- Corresponding Author:
- S. B. Sateesha
Department of Pharmaceutics, Acharya and BM Reddy College of Pharmacy, Bangalore-560 090
E-mail: sbsateesh@gmail.com
| Date of Submission | 23 September 2013 |
| Date of Revision | 17 October 2014 |
| Date of Acceptance | 15 January 2015 |
| Indian J Pharm Sci 2015;77(1):34-40 |
Abstract
Since the introduction of gliclazide in the pharmaceutical industry, a large number of research groups have been engaged in various investigations aiming to enhance its biomedical application. But, very limited efforts have been made to study polymorphism of gliclazide. Therefore, this study focuses on solvent-induced polymorphism of gliclazide and its characterization by thermal methods. Three polymorphs namely, Form-I, II and III and an amorphous powder were produced from different solvents and solvent mixtures. Crystals were analyzed using infrared spectroscopy, differential scanning calorimetry, X-ray powder diffraction and single crystal x-ray diffraction. Polymorph Form-I is found to exist in centro-symmetric triclinic P-1 space group and has endothermic peak at 162.93°. Form-II has endothermic peak from 171.2° to 172.35° and exists in centro-symmetric monoclinic P2 1 /a space group while Form-III has endothermic peak from 168.93° to 169.86° and exists in centro-symmetric monoclinic P2 1 /n space group. The equilibrium solubility values of Form-I, II, III and the amorphous form were 0.4825±0.025, 0.2341±0.042, 0.2581±0.038 and 0.5213±0.072 mg/ml, respectively. The Form-I has relatively higher solubility and similar to that of amorphous gliclazide. Form-II and Form-III are relatively most stable and least soluble. However, there was no remarkable difference in their aqueous solubility under the conditions in which study was conducted.
Keywords
Gliclazide polymorph, single crystal X-ray diffraction, centro-symmetric, monoclinic, triclinic, equilibrium solubility
Polymorphism has a greater impact on aqueous solubility[1,2] and bioavailability of a drug. Polymorphism attains a considerable influence on biopharmaceutical[3,4] and technological performance[5,6] of the drug. Polymorphism is extensive in sulfonylureas as these compounds represent chemical groups that cast hydrogen bonds and dipolar interactions, which are often convoluted in the formation of different polymorphic structures[7,8].
Gliclazide, 1-(3-azabicyclo[3.3.0]oct-3-yl)-3- tosylurea; is also noted as 1-(3-azabicyclo[3.3.0] oct-3-p-tolylsulfonylurea (fig. 1). Since its inception in the pharmaceutical industry, a large number of research groups have been intensified in various quests aiming to enhance its biomedical value[9-11]. But, very confined efforts have been made to reflect polymorphism of gliclazide. Winters et al., has conveyed solid state characterization of gliclazide polymorphs[12], but their study is confined to powder X-ray diffraction (XRPD), infrared (IR) spectroscopy and differential scanning calorimetry (DSC). Single crystal x-ray diffraction is used to study solid-state properties and crystal structure of gliclazide, but it was limited to characterization of only one polymorph[12]. There upon our studies resort on inspecting the solvent-induced polymorphism of gliclazide[13]. Solvent-induced crystallization technique is applied in this study; as a result the polarity of the recrystallisation solvent and its ability to form hydrogen bonds has a great impact on the polymorphism of gliclazide. The isolated crystal forms were delineated by their melting points, IR spectra, DSC, XRPD patterns, single crystal x-ray diffraction and equilibrium solubility.
Materials and Methods
Gliclazide BP was graciously provided as a gift sample by Zhejiang Jiuzhou Pharmaceuticals Co. Ltd. All solvents used in the study were double distilled from an all-glass still.
Preparation of gliclazide polymorphs
Polymorph-I was obtained from a gliclazide suspension in ethanol, which is kept for boiling and filtered when hot. The filtrate was retained in a refrigerator until the completion of crystallization process. This polymorphic appearance can also be accessed from binary solvent systems of water:ethanol (1:5). Polymorph-II was accessed from a super saturated solution of gliclazide in methanol at 65° and frost in ice bath for 2 h and filtered. The crystals accessed were collected and stewed under vacuum. This polymorph can also be obtained by crystallization using acetone, and binary solvent system of methanol:acetone (2:1). This polymorph-III was obtained from a super saturated solution of gliclazide in chloroform at 40°. The solution is frosted in an ice bath for 2 h and filtered. The crystals obtained were collected and dried under vacuum. Crystallization in ethyl acetate or dichloromethane or binary solvent system of chloroform:dichloromethane (4:1) also produces this polymorphic appearance. The received commercial sample of gliclazide (raw material) is found to be in the amorphous form.
