- *Corresponding Author:
- Neetu Jain
Department of Botany, S.S. Jain Subodh P.G. (Autonomous) College, Jaipur 302007, Rajasthan, India
E-mail: neetugodika@yahoo.co.in
| Date of Received | 20 June 2021 |
| Date of Revision | 23 October 2022 |
| Date of Acceptance | 16 November 2022 |
| Indian J Pharm Sci 2022;84(6):1369-1379 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
Thymus vulgaris L. commonly known as thyme or garden thyme belonging to family Lamiaceae is a pleasant smelling flowering perennial shrub, cultivating all over the world. Thyme has great importance due to the possibility of its use in various applications, as food additives, in medicines and in cosmetic industry. Thyme leaves are highly aromatic therefore commonly used fresh or dried as a seasoning in a variety of culinary applications including soups, stews, sauces, meat, fish dishes and for flavoring liqueurs, herbal tea preparations. Thymus is an important medicinal plant, highly endorse due to a broad range of therapeutic properties of their chemical components such as antirheumatic, antidermatophytic, antioxidants, antiseptic, antispasmodic, antimicrobial, cardiac, carminative, astringent, diuretic and expectorant. The plant is also useful against cough, cold, chest infections, diabetes and for digestive upset. Flavonoids have good potential as antioxidants and antifungal behavior. The present review article gives comprehensive information about various medicinal and traditional utility and pharmacological activities of the thymus plant and its constituents.
Keywords
Thymus vulgaris, bioactive constituents, biological activities
In traditional medicine, plants and their products have been used since ancient times for the protection and treatment of several infectious diseases. The medicinal plant is a principal integrant of native medical systems all over the world. In recent years, there are a gradual revival of interest in the use of herbal medicines and food supplements as an alternative therapy for infectious diseases due to their high content of secondary byproducts such as polyphenols, i.e. flavonoids, tannins and alkaloids, steroids, essential oil, etc[1,2].
The genus thymus comprises approximately 400 species, several of which are widely used in traditional medicine[3]. Thymus vulgaris (T. vulgaris) L. belonging to the family Lamiaceae is a pleasant-smelling perennial shrub, cultivated all over the world. Thyme grows well in a temperate to heat, dry, sunny climate and wherever the plants don’t seem to be shaded.
It is a bushy, woody based shrub, around 10 to 40 cm high with small and highly aromatic gray-green oval leaves containing numerous small glands with clusters of pink or purple flowers. Thyme leaves are very small, usually 2.5 to 5 mm long and vary significantly in form like linear/ linear-lanceolate, ovate or oblong and with an acute apex, obtuse base tapering into a petiole and revolute margins. The upper surface is light gray or light brownish gray to weak olive green with numerous hairs while the lower surface is grayish, pubescent and glandular-punctate. Flowers are present in the axillary whorls, the calyx are 4 mm in length and tubular, bilabiate, pubescent in shape. The corolla is about twice as long as the calyx, purplish and bilabiate. The stamens are didynamous and style is with bilobed stigma[4].
Plant extracts from T. vulgaris have been used in traditional medicine for the treatment of several respiratory diseases like bronchitis, chronic obstructive pulmonary disease, asthma and the treatment of several other pathological disorders due to various properties such as antimicrobial, antifungal, antioxidative antiseptic, antispasmodic, antitussive and antiviral[5]. Thymus oil is widely used in phytotherapy, skin infections like acne, hypertension, infections and cancers[6].
The oil is also beneficial in boosting the immune system and helps to fight colds, flu, infectious diseases and chills. It is proved to be a urinary antiseptic, being very helpful for cystitis and urethritis. Thyme is widely used for seasoning vegetables, fish, soups, poultry farms, for flavoring liqueurs, herbal tea preparations.
Chemical Constituents
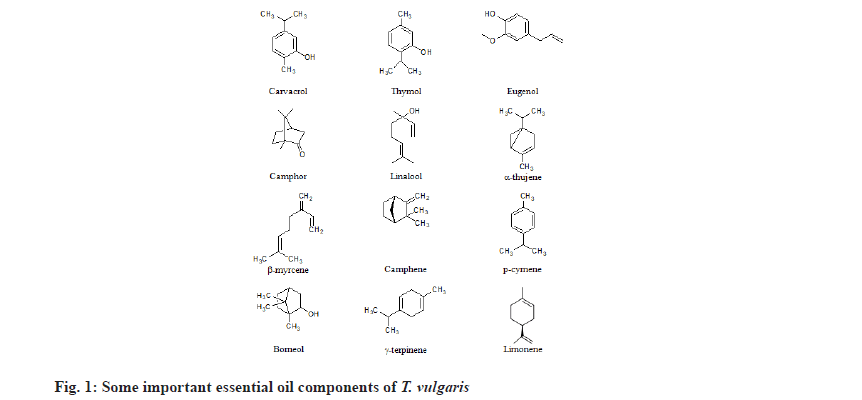
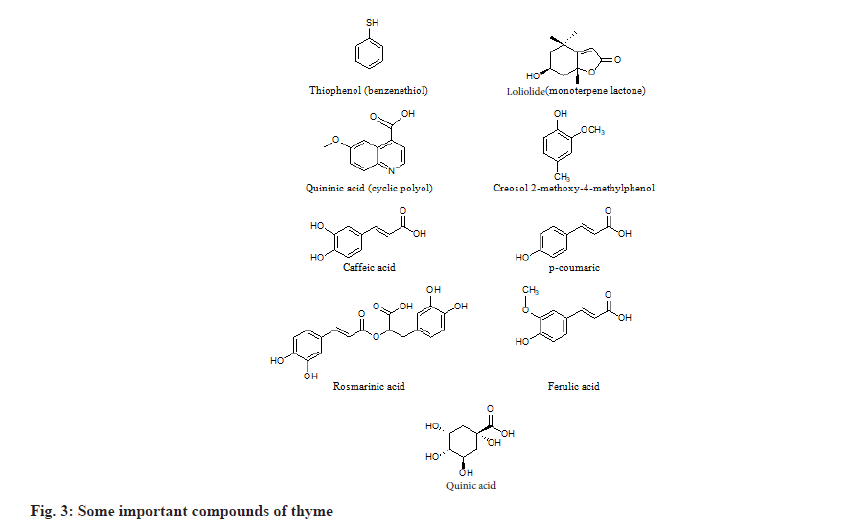
T. vulgaris L. contain a myriad of biochemical compounds such as steroids, terpenoids, flavonoids, alkaloids, tannins, saponins, etc. Thyme is one of the major sources of monoterpene phenolic compounds. They contained thymol, carvacrol, p-cymene, α-pinene, linalool, borneol and 1, 8 cineole[7]. Heidari et al.[8] identified creosol 2-methoxy-4-methylphenol, thiophenol (benzenethiol), loliolide, 3-methoxy-5-methylphenol, quininic acid through Gas Chromatography-Mass Spectrometry (GC-MS) analysis as shown in fig. 1.
