- *Corresponding Author:
- Xin Yi
Department of Human Anatomy, Medical School, Nantong University, China
E-mail: yixin@ntu.edu.cn
| Date of Received | 05 April 2021 |
| Date of Revision | 03 February 2022 |
| Date of Acceptance | 04 September 2022 |
| Indian J Pharm Sci 2022;84(5):1133-1139 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
To study the protective effect of curcumin on lipopolysaccharide-induced rat cardiomyocyte hypertrophy is the main objective of the study. Cardiomyocytes were purified and cultured by differential adherence in in vitro and then they were divided into control group, lipopolysaccharide group, lipopolysaccharide+curcumin low-dose (12.5 mg/l) group, lipopolysaccharide+curcumin medium-dose (25 mg/l) group and lipopolysaccharide+curcumin high-dose (50 mg/l) group. The morphological changes were observed under a phase-contrast inverted microscope and the cell volume was measured. The total protein content of the cell was detected by Coomassie brilliant blue kit. Enzyme-linked immunosorbent assay method was used to detect the level of tumor necrosis factor alpha. The TILL imaging system was used to observe the changes in intracellular calcium ions. Compared with the control group, lipopolysaccharide significantly increased the cardiomyocyte volume, the total cell protein content, the level of tumor necrosis factor alpha (p<0.01) and the instantaneous peak of calcium ions. Compared with the lipopolysaccharide group, 12.5, 25, 50 mg/l curcumin treatment inhibited the increase of cardiomyocyte volume, total protein content, tumor necrosis factor alpha level and the instantaneous peak of calcium ions induced by lipopolysaccharide and showed a dose-dependent effect. Curcumin has a protective effect on rat cardiomyocyte hypertrophy induced by lipopolysaccharides, which may be related to the inhibition of tumor necrosis factor alpha production and the inhibition of calcium ions overload.
Keywords
Curcumin, lipopolysaccharides, cardiomyocytes, hypertrophy, tumor necrosis factor alpha, calcium ions
Cardiomyocyte hypertrophy is an independent risk factor for many cardiovascular diseases and a common complication of these diseases[1]. Cardiomyocyte hypertrophy mainly occurs when the heart is chronically overloaded. Early cardiomyocyte hypertrophy has a certain compensatory significance and later decompensation, seriously affects cardiomyocyte function and can cause heart failure in severe cases[2]. Therefore, prevention and reversal of cardiomyocyte hypertrophy has always been one of the key topics in the field of cardiovascular research.
Lipopolysaccharide (LPS) is a component of the outer wall of the cell wall of Gram-negative bacteria and is a substance (glycolipid) composed of lipids and polysaccharides[3]. LPS is difficult to shed from the cell wall. When bacteria die, it will shed from dissolving and destroying bacteria, and exerts its toxicity by acting on the functional cells of the body. Due to this nature, it is also called endotoxin. When it acts on other biological cells of humans or animals, it will exhibit a variety of biological activities. The physiological role of LPS is triggered by the Toll-Like Receptor 4 (TLR4) present on the cell membrane surface[3,4]. When LPS is released into the blood in large quantities, it can cause endotoxemia, extensive uncontrolled inflammation and immune response in the body, which results in shock, systemic inflammation and multiple organ failure[5-8]. For the heart, LPS can weaken cardiomyocyte contractility and slower heart rate, which seriously affects heart function[9,10]. In vitro LPS can induce cardiomyocyte hypertrophy and the cell model is widely used in scientific research[11,12].
Curcumin is an active ingredient extracted from the rhizomes of turmeric. It can scavenge oxygen free radicals[13], reduce cardiomyocyte damage caused by ischemia and hypoxia[14], and inhibit ventricular remodeling after infarction[15], improve heart function[16]. However, the influence of curcumin on the cardiomyocyte hypertrophy is still unclear. In this study, rat cardiomyocyte hypertrophy was induced by LPS in vitro and cell model was treated with different concentrations of curcumin. The protective effect of curcumin on LPS-induced rat cardiomyocyte hypertrophy and its possible mechanism were explored.