DSC analysis
The thermal curves are recorded on a DSC on a Dupont DSC model in an open pan system under static conditions. The analyzer is equipped with a data acquiring station with attaining a capability to obtain the first derivative of DSC. Alumina was used as an inert reference. Each sample is sealed in an aluminum pan and the thermal readings were observed at a rate of 10°/min from room temperature to 225º.
X-ray powder diffraction analysis
The X-ray diffraction patterns are recorded for the samples with a radiation source of Cu-K alpha =1.54056 Å using a Bruker AXS advance X-ray diffractometer fitted with a scintillation counter and divergent beam monochromator. Data was collected from a range of 5°-50° at 2θ with a collection time of one second per step. Intensity ratio (I/I0, %) is calculated considering high intense peak as 100%.
FTIR spectroscopy
The IR spectra are being recorded on Bruker Optics, Tensor 27. Samples were scanned over 400–4000 cm-1 with a resolution of 4 cm-1. Samples were neither grinded nor compressed to prevent stress-induced polymorphic changes in the samples.
Single crystal X-ray diffraction
The single crystal X-ray diffraction patterns for the samples were recorded with a monochromatic radiation source of Mo-K alpha=0.71073 Å using a Bruker AXS diffractometer equipped with Oxford cryo system 700 Plus X-ray diffraction/crystallography system.
Microscopic visual analysis
Microscopic visual analysis of the polymorphs was performed using Leica DM 1000 binocular microscope. The magnification at 100X was used to study the crystal habit of the polymorphs.
Solubility measurement
Higuchi-Connors method (1965) was followed to determine the solubility of each polymorphic form[14]. An excess amount of each form (100 mg) was added to 25 ml of deionized water in a 100 ml flask with a glass stopper. The flasks were placed on a mechanical shaker and maintained at 37° in thermostatic water bath for 72 h. An aliquot (3 ml) of each solution was withdrawn and filtered through a dialysis membrane 0.45 μ Millipore filter. The amount dissolved was determined by reading the absorbance at 228 nm using a UV/Vis spectrophotometer (Mode l UV-240, Shimadzu, Japan) and using suitably constructed calibration curve.
Results and Discussion
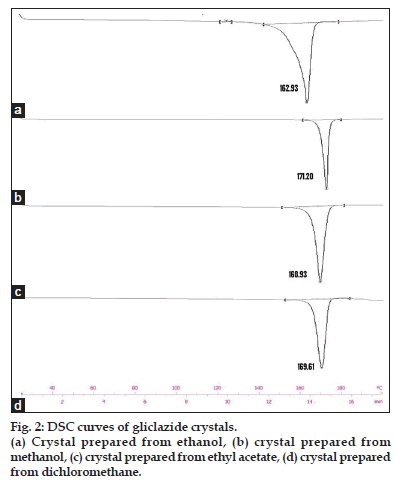
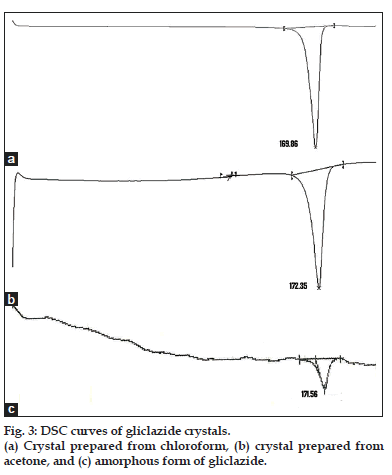
DSC is an extremely important analytical tool for measuring the wide variety of transitions in pharmaceutical materials[15]. DSC data can derive useful information in identifying the polymorphs. The DSC scans of all the crystals and raw material are shown in figs. 2 and 3. The DSC endothermic details of gliclazide crystals are presented in Table 1. The DSC curve of the gliclazide crystal prepared from ethanol shows a sharp endothermic peak at 162.93° with an insignificant peak at 124.80°. This form is designated here as the polymorph-I. The DSC curve of the polymorph-II, which is obtained by crystallization from methanol, exhibits a single sharp endotherm at 171.20°. This polymorph can also be obtained from acetone, it shows melting endothermic peak at 172.35°.
