- *Corresponding Author:
- K. R. Iyer
Department of Pharmaceutical Chemistry, Bombay College of Pharmacy, Kalina, Mumbai-400 098, India.
E-mail: krishna@bcpindia.org
| Date of Submission | 28 September 2005 |
| Date of Revision | 24 May 2006 |
| Date of Acceptance | 20 January 2007 |
| Indian J Pharm Sci,2007, 69 (1): 41-45and |
Abstract
Xanthine oxidase activity containing fractions from rat, mouse, rabbit and guinea pig livers were obtained by heat treatment and ammonium sulfate precipitation. Xanthine oxidase activity was observed in rat and mouse liver fractions, while xanthine oxidase activity was absent in rabbit and guinea pig liver fractions. Enzyme kinetic parameters, Km and Vmax, were determined for the conversion of xanthine to uric acid by rat and mouse live fractions, by both spectrophotometric and HPLC methods. The Km values obtained by either method for both animal liver fractions were in the range of 5.32-13.8 µM.
Introduction
Xanthine oxidase (XO) is cytosolic enzyme that is widely distributed among species (from bacteria to man) and within various tissues of mammals [1,2]. XO belongs to the group of enzymes collectively known as the molybdenum iron-sulfur flavin hydroxylases [1,2]. XO catalyzes the hydroxylation of purines, and in particular, hypoxanthine to xanthine and then to uric acid. Uric acid has been implicated to contribute to the antioxidant capacity of the blood. XO catalyzed oxidation involves molecular oxygen as the physiological electron acceptor. XO is an important source of oxygen free radicals since it catalysis the reduction of O2 and H2O leading to the formation of superoxide (O2-) and hydrogen peroxide (H2O2), and has thus been proposed as being part of the central mechanism of oxidative injury in some situations [1]. With regard to drug metabolism, XO is an important Phase I oxidative enzyme that contributes to the metabolism of heterocyclic structures that possess an electropositive carbon centre (generally C adjacent to a heteroatom like N, S, or O)3. Protocols for XO enzyme activity extraction have been reported from liver tissue and body fluids such as milk[1,2,4-6].
Our laboratory has been involved in the establishment of both enzyme fraction isolation protocols and enzyme assay protocols for various important drug metabolizing enzymes. The present study was initiated for isolation of crude fraction of XO from various animal species and determination of kinetic parameters of XO for substrate xanthine by both spectrophotometric and HPLC methods.
Materials and Methods
Animal livers were obtained from the Department of Pharmacology, Bombay College of Pharmacy, Mumbai.The animals and the strains used were as follows-Rat (Charles Foster), mouse (Swiss Albino), guinea pig (Albino), and rabbit (New Zealand). The animals used in this study were those that were sacrificed as part of other experiments approved by the Institutional Animal Ethics Committee. It should be noted that the potential of these experiments to alter liver function (and consequently XO content) although a possibility, was not taken into consideration since the intention of this study was to only evaluate the XO isolation procedure and XO activity determination. The livers obtained were either used fresh or stored at -70° until use. Trizma base was purchased from Sigma Chemical Co., USA. Potassium dihydrogen orthophosphate, dipotassium hydrogen orthophosphate and EDTA were purchased from S. D. Fine Chem. Ltd., Mumbai. Xanthine (for biochemistry use) was from CDH Laboratory, New Delhi. Bradford’s macro method protein estimation kit was purchased from Bangalore Genie Pvt. Ltd., Bangalore. Zinc sulphate was purchased from Merck Chemicals Ltd., Mumbai and ammonium sulphate and uric acid were purchased from Himedia Lab., Mumbai. All other chemicals and reagents used in the study were of AR grade.
