- *Corresponding Author:
- R. Adepu
Department of Pharmacy Practice, Vikas College of Pharmaceutical Sciences, Rayanigudem, Suryapet 508213, India
E-mail: adepu63@gmail.com
| Date of Received | 01 July 2020 |
| Date of Revision | 25 March 2022 |
| Date of Acceptance | 05 December 2022 |
| Indian J Pharm Sci 2022;84(6):1536-1543 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
Drug information service is the provision of written or verbal information about safe use of medications or drug therapy in response to a request from any health care professional. A prospective observational pilot study was initiated at a 1000 bed tertiary care teaching hospital to create an awareness, implementation and evaluation of the drug information services. Institutional ethics committee has approved the study and the hospital authorities permitted the service. The drug information service activity was started at clinical pharmacy department in the hospital and Pharm. D interns and the faculty was involved in the study. Drug information queries were collected during ward rounds and also through direct access. The ultimate question was analyzed after receiving the query, suitable references were reviewed and the final answer was synthesized and the approved answer was provided to the requester. Periodically the quality assurance of the information provided was assessed using quality assurance check list. A total of 154 queries were received during the study period. Most of the queries (69) belong to the category of drug of choice. Majority queries came from general medicine (45 %) and emergency medicine departments (16 %). Post graduate students (40 %) and medical interns (38 %) were the major requesters. Both verbal and written answers were provided to 136 queries. Majority queries received were during ward rounds and answers were provided within 8 h. The quality assurance score for the randomly selected queries was found 4.9 out of 5 indicate good quality service. The study findings suggest that drug information services were well utilized by the health care professionals. However, there is a need to create awareness among all health care professionals regarding usefulness of this service.
Keywords
Drug information service, health care professional, query, response, quality assurance
Society of Hospital Pharmacists of Australia (SHPA), defines drug information as “the provision of written and/or verbal information or advice about drugs and drug therapy in response to a request from other health care providers, organizations, committees, patients or members of the public”[1]. The whole objective of this service is to provide accurate, unbiased, factual information, primarily in response to patient-oriented problems. Lack of unbiased information is one of the major causes of irrational use of drugs leading to therapeutic failure and adverse drug reactions. It is very difficult for any healthcare provider to be updated with increasing therapeutic information explosion both in print and electronic media to choose appropriate medications for treatment and to provide right information to the patients about the medications prescribed. In the era of “evidence-based medicine” practice, the role of Drug Information Center (DIC) is becoming highly important and the services are appreciated[2].
Drug Information Service (DIS) is well advanced in developed countries like the United States, Australia and United Kingdom (UK). The first official DIC was started in 1962 at University of Kentucky Medical center with sole purpose of offering comprehensive information on drugs to staff physicians and dentists to evaluate and compare the information on drugs to be provided to the nursing staff[3].
But in developing countries, it is still a new concept. In India, provision of drug information is an emerging concept. The DIC in India was started in 1970 at Christian Medical College in Vellore by Rosemary Sharp, a missionary from UK with an objective to provide unbiased information on medicines to doctors, nurses, pharmacists and patients[4]. In 1997, Karnataka state pharmacy council was first to start an official DIC in Bangalore to offer unbiased information on drugs to medical professionals in Karnataka state and also encouraged and supported many other state pharmacy councils in the country to start DICs[5]. The DIC’s are usually staffed by trained clinical pharmacologists and pharmacists who review the queries, search the literature, and provide the information in a structured and evidence-based manner. Various resources such as primary (peer reviewed journals), secondary (data bases) and tertiary (text books and compendia) can be used to get the proper information to synthesize the ultimate answer[6].
Pharmacy practice departments of various pharmacy colleges in South India have started their own DICs in their affiliated hospitals and attempted to assess the functioning of their DICs. Findings of their studies suggest that there is a dire need for the quality DICs to the physicians and consultants even to the nurses for rational use of medicines and also for right selection of diluents fluids and appropriate flow rate of Intravenous (IV) administration. The findings also suggest the need for improved awareness among the health care force regarding the usefulness of DICs[7-10].
