- *Corresponding Author:
- H. Y. Zhang
Department of Biochemistry, Weihai Vocational College, Weihai City Science and Technology Park, 264210, Weihai, Shandong, China
E-mail: haiyi_zhang@163.com
| Date of Submission | 03 February 2018 |
| Date of Revision | 21 August 2018 |
| Date of Acceptance | 05 February 2019 |
| Indian J Pharm Sci 2019;81(2):266-272 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
TATm-survivin (T34A), an anticancer protein drug, displayed pro-apoptotic bioactivity against various cancer cells. It was expressed in Escherichia coli as inclusion bodies. To test the bioactivity of soluble TATm-survivin (T34A), a feasible strategy was first developed for the soluble expression and purification. Effect of zinc ion and induction temperature was investigated to improve the solubility and overall production level of TATm-survivin (T34A). High solubility (92 %) was achieved by addition of zinc ion and temperature downshift after induction. An efficient protocol of purification was established by heat release, ammonium sulphate precipitation, anion exchange and heparin affinity chromatography. Purified TATm-survivin (T34A) was obtained with a purity of 96 %, which inhibited the proliferation of human pancreas carcinoma cell lines SW1990. In comparison to denatured TATm-survivin (T34A), soluble TATm-survivin (T34A) did not exhibit higher bioactivity as expected. This study represented a novel strategy to obtain highly soluble form of TATm-survivin (T34A) and would provide useful information for production of other soluble tat-mediated fusion proteins.
Keywords
TATm-survivin (T34A), expression, solubility, purification, bioactivity, HIV Tat protein, protein drug
Survivin, a novel member of the inhibitor of apoptosis protein (IAP) family, is found to be expressed by a majority of human cancers but not in adult normal tissues [1]. Anticancer therapy targeting survivin has drawn considerable attention. It is a 16.5 kDa protein, which consisted of two defined domains including an N-terminal Zn2+-binding baculovirus IAP repeat (BIR) domain linked to an amphipathic C-terminal α-helix [2]. Survivin, which is situated on chromosome 17q25, is an unique bifunctional protein that inhibited apoptosis by suppressing caspase-3 and caspase-7, and modulating the G2/M phase of the cell cycle by associating with the mitotic spindle microtubules [3]. However, Thr34→Ala mutant survivin (T34A) was found to show anticancer properties in gene therapy, which is related to the loss of Thr34 phosphorylation [4]. Considering the safety of gene therapy, an anticancer protein drug targeting survivin, TATm-survivin (T34A), was reported by Ma [5,6]. TATm-survivin (T34A) is a HIV-TAT-fusion protein that contained N-terminal protein transduction domain (PTD) for the delivery of this drug into cancer cells. It was expressed highly as inclusion bodies in Escherichia coli BL21 (DE3) under the control of T7 promoter and displayed the pro-apoptotic bioactivity against various cancer cells [5,6].
As is known to all, several studies have demonstrated that the Tat PTD directs the transduction of heterologous proteins into cells. The recombinant Tatfused proteins could be efficiently delivered into cells either as inclusion bodies [7-10] or in soluble form [11-13]. However, much less work has been done to improve the soluble expression and investigate the potential difference in bioactivity between soluble and renatured Tat-mediated proteins.
In general, soluble expression in recombinant systems was improved by the control of parameters such as temperature [14-16], host strains [17,18], codon optimization [19], addition of specific cofactors [20], co-expression of molecular chaperones [21-23] and solubility tag-technology [24-27]. This study developed a feasible strategy to achieve expression of highly soluble of TATm-survivin (T34A) by addition of zinc ion and a temperature downshift. Subsequently, an efficient protocol of purification was established by heat release, ammonium sulphate (AS) precipitation, anion exchange and heparin affinity chromatography. The bioactivity was also identified by proliferation experiments in vitro. To date, soluble expression and purification of TATm-survivin (T34A) was reported for the first time.
