- *Corresponding Author:
- J. K. Pandit
Department of Pharmaceutics, Institute of Technology, Banaras Hindu University, Varanasi-221 005
E-mail: jkpandit@bhu.ac.in
| Date of Submission | 16 June 2005 |
| Date of Revision | 27 February 2006 |
| Date of Acceptance | 13 September 2006 |
| Indian J. Pharm. Sci., 2006, 68 (5): 626-630 |
Abstract
Soluble inserts of ciprofloxacin hydrochloride using high and low molecular weight polyvinyl alcohol alone and in various combinations were fabricated by a casting technique. The in vitro drug release from the prepared inserts was studied using a continuous flow-through model, developed in our laboratory. The antimicrobial efficacies of the prepared inserts against common ocular pathogens, viz., Staphylococcus aureus ATCC 25923 and Pseudomonas aeruginosa ATCC 27853, were evaluated using a modified in vitro microbiological model. Ciprofloxacin hydrochloride release from the inserts followed matrix diffusion kinetics showing an anomalous release mechanism (erosion-controlled) based on the calculated release exponent (n) values. Drug release increased with an increase in the proportion of high molecular weight polyvinyl alcohol in the inserts. The in vitro microbiological model demonstrated the effectiveness of the inserts against the two microorganisms. Further, the results of the in vitro release studies correlated well with that of the antimicrobial studies.
Continuous delivery of drugs to the eye offers major advantages over the conventional ocular therapies that involve administration of drug solutions or suspensions as eye drops. Eye drop administration often results in poor bioavailability and therapeutic response due to rapid pre-corneal elimination of the drug and is also associated with patient compliance problems [1,2]. An ocular sustained release dosage form would ideally eliminate these problems by providing steady pre-corneal drug levels for 12-24 h periods.
Ocular inserts of various ophthalmic drugs have been reported in the recent past [3-6] to improve poor ocular bioavailability. The use of appropriate animal models [7-9] in the development of such ocular dosage forms have become essential for better understanding and advancement of existing knowledge. Many rabbit eye models have been reported for bacterial [10-12], viral [13,14] and fungal infections. However, very stringent guidelines issued by various regulatory agencies in using animals as experimental models have called for the need to develop in vitro models that could be viewed as a potential alternative to the already existing animal models. Hence, an attempt has been made in the present study to evaluate the antimicrobial efficacy of ciprofloxacin hydrochloride (CPH) ocular inserts of polyvinyl alcohol (PVA) using an in vitro microbiological model and correlate the results with the in vitro release data.
Materials and Methods
Ciprofloxacin hydrochloride was generously gifted by Ranbaxy Research Labs (Gurgaon, India). PVA (both high and low molecular weight) was obtained commercially from S. D. Fine Chemicals (Mumbai, India). The microbiological media were purchased from Hi-Media Ltd. (Mumbai, India). All other reagents used were of analytical grade.
Preparation of ocular inserts
The inserts were prepared by casting method with minor modification [15]. Solutions of PVA [different compositions of low (14 000) and high (125 000) molecular weights, Table 1] were prepared in boiling distilled water. The mixture was stirred for 20-24 h to get a clear solution. Then, glycerol (10% w/w, with respect to PVA) was added as plasticizer to the solution and stirred for further 3 h. Weighed amounts of CPH were added after passing through #100 mesh sieve and stirred for 4 h to get a uniform dispersion. The dispersion was then degassed and cast on glass substrate and dried at 50° for 18-20 h. The dried films were carefully removed and inserts of dimensions 2 × 6 mm and average thickness of 0.2 mm were punched out, wrapped individually in aluminium foil and stored in well-closed amber-coloured glass vials in a desiccator until further use.
| Batch code | Proportion of PVAL (%) | Proportion of PVAH (%) | CPH (μg / insert) |
|---|---|---|---|
| BPC1 | 0 | 100 | 500 |
| BPC2 | 0 | 100 | 300 |
| BPC3 | 0 | 100 | 200 |
| BPC4 | 100 | 0 | 500 |
| BPC5 | 100 | 0 | 300 |
| BPC6 | 100 | 0 | 200 |
| BPC7 | 50 | 50 | 300 |
| BPC8 | 50 | 50 | 300 |
| BPC9 | 20 | 80 | 300 |
| BPC10 | 80 | 20 | 300 |
| BPC11 | 33.3 | 66.6 | 300 |
| BPC12 | 66.6 | 33.3 | 300 |
| BPC13 | 75 | 25 | 300 |
| BPC14 | 25 | 75 | 300 |
PVAL – PVA of low molecular weight (14 000), PVAH – PVA of high molecular weight (125 000)
Table 1: Composition of various batches of the prepared inserts
Uniformities of weight, thickness and drug content
The thickness of six inserts was measured at three different randomly selected spots of each insert with a screw gauge. For uniformity of weight, 10 inserts from each batch were weighed individually and their mean determined. For uniformity of drug content, 6 inserts from each batch were weighed individually and dissolved in 50 ml of 0.2 M phosphate buffer pH 7.4. The resultant solution was filtered through a G-2 glass filter. An aliquot of the filtrate was suitably diluted and analyzed for CPH content at 272 nm (Shimadzu, UV-1601, Japan).