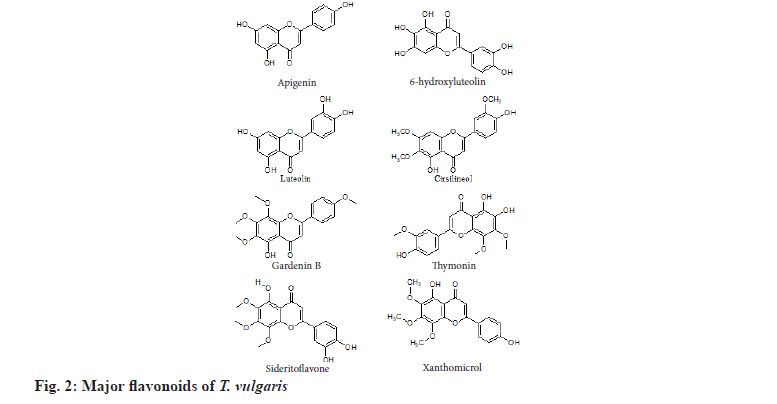
Thyme is also a rich source of flavonoids (fig. 2). Major flavones are apigenin, 6-hydroxyluteolin, luteolin, methyl-flavones like cirsilineol, 5-demethylnobiletin, 8-methoxycirsilineol, cirsimaritin, 7-methoxy-luteolin, gardenin B, thymonin, Sideritoflavone, xanthomicrol and thymosin[9]. Rubey et al.[10] explained that methanolic fraction of T. vulgaris contained phenolic acid like caffeic acid, cinnamic acid, p-coumaric acid, rosmarinic acid, ferulic acid, quinic acid, carnosic acid, caffeoylquinic acid; flavones like quercetin-7-O-glucoside; flavanones like apigenin and naringenin (fig. 3).
Thymol is major constituent of oil ranging from 42.6 %-57.8 % according to collection time and cultivation place[11]. Other components are α-cymene, carvacrol, α-thujene, α-pinene, β myrcene, trans-ocimene, γ terpinene, limonene. Prasanth et al.[12] reported high content of oxygenated monoterpene (56.53 %) and low contents of monoterpene hydrocarbons (28.69 %), sesquiterpene hydrocarbons (5.04 %) and oxygenated Sesquiterpenes (1.84 %) in the essential oil of T. vulgaris. The predominant compound among the essential oil components was thymol (51.34 %) while the amount of all other components of the oil was found to be less than 19 %. Imelouane et al.[13] classified the essential oil components of T. vulgaris into five classes, monoterpene hydrocarbons, oxygenated monoterpene, sesquiterpene hydrocarbons, oxygenated Sesquiterpenes and others. Based on GC and GC-MS analysis of the essential oil of thyme 41 components were identified, which represented 97.85 % of the total detected constituents. The major constituents of the oil were camphor (39.39 %), camphene (17.57 %), α-pinene (9.55 %), 1,8-cineole (5.57), borneol (5.03 %), β-pinene (4.32 %). Other components were present in amounts less than 2 %. The essential oil from thyme contained camphene, α pinene, β-pinene, myrcene the most important monoterpene hydrocarbons. In particular, oxygenated monoterpenes were the most abundant compound group of the oil (54.82 %). According to Uhl[14] thyme contains high concentrations of phenols, including thymol (12 %-61 %), carvacrol (0.4 %-20.6 %), 1,8-cineole (0.2 %-14.2 %), q-cymene(9.1 %-22.2 %), linalool (2.2 %-4.8 %), borneol (0.6 %-7.5 %), α-pinene (0.9 %-6.6 %) and camphor (0 %-7.3 %). Carvacrol and thymol are the main phenolic components that are primarily responsible for their antioxidative activities.
Thyme is also a good source of vitamins[15]. It is particularly rich in Vitamin A and Vitamin C. Vitamin A is an antioxidant property while Vitamin C provides resistance against infectious diseases. It is also a good source of Vitamin B6 or pyridoxine which acts as a stress buster. A trace amount of Vitamin K, Vitamin E and folic acid were also reported in this herb[16]. Thymus leaves are excellent sources of minerals like potassium, calcium, iron, manganese, magnesium and selenium. Minerals are vital for optimum growth.
Antimicrobial Properties
Sartoratto et al.[17] studied antimicrobial activates of T. vulgaris by a bio autographic method. Minimal Inhibitory Concentration (MIC) of essential oil was determined by micro dilution method. T. vulgaris was found effective against Enterococcus faecium and Salmonella cholerasuis. Rota et al.[18] studied the chemical compositions and antimicrobial activity of T. vulgaris (thymol chemotype) along with Thymus zygis (T. zygis) subsp. gracilis (thymol and two linalool chemo types) and Thymus hyemalis (T. hyemalis) Lange (thymol, thymol/linalool and carvacrol chemo types) essential oils extracted from seven plants cultivated in Murcia (Spain). They screened essential oils for their antimicrobial activities against ten pathogenic organisms and suggested that T. vulgaris possesses antimicrobial properties and are a potential source of antimicrobial ingredients for the food industry. Millezi et al.[19] studied the antimicrobial properties of T. vulgaris essential oil against five important foodborne pathogens Staphylococcus aureus (S. aureus) ATCC 25923, Escherichia coli (E. coli) ATCC 25922, Listeria monocytogenes ATCC 19117, Salmonella enterica Enteritidis S64, and Pseudomonas aeruginosa (P. aeruginosa) ATCC 27853. T. vulgaris was found to be more effective against gram-positive bacteria. Borugă et al.[20] studied the chemical composition and antimicrobial properties of the essential oil of T. vulgaris cultivated in Romania. GC and GCMS analysis showed major components were p-cymene (8.41 %), γ-terpinene (30.90 %) and thymol (47.59 %). Disc diffusion method was applied for the screening of antimicrobial activity against food deteriorates bacteria and fungi. T. vulgaris essential oil showed strong antimicrobial properties and may in the future represent a new source of natural antiseptics with applications in the pharmaceutical and food industry.
