosphate buffer of pH 7.4. The absorbance of these solutions was measured in UV/Vis spectrophotometer (ELICO SL-159). The method obeyed Beer’s law in the concentration of 0-20 μg/ml.
From each batch, 4 samples of 50 mg each were taken and analyzed for the drug mefenamic acid. Fifty milligrans of dispersions were weighed into a 100 ml volumetric flask. Methanol was added and the contents were mixed thoroughly to dissolve the drug from the dispersion. The solution was then filtered and collected carefully into another 100 ml volumetric flask. The solution was made up to volume with the solvent. The solution was suitably diluted with phosphate buffer of pH 7.4 and assayed at 279 nm for MA. The results are given in Table 2.
| SD code | Percent mefenamic acid content (x ± SD) |
|---|---|
| MA-PVP, (8:2) | 79.5 ± 0.74 (0.93) |
| MA-PJ, (1:1) | 49.4 ± 0.12 (0.25) |
| MA-PJ, (1:2) | 33.0 ± 0.19 (0.58) |
| MA-PJ, (1:4) | 19.8 ± 0.22 (1.11) |
| MA-PJ-PVP (1:3.2:0.8) | 19.2 ± 0.32 (1.26) |
Table 2: Mefenamic acid content of various solid dispersions prepared
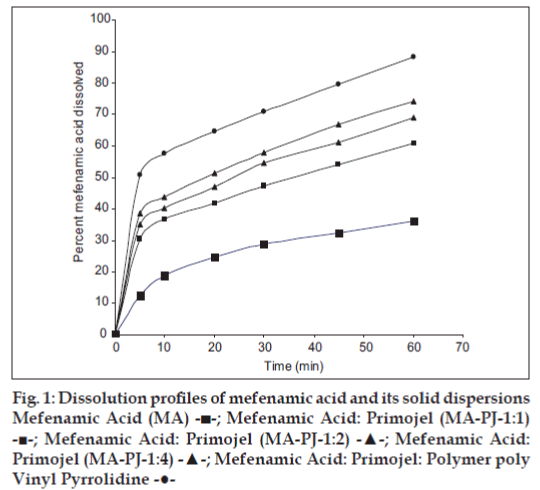
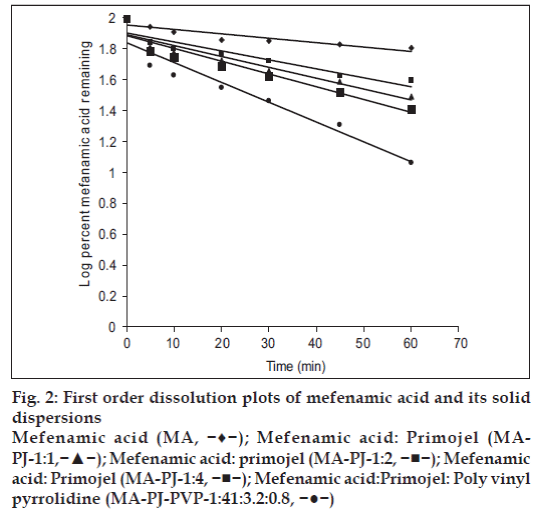
Dissolution rate of MA was studied using an USP XXIII six station dissolution rate test apparatus (Electro Lab). Paddle stirrer at a speed of 50 rpm and temperature of 37±1° were used in each test. Drug or solid dispersion of drug equivalent to 100 mg of MA was used in each dissolution rate test. Samples of dissolution medium i.e., phosphate buffer pH 7.4 (5 ml) were withdrawn through a filter (0.45 μ) at different time intervals, suitably diluted, and assayed for MA. The dissolution experiments were conducted in triplicate. The results are given in Table 3 and fig. 1. Dissolution rates of MA and its solid dispersions followed first order kinetics (Table 4 and fig. 2). Dissolution parameters such as T50, DE30, K1, Percent of MA dissolved in 10 minutes are given in Table 5.