Materials and Methods
E. coli BL21 (DE3) was purchased from Novagen, USA. The plasmid pET-22b (+) was purchased from Invitrogen (USA) and pRSET-B-TAT-survivin (T34A) was described in Ma [5,6]. The restriction enzymes and T4 DNA ligase were from Takara (Japan). Plasmid DNA extraction kit and DNA gel extraction kit were from Shanghai Sangon (China). All other chemicals were of analytical or higher grade. Human pancreas carcinoma cell lines SW1990 was purchased from Institute of Cell and Chemistry, Chinese Academy of Science and cultivated at 37° in an atmosphere with 5 % CO2 (v/v) with RMPI1640 (Gibco Inc., Eggenstein, Germany) supplemented with 10 % fetal bovine serum, 0.01 % ampicillin and streptomycin. LB (tryptone 10 g/l, yeast extract 5 g/l, NaCl 5 g/l) and modified lactose bile brilliant green broth (LBBG) medium were prepared. The LBBG medium contained tryptone 10 g/l, yeast extract 5 g/l, NaCl 5 g/l, K2HPO4 0.5 g/l, Na2HPO4·12H2O 4.5 g/l, glycerol 10 g/l.
Construction of recombinant plasmid pET-22b (+)-TAT-survivin (T34A):
The full-length of fusion gene TAT-survivin (T34A) was obtained with NdeI and XhoI cleavage from pRSET-B-TAT-survivin (T34A) and inserted into expression vector pET-22b (+). The resulting plasmid pET-22b (+)-TAT-survivin (T34A) was confirmed with restriction endonuclease cleavage and identified by sequencing in Invitrogen (Shanghai, China).
Expression of recombinant TATm-survivin (T34A) in E. coli:
Recombinant plasmid pET-22b (+)-TAT-survivin (T34A) was transformed into E. coli BL21 (DE3). Freshly transformed cells were incubated overnight in 25 ml LB medium containing ampicillin 100 mg/l and 1.5 ml of the culture was transferred to 50 ml LBBG medium containing ampicillin 100 mg/l in a 250 ml flask at 37° and 200 rpm. When cell growth reached OD600 of 1.6-1.8, the expression of the target gene was induced by addition of isopropyl beta-Dthiogalactopyranoside (IPTG) to a final concentration of 1.0 mmol/l. Cells were allowed to grow further for 6 h and then harvested by centrifugation at 7000 g for 10 min at 4°. Other cultivation conditions for solubility optimization were mentioned below.
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE):
TATm-survivin (T34A) was analyzed using a 15 % SDS-PAGE. The gel was stained with Coomassie blue, and the protein bands were quantified with an image analysis system (FuRi Co., Shanghai, China). The solubility of the expressed protein was identified as followed. The cell pellet collected as described above was suspended in Tris-ethylenediaminetetraacetic acid (EDTA) buffer (20 mmol/l Tris-HCl, 2.5 mmol/l EDTA, pH 8.5) in half of the original volume and sonicated (on for 3 s, off for 3 s, 150 cycles). A 100 μl aliquot of the lysate was centrifuged at 7000 g for 15 min at 4°. The supernatant and whole lysate were used for analysis by SDS-PAGE to test the solubility.
Purification of soluble TATm-survivin (T34A):
Cells bearing recombinant plasmid pET-22b (+)-TATsurvivin (T34A) were induced at 25° in LBBG medium for 6 h. About 250 ml of the culture was centrifuged at 7000 g for 10 min to collect the cells. The pellet was suspended in 50 ml TE buffer and boiled for 6 min at 95°. Then the lysate was immediately cooled on ice for 20 min and centrifuged at 7000 g for 15 min at 4°. The supernatant was precipitated by 20 % saturated AS followed by 50 % saturated AS and the precipitated protein was collected by centrifugation. The deposit was dissolved in 10 ml buffer A (25 mmol/l Tris- HCl, pH 8.5) followed by dialysis twice at 4° in the same buffer. The insoluble material was then removed by centrifugation at 7000 g for 20 min at 4° and the supernatant was loaded onto DEAE Sepharose fast flow column (1 ml prepared column, Amersham Pharmacia Biotech) with AKTA Explorer 100. The column was washed by 10 column volumes of equilibration buffer A. The flow-through fraction (40 ml) was collected and loaded onto a heparin affinity column (1×10 cm containing 5 ml resin, Bio-sep Fast Flow, China) with the same equilibration conditions described above. The target protein was eluted stepwise with buffer A containing different concentration of NaCl (250 and 600 mmol/l) at 1 ml/min. The eluent at a concentration of 600 mmol/l was then collected and dialyzed overnight against buffer A and concentrated by Millipore spin filter (10 kDa cutoff) for bioactivity analysis. The purity of the protein was assessed by 15 % SDS-PAGE and the protein concentration was determined by Bradford assay [28].