Fani et al.[21] studied the antimicrobial activity of thymus essential oil against 30 clinical isolates of each of Streptococcus pyogenes, Streptococcus mutans, Candida albicans (C. albicans), Porphyromonas gingivalis (P. gingivalis) and Aggregatibacter actinomycetemcomitans (A. actinomycetemcomitans). The MIC values for C. albicans, A. actinomycetemcomitans, and P. gingivalis were found to be 16.3, 32 and 32 mg/ml, respectively.
Antibacterial Properties
Nadia et al.[22] estimated the total polyphenols and flavonoids contains of T. vulgaris for antibacterial and antioxidant activities. Quercetin, luteolin, apigenin, kaempferol, chrysine were reported as major flavonoid contains. These isolated flavonoids showed moderate antibacterial activity against E. coli ATCC 25922.
A comparative analysis of the chemical composition and antioxidant and antibacterial activities of Thymus caucasicus, Thymus kotschyanus and T. vulgaris essential oils obtained from the aerial parts was carried out by Asbaghian et al.[23]. Antibacterial activity was determined by the broth dilution method. T. vulgaris essential oil showed excellent biological activity against E. coli (12.5 µg/ml) and Streptococcus faecalis (25 µg/ml). Among the Gram-positive bacteria, S. aureus was found to be the least sensitive (100 µg/ml). Among the Gram-negative bacteria, T. vulgaris oil exhibited slight activity against Salmonella typhi. Kon et al.[24] studied the antibacterial activity of T. vulgaris essential alone and in combination with 34 other essential oils against S. aureus and E. coli through the disk diffusion technique. T. vulgaris and Cinnamomum zeylonicum essential oils combination showed synergistic effect with 0.26 Fractional Inhibitory Concentration (FIC) index. Al-Balushi et al.[25] studied antibacterial and cytotoxic activity of petroleum ether, chloroform and hydro alcoholic extracts of thymus leaves collected from Oman. Antibacterial activity was measured using disc diffusion method against S. aureus, E. coli, P. aeruginosa and Klebsiella pneumonia. T. vulgaris leaves extracts showed very strong results, inhibition zones ranged from 7-20 mm.
Fadil et al.[26] studied the sensibility of Salmonella typhimurium strain, with a mixture of T. vulgaris L., Rosmarinus officinalis L. and Myrtus communis L. essential oils. T. vulgaris essential oil showed stronger antibacterial effect as compared to others. They suggested that a formulation comprising 55 % of T. vulgaris L. and 45 % of Myrtus communis L. essential oils can be considered for the increase of Salmonella typhimurium sensibility. Benameur et al.[27] evaluated the susceptibility of bla-Extended-Spectrum Beta-Lactamase (blaESBL) producing Enterobacteriaceae to T. vulgaris essential oil alone and in combination with cefotaxime. The identified strains were Multi-Drug Resistant (MDR). Thyme oil showed high activity against all MDR strains, including blaESBL producing isolates, with inhibition zones and MIC values in the range of 24-40 mm/10 μl and 2.87-11.5 μg/ml, respectively. Thyme oil in combination with Cefotaxime showed a synergistic action against blaSHV-12 producing E. coli (FICI 0.28) and additive effect vs. ESBL producing Enterobacter cloacae (FICI 0.987).
Antioxidant Properties
Antioxidants are substances that can prevent or slow damage to cells caused by free radicals, unstable molecules that the body produces as a reaction to environmental and other pressures. Therefore, antioxidants are known as free-radical scavengers which help in neutralizing free radicals in our bodies and boost overall health. The phenolic compound isolated from hexane extract of thyme, p-cymene-2,3-diol(2,3-dihydroxy-4-isopropyl-1-methylbenzene) showed strong antioxidant activity which was greater than those of a tocopherol and butylated hydroxyanisole[28]. Abdalla et al.[29] studied the antioxidant activity of acetone extract of thyme evaluated in sunflower oil and its 20 % oil-in-water emulsion. The thyme extracts inhibited the generation of hexanal and pentanal in both the oil and in the emulsion. Lee et al.[30] screened thymus and basil leave extracts for chemical composition and antioxidant properties using the aldehyde/carboxylic acid assay. Eugenol, thymol, carvacrol and 4-allylphenol showed stronger antioxidant activities. They all inhibited the oxidation of hexanal by almost 100 % for a period of 30 d at a concentration of 5 μg/ml. Their antioxidant activities were comparable to those of the known antioxidants, α-tocopherol and Butylated Hydroxytoluene (BHT). Kulisic et al.[31] also studied antioxidant activities of T. vulgaris (Thyme) and T. serpyllum L. (wild thyme) essential oils. Thyme and wild thyme essential oils showed significant in vitro antioxidant activity, while fractions of the both essential oils show antioxidant activity in the Thiobarbituric Acid Reactive Substance (TBARS) assay.
VukoviÄ et al.[32] studied antimicrobial and antiradical activity of Origanum vulgare L. and T. vulgaris essential oil. T. vulgaris essential oil (9.69 µl/ml after 30 min; 5.84 µl/ml after 60 min) showed lower antiradical activity in comparison to BHT and higher activity in comparison to ascorbic acid.
Roby et al.[33] evaluated the thyme (T. vulgaris L.), sage (Salvia officinalis L.), and marjoram (Origanum majorana (O. majorana) L.) extracts for their antioxidant activity, total phenols and phenolic compounds. They found that thyme methanol extract possessed the best antioxidative activity as compared to other plants, α-tocopherol and butylated hydroxyanisole. Miladi et al.[34] also determined antioxidant activity of thymus oil using a quantitative 1,1-Diphenyl2-Picryl Hydrazyl (DPPH) assay. Thymus exhibited effective radical scavenging capacity with 50 % Inhibitory Concentration (IC50) of 437±5.46 µg/ml and 189±2.38 µg/ml respectively and therefore acts as a natural antioxidant agent.
