- *Corresponding Author:
- D. F. Basri
School of Diagnostic and Applied Health Sciences, Faculty of Health Sciences, Universiti Kebangsaan, Jalan Raja Muda Abdul Aziz, 50300 Kuala Lumpur, Malaysia
E-mail: dayang@ukm.edu.my
| Date of Submission | 01 July 2016 |
| Date of Revision | 15 October 2016 |
| Date of Acceptance | 06 November 2016 |
| Indian J Pharm Sci 2016;78(6):748-754 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
Pterostilbene is biologically classified as a phytoalexin, which is part of the plant's defense system in response to pathogen infection. The present study is to identify the type of interaction and post-antibiotic effect time of pterostilbene in combination with linezolid against methicillin-resistant Staphylococcus aureus American type culture collection 43300 and methicillin-resistant S. aureus American type culture collection 33591. The minimum inhibitory concentration values of individual antimicrobial agents were determined using microbroth dilution technique whereas the microdilution checker board assay was employed to verify the type of interaction of the combined agents from the fractional inhibitory concentration index values. The post-antibiotic effect time of pterostilbene and linezolid and in combination treatment was evaluated using viable plate count method. The minimum inhibitory concentration value of pterostilbene against both strains were the same (31.25 μg/ml) compared to linezolid at 0.78 μg/ml and 1.56 μg/ml, against 43300 and 33591 strain, respectively. microdilution checker analysis of pterostilbene in combination with linezolid against both methicillin-resistant S. aureus strains displayed fractional inhibitory concentration value of 1.25. Despite the indifference interaction between pterostilbene and linezolid as shown by microdilution checker study, post-antibiotic effect analysis demonstrated antagonism as depicted by a decrease of post-antibiotic effect time of linezolid by pterostilbene against both methicillin-resistant S. aureus strains. The duration of post-antibiotic effect time obtained for pterostilbene were twice (3.6±0.15 h) that of linezolid (1.8±0.31 h) against methicillin-resistant S. aureus American type culture collection 43300. However, pterostilbene exhibited shorter post-antibiotic effect time of 0.3±0.10 h compared to linezolid (0.9±0.06 h) against 33591 strain. In conclusion, pterostilbene was not able to prolong the post-antibiotic effect duration of linezolid in both strains and each of the antimicrobials demonstrated a more favorable timing of doses when these agents were used singly.
Keywords
Pterostilbene, indifference, methicillin-resistant Staphylococcus aureus, minimum inhibitory concentration, fractional inhibitory concentration, post-antibiotic effect
Methicillin-resistant Staphylococcus aureus (MRSA) has emerged as an important cause of hospital-acquired and community-acquired infection worldwide. The extensively increased incidence of MRSA in the past few years merits special attention as these strains are not only resistant to methicillin but also to most clinically available antibiotics, including towards the newest antibacterial drug currently in the market[1,2]. The emergence of MRSA causes a wide range of illnesses, from skin and wound infections to pneumonia and blood stream infection that lead to significant morbidity and increased mortality[3]. Prolonged hospitalization of MRSA patient caused financial burden to government as well as to the patient[4,5].
Vancomycin and linezolid are the major antibiotics currently used against MRSA infection[6]. However, the emergence of vancomycin-resistant S. aureus (VRSA) in 2002 in the United States[7] and the first outbreak of linezolid-resistant MRSA reported in 2008[8] limits the use of these antibiotics. The resistance to these antibiotics is constantly increasing[9,10] hence, there is an urgent need for a new antimicrobial agent which will be effective to combat these multidrug-resistant bacteria.
One approach is treatment with antibacterial combination using two or more antibacterial to combat these multidrug-resistant bacteria[11]. In search of more effective chemotherapeutic agents for treating microbial infection, combination therapy becomes an important strategy as synergistic interaction can potentially increase efficacy, reduce toxicity, cure faster, prevent the emergence of resistance and provide broad-spectrum of activity than monotherapy regimen[12]. Phytochemicals have been considered as promising source of novel antibacterial[13,14] due to the successful defence mechanism developed by plants.
Pterostilbene is a phytochemical classified as a stilbene compound. A high content of these phenylpropanoid compounds, notably resveratrol and its analogue pterostilbene, identified in Vitis vinifera leaves[15], heartwood of sandalwood Pterocarpus marsupium[16] and in some species of Vaccinium berries[17] are known to have diverse pharmacological activities[18]. Pterostilbene possesses potent antioxidant activity[19], anticancer property[20], anti-inflammatory[21], antifungal[22] and to date, possess antibacterial activity against MRSA[23].