Surface pH
Surface pH of the inserts was determined by allowing them to swell in a closed Petri dish at room temperature for 30 min in 0.1 ml of double distilled water. The swollen devices were removed and placed on pH paper to determine the surface pH. After 60 s the colour developed was compared with the standard colour scale.
In vitro release studies
The inserts were evaluated for drug release kinetics by using a continuous flow-through apparatus, which mimics the continuous flow of tears to a certain extent but the constant blinking action of the eye was not attempted to be simulated [16]. The design and working of the continuous flow-through apparatus is described elsewhere [16].
In the drug release study, one insert was weighed and placed on the wire mesh support of the bottom plate. A Whatman filter paper circle (1 cm2 area) was then placed over the insert, and the components of the unit were screwed together. The peristaltic pump, connected with thermostated buffer (37±0.2°) at a flow rate of 0.8 ml/h, was started; and the eluate was collected in ambercoloured glass vials as a function of time. The eluates were analyzed spectrophotometrically for CPH content, as described earlier. After 24 h of release study, the residual drug in the insert was determined by stirring the residual inserts in 10 ml buffer for 6 h, and the contents were filtered through G 2 filter and analyzed spectrophotometrically.
In vitro microbiological studies
Suspensions of the test organisms S. aureus and P. aeruginosa were prepared so as to give 0.5 McFarland standard (106 organisms). McFarland standard (0.5) is said to have been achieved when the absorbances of the prepared suspensions of the microorganisms matched with that of a barium sulphate 0.5 McFarland [17] standard at 625 nm. Aliquots of 1, 2 and 3 ml of the 0.5 McFarland equivalent suspensions of S. aureus ATCC 25923 and P. aeruginosa ATCC 27853 were inoculated into sterile peptone water, and the volume was made up to 5 ml with peptone water. The inserts were introduced into these solutions, and the plugged tubes were incubated at 37°. At periodic time intervals, standard loopfuls from individual tubes were streaked on sterile nutrient agar plates and incubated at 37° for 24 h and observed for growth. To account for the growing number of organisms in the media at the initial times of drug release from the inserts, challenging was done at 3 h from the start of the study by further inoculation with 20% v/v of the initial inoculum of S. aureus or P. aeruginosa. Positive and negative controls were maintained throughout the study.
Results and Discussion
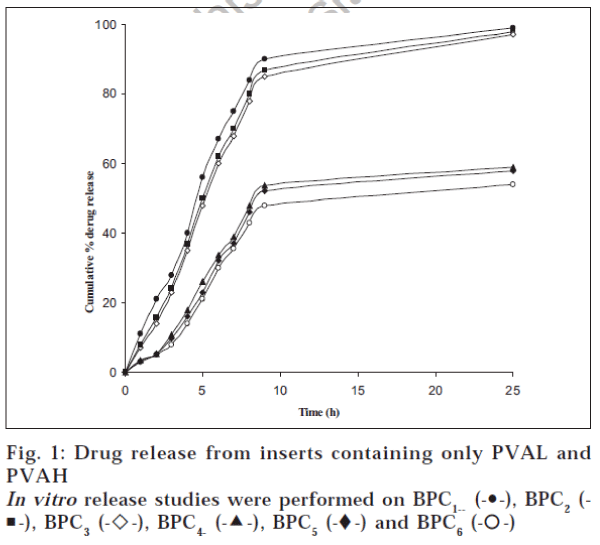
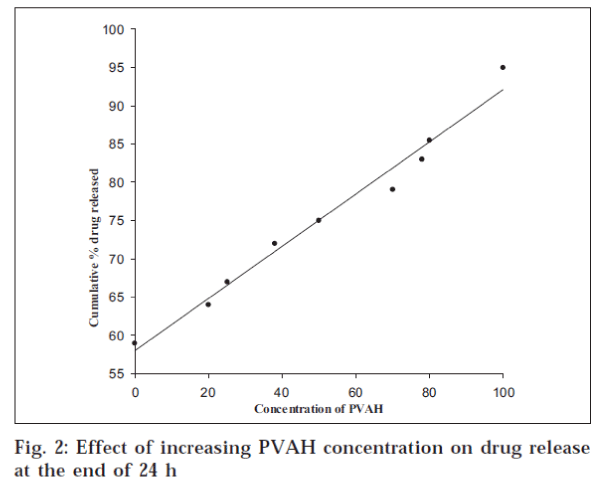
The surface pH of the prepared inserts varied between 5.5 and 7.5, indicating that the inserts did not have an irritation potential [18] as the pH is within the accepted ocular range. The weight variation and thickness of the prepared inserts were within 2%. The drug content of the inserts varied from 95.96±1.14% to 99.67±1.03% for BPC7 and BPC14 respectively. The release from the prepared inserts followed matrix diffusion kinetics in all cases, as evidenced by the regression coefficient (r2) values, which were always higher for Q vs. t1/2 (between 0.917 and 0.997) than for Q vs. t (0.774 to 0.936). Similar release kinetics was reported for flurbiprofen [19] and indomethacin [20] from PVA gels prepared