Global evaluation about the functioning of the DICs suggest that, DICs should possess well trained staff with adequate, primary, secondary and tertiary resources and able to advocate the health care professionals on appropriate use of therapeutic guidelines and influence upon the desired therapeutic outcomes[11].
In majority DICs, resources used are (tertiary resources), American Hospital Formulary System (AHFS) drug information. American Society of Health-System Pharmacists; Australian injectable drugs handbook. Society of hospital pharmacists of Australia; Drug information handbook. Lexi-Comp; Briggs GG, Freeman RK, Yaffe SJ, editors. A reference guide to fetal and neonatal risk: drugs in pregnancy and lactation; Stockley IH, editor. Drug Interactions, London: Blackwell Scientific Publications; Ellernhorn MJ, Barceloux DG. Ellenhorn's medical toxicology: Diagnosis and treatment of human poisoning. Williams and Wilkins; Martindale: The complete drug reference. London: Pharmaceutical Press, Davies DM, editor. Textbook of adverse drug reactions. New York: Oxford University Press and USP Dependency Injection (DI). Drug information for the health care professional, Vol. I. and Vol II Rockville: United States Pharmacopeia Convention.
Secondary resources include Iowa Drug Information Services (IDIS), Micromedex and Lexicomp. And primary resource includes peer reviewed journals.
The present study was conducted with an objective of initiating and evaluating the usefulness of DIS at a tertiary care teaching hospital in South Telangana.
Materials and Methods
The present study was conducted at Kamineni Institute of Medical Science (KIMS) Hospital, a 1000 bed tertiary care teaching hospital located in Narketpally, South Telangana. The study was conducted as pilot prospective observational study, for a period of 6 mo with a goal of initiating the DIS and assesses its usefulness to the health care team in the hospital. After receiving the approval from the Institutional Ethics Committee and the hospital authorities, the study was initiated in the hospital. Briefing about the DIS and its usefulness to the post graduate students, medical interns and to the chief of emergency medicine department.
The Pharm D interns have collected the queries during the ward rounds in a suitably designed Drug Information (DI) request form. The clinical pharmacy department in the hospital is equipped with all the resources, including the CliniRex® drug information and documentation database, to provide drug information and document all the DI services provided.
The clinical pharmacy faculty and the Pharm. D interns posted in DI service along with research students to analyze the DI queries and synthesize the answers using suitable DI resources and finally get the answers reviewed by the faculty in the department. Upon approval, the answers either verbally or written were provided to the requester. The clinical pharmacy department offered the DICs on weekdays from 9 am-4 pm.
Every week, the quality of the information provided was assessed using an approved quality assurance check list[11] for at least 30 % of the queries randomly picked up by the faculty. The quality assurance check list contains 8 questions. Each correct answer carries one mark. Scores of the queries categorized in to grades; A-Excellent, B-Good, C-Can improve and D-Should improve. Where A=7-8 points, B=5-6 points, C=4 points, D=3 or less. Out of 8 score, minimum of 3.5 score is expected to consider the DI answer as satisfactory.
Results and Discussion
Total of 154 queries were received during the study period. Majority queries came from general medicine (45 %) and emergency medicine departments (16 %). Post graduate students (40 %) and medical interns (38 %) were the major requesters. Majority queries received were during ward rounds and answers were provided within 8 h. More number of queries was asked for patient care and for updating the knowledge. The details of the department, requester, type of queries, within what time the answer was asked are presented in Table 1.