Bioactivity evaluation of TATm-survivin (T34A) in vitro:
The inhibitory effect of TATm-survivin (T34A) on proliferation of human pancreas carcinoma cell lines SW1990 was examined by MTT colorimetric survival assay [29]. SW1990 cells (5000 per well) were plated into 96-well plates and allowed to attach for 8 h. Then RPMI 1640 with various concentrations of TATm- survivin (T34A) (0, 18.8, 50, 75, 100, 150 and 200 μg/ml) were added in quadruplicate and cells were incubated for 24 h. The medium was removed and fresh medium containing MTT was added to each well. After 4 h, the formazan crystals formed were dissolved in dimethyl sulfoxide (100 μl) and the absorbance was measured at 490 nm with an ELISA reader. These data were presented as means±SD from three independent experiments and analyzed statistically using two-sided Student t-test.
Results and Discussion
In this study, the recombinant plasmid pET-22b (+)-TAT-survivin (T34A) was used to induce the expression of target gene based on the finding that there was an increase of 40 % in cell growth and solubility under the same conditions (LB medium, 37°) in comparison with the plasmid pRSET-B-TAT-survivin (T34A) (Table 1). Effect of zinc ion and induction temperature was investigated to improve the solubility and overall production level of TATm-survivin (T34A).
| Expression vectors | OD600 | Total TATm-survivin | Soluble TATm-survivin | Solubility (%) |
|---|---|---|---|---|
| pRSET-B-TAT-survivin (T34A) | 2.59±0.05 | 25.2±0.75 | 0 | 0 |
| pET-22b(+)-TAT-survivin (T34A) | 4.32±0.01 | 24.8±1.51 | 3.1±0.69 | 12.5±2.02 |
Optical density at 600 nm 6 h after induction (OD600), total TATm-survivin (T34A)- percentage of total cellular protein (total TATm-survivin), soluble TATm-survivin (T34A)- percentage of total cellular protein (Total TATm-survivin), the percentage of soluble to total TATm-survivin (T34A) (solubility)
Table 1: Effect of Expression Vectors on the Solubility and Overall Yield of TATm-Survivin (T34A)
Preliminary results showed that the cell growth and total production level were reduced when zinc ion concentration was above 1 mmol/l while the solubility began to fall beyond 0.5 mmol/l (Table 2). Thus, 0.5 mmol/l was chosen to be the optimum concentration | of zinc ion in the following experiments. Seen from Table 2, the supplementation of zinc ion was beneficial to the solubility and overall yield of the target protein. In this experiment, about 48 % increase in solubility was observed with the addition of zinc ion, but the yield of the soluble protein was still not high.