Materials and Methods
Primary culture of rat cardiomyocytes:
The animal experiments were approved by the Institutional Animal Care and Use Committee of Nantong University. 6 newborn, 1-2 d Sprague Dawley (SD) rats (male or female) were provided by the Experimental Animal Center of Nantong University. The hearts were removed under aseptic conditions and cleaned with D-Hank’s solution, and then the large blood vessels and atrial tissue connected to the heart were removed, and then they were cut into small tissue pieces with ophthalmological scissors. The small tissue pieces were repeatedly pipetted with 0.08 % trypsin, digested at 37° for 10 min and the supernatant were discarded. The tissue was then digested with 0.08 % trypsin for 3 to 4 times to get the cells. The cells were cultured in culture flask containing Dulbecco’s Modified Eagle Medium (DMEM), placed in a 37°, 5 % Carbon dioxide (CO2) incubator and cultured for 1.5 h. Subsequently, the non-adherent cardiomyocyte suspension was sucked out with a pipette and then was cultured in a culture flask at a cell density of 4×108/l, and placed in a 37°, 5 % CO2 incubator for 48 h.
Drug treatment:
The curcumin (Sigma, United States of America (USA)) solution was prepared with normal sodium containing 5 % ethanol. The above-mentioned primary cultured cardiomyocyte were cultured in the 24-well culture plate equipped with a cover glass and containing DMEM for 24 h. The cells were divided into control group, LPS group, LPS+curcumin low-dose group, LPS+curcumin medium-dose group and LPS+curcumin high-dose group. The DMEM of LPS group, LPS+curcumin low-dose group, LPS+curcumin medium-dose group and LPS+curcumin high-dose group contained LPS 1 mg/l and curcumin 0 mg/l (control), 12.5 mg/l, 25 mg/l, 50 mg/l respectively. The DMEM of control group and LPS group contained the equal concentration of ethanol.
Morphological observation and cell volume measurement:
After the cells were cultured for 24 h, the morphological changes of each group of cells were observed under a phase-contrast inverted microscope and filmed. Each culture well randomly selects 4 fields of 200 times and the computer cell image analysis system measures the diameter of the cell, and then calculates the volume of the cell.
Determination of total protein content in cardiomyocyte:
The total protein content in cardiomyocyte was detected by Coomassie brilliant blue kit (Beyotime, China) according to the protocol of the product manual.
Determination of Tumor Necrosis Factor alpha (TNF-α) level:
The TNF-α level in medium of each group was measured by TNF-α Enzyme-Linked Immunosorbent Assay (ELISA) detection kit (Beyotime, China).
Calcium (Ca2+) ions transient change measurement:
The culture medium of each group was replaced with DMEM medium containing Fura-2/Acetoxymethyl Ester 3 (AM3) μmol/l and 0.2 % albumin and incubated them in a 37° water bath for 0.5 h. The (Ca2+) ions transient change was detected by TILL imaging system (Martinsried, Germany). The emission wave length was 505 nm, the excitation wave length was 340 nm and 380 nm and the sampling interval was 300 ms.
Statistical analysis:
Statistical Package for the Social Sciences (SPSS) 21.0 statistical software was used for statistical analysis of the data in this study. The measurement data in this study was in accordance with the normal distribution, expressed as mean±Standard Deviation (SD), the comparison among groups was performed by one-way Analysis of Variance (ANOVA) and the difference was considered statistically significant with p<0.05.
Results and Discussion
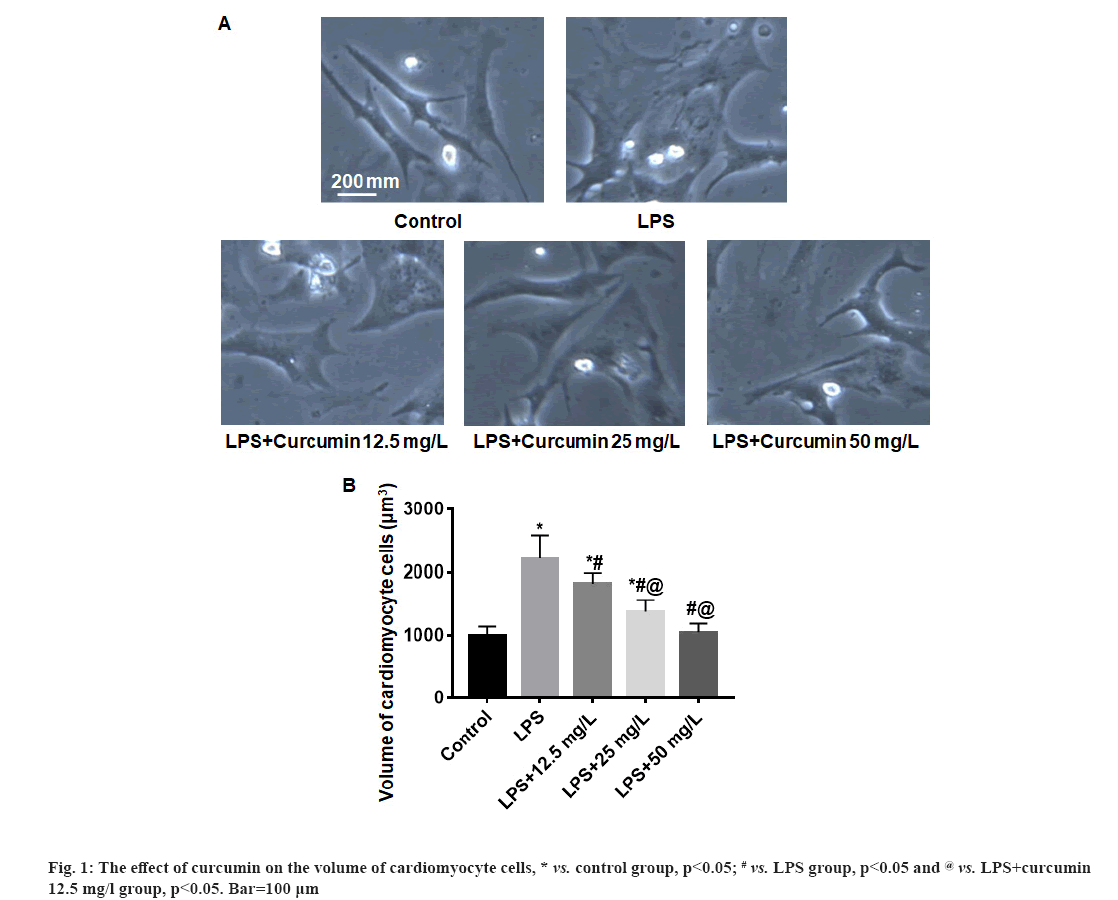
The effect of curcumin on the volume of cardiomyocyte cells were shown in fig. 1. In the white field of vision, the shape of cardiomyocytes is polygonal, fusiform or irregular triangle (fig. 1A). Compared with the control group, the volume of cardiomyocytes in the LPS group increased significantly (p<0.05). The volume of cardiomyocyte in the LPS+curcumin 12.5 mg/l, 25.0 mg/l and 50.0 mg/l groups gradually decreased with the increase of curcumin dose and the volume of cardiomyocyte in the LPS+curcumin 50.0 mg/l group were close to that of the control group (fig. 1B).
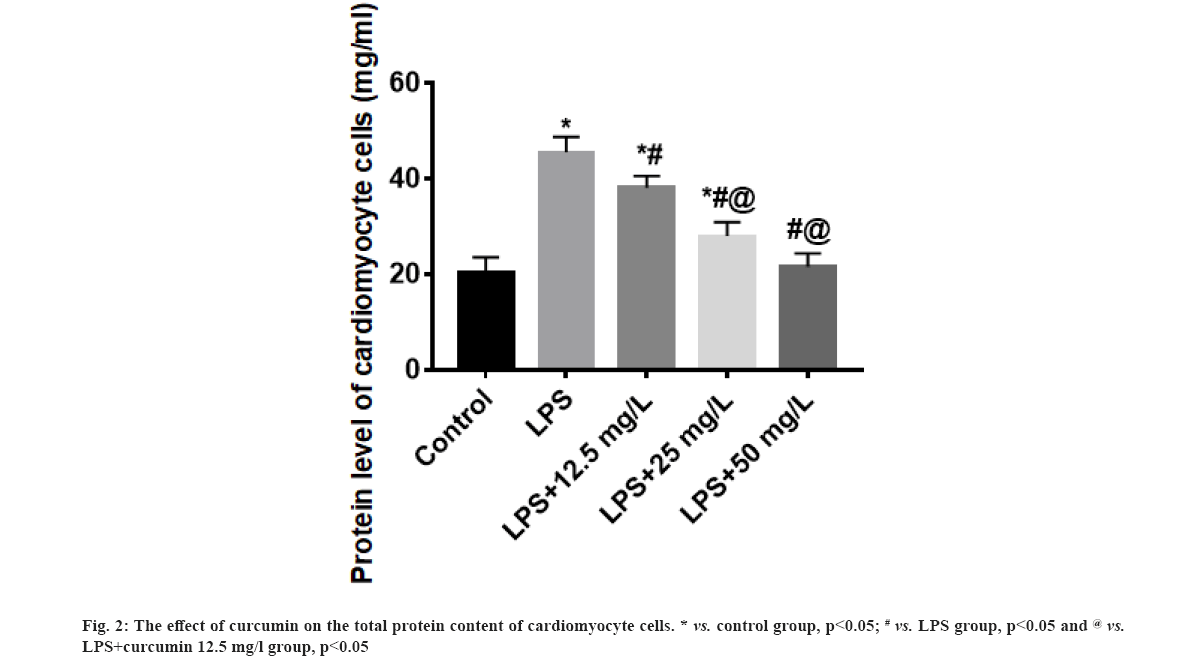
The effect of curcumin on the total protein content of cardiomyocyte cells were shown here. Compared with the control group, the total protein content of cardiomyocytes in the LPS group was significantly increased (p<0.05). The total protein content of cardiomyocytes in the LPS+curcumin 12.5 mg/l, 25.0 mg/l and 50.0 mg/l groups gradually decreased with the increase of curcumin dose and the total protein content of cardiomyocyte in the LPS+curcumin 50.0 mg/l group were close to that of the control group (fig. 2).