| Category | Number of queries (n=154) | Percentage (%) |
|---|---|---|
| Department | ||
| General medicine | 70 | 45 |
| Emergency medicine | 24 | 16 |
| General surgery | 13 | 8 |
| Gynaecology | 7 | 5 |
| Ophthalmology | 5 | 3 |
| Orthopedics | 5 | 3 |
| Pediatrics | 3 | 2 |
| Psychiatry | 14 | 9 |
| Pulmonology | 1 | 1 |
| Dermatology | 12 | 8 |
| Requester | ||
| Physician | 32 | 21 |
| Postgraduate | 62 | 40 |
| Medical intern | 30 | 39 |
| Mode of Request | ||
| During ward rounds | 112 | 73 |
| Direct access | 41 | 26 |
| 1 | 1 | |
| Time taken to provide response | ||
| Immediately | 23 | 15 |
| Within 2-4 h | 31 | 29 |
| Within 24 h | 72 | 47 |
| Within 48 h | 28 | 18 |
| Purpose of query | ||
| Patient care | 80 | 52 |
| Academic related | 26 | 17 |
| To update knowledge | 48 | 31 |
| Mode of reply | ||
| Verbal | 2 | 1 |
| Written | 16 | 11 |
| Both verbal and written | 136 | 88 |
| References used | ||
| Primary | 82 | 53 |
| Secondary | 21 | 14 |
| Tertiary | 51 | 33 |
Table 1: Details of DI Queries received.
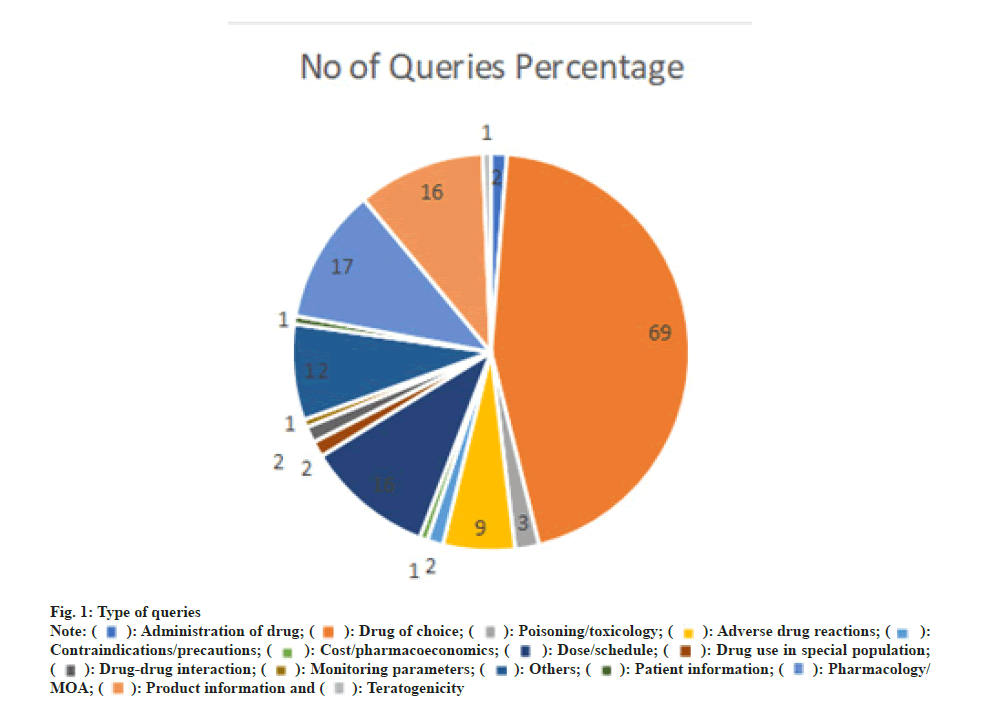
All the queries received were categorized upon the type. Majority queries belongs to the category of drug of choice (45 %) followed by contra indications (11 %) and drug of choice (11 %). The details of question category were presented in fig. 1.