Thus, the present study is to identify the interaction effect and post-antibiotic effect (PAE) of pterostilbene in combination with linezolid against MRSA. Linezolid was chosen in combination with pterostilbene as it has broad in vitro activity against antibiotic-susceptible and antibiotic-resistant Gram-positive bacteria including activity against MRSA. This approach is hope to come up new antimicrobial agent for the battle against MRSA, hence, decrease the total intake of antibiotic as well as reducing treatment cost, morbidity and mortality rate.
Materials and Methods
Pterostilbene was purchased commercially from EMD Biosciences/Calbiochem (USA) and both pterostilbene and linezolid were prepared according to the manufacturer’s recommendation which stated that dilution of these antimicrobial agents was performed depending on their weight with respect to their minimum inhibitory concentration (MIC) values. The antimicrobial solutions were mixed with 100% dimethylsulfoxide (DMSO) solvent using Autovortex mixer until the powder dissolved completely prior to the experiments.
Preparation of bacterial inoculum
In the present study, the bacteria strains used were MRSA ATCC 33591 and 43300 obtained from the American Type Culture Collection (ATCC). The stock cultures were grown on nutrient agar (Merck, Germany) slant at 37° for 24 h to obtain isolated colony. One or two single colony that grew on Mueller- Hinton agar (MHA) was cultured and transferred into the sterile Mueller-Hinton broth (MHB) and incubated at 37° for 24 h. The inoculum size of test strain was standardized to absorbance reading ranging from 0.08- 0.13 equivalent to 0.5 McFarland’s standard using spectrophotometer at a wavelength of 625 nm. The 0.5 McFarland suspension was then diluted to 1:100 in MHB resulting to a final bacterial inoculum size of approximately 1×106 colony forming units (CFU)/ml.
Determination of MIC
MIC values of pterostilbene and linezolid against both MRSA strains were determined by microdilution technique using 96-well microtiter plate at final concentration ranging from 0.195 to 100 μg/ml and 0.98 to 500 μg/ml, respectively. The tested compound in MHB was used as negative control to ensure medium sterility while inoculum in MHB was used to control the adequacy of the broth for bacterial growth in positive control well. For confirmation, 40 μl of triphenyl tetrazolium chloride (TTC) at 2 mg/ml was added into each well to allow detection of viability. The MIC values were taken as the lowest concentration of the tested agents in the well of the microtiter plate that inhibited the visible growth of organism after 24 h of incubation at 37°. This assay was done in triplicate for consistency.
Determination of fractional inhibitory concentration (FIC)
Interaction study between pterostilbene and linezolid against MRSA ATCC 43300 and 33591 were determined from the FIC index using microdilution checkerboard (MDC) method. The tested antimicrobials were prepared in the well of the microtiter plate at six different concentrations namely, 1MIC, 1/2×MIC, 1/4×MIC, 1/8×MIC, 1/16×MIC and 0×MIC in MHB before the diluted bacterial suspension at a final inoculum of 106 CFU/ml was added. FIC values for pterostilbene or linezolid were determined by dividing the concentration of the antimicrobial agents necessary to inhibit growth in a given row or column by the MIC value of the test organism for that individual agents alone. To characterize the interaction between each combination, FIC index was calculated using the formula[24], FICindex (Σ FIC)=FICA+FICB=A/MICA+B/ MICB, where, A is the MIC of drug A in combination, B is the MIC of drug B in combination, MICA is the MIC of drug A alone, MICB is the MIC of drug B alone.
The FIC index was then used to determine if synergy, antagonism, or indifference occurred as a result of interaction between the combinations. The FIC index range of between 0.5 and 4.0 is commonly used to define the type of combination whether they are synergistic (<0.5), additive (0.5-1.0), indifference (1.0- 4.0) and antagonism (>4.0)[25].
Determination PAE time
PAE time of pterostilbene, linezolid and pterostilbene in combination with linezolid were determined using the viable plate count method in both the MRSA ATCC 43300 and 33591. The treatment groups at concentration 10×MIC in the bacterial suspension at 106 bacteria/ml were incubated for 1 h at 37°. The control group comprised MHB and the bacterial suspension. After 1 h incubation, dilution at 1:1000 was performed in MHB. A volume of 2 μl of the diluted sample was streaked onto MHA at 0, 2, 4, 6, 8, 10 and 24 h in order to count the number of colonies presented after 24 h of incubation at 37°. This was performed in triplicate aseptically. Graph of log10 CFU/ml against time was plotted where the duration of PAE time was calculated from the graph using the equation PAE=T-C[26], where T is the time required for the treated organism to increase 1 log10 CFU/ml after removal of treatment agents and C is the time required for the control organism to increase 1 log10 CFU/ml after completion of the same procedure.