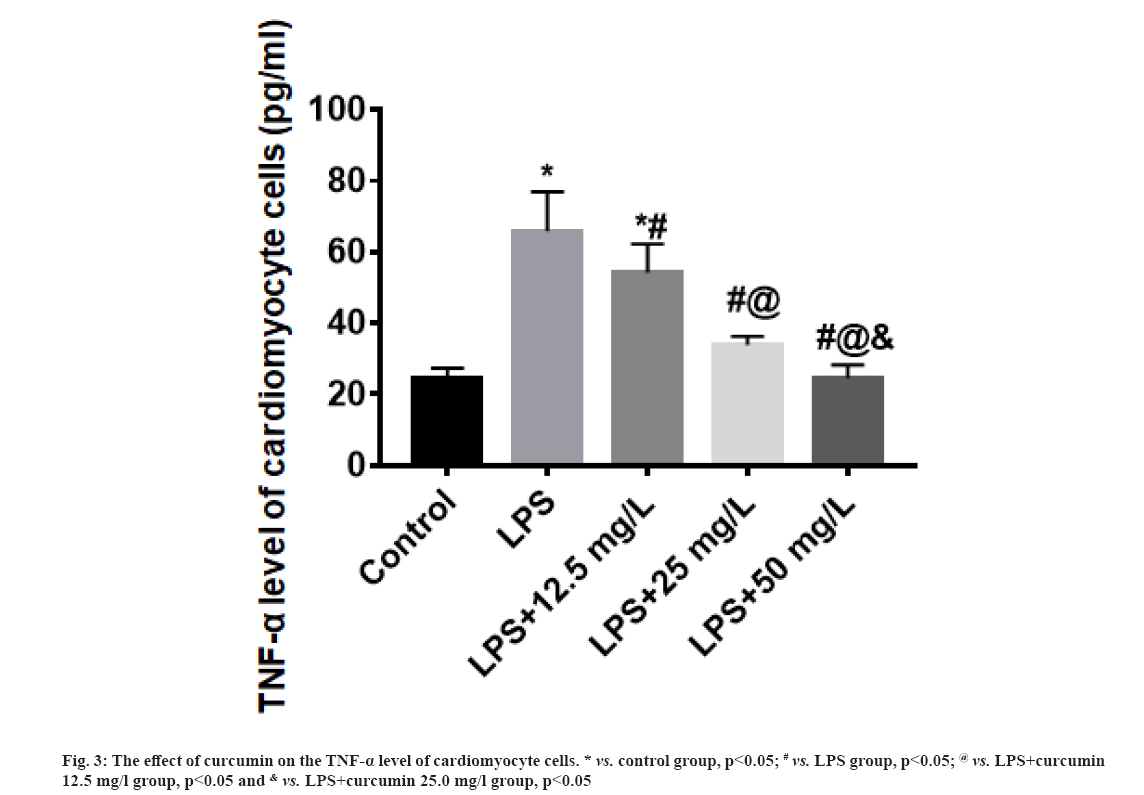
The effect of curcumin on the TNF-α level of cardiomyocyte cells were explained here. Compared with the control group, the TNF-α level of cardiomyocytes in the LPS group was significantly increased (p<0.05). The TNF-α level of cardiomyocytes in the LPS+curcumin 12.5 mg/l, 25.0 mg/l and 50.0 mg/l groups gradually decreased with the increase of curcumin dose, and the TNF-α level of cardiomyocyte in the LPS+curcumin 50.0 mg/l group were close to that of the control group (fig. 3).