by freeze-thawing and PVA disks respectively. However, the calculated diffusion coefficient (n) values ranged between 0.737 and 0.851, indicating the prevalence of an anomalous release behaviour / mechanism, which seems to be dependent on both matrix swelling and erosion [21]. The minimal initial burst effect seen from the inserts (fig. 1) containing both PVAL and PVAH in varying proportions could be attributed to the more soluble nature of PVAH in comparison to PVAL. Further, the amount of drug released increased with an increase in the proportion of PVAH in the inserts. Perhaps, the presence of higher proportions of PVAH could have resulted in more pronounced matrix swelling and erosion. This was further confirmed by the fact that the drug release from batches containing PVAH alone (BPC1 to BPC3) was significantly (P < 0.05, t-test) higher than the corresponding batches containing only PVAL (fig. 2). Similar results were reported for indomethacin ocular inserts containing different proportions of PVAH and PVAL15. The effects of three different drug loadings were studied from inserts containing either PVAH or PVAL. The release profiles of the inserts containing PVAH indicated that there was no significant difference between the amounts of drug released at the end of 24 h. However, during the initial phases, a marked decrease in the amount of released drug was observed with decrease in drug loading, presumably due to the presence of lesser amount of the drug at the periphery with decrease in drug loading. Similar behaviour was observed from the batches containing PVAL (fig. 1). The rate of release was however proportional to the drug loading, showing a significant increase with increase in drug loading.
Since not much difference was observed in the release profiles from either of the inserts (PVAL and PVAH), the matrix residue was weighed, after drying on tissue paper, at the end of the dissolution studies. The PVAH inserts showed 35-38% reduction from their original weight, while the PVAL inserts showed a reduction of 22-25%. This was again indicative of the fact that the drug release was controlled by swelling /erosion of the matrix along with diffusion, which explained the independence of drug release to drug loading.
The plasticizer is the most important component that may affect the mechanical properties of the films as it lowers the glass-transition temperature [22] of the polymer. Hence, drug release from the plasticized insert (BPC7) was compared with the corresponding unplasticized insert (BPC8). In these two batches, both PVAL and PVAH are present in equal proportions, and any difference in the release pattern could be directly related to the presence or absence of the plasticizer. PVA could be effectively plasticized by a variety of plasticizers like PEG 200 and 400, propylene glycol, glycerol, etc. In this study, glycerol at 10% concentration was selected as it gave sufficiently pliable films to allow for uniform subdivision into inserts without breaking the film. The amount of drug released from BPC7 was significantly higher than the amount released from the unplasticized insert (BPC8). The presence of glycerol in BPC7 enhanced the hydrophilicity of the PVA matrix. This in turn resulted in enhanced permeability of the inserts, resulting in enhanced swelling and the consequent increase in the porosity of the matrix, thus accounting for higher drug release.