Chizzola et al.[35] studied leaves extract of thymus and commercial thyme rich in thymol for antioxidative potential in various extracts. The assays for antioxidative activity were the total phenolic according to the Folin-Ciocalteu method, DPPH decoloration and Ferric Reducing Antioxidant Power (FRAP). The best results were obtained with 60 % ethanol as an extractant. Essential oils with high proportions of phenolic compounds like thymol and Carvacrol exhibited excellent antioxidant activity. El-Nekeety et al.[36] experimented to work out the elements of T. vulgaris L. oil and to evaluate the protecting effects of this oil against aflatoxin induce oxidative stress in rats. Gedikoğlu et al.[37] obtained the essential oils T. vulgaris (thyme) and Thymbra spicata (zahter) by Hydro Distillation (HD) and Microwave-Assisted Extraction (MAE) methods, and studied free radical scavenging activity (IC50), FRAP value and antimicrobial properties. Thyme essential oil had higher antioxidant capacity for both HD and MAE in comparison with zahter essential oil. Methanol extracts of both thyme and zahter showed higher phenolic composition in comparison with their ethanol extracts. Antioxidant activity was determined by two assays the DPPH free radical scavenging activity and the FRAP. The free radical scavenging activity of thyme essential oil obtained with MAE (93.77±13.0 µg/ml) was significantly (p<0.05) higher than that found with HD (159.59±12.79 µg/ml). According to Assiri et al.[38] thymus oil had strong antiradical action wherein 65 % of DPPH radicals and 55 % of galvinoxyl radical were quenched after 60 min of incubation.
Antifungal Properties
Thymus oils are found to be the greatest inhibitors of fungal pathogens because of the presence of phenolic compounds such as thymol which might disrupt the fungal cell membrane. Numpaque et al.[39] studied the behavior of the antifungal nature of thymol. According to them thymol altered the hyphal morphology and caused hyphal aggregates, resulting in reduced hyphal diameters and lyses of hyphal wall.
Giordani et al.[40] studied the antifungal potential of essential oils isolated from various chemo types of T. vulgaris against C. albicans. The essential oil of the thymol chemo types of T. vulgaris was the most potent, with a MIC 80 % of 0.016 µl/ml, where the efficacy was mainly due to the high level of thymol (63.2 %). Šegvić Klarić et al.[41] studied the antifungal activities of essential oil of thyme (T. vulgaris L.) and pure thymol, as comparative substance, on different mold species like Aspergillus, Penicillium, Cladosporium, Trichoderma, Mucor and Rhizopus isolated from damp dwellings. MIC of both thymol and essential oil were below 20 μg/ml, except for Mucor spp. (50.2 μg/ml). Thymol exhibited approximately three times stronger inhibition than essential oil of thyme. The vaporous phase of the thyme essential oil (82 μg/ml) in glass chambers strongly suppressed the sporulation of molds during 60 d of exposure. According to them, thymol is lipophilic component, enabling it to interact with the cell membrane of fungus cells, altering cell membrane permeability by permitting the loss of macromolecules. Similar results were also reported by various researchers[21,42,43].
Mota et al.[44] studied the antifungal activity of T. vulgaris essential oil and their component against Rhizopus oryzae. Moghtader[45] studied the antifungal effects of the T. vulgaris L. essential oil and comparison with synthetic thymol on Aspergillus niger. The results showed that thymus oil 1, 1/2 and 1/4 oil dilutions exhibit strong antifungal activity than streptomycin sulphate (72 % SP) and gentamycin (8 mg/ml) antibiotics and that exhibited on strong synthetic thymol was at 10 % dilution. The high antifungal properties of thymus oil were due to the presence of natural monoterpene phenol thymol.
Gucwa et al.[46] studied the activities of T. vulgaris, Citrus limonum, Pelargonium graveolens, Cinnamomum cassia, Ocimum basilicum and Eugenia caryophyllus essential oils distributed against a group of 183 clinical isolates of C. albicans and 76 isolates of Candida glabrata. All of the oils exhibited both fungistatic and fungicidal activity toward C. albicans and C. glabrata isolates. The highest thyme oil MIC concentration established for both C. glabrata and C. albicans was 5.731 mg/ml (0.625 % (v/v)), but predominant values were 0.734 mg/ml (0.08 % (v/v)) or less than 0.046 mg/ml (0.005 % (v/v)).
Cytotoxicity
Miladi et al.[34] screened T. vulgaris L. and Rosmarinus officinalis L essential oils for their in vitro cytotoxic effects against human respiratory epithelial cell line (A549). Cytotoxicity was measured using 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphynyltetrazolium Bromide (MTT) colorimetric assay. Dose-dependent studies revealed IC50 of 8.50±0.01 µg/ml and 10.50±0.01 µg/ml after 72 h on the A549 cells these extracts were not cytotoxic towards A549 cell line in all tested concentrations. Al-Balushi et al.[25] studied antibacterial and cytotoxic activities of petroleum ether, chloroform and hydro alcoholic extracts of thymus leaves collected from Oman. A brine shrimp test was used to estimate cytotoxic activity. Petroleum ether and chloroform extracts have almost killed all the shrimp larvae at higher concentrations of 1000 μg/ml. Lethal Concentration 50 (LC50) values for the two extracts were found to be 85.2 and 95.8 μg/ml, respectively. Polar fractions like hydro alcoholic extract had displayed very low cytotoxic activities.
Antispamodic Activity
T. vulgaris L. (Lamiaceae) is well known medicine with broncholytic and secretomotoric activity. According to van den Broucke et al.[47,48] phenols may be of minor significance for the antispasmodic effect on smooth muscles due to their small contents. They also studied the effect of flavonoids (luteolin, luteolin glycosides, apigenin, cirsilineol, 8-methoxycirsilineol and thymin) as possible spasmolytic compounds.
Begrow et al.[49] examined various extract preparations of thyme; those with normal and very low thymol (and carvacrol) concentrations. Also, thymol and carvacrol alone were investigated. Functional studies on two different organs (ileum and trachea contraction and relaxation) were used as well as mucociliary clearance (velocity of ciliary transport) in vivo. Thymol and Carvacrol were found effective in smooth muscles such as rat ileum and trachea independent of the type of stimulation (acetylcholine, K+ or Ba++). While thyme extract with low thymol contents reduced contractions by ~40 % when Ba++ or endothelin-1 were used as stimulants indicated that thymol is not the compound of major importance.
Micucci et al.[50] chemically analyzed a new T. vulgaris L. solid essential oil formulation composed of liquid essential oil linked to solid excipients and evaluated for its intestinal spasmolytic and anti spastic effects in ex vivo ileum and colon of guinea pig and compared with liquid essential oil and excipients. The essential oil was found most effective in decreasing basal contractility in ileum and colon while excipients addiction permitted normal contractility pattern in solid linked essential oil. In ileum and colon, the solid formulation of T. vulgaris L. exerted the relaxant activity on K+-depolarized intestinal smooth muscle as well as liquid essential oil. Engelbertz et al.[51] explained that thymol alone did no effect on endothelia-induced trachea contraction.