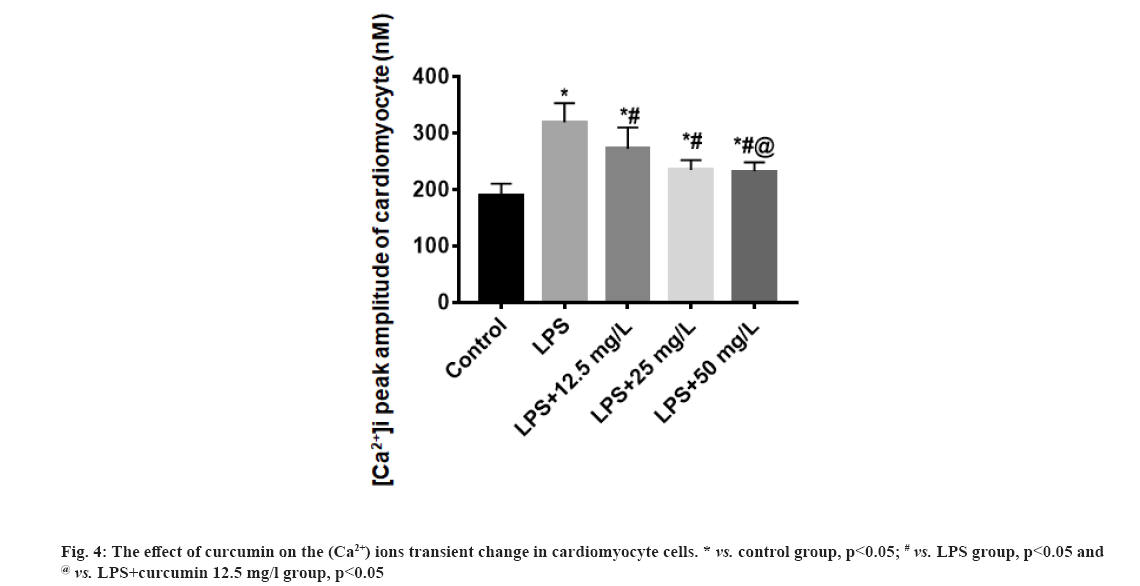
The effect of curcumin on the (Ca2+) ions transient change in cardiomyocyte cells were explained here. Compared with the control group, the (Ca2+) ions peak amplitude of cardiomyocytes in the LPS group increased (p<0.05); the (Ca2+) ions peak amplitude of cardiomyocytes in the LPS+curcumin 12.5 mg/l group, LPS+curcumin 25 mg/l and LPS+curcumin 50 mg/l was lower than that in the LPS group (p<0.05). With the increase of curcumin dose, the (Ca2+) ions peak amplitude of cardiomyocytes decreased (fig. 4).
LPS or endotoxin is a component in the cell wall of Gram-negative bacteria and is the main pathogenic component. LPS can activate the immune and inflammatory response system in the body, causing extensive damage to the body and the heart is the most vulnerable organ[16]. Most patients with endotoxemia, have symptoms of cardiac insufficiency, which lead to cardiomyocyte hypertrophy[17]. Many studies have shown that LPS can induce cardiac hypertrophy, increase the total protein content of cardiomyocyte and increase the volume of cardiomyocyte[18,19]. The results of this study also showed that LPS induced the rat cardiomyocyte hypertrophy in vitro, which was consistent with other studies.
It is currently believed that the pathogenic mechanism of the body damage caused by LPS is involved in multiple signal transduction pathways mediated by TLR4[20,21]. TNF-α is one of its downstream signals[22,23]. The results of this study showed that, while LPS induced rat cardiomyocyte hypertrophy, it also promoted the high expression of TNF-α in cardiomyocyte. TNF-α has a variety of biological effects and can induce cardiomyocyte hypertrophy through the biological pathways as followed: It interacts with the renin-angiotensin system and causes the proliferation of reactive oxygen intermediates, thereby inducing cardiomyocyte hypertrophy[24]; it can also interact with Interleukins (IL-1 and IL-6), etc., to cause cardiomyocyte hypertrophy[25].