Gliclazide sample crystallized in dichloromethane, ethyl acetate and chloroform is designated here as the polymorph-III. This form has melting endotherm between 168.93° to 169.86° depending upon the solvent system used. The DSC scans of the commercial sample (untreated gliclazide) shows (fig. 3) an endothermic peak at 171.56°. The peak is short and not sharp like other crystalline forms indicating its amorphous nature[16].
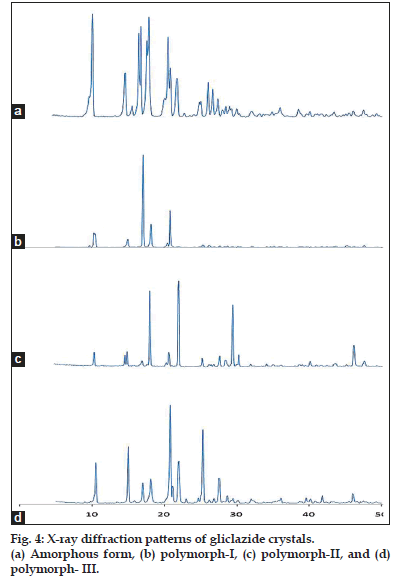
Main XRPD peak angles and intensity ratio (I/I0) of all the gliclazide crystals are presented in Table 2. The diffractograms of the entire crystalline forms exhibit a series of intense lines except the commercial form. The number of diffractions and intensity of peaks exhibited by the polymorph-III are more than the diffractions of the polymorph-I and II. The polymorph-II shows more diffractions than those of the polymorph-I. Further, the diffractions are different with respect to their position and intensity. Based on the number and intensity of diffractions, it is confirmed that the sample obtained from methanol (polymorph-II) and chloroform (polymorph-III) is more crystalline than the sample obtained from ethanol (polymorph-I). The spectral lines observed for the commercial form are less intense and relatively broad which further indicates that the sample is amorphous in nature[17,18]. The X-ray diffraction patterns of all polymorphs are shown in fig. 4.
| Solvent system and | Integral Normalized Onset | Peak | Endset | ||
|---|---|---|---|---|---|
| temperature conditions | (mJ) | (J/g) | (°) | (°) | (°) |
| Ethyl acetate, 4° | −468.41 | −143.24 | 165.62 | 168.93 | 172.88 |
| Acetone, 4° | −117.34 | −38.6 | 167.08 | 172.35 | 175.82 |
| Chloroform, 4° | −413.04 | −134.98 | 165.39 | 169.85 | 173.11 |
| DCM, 10° | −452.57 | −136.73 | 166.19 | 169.61 | 173.57 |
| Ethanol, 4° | −352.12 | −107.66 | 158.28 | 162.93 | 166.18 |
| Methanol, 4° | −449.74 | −145.08 | 168.92 | 171.2 | 173.6 |
| Commercial sample | - | - | 168.08 | 163.43 | - |
Table 1: Dsc Endothermic Details Of Gliclazide Crystals Prepared Under Various Conditions.
| Angle θ | Polymorph-I | Polymorph-II | Polymorph-III | Amorphous form | ||||
|---|---|---|---|---|---|---|---|---|
| d-spacing | I/I0 (%) | d-spacing | I/I0 (%) | d-spacing | I/I0 (%) | d-spacing | I/I0 (%) | |
| 10.5 | 8.418 | 13.850 | 8.418 | 18.164 | 8.418 | 41.687 | 8.418 | 100 |
| 15 | 5.902 | 8.123 | 5.902 | 19.045 | 5.903 | 58.206 | 5.902 | 44.07 |
| 16.8 | - | - | - | - | - | - | 5.276 | 82.459 |
| 17.1 | 5.183 | 100 | - | - | - | - | 5.183 | 88.751 |
| 17.9 | - | - | - | - | - | - | 4.953 | 74.75 |
| 18.1 | - | - | 4.901 | 89.148 | 4.901 | 25.595 | - | - |
| 18.2 | 4.872 | 23.477 | - | - | - | - | 4.872 | 96.928 |
| 20 | - | - | 4.437 | 17.966 | - | - | - | - |
| 20.8 | 4.267 | 37.298 | - | - | 4.267 | 100 | 4.267 | 78.52 |
| 21.88 | - | - | - | - | 4.037 | 43.444 | - | - |
| 22 | - | - | - | - | - | - | 4.037 | 39.039 |
| 22.1 | - | - | 4.020 | 100 | - | - | - | - |
| 25.3 | - | - | - | - | 3.518 | 74.725 | 3.518 | 16.9 |
| 26.3 | - | - | - | - | - | - | 3.385 | 34.671 |
| 26.9 | - | - | - | - | - | - | 3.313 | 28.509 |
| 27.5 | - | - | - | - | 3.242 | 26.254 | - | - |
| 29.5 | - | - | 3.025 | 72.306 | - | - | - | - |
| 46.1 | - | - | 1.967 | 26.539 | - | - | - | - |
Table 2: Xrpd Peak Angles D (A°) And Intensity Ratios (I/I0) Of Gliclazide Polymorphs Andamorphous Form.