Antidermatophytic Activity
Sokovic et al.[52] of studied the effect of thymus oil against human pathogenic fungi like Trichophyton mentagrophytes, Trichophyton rubrum and Trichophyton tonsurans. The therapeutic efficacy of a 1 % solution of the essential oil of T. vulgaris and thymol as well as the commercial preparation bifonazole was evaluated. All animals were cured after 37 d of observation period.
Jain et al.[11] reported strong antidermatophytic activity of T. vulgaris essential oil against all selected filamentous fungi namely Trichophyton mentagrophytes, Trichophyton rubrum, Trichophyton tonsurans, T. soudanense, Microsporum fulvum and Microsporum gypseum. MIC and Minimum Fungicidal Concentration (MFC) were determined by semi solid agar antifungal susceptibility testing method. MIC was ranged between 0.020±0.000 to 0.1±0.033 μl/ml. MFC were ranged from 0.020±0.00 μl/ml to 0.2±0.000 μl/ml. These promising antifungal activities against all selected pathogenic fungi may be due to the presence of a high concentration of phenolic compounds.
Cardiovascular Treatment
Ramchoun et al.[53] studied the antioxidant, hypercholesterolemia and hypotriglyceridemic activities of T. vulgaris aqueous extracts. Screening of antioxidant activity of polyphenol-rich extracts was carried out by using the radical scavenging activity method, FRAP assay and by the inhibition of the 2,2'-azobis (2-amidinopropane) hydrochloride induced oxidative erythrocyte hemolysis. Intraperitoneal injection of Triton WR-1339 (at a dose of 200 mg/kg body weight) was used for the induction of hyperlipidemia in rat. After 24 h of treatment with polyphenol-rich extract of T. vulgaris no significant effect on both plasma total cholesterol and triglycerides profiles was observed.
Ocaña A et al.[54] studied the effects of thyme extract oils (from T. vulgaris, T. zygis and T. hyemalis) on cytokine production and gene e of oxidized Low-Density Lipoproteins (oxLDL)-stimulated THP-1-macrophages. These cells were incubated with the thyme fraction oils and the productions and gene expressions of the inflammatory mediators Tumor Necrosis Factor-Alpha (TNF-α), Interleukin (IL)-1B, IL-6, and IL-10 were determined. Thyme extracts significantly reduced production and gene expression of the proinflammatory mediators TNF-α, IL-1B, and IL-6 and highly increased these parameters on the anti-inflammatory IL-10 cytokine.
Anti-Inflammatory
Vigo et al.[55] reported that thyme oil reduced nitric oxide production by lipopolysaccharide and interferon-gamma in a dose-dependent manner in murine macrophage cell line J774A.1. They suggested that the inhibition of net nitric oxide production may be due to their nitric oxide scavenging activity and their inhibitory effects on inducible nitric oxide synthase gene expression. Thymol is the major constituent responsible for anti-inflammatory activity[56].
Antiviral Activity
Rezatofighi et al.[57] evaluated T. vulgaris extracts against Newcastle disease virus in Ovo. Egg toxicity assay was performed using embryonated eggs to determine the maximum non-toxic concentration. Inhibition percentage was determined as 10 (1.75), which indicated the ability of the extracts to reduce the viral potency by more than 56 folds.
Kaewprom et al.[58] studied the antiviral activity of T. vulgaris and Nepeta cataria hydrosols against porcine reproductive and respiratory syndrome virus. They revealed that T. vulgaris hydrosol significantly reduced the Porcine Reproductive and Respiratory Syndrome Virus (PRRSV) load. The anti-PRRSV activity occurred in both pre-entry and post-entry steps.
Nolkemper et al.[59] examined aqueous extracts isolated from species of the Lamiaceae family for their antiviral activity against Herpes Simplex Virus (HSV). Among all extracts T. vulgaris extracts exhibited inhibitory activity against HSV-1, type 2 (HSV-2) and an acyclovir-resistant strain of HSV-1 which were tested in vitro on RC-37 cells in a plaque reduction assay.
Vimalanathan et al.[60] evaluated several essential oils and some of their major constituents for their possible anti-influenza virus properties in both liquid and vapor phases. T. vulgaris displayed 100 % inhibitory activity at 3.1 µl/ml concentration in liquid phase.
Insecticidal Activity and Larvicidal Activity
El-Akhal et al.[61] evaluated the properties of larvicidal activity of essential oils of T. vulgaris and O. majorana family of Lamiaceae collected at Taounate province in the North East of Morocco, against the larvae of the malaria vector Anopheles labranchiae (Diptera: Culicidae). The mortality percentages were determined after 24 h. LC50 and LC90 were calculated and measured. They were respectively of the order of 351.63 µg/ml and 621.34 µg/ml for the essential oil of T. vulgaris whereas O. majorana were found of the order of LC50=107.13 µg/ml and LC90=365.9 μg/ml.
Szczepanik et al.[62] studied insecticidal activities of T. vulgaris essential oil and its components (thymol and Carvacrol) against larvae of lesser mealworm, Alphitobius diaperinus (A. diaperinus). The insecticidal activity of thyme volatile oil, thymol and Carvacrol were evaluated against completely different larval stages of lesser mealworm. The sooner and later larval stages were reared on diets containing one or two acetone solutions of tested compounds. Insecticidal activity of thyme volatile oil and pure monoterpenes against A. diaperinus larvae relied on the dose and age of larvae. The growth of younger larvae was considerably affected, whereas those of the older larval stage were less influenced and only by pure oil components. In young larvae the application 1 % thyme oil, thymol and Carvacrol, caused mortality of 50.0, 86.67 and 85 %, respectively. Saroukolai et al.[63] also examined insecticidal properties of Trigonodactylus persicus essential oil against Tribolium castaneum and Sitophilus oryzae. Rodriguez et al.[64] studied the larvicidal and cytotoxic activities of extracts from 11 native plants from North Eastern Mexico and found excellent results.
Anticancer Activity
Naturally-occurring mixtures of phytochemicals present in plant foods are proposed to possess tumor-suppressive activities. Kubatka et al.[65] studied the antitumor effects of T. vulgaris L. in vivo and in vitro mammary carcinoma models. Dried T. vulgaris was continuously administered at two concentrations of 0.1 % and 1 % in the diet in a chemically-induced rat mammary carcinomas model and a syngeneic 4T1 mouse model. After an autopsy, histopathological and molecular analyses of rodent mammary carcinomas were performed. In mice, T. vulgaris at both doses reduced the volume of 4T1 tumors by 85 % (0.1 %) and 84 % (1 %) compared to the control, respectively. Treated tumors showed a substantial decrease in necrosis/tumor area ratio and mitotic activity index. T. vulgaris L. demonstrated significant chemo preventive and therapeutic activities against experimental breast carcinoma.