S. aureus ATCC 25923, the most common ocular pathogen; and P. aeruginosa ATCC 27853, the most opportunistic ocular pathogen, were used as model organisms in the study. The 0.5 McFarland standard suspensions of S. aureus and P. aeruginosa revealed a count of 1.02×106 and 1.43×106 organisms/ml respectively. The antimicrobial effectiveness of the inserts was tested by varying the volume of the inoculum. The inserts were more effective in inhibiting the growth of P. aeruginosa when compared to S. aureus (Table 2, data shown for 30 and 48 h only). Challenging was done with 20% v/v of the initial inoculum size after 3 h to simulate the number of growing organisms.
| Batch code |
Inoculum volume (ml) | Time (h) | |||
|---|---|---|---|---|---|
| P. aeruginosa | S. aureus | ||||
| 30 | 48 | 30 | 48 | ||
| BPC1 | 2 | -5 | - 5 | -3 (+2) | -3 (+2) |
| 3 | -5 | - 5 | -3 (+2) | -3 (+2) | |
| BPC2 | 2 | -4 (+1) | - 5 | -4 (+1) | -3 (+2) |
| 3 | -5 | -5 | -4 (+1) | -3 (+2) | |
| BPC3 | 2 | -5 | -5 | -4 (+1) | -3 (+2) |
| 3 | -5 | -4 (+1) | -3 (+2) | -3 (+2) | |
| BPC4 | 2 | -5 | - 5 | -4 (+1) | -4 (+1) |
| 3 | -5 | - 5 | -4 (+!) | -4 (+1) | |
| BPC5 | 2 | -5 | -5 | -5 | -4 (+1) |
| 3 | -5 | -5 | -5 | -4 (+1) | |
| BPC6 | 2 | -5 | -5 | -4 (+1) | -4 (+1) |
| 3 | -5 | -5 | -3 (+2) | -3 (+2) | |
| BPC7 | 2 | -5 | -5 | -5 | -4 (+1) |
| 3 | -5 | -5 | -5 | -4 (+1) | |
| BPC8 | 2 | -5 | -5 | -4 (+1) | -3 (+2) |
| 3 | -5 | -5 | +5 | +5 | |
| BPC9 | 2 | -5 | -5 | -5 | -5 |
| 3 | -5 | -5 | -4 (+1) | -4 (+1) | |
| BPC10 | 2 | -5 | -5 | -4 (+1) | -4 (+1) |
| 3 | -5 | -5 | -3 (+2) | -3 (+2) | |
| BPC11 | 2 | -5 | -4 (+1) | -4 (+1) | -4 (+1) |
| 3 | -5 | -4 (+1) | -4 (+1) | -3 (+2) | |
| BPC12 | 2 | -5 | -5 | -4 (+1) | -4 (+1) |
| 3 | -4 (+1) | -3 (+2) | -4 (+1) | -4 (+1) | |
| BPC13 | 2 | -5 | -5 | -3 (+2) | -3 (+2) |
| 3 | -5 | -5 | -4 (+1) | -3 (+2) | |
| BPC14 | 2 | -5 | -5 | -5 | -4 (+1) |
| 3 | -5 | -5 | -2 (+3) | -2 (+3) | |
At 1 ml of inoculum level, inserts were able to inhibit test organism. ± indicates presence/absence of growth and the number indicates the number of tubes in which growth was present or absent. (n = 5)
Table 2: in vitro microbiological efficacies of the inserts against p. aeruginosa and s. aureus
When S. aureus was the organism, batches BPC1 and BPC4 containing 0.5 mg of CPH were effective in inhibiting the growth in all the tubes for 24 h, beyond which growth was observed in two tubes each at 30 and 48 h for batch BPC1 at inoculum volume of 2 and 3 ml. In case of batch BPC4, growth was observed in one tube each at 48 h at an inoculum volume of 2 and 3 ml, indicating that the batch BPC4 composed of only PVAL sustained the drug release more effectively than BPC1 containing the same amount of PVAH. Decrease in the drug loading in the inserts composed of either of the PVA did not produce any remarkable change in the effectiveness of the inserts against the two microorganisms.
The unplasticized insert (BPC8) was unable to inhibit the growth of S. aureus in all the tubes from the 24th hour (at 3 ml inoculum level) onwards, whereas in contrast the corresponding plasticized insert (BPC7) inhibited the growth of S. aureus in all the tubes for 30 h. This indicated the propensity of the plasticized insert to release higher amount of the drug due to the channelling effect of the plasticizer, resulting in faster and higher amount of drug release.
Overall, when S. aureus was the organism, the inserts were able to inhibit the growth for the entire duration of the study (48 h) when the inoculum volume was 1 ml. At higher inoculum volumes (2 and 3 ml), the inserts were unable to inhibit the growth in all the tubes, showing an efficiency of 60-80% from the 24th hour onwards. Introduction of one more insert at the 24th hour would have resulted in the inhibition of the growth in all the tubes beyond the 24th hour. In other words, and by implication, the developed inserts have the potential of once-a-day application in the treatment of ocular infections, the various ocular barriers in the ocular domain notwithstanding.