| Time (min) | Percent MA dissolved (x ± SD, n=3) | ||||
|---|---|---|---|---|---|
| MA | MA-PJ (1:1) | MA-PJ (1:2) | MA-PJ (1:4) | MA-PJ-PVP (1:3.2:0.8) | |
| 5 | 12.39 ± 0.63 | 30.42 ± 1.04 | 35.11 ± 1.66 | 38.58 ± 1.48 | 50.82 ± 1.11 |
| 10 | 18.66 ± 0.38 | 36.72 ± 1.70 | 40.17 ± 2.82 | 43.77 ± 0.66 | 57.62 ± 1.67 |
| 20 | 24.2 ± 0.56 | 41.91 ± 1.33 | 47.11 ± 1.11 | 51.31 ± 1.25 | 64.65 ± 1.65 |
| 30 | 28.83 ± 0.69 | 47.26 ± 1.82 | 54.53 ± 2.22 | 57.99 ± 1.32 | 70.86 ± 1.33 |
| 45 | 32.25 ± 0.71 | 54.04 ± 2.23 | 61.08 ± 1.13 | 66.75 ± 1.48 | 79.51 ± 1.49 |
| 60 | 36.05 ± 0.54 | 60.84 ± 1.69 | 69.12 ± 1.06 | 74.07 ± 1.75 | 88.42 ± 1.36 |
Table 3: Dissolution profiles of mefenamic acid solid dispersions
| Solid dispersion | Correlation coefficient (r) value | ||
|---|---|---|---|
| Zero order | First order | Hixson-crowell | |
| Pure drug | 0.9875 | 0.9940 | 0.9920 |
| MA-PVP, (8:2) | 0.8763 | 0.9075 | 0.9061 |
| MA-PJ, (1:1) | 0.8615 | 0.9282 | 0.9090 |
| MA-PJ, (1:2) | 0.8652 | 0.9430 | 0.9220 |
| MA-PJ, (1:4) | 0.8601 | 0.9451 | 0.9262 |
| MA–PJ-PVP, (1:3.2:0.8) | 0.8180 | 0.9260 | 0.9231 |
Table 4: The correlation coefficient (r) values in the analysis of dissolution data of mefenamic acid solid dispersions as per zero order, first order and hixson crowell cube root models
| Solid dispersion | Dissolution parameter | |||
|---|---|---|---|---|
| T50(min) | % Dissolved in10 min | DE30(%) | K1(min–1) | |
| Mefenamic acid | >60 | 10.63 | 19.60 | 0.0072 |
| MA-PJ, (1:1) | 36 | 31.85 | 36.09 | 0.0128 |
| MA-PJ, (1:2) | 24 | 35.61 | 40.68 | 0.0164 |
| MA-PJ, (1:4) | 14 | 38.88 | 44.14 | 0.0188 |
| MA-PJ-PVP, | 4.57 | 57.62 | 56.25 | 0.0296 |
| MA-PVP, (8:2) | >60 | 26.20 | 26.81 | 0.0087 |
DE30: Dissolution efficiency index per 30 min, K1: Hixson-Crowell constant/min
Table 5: Dissolution parameters of mefenamic acid and its solid dispersions in superdis integrants
Figure 2: First order dissolution plots of mefenamic acid and its solid
dispersions
Mefenamic acid (MA, −♦−); Mefenamic acid: Primojel (MAPJ-1:1,−▲−); Mefenamic acid: primojel (MA-PJ-1:2, −■−); Mefenamic acid: Primojel (MA-PJ-1:4, −■−); Mefenamic acid:Primojel: Poly vinyl pyrrolidine (MA-PJ-PVP-1:41:3.2:0.8, −●−)
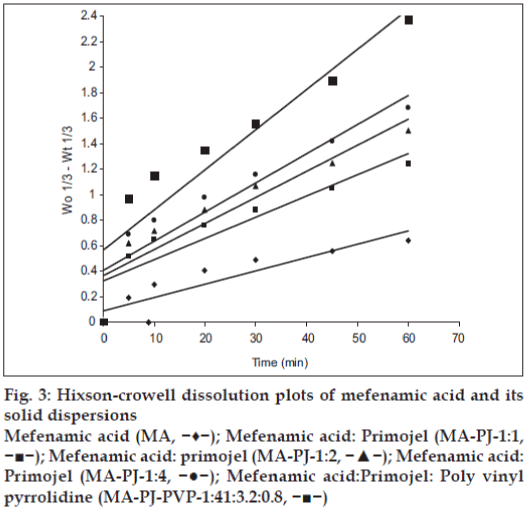
The dissolution data of MA and their solid dispersions
were also analyzed as per Hixson-Crowell’s [7] cube
root equation. Hixson-Crowell introduced the concept
of changing surface area during dissolution and
derived the “cube-root law” to nullify the effect of
changing surface area and to linearize the dissolution
curves. Hixson-Crowell’s cube root law is given by
the following equation. (Wo)1/3–(Wt)1/3= Kt, where
Wo is initial mass and Wt is the mass remained at
time ‘t’. The cube root equation is applicable to
the dissolution of monodisperse powder consisting
of uniform sized particles. A plot of (Wo)1/3–(Wt)1/3 versus time will be linear when dissolution occurs
from monodisperse particles of uniform size. Hixson-
Crowell plots of the dissolution data were found
to be linear (fig. 3) with all solid dispersions.
This observation indicated the drug dissolution
from all the solid dispersions is occurring from
discretely suspended or deposited (monodisperse)
particles. This might have also contributed to the
enhanced dissolution rate of the solid dispersions.
The correlation coefficient (r) values of the first
order release model are found to be (0.9075 to
0.9940) slightly higher when compared to the Hixson-
Crowell’s cube root model. Hence the release of
drug from the preparations followed predominantly first order kinetics compared to Hixson-Crowell
cube root law. Correlation coefficient values in the
analysis of dissolution data as per zero order, first
order and Hixson-Crowell cube root are given in Table 3. Another parameter suitable for evaluation
of in vitro dissolution has been suggested by Khan [8]
is, dissolution efficiency (DE). DE is defined as
the area under the dissolution curve up to a certain
time ‘t’ expressed as percentage of the area of the
rectangle described by 100% dissolution in the same
time. Dissolution efficiency 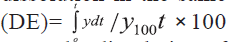 and the index DE30 would relate to the dissolution of
drug from a particular formulation after 30 min and
could be compared with DE30 of other formulations.