| Identification | Polymorphic Form | ||
|---|---|---|---|
| code | I | II | III |
| Space group | P-1 | P21/a | P21/n |
| a (A°) | 10.517 | 10.839 | 18.215 |
| b (A°) | 23.205 | 14.248 | 4.4354 |
| c (A°) | 10.41 | 10.666 | 9.869 |
| α(°) | 89.96 | 90 | 90 |
| β (°) | 118.55 | 105.23 | 112.68 |
| γ (°) | 91.39 | 90 | 90 |
| Volume (A3) | 2230.49 | 1589.619 | 735.796 |
| Index ranges | −1≤h≤4 | −3≤h≤2 | −9≤h≤2 |
| - | −4≤k≤5 | 0≤k≤3 | 0≤k≤1 |
| - | 0≤l≤3 | 0≤l≤2 | 0≤l≤5 |
| Crystal system | Triclinic | Monoclinic | Monoclinic |
Table 3: Crystallographic Data And Structure Refinement Parameters Of Gliclazide Polymorph-I, Ii And Iii
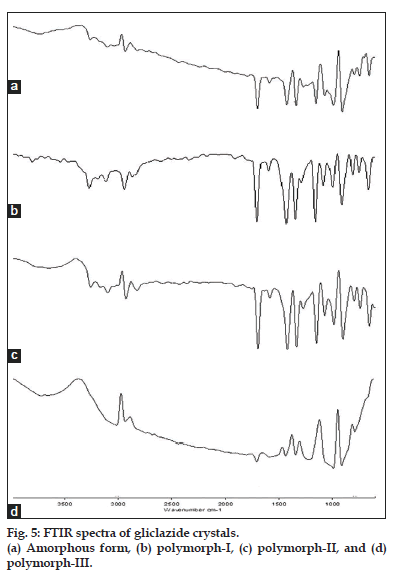
The IR spectra of the three crystal forms are presented in fig. 5. An IR spectrum reveals the existence of gliclazide in three polymorphic forms. Significant differences exist between polymorph-I, II and III. An additional peak at 3266 cm-1 is observed with the polymorph-II, which is lacking in the polymorph-I. Many peaks, which are distinct with the polymorph-II and polymorph-III (3107, 1705, 2440, 2909, 1432) are only rudimentary with the polymorph-I. This informs polymorph-I is different from other two crystals. However, no differences were observed between polymorph-II and polymorph-III in the number and position of peaks.
Single crystal x-ray diffraction study can provide unambiguous atomic positions and complete crystal information. This study infers three-dimensional structure of all the crystals including crystal cell volume, space group, bond lengths and bond angles which helps in refining crystallographic structure[19]. Crystallographic data and structure refinement parameters of gliclazide polymorph-I, II and III are shown Table 3.
Based on the results obtained from single crystal x-ray diffraction studies, the polymorph-II and III is found to form a rectangular prism with a parallelogram as its base. Hence, centro-symmetric monoclinic space group is suggested as a probable space group for the polymorph-II and III[20,21].
According to Kitaigorodsky for molecules with center of symmetry, the possible space groups are P, P21C, C2/c and Pbca for a 2-fold axis[22]. Also, in both the polymorphs the two pairs of vectors are perpendicular, while the third pair makes an angle other than 90° (α = γ=90°, β≠90°) and the crystal is described by vectors of unequal length (a≠b≠c). Based on these features centro-symmetric monoclinic space group P21/a, P21/n are suggested for polymorph-II and polymorph-III, respectively[23]. The settings P21/a, P21/n, are just different settings of the most common space group; P21/c (space group no.14) and these are indeed (with couple of exceptions) very common space groups. P21/a indicates a primitive monoclinic unit cell with a twofold screw axis along b, and a glide plane to this with a transition of a/2[24].