Isolation of xanthine oxidase fractions
Crude fractions of XO were isolated from animal livers by previously reported procedures [5,6]. Briefly, 10 g of liver was homogenized in 5 volumes of 0.01 M Tris-HCL buffer, pH 8.0, containing 1 mM EDTA for 5 min. in a Potter glass homogenizer equipped with a Teflon pestle. The homogenate was then rapidly heated to 55° on a water bath, maintained at this temperature for 5 min and then cooled quickly to below 10° in an ice bath. During both the heating and cooling steps the homogenate was stirred. The heat-treated and cooled homogenate was centrifuged at 16 000×g for 15 min. and the precipitate discarded. Solid ammonium sulphate was added to the supernatant to a final concentration of 30% saturation (18.78 g/100 ml), the mixture centrifuged at 16 000×g for 115 min. and the precipitate discarded. XO in the supernatant was precipitated by addition of solid ammonium sulphate to a final concentration of 60% saturation (37.56 g/100 ml). After centrifugation at 16 000×g for 15 min, the pellet was suspended in 10 ml of 0.05 M potassium phosphate buffer, pH 7.5, containing 0.3 mM EDTA and stored at -70° for further use. Three different samples of XO fractions were obtained from different liver samples of each animal and all of the subsequent estimations were done in duplicate for each of the three isolated XO fractions per animal.
Enzyme assay
XO activity assay was performed according to the reported method7. A Shimadzu spectrophotometer (UV 160 A) with matched 10 mm cuvettes was used for the assay. The assay mixture contained 50 mM potassium phosphate buffer, pH 7.5, containing 0.3 mM EDTA, 50 µM xanthine and 50 µl of enzyme sample in final volume of 3 ml, in a 10 mm quartz cuvette. The blank cuvette consisted of all the components listed above except enzyme. XO activity determination was initiated by addition of enzyme and was monitored by analyzing the amount of uric acid formed at 292 nm at ambient temperature. The velocity of reaction was determined as nmol of uric formed per 3 ml per min using an extinction coefficient (e) of 12.2 mM-1 cm-1. One unit of enzyme activity was defined as the amount of enzyme that catalyzed the formation of 1 nmol of uric acid per min under the stated conditions [8].
Protein determination
Protein concentrations of partially purified XO fractions were determined according to the Bradford method with Bradford macro method kit, using bovine serum albumin as standard, according to the manufacturer’s instructions.
Spectrophotometric determination of XO enzyme kinetic parameters
Km (Michaelis-Menten constant) and Vmax (maximum velocity) values for the oxidation of xanthine to uric acid with rat and mouse liver XO fractions were determined [9]. Initial experiments to determine linearity of reaction with respect to both enzyme amount and time were conducted (data not shown). Product formation was kept to 10% or below for adherence to Michaelis-Menten assumptions. The assay methodology used was similar to described above for enzyme assay, except that different concentrations of xanthine i.e. 1, 2, 5, 7.5, 10, 12.5, 15, 17.5, 20, 25 and 50 µM were used. The velocity of reaction was determined as the nmol of uric formed per ml per min using extinction coefficient (e) of 12.2 mM-1 cm-1
HPLC determination of XO enzyme kinetic parameters
Km (Michaelis-Menten constant) and Vmax (maximum velocity) values for the oxidation of xanthine to uric acid with rat and mouse liver XO fractions were determined by HPLC. Initial experiments to determine linearity of reaction with respect to both enzyme amount and time were conducted (data not shown). Product formation was kept to 10% or below for adherence to Michaelis-Menten assumptions. For HPLC analysis, XO incubations were done as mentioned in the ‘spectrophotometric determination of XO enzyme kinetic parameters’ section except for the following changes. Incubations were performed in 10 ml vials which were placed in shaking water bath and the assay itself was conducted at 37°. The incubations were stopped after 5 min by addition 1 ml of 2% w/v ZnSO4 solution (in 50:50 methanol:water) as quenching agent. Samples were centrifuged in microcentrifuge for 5 min at Samples were centrifuged in microcentrifuge for 5 min at 7225×g and supernatants analyzed by HPLC. HPLC analysis was carried out using a Jasco HPLC system with PU-980 intelligent pump and UV 975 detector. Chromatographic separation was achieved using Supelcosil C18, 5 µm (150×4.6 mm) column. The mobile phase was 50 mM monobasic potassium phosphate:acetonitrile (97.5:2.5) and the flow rate was 0.5 ml/min [10]. Oxidized metabolite i.e. uric acid, was identified by comparison of retention time with standard uric acid. Calibration curve for uric acid was generated in the range of 0.1 µM - 40 µM for the quantitative determination of uric acid formed. The velocity of reaction was determined as the nmol of uric formed per ml per min.