| Factor | OD600 | Total TATm-survivin | Soluble TATm-survivin | Solubility (%) | |
|---|---|---|---|---|---|
| Zinc ion(mmol l-1) | 0 | 7.68±0.08 | 31.5±0.81 | 3.6±1.08 | 11.4±3.13 |
| 0.5 | 7.53±0.01 | 35.7±1.51 | 7.9±0.69 | 22.1±1.00 | |
| Temp (°) | 37 | 7.84±0.04 | 35.5±0.48 | 7.5±1.16 | 21.1±2.98 |
| 30 | 7.78±0.07 | 28.3±1.05 | 15.5±1.36 | 54.8±2.77 | |
| 25 | 7.45±0.12 | 27.4±0.87 | 25.2±1.39 | 92.0±2.15 | |
| 20 | 5.24±0.01 | 19.1±1.23 | 18.2±0.88 | 95.3±1.53 | |
Optical density at 600 nm 6 h after induction (OD600), total TATm-survivin (T34A)- percentage of total cellular protein (total TATm-survivin), soluble TATm-survivin (T34A)- percentage of total cellular protein (total TATm-survivin), the percentage of soluble to total TATm-survivin (T34A) (solubility). Data are expressed as the mean of triplicate±SD except for the solubility
Table 2: Effect of Zinc Ion and Temperature on the Solubility and Overall Yield of Tatm-Survivin (T34A)
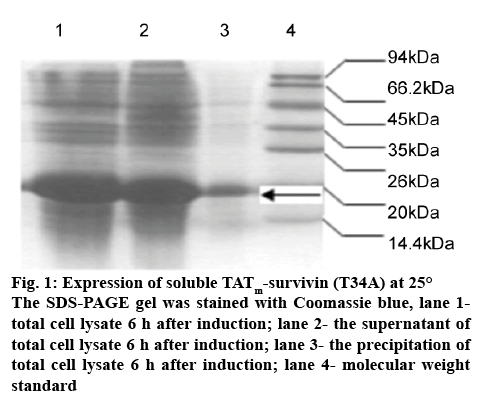
Lower temperature induction is an alternative method to prevent the aggregation of recombinant proteins. Thus, after zinc ion was added, the temperature was decreased to 30, 25, 20°, respectively. The results are shown in Table 2. When cells were induced at 30°, the soluble yield and solubility were more than twice as much as that at 37°. With further decrease in temperature, highly soluble TATm-survivin (T34A) was produced (>92±2.15 % in solubility) when induced at 25 and 20°. Figure 1 showed the electrophoretic diagram of soluble protein expression at 25° for 6 h. Seen from figure 1, the most part of TATm-survivin (T34A) was found in the supernatant of total cell lysate (figure 1 lane 1-3). At 37°, cells displayed the optimum growth and expression of foreign gene. After temperature downshift, overall production level was evidently reduced and cell growth was slightly affected (30 and 25°). However, at 20°, the cell density and total TATm-survivin (T34A) level were decreased by 33.1 and 46.1 %, respectively. Although higher solubility was obtained at 20°, it was decided to perform the soluble expression of TATm-survivin (T34A) at 25° from the viewpoint of higher cell density and soluble production level.
Figure 1: Expression of soluble TATm-survivin (T34A) at 25°
The SDS-PAGE gel was stained with Coomassie blue, lane 1- total cell lysate 6 h after induction; lane 2- the supernatant of total cell lysate 6 h after induction; lane 3- the precipitation of total cell lysate 6 h after induction; lane 4- molecular weight standard
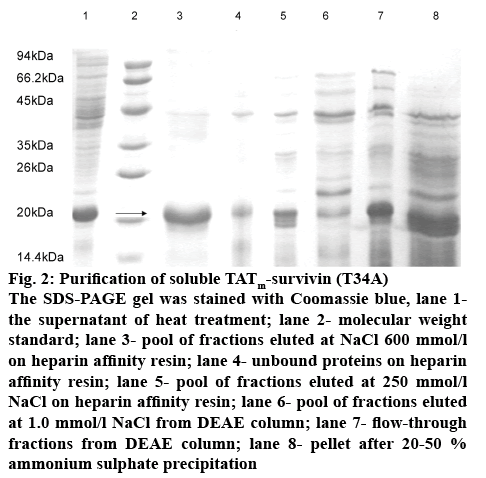
In this study, heat release of the target protein was attempted and attained good results (figure 2, lane 1, Table 3). When cells were treated via heat for 6 min at 95°, the content of TATm-survivin (T34A) was increased to 46 % of total cellular protein. At the same time, no obvious viscosity in the supernatant was observed, indicating that nucleic acids released were mostly precipitated. Heat treatment was found to lead to an increase of 50 % in the content of TATm-survivin (T34A).