Intracellular Ca2+ plays a central role in cardiac hypertrophy. Studies have shown that the increase of intracellular Ca2+ signal can activate many downstream signal factors, promote the expression of related genes and produce cardiomyocyte hypertrophy[26]. The results of this study showed that LPS induced an increase in the instantaneous peak of (Ca2+) ions in cardiomyocytes. It may be related to the decrease in the activity of Sodium-Potassium Adenosine Triphosphatase (Na+-K+-ATPase) in cardiomyocytes caused by LPS and cause the retention of Sodium ions (Na+) in the cell. Due to the increase in the intracellular Na+, a large amount of Ca2+ flows into the cell through the exchange of Na+-Ca2+-ATPase and intracellular Ca2+ pumping decreases due to the Ca2+-ATPase activity decreases, which eventually leads to intracellular Ca2+ overload. In addition, studies have shown that LPS induced high expression of TNF-α in cardiomyocytes which can also promote the increase of intracellular Ca2+ concentration[27].
Curcumin is an active ingredient extracted from the rhizome of turmeric. It belongs to polyphenols and consists of two O-methylated phenols and one beta-diketone (β-diketone)[28]. As a traditional Chinese medicine component, curcumin can regulate numerous cellular metabolic pathways and signal transduction pathways. A large number of clinical medical studies have shown that curcumin has a wide range of pharmacological effects such as anti-tumor[29], lowering blood lipids[30], anti-inflammatory[31], anti-oxidation[32], anti-atherosclerosis[33] and scavenging free radicals[13]. It also has preventive effects on many diseases. Its attribute is mild, so there is no obvious toxic and side effects after long-term use[34-37]. The results of this study showed that compared with the LPS group, the treatment of 12.5, 25 and 50 mg/l of curcumin could inhibit the increase of cardiomyocyte cell volume, reduce the increase of total cell protein content and inhibit the increase of TNF-α expression level and increase in the instantaneous peak of (Ca2+) ions induced by LPS, and showed a dose-dependent effect. The specific mechanism needs further study in the following days.
In summary, curcumin has a protective effect on rat cardiomyocyte hypertrophy induced by LPS, which may be related to the inhibition of TNF-α production and the inhibition of Ca2+ overload. This provides an experimental basis for applying curcumin to treat cardiomyocyte hypertrophy.
Ethical approval:
The animals used in this experiment were approved by the Institutional Animal Care and Use Committee of Nantong University (S20200302-039).
Acknowledgements:
This work was supported by Nantong Science and Technology Project (JC2019079, YYZ17097).
Conflict of interests:
The authors declared no conflict of interest.
References
- Shakaryants GA, Kozhevnikova MV, Kaplunova VY, Privalova EV, Lishuta AS, Korobkova EO, et al. Focus on the myocardial hypertrophy from the perspective of transcriptomics and metabolomics. Kardiologiia 2020;60(4):120-9.
[Crossref] [Google scholar] [PubMed]
- Ma D, Zhang J, Zhang Y, Zhang X, Han X, Song T, et al. Inhibition of myocardial hypertrophy by magnesium isoglycyrrhizinate through the TLR4/NF-κB signaling pathway in mice. Int Immunopharmacol 2018;55:237-44.
[Crossref] [Google scholar] [PubMed]
- Lasselin J, Schedlowski M, Karshikoff B, Engler H, Lekander M, Konsman JP. Comparison of bacterial lipopolysaccharide-induced sickness behavior in rodents and humans: Relevance for symptoms of anxiety and depression. Neurosci Biobehav Rev 2020;115:15-24.
[Crossref] [Google scholar] [PubMed]
- Mazgaeen L, Gurung P. Recent advances in lipopolysaccharide recognition systems. Int J Mol Sci 2020;21(2):379.
[Crossref] [Google scholar] [PubMed]
- Huszczynski SM, Lam JS, Khursigara CM. The role of Pseudomonas aeruginosa lipopolysaccharide in bacterial pathogenesis and physiology. Pathogens 2019;9(1):6.
[Crossref] [Google scholar] [PubMed]
- Alexandrov P, Zhao Y, Li W, Lukiw W. Lipopolysaccharide-stimulated, NF-kB, miRNA-146a and miRNA-155-mediated molecular-genetic communication between the human gastrointestinal tract microbiome and the brain. Folia Neuropathol 2019;57(3):211-9.
[Crossref] [Google scholar] [PubMed]
- Batista CR, Gomes GF, Candelario-Jalil E, Fiebich BL, de Oliveira AC. Lipopolysaccharide-induced neuroinflammation as a bridge to understand neurodegeneration. Int J Mol Sci 2019;20(9):2293.