Polymorph-I is described by vectors of unequal length (a≠b≠c) and angle (a≠b≠g), as in the orthorhombic system. In addition, all three vectors are not mutually orthogonal (α=89.96°, β=118.55°, γ=91.39°)[25]. Therefore, centro-symmetric triclinic P-1 space group is proposed for this crystal. The commercial sample of gliclazide exhibit weak scattering to a single crystal X-ray diffraction, which clearly confirms its amorphous nature.
The equilibrium solubility values of the three crystalline polymorphs, I, II, III, and an amorphous form were 0.4825±0.025, 0.2341±0.042, 0.2581±0.038 and 0.5213±0.072 mg/ml, respectively. According to ideal solution theory, the solubility of a drug is related to two important thermodynamic parameters i.e. heat of fusion and melting point[26]. High melting and hard crystalline materials with large enthalpies of fusion are less soluble than low melting compounds[27]. Because, compounds with strong crystal lattices (high melting points) will readily crystallize in solvent hence the solubility benefit will be low[28]. Compounds with weak crystal lattices (low melting points) are less prone to crystallize and therefore amount in solution will remain high[29,30]. Microscopic visual analysis infers needle shape (fig. 6) for polymorph-II and III, which is the most crystalline state. These forms showed greater heat of fusion, which is clearly evident from DSC results. Therefore they are more stable and less soluble. polymorph-I, the most energetic exhibits a less crystalline (fig. 6) and lower heat of fusion and therefore has relatively higher solubility and is close to the amorphous gliclazide[31-33].
In conclusion, the three polymorphic forms of gliclazide were elucidated by solvent-changing method. All known polymorphs are least soluble but sufficiently stable. Polymorph-I was found to exist in centro-symmetric triclinic P-1 space group. This form has relatively higher solubility and similar to that of amorphous gliclazide. Polymorph-II and III exist as centro-symmetric monoclinic space group P21/a, P21/n and they are relatively more stable and less soluble. However there was no remarkable difference in their aqueous solubility under the conditions in which study was undertaken.
Acknowledgements
We are grateful to the Management, Acharya Institutes and the Principal, Acharya and B. M. Reddy College of Pharmacy, Bangalore, for providing research facilities. We are also thankful to Dr. L. V. G. Nargund Chairman, Nargund Research Foundation, Bangalore, for analysis of crystals and crystallographic studies.
References
- Blagden N, Matas DM, Gavan PT, York P. Crystal engineering of active pharmaceutical ingredients to improve solubility and dissolution rates. Adv Drug Deliver Rev 2007;59:617-30.
- Tiwary AK. Modification of crystal habit and its role in dosage form performance. Drug DevInd Pharm 2001;27:699-709.
- Grant DW. Theory and origin of polymorphism. In: Brittain HG, editors. Polymorphism in Pharmaceutical Solids. New York: Taylor and Francis; 1999. p. 1-34.
- Salehifar E, Zohrabi M, Eshghi S, Saeedi M, Ebrahimi P. Different pharmacokinetic parameters of phenytoin in Iranian Outpatients: Need to optimize the current dosage administration. Iranian J Pharm Res 2009;8:37-45.
- Barbas R, Prohens R, Cristina P. A new polymorph of norfloxacin complete characterization and relative stability of its trimorphic system. J Therm Anal Calorim 2007;89:687-92.
- Nokhodchi A, Maghsoodi M, Hassanzadeh D. An Improvement of Physicomechanical Properties of Carbamazepine Crystals. Iran J Pharm Res 2007;6:83-93.
- Zornoza A, de Nó C, Martín C, Goñi MM, MartínezOhárriz MC, Vélaz I. Evidence for polymorphism in glisentide. Int J Pharm 1999;186:199-204.
- Panagopoulou-Kaplani A, Malamataris S. Preparation and characterization of a new insoluble polymorphic form of glibenclamide. Int J Pharm 2000;195:239-46.
- Rang R, Dale MM, Ritter JM, Flower RJ, editors. In: Rang and Dale’s Pharmacology. 7th ed. New York: Churchill Livingstone; 2007. p. 405-6.