Summation of the large dissolution data into a single
figure DE enables ready comparison to be made
between a large numbers of formulations.
and the index DE30 would relate to the dissolution of
drug from a particular formulation after 30 min and
could be compared with DE30 of other formulations.
Summation of the large dissolution data into a single
figure DE enables ready comparison to be made
between a large numbers of formulations.
Figure 3: Hixson-crowell dissolution plots of mefenamic acid and its solid dispersions
Mefenamic acid (MA, −♦−); Mefenamic acid: Primojel (MA-PJ-1:1, −■−); Mefenamic acid: primojel (MA-PJ-1:2, −▲−); Mefenamic acid: Primojel (MA-PJ-1:4, −●−); Mefenamic acid:Primojel: Poly vinyl pyrrolidine (MA-PJ-PVP-1:41:3.2:0.8, −■−)
All the dissolution parameters given in Table 2 indicated rapid and higher dissolution of MA from all solid dispersions when compared to MA pure drug. MA-PVP (8:2) solid dispersion gave rapid and higher dissolution than the pure drug. A 1.21 fold increase in the dissolution rate of MA was obtained with this solid dispersion when compared to pure drug. Water dispersible superdisintegrants gave much higher enhancement in the dissolution rate of mefenamic acid than water-soluble carriers. Solid dispersions of superdisintegrants gave rapid and higher dissolution of MA when compared to pure drug as well as its solid dispersion in water soluble PVP. In each case, the K1 and DE30 values were increased as the concentration of carrier (superdisintegrant) in the solid dispersion was increased. At 1:4 ratio of MA:PJ, the order of increasing dissolution rate with various superdisintegrants was 1:4>1:2>1:1. A 2.61 fold increase in the dissolution rate of MA was observed with MA-PJ (1:4) solid dispersion. All the solid dispersions in combined carriers gave much higher rates of dissolution, several times higher than the dissolution rate of pure drug. PVP combined super disintegrants gave higher dissolution rates than superdisintegrants alone. MA-PJ-PVP solid dispersion gave a 4.11 fold increase in the dissolution rate of MA whereas solid dispersion of MA in PJ lone (MA-PJ 14 solid dispersion) gave only 2.61 fold increase. Thus combination of superdisintegrants with water soluble carrier PVP resulted in a greater enhancement in the dissolution rate of MA. Hladon et al. [9], the inclusion complex of MA with b-cyclodextrin was obtained by the method of coprecipitation from diethyl ether. The product was identified by the thermogravimetric and X-ray methods. The complex stability constants were determined by the potentiometric method. The effect of β-CD on the solubility and stability of MA was analysed. Thus superdisintegrant, PJ was found to be useful as a carrier in MA solid dispersions alone and in combination with PVP to enhance the solubility, dissolution rate and dissolution efficiency.
Acknowledgements
The authors would like to express sincere thanks to the Management of D. C. R. M. Pharmacy College for their encouragement and providing necessary facilities in carryout this research work. The authors would also express sincere thanks to M/s. Sigma Laboratories, Mumbai for generous gift of mefenamic acid samples.
References
- Sweetmann SC. Martindale- The Extra Pharmacopoeia, London: The Pharmaceutical Press; 2005.
- Kato F, Otsuka M, Matsuda Y. Kinetic study of the transformation of mefenamic acid polymorphs in various solvents and under high humidity conditions. Int J Pharm 2006;321:18-26.
- Fang L, Numajiri S, Kobayashi D, Ueda H, Nakayama K, Miyamae H, et al. Physicochemical and crystallographic characterization of mefenamic acid complexes with alkanolamines. J Pharm Sci 2004;93:144-54.
- Guirguis M, Jammali F. Disease-drug interaction: Reduced response to propranolol despite increased concentration in the rat with inflammation. J Pharm PharmSci 2001;4:1077-84.
- Vasconcelos T, Sarmento B, Costa P. Solid dispersions as strategy toimprove oral bioavailability of poor water soluble drugs. Drug Discov Today 2007;12:1068-75.
- Sekiguchi K, Obi N. Studies on absorption of eutectic mixture-I. Chem Pharm Bull 1961;9:866-72.
- Hixon AW, Crowell JH. Dependence of reaction velocity upon surface and agitation: I-theoretical consideration. IndEngChem 1931;23: 923-31.
- Khan KA, Rhodes CT. Effect of compaction pressure on the dissolution efficiency of direct compression systems. Pharm ActaHelv 1972;47:594-607.
- Hładoń T, Pawlaczyk J, Szafran B. Stability of mefenamic acid in the inclusion complex with β-cyclodextrin in the solid phase. J Inclusion Phenomena MacrocyclicChem 1999;35:497-506.