[Crossref] [Google scholar] [PubMed]
- Kwiatkowska K, Ciesielska A. Lipid-mediated regulation of pro-inflammatory responses induced by lipopolysaccharide. Postepy Biochem 2018;64(3):175-82.
[Crossref] [Google scholar] [PubMed]
- Tan Y, Wan HH, Sun MM, Zhang WJ, Dong M, Ge W, et al. Cardamonin protects against lipopolysaccharide-induced myocardial contractile dysfunction in mice through Nrf2-regulated mechanism. Acta Pharmacol Sin 2021;42(3):404-13.
[Crossref] [Google scholar] [PubMed]
- Zhang J, Wang M, Ye J, Liu J, Xu Y, Wang Z, et al. The anti-inflammatory mediator resolvin E1 protects mice against lipopolysaccharide-induced heart injury. Front Pharmacol 2020;11:203.
[Crossref] [Google scholar] [PubMed]
- Jan RL, Yang SC, Liu YC, Yang RC, Tsai SP, Huang SE, et al. Extracellular heat shock protein HSC70 protects against lipopolysaccharide-induced hypertrophic responses in rat cardiomyocytes. Biomed Pharmacother 2020;128:110370.
[Crossref] [Google scholar] [PubMed]
- Li X, Liu J, Wang J, Zhang D. Luteolin suppresses lipopolysaccharide‑induced cardiomyocyte hypertrophy and autophagy in vitro. Mol Med Rep 2019;19(3):1551-60.
[Crossref] [Google scholar] [PubMed]
- Zheng D, Huang C, Huang H, Zhao Y, Khan MR, Zhao H, et al. Antibacterial mechanism of curcumin: A review. Chem Biodivers 2020;17(8):e2000171.
[Crossref] [Google scholar] [PubMed]
- Miriyala S, Panchatcharam M, Rengarajulu P. Cardioprotective effects of curcumin. Adv Exp Med Biol 2007;595:359-77.
[Crossref] [Google scholar] [PubMed]
- Cao Q, Zhang J, Gao L, Zhang Y, Dai M, Bao M. Dickkopf‑3 upregulation mediates the cardioprotective effects of curcumin on chronic heart failure. Mol Med Rep 2018;17(5):7249-57.
[Crossref] [Google scholar] [PubMed]
- Banez MJ, Geluz MI, Chandra A, Hamdan T, Biswas OS, Bryan NS, et al. A systemic review on the antioxidant and anti-inflammatory effects of resveratrol, curcumin and dietary nitric oxide supplementation on human cardiovascular health. Nutr Res 2020;78:11-26.
[Crossref] [Google scholar] [PubMed]
- Guido MC, Clemente CF, Moretti AI, Barbeiro HV, Debbas V, Caldini EG, et al. Small interfering RNA targeting focal adhesion kinase prevents cardiac dysfunction in endotoxemia. Shock 2012;37(1):77-84.
[Crossref] [Google scholar] [PubMed]
- Chao CN, Lai CH, Badrealam KF, Lo JF, Shen CY, Chen CH, et al. CHIP attenuates lipopolysaccharide‐induced cardiac hypertrophy and apoptosis by promoting NFATc3 proteasomal degradation. J Cell Physiol 2019;234(11):20128-38.
[Crossref] [Google scholar] [PubMed]
- Magi S, Nasti AA, Gratteri S, Castaldo P, Bompadre S, Amoroso S, et al. Gram-negative endotoxin lipopolysaccharide induces cardiac hypertrophy: Detrimental role of Na+-Ca2+ exchanger. Eur J Pharmacol 2015;746:31-40.
[Crossref] [Google scholar] [PubMed]
- Tang F, Fan K, Wang K, Bian C. Atractylodin attenuates lipopolysaccharide-induced acute lung injury by inhibiting NLRP3 inflammasome and TLR4 pathways. J Pharmacol Sci 2018;136(4):203-11.
[Crossref] [Google scholar] [PubMed]
- El-Gaphar OA, Abo-Youssef AM, Halal GK. Levetiracetam mitigates lipopolysaccharide-induced JAK2/STAT3 and TLR4/MAPK signaling pathways activation in a rat model of adjuvant-induced arthritis. Eur J Pharmacol 2018;826:85-95.
[Crossref] [Google scholar] [PubMed]
- Silva CR, Melo BM, Silva JR, Lopes AH, Pereira JA, Cecilio NT, et al. S100A9 plays a pivotal role in a mouse model of herpetic neuralgia via TLR4/TNF pathway. Brain Behav Immun 2020;88:353-62.