Data analysis
Km and Vmax values were determined using the Lineweaver-Burk, Eadie-Hofstee and Hanes plotting methods. The line of best fit through the points on the plot was determined using linear regression by least squares method using Microsoft Excel (Microsoft Office XP).
Results and Discussion
Unit activity and specific activity of XO fractions from different animal liver cytosolic fractions were measured spectrophotometrically using xanthine (50 µM) as the specific probe substrate for XO. The mean unit XO activities and specific XO activities are listed in Table 1. XO activity was present in rat and mouse isolated liver XO fractions. XO activity was however absent in rabbit and guinea pig liver XO fractions. Further enzyme kinetic parameter estimations were therefore done only with rat and mouse liver XO fractions.
| Animal liver | Mean unit activity (units/ml of enzyme solution) | Mean specific activity (units/mg protein) |
|---|---|---|
| Rat | 106±20.7 | 20.8±4.74 |
| Mouse | 85.1±20.1 | 18.3±10.1 |
| Rabbit | Absent | Absent |
| Guinea pig | Absent | Absent |
One unit of enzyme activity was defined as the amount of enzyme that catalyzed the production of 1 nmol of uric acid per min under the stated conditions, when xanthine was used as the substrate. The values listed are the mean values ± standard deviation, obtained from three liver fractions for each animal, each experiment being conducted in duplicate
Table 1: Unit Activity And Specific Activity Of Xanthine Oxidase In Rat, Mouse, Rabbit And Guinea Pig Liver Fractions
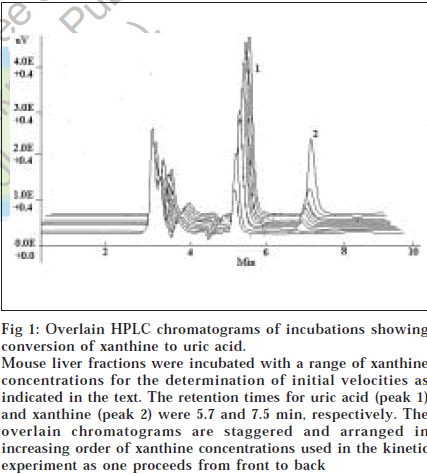
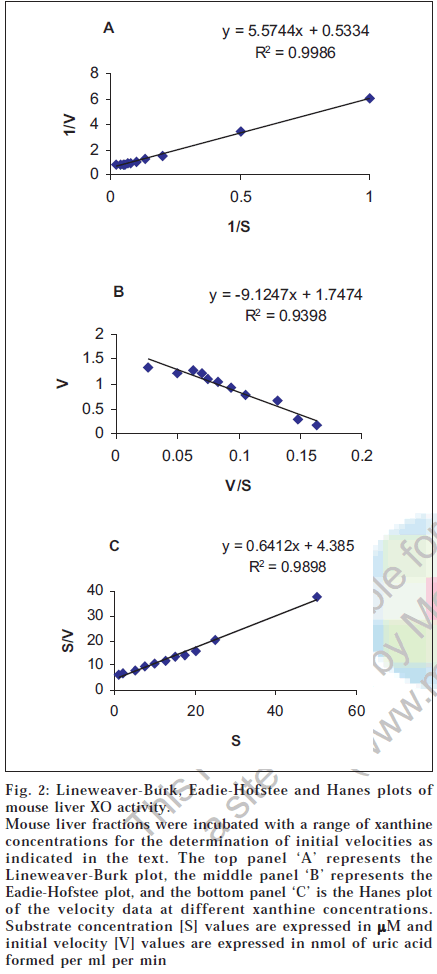
Km and Vmax values for the oxidation of xanthine to uric acid by xanthine oxidase fractions were measured by both spectrophotometric and HPLC methods, for rat and mouse liver XO liver fractions. Mean values of K m and Vmax of xanthine oxidase by spectrophotometric method as determined using three different plotting methods viz. Lineweaver-Burk plot, Eadie-Hofstee plot and Hanes plot are listed in Table 2 [9,11,12]. The typical chromatogram of xanthine oxidation incubation to uric acid as observed by HPLC analysis is depicted in fig. 1. The retention times for uric acid and xanthine were 5.73 and 7.52 min, respectively. Mean values of Km and Vmax of xanthine oxidase by HPLC method were also determined by three different plotting methods viz. Lineweaver-Burk plot, Eadie-Hofstee plot and Hanes plot and are given in Table 2.