Figure 2: Purification of soluble TATm-survivin (T34A)
The SDS-PAGE gel was stained with Coomassie blue, lane 1-the supernatant of heat treatment; lane 2- molecular weight standard; lane 3- pool of fractions eluted at NaCl 600 mmol/lon heparin affinity resin; lane 4- unbound proteins on heparin affinity resin; lane 5- pool of fractions eluted at 250 mmol/l NaCl on heparin affinity resin; lane 6- pool of fractions eluted at 1.0 mmol/l NaCl from DEAE column; lane 7- flow-through fractions from DEAE column; lane 8- pellet after 20-50 %
ammonium sulphate precipitation
| Purification steps | Total protein (mg) | Target protein (mg) | Yield(%) | Purity(%) |
|---|---|---|---|---|
| Heat release | 105.6 | 48.6 | 100 | 46 |
| Ammonium sulphate (50%) precipitation | 68.2 | 35.4 | 72.8 | 52 |
| Anion-exchange flowthrough | 46.7 | 27 | 76.2 | 57.8 |
| Heparin affinity chromatography | 4.1 | 3.8 | 14.1 | 96.1 |
Total protein was determined by Bradford assay with bovine serum albumin as a standard. Target protein was determined by densitometry scan. Yield was calculated based on the amount of the target protein. Target protein percentage of total protein (purity)
Table 3: Purification of Recombinant Tatm-Survivin (T34A)
The supernatant obtained by heat release was purified by AS fractional precipitation. SDS-PAGE analysis showed that some cellular proteins with high molecular mass (above 45 kDa) were mostly removed (figure 2, lane 8), accompanied by co-precipitation of a small proportion of the target protein. As a result, this step yield was calculated to about 72.8 % with a purity of approximately 52 % (Table 3). Subsequently, the sediment was dissolved in buffer A, dialyzed twice against the same buffer and then further purified by anion exchange chromatography. Although the theoretical isoelectric point (7.99) of TATm-survivin (T34A) was below the actual pH (8.5), the target protein was detected mostly in the flow-through fraction and the small amount was absorbed onto the anion column (figure 2, lane 7 and 6). In this step, more cellular proteins were removed and the purity of 57.8 % was achieved (Table 3).
Afterwards, heparin affinity chromatography was employed for the following purification. SDS-PAGE analysis (figure 2 lane 3) indicated that the purity of the target protein was about 96 % (Table 3). Other cellular proteins bound via ionic interaction were easily removed by a low concentration of NaCl (figure 2, lane 5).
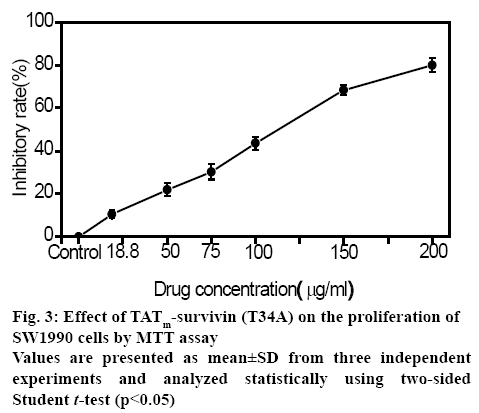
The inhibitory effect of soluble TATm-survivin (T34A) was evaluated on the proliferation of human pancreas carcinoma cell lines SW1990 using the MTT assay. It was observed that TATm-survivin (T34A) inhibited proliferation of SW1990 cells significantly in a dose-dependent manner (figure 3). The rate of inhibition increased from 10.5±1.7 to 79.9±3.2 % after treatment for 24 h.
Survivin contained N-terminal zinc-binding BIR domain tetrahedrally coordinated by Cys 57, Cys 60, His 77 and Cys 84 [2], which meant zinc ion was very important in stabilizing the correct structure. In this study, the overall yield of the target protein and solubility could be improved by supply of zinc ion after induction, which was assumed that the transport of zinc ion could not meet the demand of rapid protein production under the control of strong T7 promoter. At high protein synthetic rates, zinc ion uptake rate was likely to be a limiting step, resulting in the accumulation of misfolded protein in the cytoplasm. The positive effect of zinc ion was also found on the soluble expression of an apoptosis-inducing ligand (Apo2L/TRAIL) [30]. In addition, it was observed that the solubility was strongly dependent of temperature, which was presumed that lower temperature reduced the rate of protein synthesis and allowed more sufficient time for the nascent peptide chains to fold properly.