This study describes the complete methodology for the development of the CPH ocular insert, including the in vitro release studies on a flow-through apparatus and analysis of drug release kinetics. The studies also demonstrated the effectiveness of the prepared inserts against two of the most common ocular pathogens (S. aureus and P. aeruginosa). Further, the in vitro release data correlated well with the microbiological efficacy data without having to simultaneously use the rabbit eye model, which, in our opinion, is a substantial improvement over an earlier reported method [23]. These inserts have the potential to form the basis of a once-daily therapy of ocular infections. However, validation of this model with other categories of antibacterials using other ocular pathogens and correlation with in vivo models are required before it could be viewed as a potential alternative to the in vivo models.
References
- Schoenwald, R.D. Clin. Pharmacokinet., 1990, 18, 255
- Hume, L.R., Lee, H.K., Benedetti, L., Sanzgiri, Y.D., Topp, E.M. andStella, V.J., Int. J. Pharm., 1994, 111, 295.
- DiColo, G., Burgalassi, S., Chetoni, P., Fiashi, M.P., Zambito, Y. andSaettone, M.F., Int. J. Pharm., 2001, 215, 101.
- Kawakami, S., Nishida, K., Mukai, T., Yamamura, K., Nakamura, J.,Sakeda, T., Nakashima, M. and Sasaki, H., J. Control. Release,2001, 76, 255.
- Sasaki, H., Tie, C., Nishida, K. and Nakamura, J., J. Control.Release, 1993, 27, 127.
- Baleens, V., Catalos, V., Boisrame, B., Varesio, E. and Gurny, R., J.Control. Release, 1998, 52, 215.
- Lee, V.H.K. and Robinson, J.R., Int. J. Pharm., 1989, 53, 219.
- Lee, Y.C., Simamaora, P. and Yalkowsky, S.H., J. Pharm. Sci.,1997, 86, 430.
- Wood, R., W. Lee, V.H.K., Kreiuter, J. and Robinson, J.R., Int. J.Pharm., 1985, 23, 175.
- Badenoch, P.B., Hay, G.J., McDonald, P.J. and Coster, D.J.A., Arch.Ophthalmol., 1985, 103, 718.
- Hobden, J.A., Reidy, J.J., O’Callaghan, R.J. and Hill, J.M., Arch.Ophthalmol., 1988, 106, 1605.
- Aswad, M.I., Barza, M. and Baum, J., Arch. Ophthalmol., 1989,107, 1667.
- Pavan-Langston, D., Langston, R.H.S. and Geary, P.A., Arch.Ophthalmol., 1975, 93, 1349.
- Troudale, M.D., Barlow, W.E. and McGuigan, L.J.B., Arch.Ophthalmol., 1989, 107, 1664.
- Kim, H., Robinson, M.R., Lizak, J.M., Tansey, G., Lutz, R.J., Yuan, P.,Wang, N.S. and Csaky, K.G., Inves. Ophtalmol. Vis. Sci., 2004,45, 2722.
- Pandit, J.K., Hari Kumar, S.L., Mishra, D.N. and Balasubramaniam, J.,Indian J. Pharm. Sci., 2003, 65, 146.
- Jorgensen, J.H., Turnidge, J.D. and Washington, J.A. In: Murray, P.R.,Pfaller, M.A., Tenover, F.C., Baron, E.J. and Yolken, R.H., Eds.,Mannual of Clinical Microbiology, 7th Edn., ASM Press, Washington,DC, 1999, 1526.
- Balasubramaniam, J., ThilekKumar, M., Pandit, J.K. and Kant, S.,Pharmazie., 2001, 56, 793.
- Takamura, A., Ishii, F. and Hikada, S., J. Control. Rel., 1992, 20, 21.
- Thanoo, B.C., Sunny, M.C. and Jayakrishnan, A., J. Pharm. Pharmacol., 1993, 45, 16.
- Vazquez, M.J., Perez Macros, B., Gomez-Amoza, J.L., Martinez-Pacheo, R., Souto, C.fromandConchrio, A., Drug. Dev. Ind. Pharm.,1992, 18, 1355.
- Deamin, R.D., In; Seymour, R.B. Eds., Additives of Plasticizers,Academic Press, New York, 1978, 203.
- Devarajan, P.V., Bhogte, C.P., Majali, A.B., and Sabharwal, S., DrugDevelop. Ind. Pharm., 1999, 25, 781.