| Animal | LB plot | EH plot | Hanes plot | |||
|---|---|---|---|---|---|---|
| Km | V max | Km | V max | Km | V max | |
| Rat (UV) | 9.71±5.03 | 2.00±1.05 | 7.44±3.25 | 1.10±0.463 | 5.32±2.74 | 1.57±0.726 |
| Rat (HPLC) | 13.8±4.24 | 2.55±0.894 | 9.82±1.95 | 2.13±0.611 | 8.17±1.42 | 1.96±0.538 |
| Mouse (UV) | 12.0±3.33 | 1.60±1.05 | 8.69±2.37 | 1.36±0.886 | 7.24±2.74 | 1.27±0.843 |
| Mouse (HPLC) | 9.67±3.28 | 2.15±0.914 | 7.88±2.84 | 2.00±0.940 | 5.73±4.00 | 1.848±0.962 |
The values listed are the mean values ± standard deviation, obtained from three liver fractions for each animal, each experiment being conducted in duplicate. LB – Lineweaver-Burk, EH – Eadie Hofstee. Km values are expressed in μM and V max values are expressed in nmol of uric acid formed per ml per min
Table 2: Values Of Km And Vmax Of Xo For Xanthine By Uv And Hplc Assays Using Lb, Eh, And Hanes Plots
Fig 1: Overlain HPLC chromatograms of incubations showing conversion of xanthine to uric acid. Mouse liver fractions were incubated with a range of xanthine concentrations for the determination of initial velocities as indicated in the text. The retention times for uric acid (peak 1) and xanthine (peak 2) were 5.7 and 7.5 min, respectively. The overlain chromatograms are staggered and arranged in increasing order of xanthine concentrations used in the kinetic experiment as one proceeds from front to back
In present study, high xanthine oxidase activity was detected in rat and mouse liver whereas, XO activity was absent in rabbit and guinea pig livers. This was expected as previous studies have indicated that little or no XO is present in rabbit and guinea pig livers [13,14]Our study also indicates that the standard deviation values associated with the K m values by HPLC method are less as compared to spectrophotometric method, suggesting that the HPLC method is more precise than the spectrophotometric method. The kinetic parameters i.e. K m and V max were determined using three Lineweaver-Burk plot, Eadie-Hofstee plot and Hanes plot. Of the three plotting methods, it is suggested that the Eadie-Hofstee plot is superior to the other two methods of plotting data [9,11,12]. This is due to use of low velocity values (that have most errors) without any transformation, rather than in reciprocal form as in Lineweaver-Burk and Hanes methods. Further, the Eadie-Hofstee plot is more suited to detection of both, deviation from linearity with changing substrate concentrations and detection of data of lower quality [10-12]. One representative mouse liver fraction XO kinetic parameter estimation data plotted by the three methods is shown in fig. 2. As seen in fig. 2, the superiority of EH plot is evident by the lower correlation coefficient value (0.9398) obtained as compared to the other two methods (0.9986 and 0.9898) due to the inherent stringency of this plotting method. The reported Km value for xanthine oxidation by XO ranges from 1-14 µM [15,16]. For bovine milk xanthine oxidase the reported Km by spectrophotometric method using Lineweaver-Burk plot is 8 µM [15,16]. The results obtained in this study shows that the K m values as determined by Lineweaver-Burk plot, for rat and mouse liver XO fractions were 9.71 ± 5.03 µM and 12.0 ± 3.33 µM, respectively, which are in reasonable agreement with the reported values, since XO structure is fairly conserved across species. The values of Kdetermined by different plotting methods by both UV and HPLC are also in the same range (5.32-13.8 µM). Further, the values for K m as obtained by spectrophotometric and HPLC methods are also in close agreement even though HPLC assays were done at 37° as opposed to ambient temperature for UV assays. Further, the estimated Vmax values were consistently higher in HPLC based assays. This is expected since incubations at higher temperatures generally yield higher Vmax values due to enhanced collision events between enzyme and substrate.