- Fronzo DRA. Pharmacologic therapy for type 2 diabetes mellitus. Ann Intern Med 1999;131:281-303.
- Mizuno CS, Chittiboyina AG, Kurtz TW, Pershadsingh HA, Avery MA. Type 2 diabetic and oral antihyperglycemic drugs. Curr Med Chem 2008;15:61-4.
- Winters CS, Shields L, Timmins P, York P. Solid-state properties and crystal structure of gliclazide. J Pharm Sci 1994;83:300-4.
- Bernstein J. Analytical techniques for studying and characterizing polymorphs. In: Polymorphism in molecular crystals. London: Clarendon Press; 2002. p. 94-150.
- Higuchi T, Connors KA. Phase solubility techniques. Adv Anal ChemInstrum 1965;4:117-212.
- Datta S, Grant DJ. Crystal structures of drugs: Advances in determination prediction and engineering. Nat Rev Drug Discov 2004;3:42-57.
- Amelia RD, Franks T, William F, Nirode. Introduction of differential scanning calorimeters in a general chemistry laboratory course: Determination of thermal properties of organic hydrocarbons. J ChemEduc 2007;84:453.
- Margiolaki I, Wright JP. Powder crystallography on macromolecules. Acta Crystallogr 2008;64:169-80.
- De Villiersde MM, Terblanche RJ, Liebenberg W, Swanepoel E, Dekker TG, Song M. Variable temperature X-ray powder diffraction analysis of the crystal transformation of the pharmaceutically preferred polymorph C of mebendazole. J Pharm Biomed Anal 2005;38:435-41.
- David WIF, Shankland K. Structure determination from powder diffraction data. Acta Crystallogr 2008;64:52-64.
- Kelmann RG, Kuminek G, Teixeira HF, Koester LS. Preliminary study on the development of nanoemulsions for carbamazepine intravenous delivery: An investigation of drug polymorphic transition. Drug DevInd Pharm 2008;34:53-8.
- Mighell AD, Ondik HM, Molino BB. Crystal data space group tables. J PhysChem Ref Data 1977;6:675-829.
- Kitaigorodsky AI. Molecular crystals and molecules. In; Physical Chemistry: A series of Monographs. 2nd ed. New York: Academic Press; 1973. p. 273-43.
- Harp JM, Hanson, BL, Timm DE, Bunick GJ. Macromolecular crystal annealing: Evaluation of techniques and variables. Acta Crystallogr D BiolCrystallogr 1999;55:1329-34.
- Keeffe OM, Hyde BG. Crystal Structures; I. Patterns and Symmetry. Washington, DC: Mineralogical Society of America, Monograph Series; 1996.
- Zachariasen WH. Theory of X-ray Diffraction in Crystals. J ChemEduc 1945;22:364.
- Pudipeddi M, Serajuddin AT. Trends in solubility of polymorphs. J Pharm Sci 2005;94:929-39.
- Park K, Evants JMB, Myerson AS. Determination of solubility of polymorphs using differential scanning calorimetry. Cryst Growth Des 2003;3:991-5.
- Martin A, Carstensen J. Extended solubility approach: Solubility parameters for crystalline compounds. J Pharm Sci 1981;70:170-2.
- Hoye JA, Gupta A, Myrdal PB. Solubility of solid solutes in HFA-134a with a correlation to physico-chemical properties. J Pharm Sci 2008;97:198-208.
- Mallick S, Pattnaik S, Swain K, De PK. Current perspectives of solubilization: Potential for improved bioavailability. Drug DevInd Pharm 2007;33:865-73.
- Palmer KJ, Brogden RN. Gliclazide: An update of its pharmacological properties and therapeutic efficacy in non-insulin dependent diabetes mellitus. Drugs 1993;46:92-125.
- Talari R, Varshosaz J, Mostafavi SA, Nokhodchi A. Dissolution Enhancement of gliclazide using pH change approach in presence of twelve stabilizers with various physico-chemical properties. J Pharm Pharm Sci 2009;12:250-65.
- Hong S, Lee S, Lee Y, Chung S, Lee M, Shim CK. Accelerated oral absorption of gliclazide in human subjects from a soft gelatin capsule containing a PEG 400 suspension of gliclazide. J Control Release 1998;51:185-92.