The purification of soluble TATm-survivin (T34A) from the general pool of cytoplasmic proteins seems to be a difficult task because this compartment contains the vast majority of cellular proteins. In general, sonication is used to obtain soluble protein products. However, the complete release of host proteins by this method can increase the difficulty and complexity in subsequent purification procedures, especially for those recombinant products without purification tags. Heat release was widely used for some thermostable recombinant proteins in the initial purification steps, which contributed to the precipitation of host proteins and the reduction of the potential degradation of the target product by thermal deactivation of E. coli and its proteases [31-34]. In this study, it was revealed for the first time that heat treatment was applicable for the initial purification of TATm-survivin (T34A), leading to a 50 % increase in the content of TATm-survivin (T34A). In addition, TATm-survivin (T34A) also displayed the ability to bind heparin, suggesting that it contained specific binding motifs. Tat-PTD peptide has been found to specifically bind heparin, which was characterized by an arginine-rich basic domain (49-57aa)[35-37]. TATm- survivin (T34A) contained the mutant Tat-PTD, where two arginine residues were substituted by alanine, but it probably contributed to the binding to heparin. Also, three-dimensional structure of survivin revealed an extensive basic patch that was also responsible for the binding to negatively charged heparin [2]. Although this study provided a feasible strategy for the purification of soluble TATm-survivin (T34A), the overall yield was relatively low. Future experiments are required to optimize the purification conditions.
The cell proliferation experiments primarily demonstrated the feasibility of purification procedures. It was worth noting that the inhibitory rate of soluble TATm-survivin (T34A) was lower than that of renatured TATm-survivin (T34A) under the comparable drug concentration in the study by Ma [5,6]. This phenomenon may be partially explained by the reduction in the amounts of soluble TATm-survivin (T34A) that was successfully delivered into the cancer cells and correctly folded.
Generally, soluble proteins are expected to be highly functional. But, for tat-mediated proteins, Bonifaci [38] suggested that an unfolding step is required for the internalization of exogenous Tat-mediated proteins. Also, if the PTD fusion product was denatured, subsequent entry into the cell would be facilitated [39]. In this context, it was assumed that the folding state of renatured TATm-survivin (T34A) was more prone to unfold compared to the soluble protein during membrane translocation. Accordingly, for soluble TATm-survivin (T34A), the low efficiency of intracellular delivery might be an obstacle, which resulted in a decrease of bioactivity. On the other hand, the result of bioactivity indicated that soluble TATm-survivin (T34A) was not completely blocked into living cells, whereas a portion of it could effectively enter the cells, which was speculated to be the existence of different conformational types that were already confirmed in the soluble VP1-fused green fluorescent protein [40]. Therefore, future efforts should be taken to investigate the possible leading factors for lower bioactivity in soluble TATm-survivin (T34A).
In this study, TATm-survivin (T34A) as an anticancer protein drug, was successfully expressed and purified as a soluble protein in E. coli. The result of bioactivity primarily revealed the inhibitory effect of the soluble protein on the cell proliferation. In addition, this study suggested that tat-mediated proteins as inclusion bodies probably showed higher bioactivity than those in soluble form, which would provide some useful information for other soluble tat-mediated fusion proteins.
Acknowledgements
The author thanks Mr. Dongzhi Wei and Mr. Xingyuan Ma for technical assistance and helpful comments.
Conflict of interest
The authors declare no conflict of interest.
References
- Altieri DC. The molecular basis and potential role of survivin in cancer diagnosis and therapy. Trends Mol Med 2004;7:542-7.
- Verdecia MA, Huang HK, Dutil E, Kaiser DA, Hunter T, Noel JP. Structure of the human anti-apoptotic protein survivin reveals a dimeric arrangement. Nat Struct Biol 2000;7(7):602-8.
- Yamamoto T, Tanigawa N. The role of survivin as a new target of diagnosis and treatment in human cancer. Med Electron Microsc 2001;34(4):207-12.
- O'Connor DS, Grossman D, Plescia J, Li FZ, Zhang H, Villa A, et al. Regulation of apoptosis at cell division by p34cdc2 phosphorylation of surviving. Proc Natl Acad Sci 2000;97(24):13103-7.