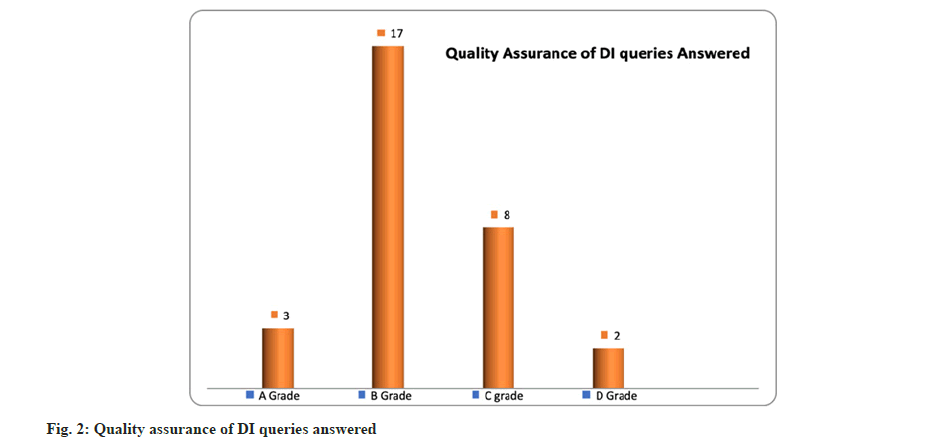
As per the scores of quality assurance check list, 30 questions randomly picked up from all the queries were subjected for quality assurance. 3 questions scored A grade, 17 queries received B grade, 8 queries scored C grade and only 2 queries scored D grade with an average score of 5 suggesting the quality of service is good. The details are presented in fig. 2.
The present study was carried out in KIMS hospital as a pilot study for about 6 mo to assess the usefulness of DIS to clinicians and patients. The college has signed a memorandum of understanding with KIMS Hospital for providing clinical pharmacy services to patients and clinicians and to train clinical pharmacy students at bed side for clinical case management learning. One of the responsibilities of clinical pharmacist is to offer unbiased DICs. Thus, this study was initiated to create awareness about DICs to clinicians and assist them to make use of clinical pharmacy services.
Before initiating the DIS activity, Medical Superintendent and General Manager of the Hospital were explained about the purpose and usefulness of the services in better patient care. Upon their approval, team of consultants and postgraduates were briefed about the DI service, how to send their query, how much time will be the wait period to get the answer and mode of receiving the requested information.
During the study period, a total number of 112 queries were received in the ward rounds and 41 queries through direct access in the department. About 45 % of queries were received from General Medicine department followed by 16 % from emergency medicine. The reason for more queries from these departments is due to high use of medicines to manage the clinical conditions. Many previous studies have also corroborated these type findings. Medicine department use medicines while treating the patients.
For practicing the evidence-based medicine, well reviewed and referenced unbiased information helps better in patient care process. In the present study, majority queries were related to the drug of choice 45 %, pharmacology related queries 11 %, product and dose related queries was 10 %. Majority queries (77 %) asked were the purpose of updating the requester’s knowledge. In order to answer the received queries, primary, secondary and tertiary information resources were used. Among them, 53 % queries were answered by using primary resources such as journals. About 47 % of the queries received were answered within 24 h. Average score of quality assurance of the answers provided to the queries is 4.9 and ensures the highest quality of the information and service. In our study, the resources used were peer reviewed online journals, CliniRex® database, AHFS, Australian drug information Handbook, Martindale and other pharmacology and pharmacotherapeutics text books.
In 1962, the first DIC was established at the University of Kentucky Medical Center, United States of America (USA) with an intension to utilize as a source of selected, comprehensive drug information for staff physicians and dentists to allow them to evaluate and compare the information about the drugs besides catering to the information needs of nursing staff[12].
As per a report published in 1995, about 120 DICs were established in USA to serve the health care professional’s medicines information requirement. The major objective of DIS is to provide unbiased interpretative information on the medicines to the requesters. The most frequent type of inquiries is reported to involve information on therapeutic use, dosage, product identification (US and foreign) and American Depositary Receipts (ADRs). In US, it was documented that the mean percentage of questions requiring judgmental responses synthesized from the integration of data, knowledge and experience has increased from 5 % in 1975 to 52 % in 2004 however the quality assurance level of the information provided was found decreased in some centers[13]. Our study findings also reveal that, the initiation DI services is with the similar objectives.