Solar Ultraviolet (UV) radiation-induced reactive oxidative species is mainly responsible for the development of photo aging. Sun et al.[66] extracted rosmarinic acid from T. vulgaris. T. vulgaris remarkably prevented the UVB-induced reactive oxygen species and lactate dehydrogenase. The researcher found that NF-E2-related factor 2 expression was regulated by dihydrolipoamide dehydrogenase, which was a tricarboxylic acid cycle-associated protein that decreased after UVB exposure. Besides, T. vulgaris significantly diminished UVB induced phosphorylation of mitogen activated protein kinases pathway, containing extracellular signal-regulated kinase, Jun N‐terminal kinase and p38, which consequently reduced phosphorylated c-fos and c-jun.
Anxiety
Komaki et al.[67] investigated the effects of extract of T. vulgaris on rat behavior in the Elevated Plus-Maze (EPM). During the experiments they studied the total distance covered by animals, the number of open and closed-arm entries and the time spent in open and closed arms. Researcher suggested that T. vulgaris may have an anxiolytic profile in rat behavior in the EPM test, which is not influenced by the locomotors activity.
Based upon available literature T. vulgaris emerged as a source of several potential biological components which showed broad spectrum pharmacological activities like antibacterial, antioxidant, antifungal, anti-inflammatory, antispasmodic, cytotoxicity, anticarcinogenic, nematicidal, larvicidial, and antiviral. The oil is also beneficial in boosting the immune system and helps to fight various ailments. Thyme is widely used for seasoning of vegetables, fish, soups and poultry farms, for flavoring liqueurs, herbal tea preparations.
Furthermore, considering its multifaceted medicinal uses, there is wide scope for future research especially in the field of antitumor and cytotoxic properties of essential oil and aqueous extracts.
The estimation of correct dosage of different plant fractions and their toxicological effect should be determined. Application of chemical constituents of T. vulgaris L. and their mixer for treatment of various illnesses can expose the doors to development of effective therapeutic agents.
Conflict of interests:
The authors declared no conflict of interests.
References
- Jain N, Sharma M, Kumar P. Regulatory effect of some plant extracts on the growth of dermatophytic fungi. Indian J Microbiol 2004;44:59-62.
- Jain N, Sharma M. Ethanobotany, phytochemical and pharmacological aspects of Thuja orientalis. Rev Int J Pure Appl Biosci 2017;5(3):73-83.
- Javadi H, Hesamzadeh HS, Shikhbaba BM. Chromosome reports on two species of thymus (Lamiaceae). Iran J Bot 2011;18:108-11.
- Lester A. Wilson. Encyclopedia of food and health. 2016;84-92.
- Ocaña A, Reglero G. Effects of thyme extract oils (from Thymus vulgaris, Thymus zygis, and Thymus hyemalis) on cytokine production and gene expression of oxLDL-stimulated THP-1-macrophages. J Obes 2012;2012:104706.
[Crossref] [Google Scholar] [PubMed]
- Zarzuelo A, Crespo E. The medicinal and non-medicinal uses of thyme. In: Thyme 2002;277-306.
- Burt S. Essential oils: Their antibacterial properties and potential applications in foods—a review. Int J Food Microbiol 2004;94(3):223-53.
[Crossref] [Google Scholar] [PubMed]
- Heidari Z, Salehzadeh A, Sadat Shandiz SA, Tajdoost S. Anti-cancer and anti-oxidant properties of ethanolic leaf extract of Thymus vulgaris and its bio-functionalized silver nanoparticles. 3 Biotech 2018;8(3):1-4.
[Crossref] [Google Scholar] [PubMed]
- Kuete V. Thymus vulgaris. Medicinal spices and vegetables from Africa. 2017:599-609.
- Roby MH, Sarhan MA, Selim KA, Khalel KI. Evaluation of antioxidant activity, total phenols and phenolic compounds in thyme (Thymus vulgaris L.), sage (Salvia officinalis L.), and marjoram (Origanum majorana L.) extracts. Ind Crops Prod 2013;43:827-31.
- Jain N, Sharma M. Screening of Thymus vulgaris essential oil against fungi causing dermatophytosis in human beings. Int J Pharm Pharm Sci 2017:236-9.
- Prasanth Reddy V, Ravi Vital K, Varsha PV, Satyam S. Review on Thymus vulgaris traditional uses and pharmacological properties. Med Aromat Plants 2014;3(164):2167-412.
- Imelouane B, Amhamdi H, Wathelet JP, Ankit M, Khedid K, El Bachiri A. Chemical composition and antimicrobial activity of essential oil of thyme (Thymus vulgaris) from Eastern Morocco. Int J Agric Biol 2009;11(2):205-8.
- Uhl SR. Handbook of Spices, Seasonings and Flavorings. Tecnomic publishing: Lancaster; 2000.
- Dauqan EM, Abdullah A. Medicinal and functional values of thyme (Thymus vulgaris L.) herb. J Appl Biol Biotechnol 2017;5(2):17-22.
- Vineetha. 22 Amazing benefits and uses of Thyme. Health Beckon. 2014.
- Sartoratto A, Machado AL, Delarmelina C, Figueira GM, Duarte MC, Rehder VL. Composition and antimicrobial activity of essential oils from aromatic plants used in Brazil. Braz J Microbiol 2004;35(4):275-80.
- Rota MC, Herrera A, Martínez RM, Sotomayor JA, Jordán MJ. Antimicrobial activity and chemical composition of Thymus vulgaris, Thymus zygis and Thymus hyemalis essential oils. Food Control 2008;19(7):681-7.
- Millezi AF, Caixeta DS, Rossoni DF, Cardoso MD, Piccoli RH. In vitro antimicrobial properties of plant essential oils Thymus vulgaris, Cymbopogon citratus and Laurus nobilis against five important foodborne pathogens. Food Sci Technol 2012;32(1):167-72.
- Borugă O, Jianu C, Mişcă C, Goleţ I, Gruia AT, Horhat FG. Thymus vulgaris essential oil: Chemical composition and antimicrobial activity. J Med Life 2014;7(3):56.
- Fani M, Kohanteb J. In vitro antimicrobial activity of Thymus vulgaris essential oil against major oral pathogens. J Evid Based Complement Altern Med 2017;22(4):660-6.