Fig. 2: Lineweaver-Burk, Eadie-Hofstee and Hanes plots of mouse liver XO activity. Mouse liver fractions were incubated with a range of xanthine concentrations for the determination of initial velocities as indicated in the text. The top panel ‘A’ represents the Lineweaver-Burk plot, the middle panel ‘B’ represents the Eadie-Hofstee plot, and the bottom panel ‘C’ is the Hanes plot of the velocity data at different xanthine concentrations. Substrate concentration [S] values are expressed in µM and initial velocity [V] values are expressed in nmol of uric acid formed per ml per min
Overall, this study presents a comparison of two methods for the determination of enzyme kinetic parameters in crude fractions of XO (isolated using reported methods), with xanthine as the model substrate. Such assays are of utility is the establishment of drug metabolism study protocols in drug metabolism research.
The authors would like to extend special thanks to Dr. M. L. Kubal, Wockhardt Ltd., Aurangabad for providing HPLC columns used in this work. The authors also wish to thank AICTE (File No. 8019/RDII/R&D/PHA (213) 2000-01) and DST-“FIST Program”-2003 [485] for providing the research grant for this work.
References
- Garattini, E., Mendel, R., Romao, M.J., Wright, R. and Terao, M., Biochem. J., 2003, 372, 15.
- Garry, R.B. and Larry, W.O., 2001, http: // www. medicine.uiowa.edu /frrb /education /freeradicalSp01 /paper /203 /Lilpaper3. pdf.
- Dambrova, M., Uhlen, S., Nelch C.J. and Wikberg, J.E.S., Eur. J. Biochem., 1998, 257, 178.
- Evans, C.E., Mohammed, A.V. and Patience, O.E., Biokemistri, 2005, 17, 1.
- David, J.T., J. Biol. Chem., 1994, 269, 27932.
- Maia, L. and Mira, L., Arch. Biochem. Biophys., 2002, 400, 48. Calazyme Manufacturers of Enzyme, Xanthine oxidase, June-2004, pg 1-2, http://www.freedoniagroup.com/pdf/1353smwe.pdf.
- Carpani, G., Racchi, M., Ghezzi, P., Terao, M. and Garattini, E., Arch. Biochem. Biophys., 1990, 279, 237.
- Cell and Molecular Biology Lab, Part-3, Xanthine oxidase Km and Vmax, BIL-256, Spring-2005, http://fig.cox.miami.edu/~ddiresta/bil256/ lab31.pdf.
- Cooper, N., Brown, P., Xu, L., DiGiorgio, A., Zhu, L., Lin, P., Lee, J., Khosravan, R. and Bopp, B., Bioanalysis of xanthine, hypoxanthine and uric acid in urine by HPLC and LC/MS/MS method: Method Comparison for pharmacodynamic biomarkers., 2001, www.mdsps.com.
- Houston, J.B., Kenworthy, K.E. and Galetin, A., In; Lee, J.S., Obach, R.S. and Fisher, M.B., Eds., Drug Metabolizing Enzymes Cytochrome P450 and Other Enzymes in Drug Discovery and Development, FontisMedia S.A. and Marcel Dekker Inc., Netherlands and NY-USA, 2003, 211.
- Dixon, M. and Webb, E.C., Eds., Enzymes, 3rd Edn., Academic Press Inc., NY, USA, 1979, 60.
- Panoutsopouios, G.I., Kouretas, D., Gounaris, E.G. and Beedham, C., Basic Clin. Pharmacol. Toxicol., 2004, 95, 273.
- Lang, D. and Kalgutkar, A.S., In; Lee, J.S., Obach, R.S. and Fisher, M.B., Eds., Drug Metabolizing Enzymes – Cytochrome P450 and
- Other Enzymes in Drug Discovery and Development, FontisMedia S.A. and Marcel Dekker Inc., Netherlands and NY-USA, 2003, 483.
- Rashidi, M.R., Smith, J.A., Clarke, S.E. and Beedham, C., Drug Metab. Dispos., 1997, 25, 805.
- Krenitsky, T.A., Hall, W.W., Miranda, P., Beauchamp, L.M., Schaeffer, H.J. and Whiteman, P.D., Proc. Natl. Acad. Sci. USA., 1984, 81, 3209.