In many European countries and in Sweden, DIS was developed for encouraging a co-operative approach between department of pharmacology, General Practitioners (GP), pharmacists and a drug and therapeutics committee. Such ‘academic drug detailing’ provides drug information from project to practice[14,15]. Academic drug detailing is an interactive educational outreach to prescribers to present unbiased, non-commercial, evidence-based information, mostly about medications, with the goal of improving patient care.
Dyrkorn et al. had conducted a study in Norway about the usefulness of academic drug detailing by the pharmacists to GP. In this study, trained academic detailing pharmacists offered one to one discussion with about 213 GPs regarding the evidence based and manufacturer independent information and rational use of Non-Steroidal Anti-inflammatory Drugs (NSAID). After this intervention, the study findings suggest a drastic decrease in unnecessary use of NSAID in the prescriptions[16].
In India, the concept of DIS heard since 1997. The first DIC was established at department of clinical pharmacy practice, JSS Hospital, Mysore in 1997 as part of department service to the doctors and nurses in the hospital. A review was conducted by Nibu et al. about the DIS at JSS Hospital; the findings suggest that, about 82 % of queries were received from doctors followed by 16 % nurses. Majority queries were asked for the purpose of dosage or administration (30 %), ADR (27 %) and efficacy (19 %). The overall rating by the doctors and nurses regarding the usefulness of the DIS was “Highly Useful”[7].
In another study conducted by George and Rao at Kasturba Medical College Hospital reveals that, about 98 % queries received were from the doctors and the major purpose of using the services is for the better patient care. Majority questions were from medicine department. The reason observed for receiving more questions from medicine department was mainly due to medicines was being used often as primary line of management. During the study period, majority questions were asked to know about dosage and administration (27 %), followed by adverse drug reactions (26 %) and drug therapy (15 %). The results of quality assurance of DIS had shown that majority queries scored 5 out of 5[17].
In another study conducted by Mallayasamy et al. in quality assurance of the DI responses provided by the DI pharmacist, the evaluation results showed that both judgmental and non-judgmental queries had rating of very good or above. When enquirer’s perspective was evaluated, around 70 % of clinicians used services of the center and around 70 % of the people felt that services were very good. 98 % of surveyed physicians opined that there is a need for DIC in the hospital. Evaluation of DIC quality based both on providers as well as enquirer’s perspective showed that they were of good quality[4]. The quality assurance score of our DI service is also very high and comparable with above studies.
In another study conducted by Rajanandh et al. evaluating the DISs at a tertiary care teaching hospital in Tamil Nadu. The findings suggest that majority queries were from interns and physicians (70 %) and from general medicine department (56 %). The quality of information provided was highly satisfied (98 %)[18].
In another study conducted in a tertiary teaching hospital in South India by Peter et al., with respect to assessment and utilization of DIS and creating awareness for enhanced utilization of DIC in a tertiary care teaching hospital, the findings conclude that the DIS provided were useful to various healthcare professionals to maintain rational drug therapy by giving unbiased and well-reviewed information[19].
In another study conducted, Patel et al. assessing the active contribution of clinical pharmacists towards drugs and therapeutics information services to the health care professionals in JSS Hospital, the findings reveal that, during the study period, a total of 1204 queries were answered by the clinical pharmacists. Among them post-graduate medical students made the highest (48.17 %) requests followed by physicians (25.70 %), interns (8.06 %), surgeons (5.06 %) and dermatologists (5.06 %) and nephrologists (4.15 %). Most (63.95 %) of the DI queries were answered immediately followed by within 2-4 h (23.34 %), whereas around 10.38 % of queries were answered within a day and 2.33 % of queries were answered within 1-2 d of time. When the quality of DI service was assessed, 64.5 % of them was rated as excellent, 35.8 % of them were rated as good, whereas 3.73 % of queries needed an improvement in the quality of provision of DI. The highest number of DI requests during ward rounds indicates active presence and routine contribution of clinical pharmacist’s in-patient wards is for better patient care. Their presence during the rounds not only ensures the provision of medicines related information quickly to prescribers for rational drug therapy and also assist clinicians in identifying and fixing the drug related problems in the prescriptions[20].