[Crossref] [Google Scholar] [PubMed]
- Nadia Z, Rachid M. Antioxidant and antibacterial activities of Thymus vulgaris L. Med Aromatic Plant Res J 2013;1(1):5-11.
- Asbaghian S, Shafaghat A, Zarea K, Kasimov F, Salimi F. Comparison of volatile constituents, and antioxidant and antibacterial activities of the essential oils of Thymus caucasicus, Thymus kotschyanus and Thymus vulgaris. Nat Prod Commun 2011;6(1):137-40.
- Kon K, Rai M. Antibacterial activity of Thymus vulgaris essential oil alone and in combination with other essential oils. Nusantara Biosci 2012;4(2):50-6.
- Al-Balushi AH, Aljabri M, Akhtar MS, Said S, Weli A, Al-Riyami Q, et al. Antibacterial and cytotoxic activities of Thymus vulgaris leaves grown in Oman. Int J Pharm Sci Res 2013;4(11):4253.
- Fadil M, Fikri-Benbrahim K, Rachiq S, Ihssane B, Lebrazi S, Chraibi M, et al. Combined treatment of Thymus vulgaris L., Rosmarinus officinalis L. and Myrtus communis L. essential oils against Salmonella typhimurium: Optimization of antibacterial activity by mixture design methodology. Eur J Pharm Biopharm 2018;126:211-20.
[Crossref] [Google Scholar] [PubMed]
- Benameur Q, Gervasi T, Pellizzeri V, Pľuchtová M, Tali-Maama H, Assaous F, et al. Antibacterial activity of Thymus vulgaris essential oil alone and in combination with cefotaxime against bla ESBL producing multidrug resistant Enterobacteriaceae isolates. Nat Prod Res 2019;33(18):2647-54.
[Crossref] [Google Scholar] [PubMed]
- Schwarz K, Ernst H, Ternes W. Evaluation of antioxidative constituents from thyme. J Sci Food Agric 1996;70(2):217-23.
- Abdalla AE, Roozen JP. Effect of plant extracts on the oxidative stability of sunflower oil and emulsion. Food Chem 1999;64(3):323-9.
- Lee SJ, Umano K, Shibamoto T, Lee KG. Identification of volatile components in basil (Ocimum basilicum L.) and thyme leaves (Thymus vulgaris L.) and their antioxidant properties. Food Chem 2005;91(1):131-7.
- Kulisic T, Radonic A, Milos M. Antioxidant properties of thyme (Thymus vulgaris L.) and wild thyme (Thymus serpyllum L.) essential oils. Italian J Food Sci 2005;17(3):315.
- VukoviÄ N, Bobkovà A, Pavelkovà A, Rovnà K, Arpà H. Antimicrobial and antiradicals activity of Origanum vulgare L. and Thymus vulgaris essential oils. J Microbiol Biotech Food Sci 2012;2(1):263-71.
- Roby MH, Sarhan MA, Selim KA, Khalel KI. Evaluation of antioxidant activity, total phenols and phenolic compounds in thyme (Thymus vulgaris L.), sage (Salvia officinalis L.), and marjoram (Origanum majorana L.) extracts. Indust Crops Prod 2013;43:827-31.
- Miladi H, Slama RB, Mili D, Zouari S, Bakhrouf A, Ammar E. Essential oil of Thymus vulgaris L. and Rosmarinus officinalis L.: Gas chromatography-mass spectrometry analysis, cytotoxicity and antioxidant properties and antibacterial activities against foodborne pathogens. Nat Sci 2013;5(6):729-39.
- Chizzola R, Michitsch H, Franz C. Antioxidative properties of Thymus vulgaris leaves: Comparison of different extracts and essential oil chemo types. J Agric Food Chem 2008;56(16):6897-904.
[Crossref] [Google Scholar] [PubMed]
- El-Nekeety AA, Mohamed SR, Hathout AS, Hassan NS, Aly SE, Abdel-Wahhab MA. Antioxidant properties of Thymus vulgaris oil against aflatoxin-induce oxidative stress in male rats. Toxicon 2011;57(7-8):984-91.
[Crossref] [Google Scholar] [PubMed]
- Gedikoğlu A, Sökmen M, Çivit A. Evaluation of Thymus vulgaris and Thymbra spicata essential oils and plant extracts for chemical composition, antioxidant, and antimicrobial properties. Food Sci Nutr 2019;7(5):1704-14.
[Crossref] [Google Scholar] [PubMed]
- Assiri AM, Elbanna K, Abulreesh HH, Ramadan MF. Bioactive compounds of cold-pressed thyme (Thymus vulgaris) oil with antioxidant and antimicrobial properties. J Oleo Sci 2016;65(8):629-40.
[Crossref] [Google Scholar] [PubMed]
- Numpaque MA, Oviedo LA, Gil JH, García CM, Durango DL. Thymol and carvacrol: biotransformation and antifungal activity against the plant pathogenic fungi Colletotrichum acutatum and Botryodiplodia theobromae. Tropical Plant Pathol 2011;36:3-13.
- Giordani R, Regli P, Kaloustian J, Mikail C, Abou L, Portugal H. Antifungal effect of various essential oils against Candidaalbicans. Potentiation of antifungal action of amphotericin B by essential oil from Thymus vulgaris. Phytother Res 2004;18(12):990-5.
[Crossref] [Google Scholar] [PubMed]
- Šegvić Klarić M, Kosalec I, Mastelić J, Piecková E, Pepeljnak S. Antifungal activity of thyme (Thymus vulgaris L.) essential oil and thymol against moulds from damp dwellings. Lett Appl Microbiol 2007;44(1):36-42.
[Crossref] [Google Scholar] [PubMed]
- Rus C, Sumalan RM, Alexa E, Copolovici DM, Pop G, Botau D. Study on chemical composition and antifungal activity of essential oils obtained from representative species belonging to the Lamiaceae family. Plant Soil Env 2015;61(7):297-302.
- Sakkas H, Gousia P, Economou V, Petsios S, Papadopoulou C. Antifungal activity of four essential oils against Candida clinical isolates. Asian J Ethnopharmacol Med Foods 2016;2(1):22-5.
- Mota KS, Pereira FD, De Oliveira WA, Lima IO, Lima ED. Antifungal activity of Thymus vulgaris L. essential oil and its constituent phytochemicals against Rhizopus oryzae: Interaction with ergosterol. Molecules 2012;17(12):14418-33.
[Crossref] [Google Scholar] [PubMed]
- Moghtader M. Antifungal effects of the essential oil from Thymus vulgaris L. and comparison with synthetic thymol on Aspergillus niger. J Yeast Fungal Res 2012;3(6):83-8.