- Ma X, Zheng W, Wei D, Ma Y, Wang T, Wang J, et al. Construction, expression, and purification of HIV–TAT-survivin (T34A) mutant: A pro-apoptosis protein in Escherichia coli. Protein Expr Purif 2006;47(1):36-44.
- Ma X, Zheng W, Wei D, Ma Y, Wang T, Wang J, et al. High-level expression, purification and pro-apoptosis activity of HIV-TAT-survivin (T34A) mutant to cancer cells in vitro. J Biotechnol 2006;123(3):367-78.
- Shen C, Liu W, Buck AK, Reske SN. Pro-apoptosis and anti-proliferation effects of a recombinant dominant-negative survivin-T34A in human cancer cells. Anticancer Res 2009;29(4):1423-8.
- Shang X, Bao Y, Chen S, Ren H, Huang H, Li Y. Expression and purification of TAT-fused carbonic anhydrase III and its effect on C2C12 cell apoptosis induced by hypoxia/reoxygenation. Arch Med Sci 2012;8(4):711-8.
- Lan MS, Chen C, Saunee NA, Zhang T, Breslin MB. Expression of biologically active TAT-fused recombinant islet transcription factors. Life Sci 2014;114(1):45-50.
- Sohn EJ, Shin MJ, Kim DW, Ahn EH, Jo HS, Kim DS, et al. Tat-fused recombinant human SAG prevents dopaminergic neurodegeneration in a MPTP-induced Parkinson's disease model. Mol Cells 2014;37(3):226-33.
- Caldinelli L, Albani D, Pollegioni L. One single method to produce native and Tat-fused recombinant human ɑ-synuclein in Escherichia coli. BMC Biotechnol 2013;13:32-9.
- Zhao X, Dong Y, Zhao Z, Guo J, Liu J, Huang P, et al. Intracellular delivery of artificial transcription factors fused to the protein transduction domain of HIV-1 Tat. Protein Expr Purif 2013;90(1):27-33.
- Yu GR, Qin WW, Li JP, Hua W, Meng YL, Chen R, et al. HIV-TAT-fused FHIT protein functions as a potential pro-apoptotic molecule in hepatocellular carcinoma cells. Biosci Rep 2012;32(3):271-9.
- Vasina JA, Baneyx F. Expression of Aggregation-Prone Recombinant Proteins at Low Temperatures: A Comparative Study of the Escherichia coli cspA and tac Promoter Systems. Protein Expr Purif 1997;9(2):211-8.
- Vera A, González-Montalbán N, Arís A, Villaverde A. The conformational quality of insoluble recombinant proteins is enhanced at low growth temperatures. Biotechnol Bioeng 2007;96(6):1101-6.
- San-Miguel T, Pérez-Bermúdez P, Gavidia I. Production of soluble eukaryotic recombinant proteins in E. coli is favoured in early log-phase cultures induced at low temperature. Springerplus 2013;2(1):89-92.
- Miroux B, Walker JE. Over-production of Proteins in Escherichia coli: Mutant Hosts that Allow Synthesis of some Membrane Proteins and Globular Proteins at High Levels. J Mol Biol 1996;260(3):289-98.
- Cano-Garrido O, Rueda FL, Sànchez-García L, Ruiz-Ávila L, Bosser R, Villaverde A, et al. Expanding the recombinant protein quality in Lactococcus lactis. Microb Cell Fact 2014;13:167-73.
- Rabhi-Essafi I, Sadok A, Khalaf N, Fathallah DM. A strategy for high-level expression of soluble and functional human interferon alpha as a GST-fusion protein in E. coli. Protein Eng Des Sel 2007;20(5):201-9.
- Yang Q, Xu J, Li M, Lei X, An L. High-level expression of a soluble snake venom enzyme, gloshedobin, in E. coli in the presence of metal ions. Biotechnol Lett 2003;25(8):607-10.
- Mogk A, Mayer MP, Deuerling E. Mechanisms of protein folding: molecular chaperones and their application in biotechnology. Chembiochem 2002;3(9):807-14.