A survey was conducted by Wen et al. in Egypt assessing the quality and the essential need for drug information centres in Egypt. The findings suggest that, DICs performed the activities include drug information answering service (100 %), adverse drug reaction reporting (100 %), issuing bulletins (83.3 %), education (83.3 %), drug use evaluation (50 %) and Pharmacy and Therapeutics (P&T) committees (33.3 %). The most frequent question categories asked were dosing, side effects, treatment guidelines and drug interactions. More than half of the DICs were attached to the hospitals. However, an observation made during the study was lacking of confidence levels of the DIC professionals on the professionalism and DIC services and its impact on patient care. It was also identified that, the DICs has a problem in identifying the ultimate question in the DI request. The most accurately answered request was adverse drug reactions. All surveyed groups considered that it is very important to have DIC services available to the public through community pharmacies at free of cost. It is also important to devise suitable guidelines for functioning of the DIC in hospitals and community[21].
Medication utilization evaluation is an important tool in assessing the appropriateness of medication prescribing leading to achievement of therapeutic goals within the given budgets. DIC do have responsibility in ensuring the practice of evidence-based medicine. A study was conducted by Alomi et al. in Saudi Arabia MOH hospitals having DICs to assess the medication utilization evaluation systems in the DICs of ministry of health hospitals.
The investigators have used the approved questionnaire having various domains of medication use evaluation questions. The findings suggest that, established criteria, guidelines, treatment protocols and standards of care for specific medications and medications use processes found only in 60 %-65 % hospitals. The study findings reiterate that, it is very important for DICs to play advocacy role in educating healthcare professionals to practice the therapeutic guidelines, treatment protocols and standards of care to improve the therapeutic outcomes and minimize the unnecessary health care expenditure[22].
Majority DICs, worldwide use similar resources for providing answers to the requesters. In our study apart from the text books and journals, we have used CliniRex® a clinical decision support system platform developed in India. This is a user friendly and provides information accurate, specific to the point based on patient clinical condition.
Though the concept of DIS was accepted well, many clinicians are reluctant to use pharmacist mediated DIS due to quick information available online and lack of data showing the usefulness of pharmacist provided DI services in terms of achieving definite therapeutic outcomes and saving of unnecessary health care expenditure and worthiness of the service. However, consistent advocacy about DI service to the interns, PGs and junior doctors may motivate them to use this service.
Our study findings suggest that, starting DI services under the supervision of a trained clinical pharmacist will motivate clinicians to use DI services regularly and partnering them in ensuring the quality assurance will help in establishing the DI services in hospital.
Acknowledgements:
Authors express their sincere gratitude to the Dr. K. Ramadas Chairman, Sri. Sadineni Srinivasa Rao, Correspondent, Management of Vikas College of Pharmaceutical Sciences, Suryapet, Dr. ME Luther, Medical Superintendent, KIMS Hospital, Narketpally for their continuous support in completing this project.
Conflict of interests:
The authors declared no conflict of interests.
References
- Society of Hospital Pharmacists of Australia. Standards of Practice for Drug Information Services. SHPA practice standards and definitions; 1998.
- Parthasarathi G, Ramesh M, Nyfort HK, Nagavi BG. The evaluation of the quality of the drug information center based on providers and enquirers perspective. Clinical pharmacy in a South Indian showed that the service provided by the drug information teaching hospital. Ann Pharmacother 2002;36(5):927-32.
- Suresh CP. The performance of drug information center at the university of kansas medical center, Kansas city, USA-Experiences and evaluations. Indian J Pharmacol 2002;34(2):123-9.
- Mallayasamy SR, Fayazkhan M, Kishore GS, Acharya LD, Rao PG. Evaluation of drug information service provided by clinical pharmacy department based on provider and enquirer’s perspective. Indian J Pharm Prac 2008;1(1):37-44.