- Gucwa K, Milewski S, Dymerski T, Szweda P. Investigation of the antifungal activity and mode of action of Thymus vulgaris, Citrus limonum, Pelargonium graveolens, Cinnamomum cassia, Ocimum basilicum, and Eugenia caryophyllus essential oils. Molecules 2018;23(5):1116.
[Crossref] [Google Scholar] [PubMed]
- van den Broucke CO, Lemli JA. Pharmacological and chemical investigation of thyme liquid extracts. Planta Med 1981;41(2):129-35.
[Crossref] [Google Scholar] [PubMed]
- Van Den Broucke CO, Lemli JA. Spasmolytic activity of the flavonoids from Thymus vulgaris. Pharm Weekb Sci 1983;5(1):9-14.
[Crossref] [Google Scholar] [PubMed]
- Begrow F, Engelbertz J, Feistel B, Lehnfeld R, Bauer K, Verspohl EJ. Impact of thymol in thyme extracts on their antispasmodic action and ciliary clearance. Planta Med 2010;76(4):311-8.
[Crossref] [Google Scholar] [PubMed]
- Micucci M, Protti M, Aldini R, Frosini M, Corazza I, Marzetti C, et al R. Thymus vulgaris L. essential oil solid formulation: Chemical profile and spasmolytic and antimicrobial effects. Biomolecules 2020;10(6):860.
[Crossref] [Google Scholar] [PubMed]
- Engelbertz J, Schwenk T, Kinzinger U, Schierstedt D, Verspohl EJ. Thyme extract, but not thymol, inhibits endothelin-induced contractions of isolated rat trachea. Planta Med 2008;74(12):1436-40.
[Crossref] [Google Scholar] [PubMed]
- Soković M, Glamočlija J, Ćirić A, Kataranovski D, Marin PD, Vukojević J, Brkić D. Antifungal activity of the essential oil of Thymus vulgaris L. and thymol on experimentally induced dermatomycoses. Drug Dev Indu Pharm 2008;34(12):1388-93.
[Crossref] [Google Scholar] [PubMed]
- Ramchoun M, Harnafi H, Alem C, Benlyas M, Elrhaffari L, Amrani S. Study on antioxidant and hypolipidemic effects of polyphenol-rich extracts from Thymus vulgaris and Lavendula multifida. Pharmacogn Res 2009;1(3).
- Ocaña A, Reglero G. Effects of thyme extract oils (from Thymus vulgaris, Thymus zygis, and Thymus hyemalis) on cytokine production and gene expression of oxLDL-stimulated THP-1-macrophages. J Obes 2012;2012:104706.
[Crossref] [Google Scholar] [PubMed]
- Vigo E, Cepeda A, Perez-Fernandez R, Gualillo O. In vitro anti-inflammatory effect of Eucalyptus globulus and Thymus vulgaris: Nitric oxide inhibition in J774A.1 murine macrophages. J Pharm Pharmacol 2004;56(2):257-63.
[Crossref] [Google Scholar] [PubMed]
- Braga PC, Dal Sasso M, Culici M, Bianchi T, Bordoni L, Marabini L. Anti-inflammatory activity of thymol: Inhibitory effect on the release of human neutrophil elastase. Pharmacology 2006;77(3):130-6.
[Crossref] [Google Scholar] [PubMed]
- Rezatofighi SE, Seydabadi A, Nejad SM. Evaluating the efficacy of Achillea millefolium and Thymus vulgaris extracts against Newcastle disease virus in Ovo. Jundishapur J Microbiol 2014;7(2):e9016.
[Crossref] [Google Scholar] [PubMed]
- Kaewprom K, Chen YH, Lin CF, Chiou MT, Lin CN. Antiviral activity of Thymus vulgaris and Nepeta cataria hydrosols against porcine reproductive and respiratory syndrome virus. Thai J Veterinary Med 2017;47(1):25.
- Nolkemper S, Reichling J, Stintzing FC, Carle R, Schnitzler P. Antiviral effect of aqueous extracts from species of the Lamiaceae family against Herpes simplex virus type 1 and type 2 in vitro. Planta Med 2006;72(15):1378-82.
[Crossref] [Google Scholar] [PubMed]
- Vimalanathan S, Hudson J. Anti-influenza virus activity of essential oils and vapors. Am J Essential Oils Nat Prod 2014;2(1):47-53.
- El-Akhal FO, Guemmouh RA, Maniar S, Taghzouti K, El Ouali Lalami A. Larvicidal activity of essential oils of Thymus vulgaris and Origanum majorana (Lamiaceae) against of the malaria vector Anopheles labranchiae (Diptera: Culicidae). Int J Pharm Pharm Sci 2016;8(3):372-6.
- Szczepanik M, Zawitowska B, Szumny A. Insecticidal activities of Thymus vulgaris essential oil and its components (thymol and carvacrol) against larvae of lesser mealworm, Alphitobius diaperinus Panzer (Coleoptera: Tenebrionidae). Allelopathy J 2012;30(1):129-42.
- Saroukolai AT, Moharramipour S, Meshkatalsadat MH. Insecticidal properties of Thymus persicus essential oil against Tribolium castaneum and Sitophilus oryzae. J Pest Sci 2010;83(1):3-8.
- De La Torre Rodriguez YC, Estrada FR, Suarez AE, De Torres NW, Aranda RS. Larvicidal and cytotoxic activities of extracts from 11 native plants from northeastern Mexico. J Med Entomol 2013;50(2):310-3.
[Crossref] [Google Scholar] [PubMed]
- Kubatka P, Uramova S, Kello M, Kajo K, Samec M, Jasek K, et al. Anticancer activities of Thymus vulgaris L. in experimental breast carcinoma in vivo and in vitro. Int J Mol Sci 2019;20(7):1749.
[Crossref] [Google Scholar] [PubMed]
- Sun Z, Park SY, Hwang E, Zhang M, Seo SA, Lin P, et al. Thymus vulgaris alleviates UVB irradiation induced skin damage via inhibition of MAPK/AP‐1 and activation of Nrf2-ARE antioxidant system. J Cell Mol Med 2017;21(2):336-48.
[Crossref] [Google Scholar] [PubMed]
- Komaki A, Hoseini F, Shahidi S, Baharlouei N. Study of the effect of extract of Thymus vulgaris on anxiety in male rats. J Tradit Complement Med 2016;6(3):257-61.
[Crossref] [Google Scholar] [PubMed]