- Schlieker C, Bukau B, Mogk A. Prevention and reversion of protein aggregation by molecular chaperones in the E. coli cytosol: implications for their applicability in biotechnology. J Biotechnol 2002;96(1):13-21.
- de Marco A, Deuerling E, Mogk A, Tomoyasu T, Bukau B. Chaperone-based procedure to increase yields of soluble recombinant proteins produced in E. coli. BMC Biotechnol 2007;7:32-40.
- Sørensen HP, Mortensen KK. Advanced genetic strategies for recombinant protein expression in Escherichia coli. J Biotechnol 2005;115(2):113-28.
- Makrides SC. Strategies for achieving high-level expression of genes in Escherichia coli. Microbiol Rev 1996;60(3):512-38.
- Costa S, Almeida A, Castro A, Domingues L. Fusion tags for protein solubility, purification, and immunogenicity in Escherichia coli: the novel Fh8 system. Front Microbiol 2014;5:63-82.
- Cheng CH, Lee WC. Protein solubility and differential proteomic profiling of recombinant Escherichia coli overexpressing double-tagged fusion proteins. Microb Cell Fact 2010;9:63-75.
- Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976;72:248-54.
- Singh N, Sun Y, Nakamura K, Smith MR, Colburn NH. C-JUN/AP-1 as possible mediators of tumor necrosis factor-alpha-induced apoptotic response in mouse JB6 tumor cells. Oncol Res 1995;7(7-8):353-62.
- Sun AY, Shen YL, Yin JC, Zhang H, Tang YN, Wei DZ. Improvement of Expression Level and Bioactivity of Soluble Tumor Necrosis Factor-Related Apoptosis-Inducing Ligand (Apo2L/TRAIL) by a Novel Zinc ion Feeding Strategy. Biotechnol Lett 2006;28(15):1215-9.
- Collis MA, O’Neill BK, Thomas CJ, Middelberg AP. The effect of thermal deactivation on the properties and processing characteristics of E. coli. Bioseparation 1996;6:55-63.
- Desai UJ, Pfaffle PK. Single-step purification of a thermostable DNA polymerase expressed in Escherichia coli. Biotechniques 1995;19(5):780-4.
- Jonasson P, Nygren PÅ, Jörnvall H, Johansson BL, Wahren J, Uhlén M, et al. Integrated bioprocess for production of human proinsulin C-peptide via heat release of an intracellular heptameric fusion protein. J Biotechnol 2000;76(2-3):215-26.
- Wolfrum A, Brock S, Mac T, Grillenbeck N. Expression in E. coli and purification of Thermus thermophilus translation initiation factors IF1 and IF3. Protein Expr Purif 2003;29(1):15-23.
- Goda N, Tenno T, Inomata K, Iwaya N, Sasaki Y, Shirakawa M, et al. LBT/PTD dual tagged vector for purification, cellular protein delivery and visualization in living cells. Biochim Biophys Acta 2007;1773(2):141-6.
- Tyagi M, Rusnati M, Presta M, Giacca M. Internalization of HIV-1 tat requires cell surface heparan sulfate proteoglycans. J Biol Chem 2001;276(5):3254-61.
- Marty C, Meylan C, Schott H, Ballmer-Hofer K, Schwendener RA. Enhanced heparan sulfate proteoglycan-mediated uptake of cell-penetrating peptide-modified liposomes. Cell Mol Life Sci 2004;61(14):1785-94.
- Bonifaci N, Sitia R, Rubartelli A. Nuclear translocation of an exogenous fusion protein containing HIV Tat requires unfolding. AIDS 1995;9(9):995-1000.
- Leifert JA, Harkins S, Whitton JL. Full-length proteins attached to the HIV tat protein transduction domain are neither transduced between cells, nor exhibit enhanced immunogenicity. Gene Ther 2002;9(21):422-8.
- Martínez-Alonso M, González-Montalbán N, García-Fruitós E, Villaverde A. The functional quality of soluble recombinant polypeptides produced in Escherichia coli is defined by a wide conformational spectrum. Appl Environ Microbiol 2008;74(23):7431-3.