- Lakshmi PK, Rao DG, Gore SB, Bhaskaran SH. Drug information services to doctors of Karnataka, India. Indian J Pharmacol 2003;35(4):245-7.
- Lierena A, Ohman B, Alvan G. References used in drug information centers. Eur J Clin Pharmacol 1995;49(1):87-9.
- Nibu P, Ramesh M, Parthasarathi G. Review of a drug information service in an Indian teaching hospital. Aust J Hosp Pharm 2001;31(2):144-5.
- Puttegowda SK, Lakshminarayana SY, Ramarathnam NM. Assessing the pattern of drug information queries in a rural South Indian tertiary care teaching hospital. Malaysian J Pharm Sci 2010;8(2):1-9.
- Nathan JP, Gim S. Responding to drug information requests. Am J Health Syst Pharm 2009;66(8):706-11.
- Ghaibi S, Ipema H, Gabay M. ASHP guidelines on the pharmacist’s role in providing drug information. Am J Health Syst Pharm 2015;72(7):573-7.
[Crossref] [Google Scholar] [PubMed]
- Patel H, Churi S, Gurumurthy P, Madhan R. Quality assurance of drug information service and drug therapy review provided by clinical pharmacists in an Indian teaching hospital. Indian J Pharm Prac 2015;8(3).
- Rosenberg JM, Fuentts RJ, Starr CH, Kirschenbaum HL, McGuire H. Pharmacist-operated drug information centers in the United States. Am J Health Syst Pharm 1995;52(9):991-6.
[Crossref] [Google Scholar] [PubMed]
- Rosenberg JM, Koumis T, Nathan JP, Cicero LA, McGuire H. Current status of pharmacist-operated drug information centers in the United States. Am J Health Syst Pharm 2004;61(19):2023-32.
[Crossref] [Google Scholar] [PubMed]
- Taggiasco N, Sarrut B, Doreau CG. European survey of independent drug information centers. Ann Pharmacother 1992;26(3):422-8.
[Crossref] [Google Scholar] [PubMed]
- Lundborg CS, Hensjö LO, Gustafsson LL. “Academic drug-detailing”: From project to practice in a Swedish urban area. Eur J Clin Pharmacol 1997;52(3):167-72.
[Crossref] [Google Scholar] [PubMed]
- Dyrkorn R, Langaas HC, Giverhaug T, Espnes KA, Rowett D, Spigset O. Academic detailing as a method of continuing medical education. Adv Med Educ Prac 2019;10:717-25.
[Crossref] [Google Scholar] [PubMed]
- George B, Rao PG. Assessment and evaluation of drug information services provided in a South Indian teaching hospital. Indian J Pharmacol 2005;37(5):315.
- Rajanandh MG, Ruby V, Ramasamy C. Evaluation of drug information services in a tertiary care hospital in kanchipuram district and community pharmacies in neighboring areas. Indian J Pharm Pract 2011;4(2):51-6.
- Peter AV, Murali A, Tomy T, Londhe SP. Assessment and utilization of drug information services and creating awareness for enhanced utilization of drug information center in a tertiary care teaching hospital. Asian J Pharm Clin Res 2017;10(5):270-4.
- Patel H, Adepu R, Sapthagiri R, Gurumurthy P. Drugs and therapeutic information service provided by clinical pharmacists for an improved patient care: An experience from a tertiary care teaching hospital. Asian J Pharm Clin Res 2015;8(6):175-8.
- Wen MM, Aref H, Abozaid A, Kandil NH, Elsobky YH. Quality evaluation and survey of the essential need for drug information centers. Int J Pharm Pharm Sci 2016;8(11):137-43.
- Alomi YA, Alghamdi SJ, Alattyh RA. National survey of drug information centers practice in Saudi Arabia: Medication-use evaluation system at ministry of health hospitals. J Pharmacovigil 2017;5(5):245-9.
 .
.