[Crossref] [Google scholar] [PubMed]
- Yoshizaki S, Kijima K, Hara M, Saito T, Tamaru T, Tanaka M, et al. Tranexamic acid reduces heme cytotoxicity via the TLR4/TNF axis and ameliorates functional recovery after spinal cord injury. J Neuroinflammation 2019;16(1):1-5.
- Sriramula S, Cardinale JP, Francis J. Inhibition of TNF in the brain reverses alterations in RAS components and attenuates angiotensin II-induced hypertension. PLoS One 2013;8(5):e63847.
[Crossref] [Google scholar] [PubMed]
- Hedayat M, Mahmoudi MJ, Rose NR, Rezaei N. Proinflammatory cytokines in heart failure: Double-edged swords. Heart Fail Rev 2010;15(6):543-62.
[Crossref] [Google scholar] [PubMed]
- Cruz Junho CV, Trentin-Sonoda M, Alvim JM, Gaisler-Silva F, Carneiro-Ramos MS. Ca2+/Calmodulin-dependent kinase II delta B is essential for cardiomyocyte hypertrophy and complement gene expression after LPS and HSP60 stimulation in vitro. Braz J Med Biol Res 2019;52(7):e8732.
[Crossref] [Google scholar] [PubMed]
- Wang GJ, Wang HX, Yao YS, Guo LY, Liu P. The role of Ca2+/calmodulin-dependent protein kinase II and calcineurin in TNF-α-induced myocardial hypertrophy. Braz J Med Biol Res 2012;45:1045-51.
[Crossref] [Google scholar] [PubMed]
- Zielińska A, Alves H, Marques V, Durazzo A, Lucarini M, Alves TF, et al. Properties, extraction methods and delivery systems for curcumin as a natural source of beneficial health effects. Medicina 2020;56(7):1-19.
[Crossref] [Google scholar] [PubMed]
- Rutz J, Janicova A, Woidacki K, Chun FK, Blaheta RA, Relja B. Curcumin-a viable agent for better bladder cancer treatment. Int J Mol Sci 2020;21(11):3761.
[Crossref] [Google scholar] [PubMed]
- Qin S, Huang L, Gong J, Shen S, Huang J, Ren H, et al. Efficacy and safety of turmeric and curcumin in lowering blood lipid levels in patients with cardiovascular risk factors: A meta-analysis of randomized controlled trials. Nutr J 2017;16(1):1-10.
[Crossref] [Google scholar] [PubMed]
- Hasanzadeh S, Read MI, Bland AR, Majeed M, Jamialahmadi T, Sahebkar A. Curcumin: An inflammasome silencer. Pharmacol Res 2020;159:104921.
[Crossref] [Google scholar] [PubMed]
- Bahrami A, Sathyapalan T, Moallem SA, Sahebkar A. Counteracting arsenic toxicity: Curcumin to the rescue? J Hazard Mater 2020;400:123160.
[Crossref] [Google scholar] [PubMed]
- Mohammadian Haftcheshmeh S, Karimzadeh MR, Azhdari S, Vahedi P, Abdollahi E, Momtazi‐Borojeni AA. Modulatory effects of curcumin on the atherogenic activities of inflammatory monocytes: Evidence from in vitro and animal models of human atherosclerosis. Biofactors 2020;46(3):341-55.
[Crossref] [Google scholar] [PubMed]
- Oliveira S, Monteiro-Alfredo T, Silva S, Matafome P. Curcumin derivatives for Type 2 Diabetes management and prevention of complications. Arch Pharm Res 2020;43(6):567-81.
[Crossref] [Google scholar] [PubMed]
- Zhang Y, Li L, Zhang J. Curcumin in antidepressant treatments: An overview of potential mechanisms, pre‐clinical/clinical trials and ongoing challenges. Basic Clin Pharmacol Toxicol 2020;127(4):243-53.
[Crossref] [Google scholar] [PubMed]
- Ashrafizadeh M, Najafi M, Makvandi P, Zarrabi A, Farkhondeh T, Samarghandian S. Versatile role of curcumin and its derivatives in lung cancer therapy. J Cell Physiol 2020;235(12):9241-68.
- Askarizadeh A, Barreto GE, Henney NC, Majeed M, Sahebkar A. Neuroprotection by curcumin: A review on brain delivery strategies. Int J Pharm 2020;585:119476.