Results and Discussion
The results of the microbroth dilution assay are shown in Tables 1 and 2. It can be seen from Table 1 that the MIC values of pterostilbene against both MRSA strains were the same i.e. 31.25 μg/ml. On the other hand, the MIC of linezolid (Table 2) against MRSA ATCC 43300 and 33591 were 0.78 and 1.56 μg/ml, respectively. The antimicrobial activity of pterostilbene was 40 and 20 times less potent than that of linezolid, against MRSA ATCC 43300 and 33591, respectively. The MIC values of the combination of antimicrobials were equal to the MIC values of the single substances as can be concluded from Table 3 which showed the results of microdilution checkerboard assay. The present finding demonstrated that the FIC value between pterostilbene and linezolid was the same against MRSA ATCC 43300 and 33591 at FIC index value of 1.25. This indicated that the interaction between the two antimicrobials displayed indifference against both MRSA strains.
|
Concentration (μg/ml) |
ATCC 43300 | ATCC 33591 | ||||
|---|---|---|---|---|---|---|
| Pterostilbene | Positive control |
Negative control |
Pterostilbene | Positive control |
Negative control |
|
| 500 | - | + | - | - | + | - |
| 250 | - | + | - | - | + | - |
| 125 | - | + | - | - | + | - |
| 62.50 | - | + | - | - | + | - |
| 31.25 | - | + | - | - | + | - |
| 15.63 | + | + | - | + | + | - |
| 7.81 | + | + | - | + | + | - |
| 3.91 | + | + | - | + | + | - |
| 1.95 | + | + | - | + | + | - |
| 0.98 | + | + | - | + | + | - |
Table 1: Determination of Mic Value of Pterostilbene Against Mrsa Atcc 43300 and 33591
|
Concentration (μg/ml) |
ATCC 43300 | ATCC 33591 | ||||
|---|---|---|---|---|---|---|
| Linezolid | Positive control |
Negative control |
Linezolid | Positive control |
Negative control |
|
| 100 | - | + | - | - | + | - |
| 50 | - | + | - | - | + | - |
| 25 | - | + | - | - | + | - |
| 12.50 | - | + | - | - | + | - |
| 6.25 | - | + | - | - | + | - |
| 3.13 | - | + | - | - | + | - |
| 1.56 | - | + | - | - | + | - |
| 0.78 | - | + | - | + | + | - |
| 0.39 | + | + | - | + | + | - |
| 0.195 | + | + | - | + | + | - |
Table 2: Determination of Mic Value of Linezolid Against Mrsa Atcc 43300 and Mrsa Atcc 33591
| Strains | MIC (μg/ml) in combination |
FIC index | |||||
|---|---|---|---|---|---|---|---|
| Antibiotic (A) |
MIC (A) |
Phytochemical (B) |
MIC (B) |
FIC A | FIC B | Outcome | |
| ATCC 43300 | Linezolid | 0.78 | Pterostilbene | 31.25 | 0.195 | 31.25 | 1.25 (IN) |
| ATCC 33591 | Linezolid | 1.56 | Pterostilbene | 31.25 | 0.39 | 31.25 | 1.25 (IN) |
PS denotes partial synergism (FIC>0.5<1); IN denotes indifference (FIC>1≤4); AD denotes additive (FIC=1)
Table 3: Fic Index Values for Pterostilbene in Combination with Linezolidagainst two Mrsa Strains
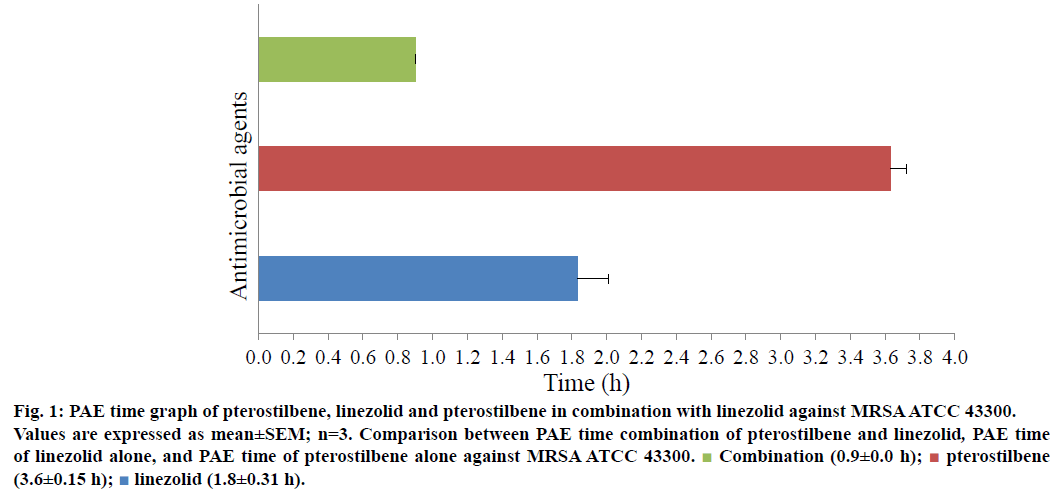
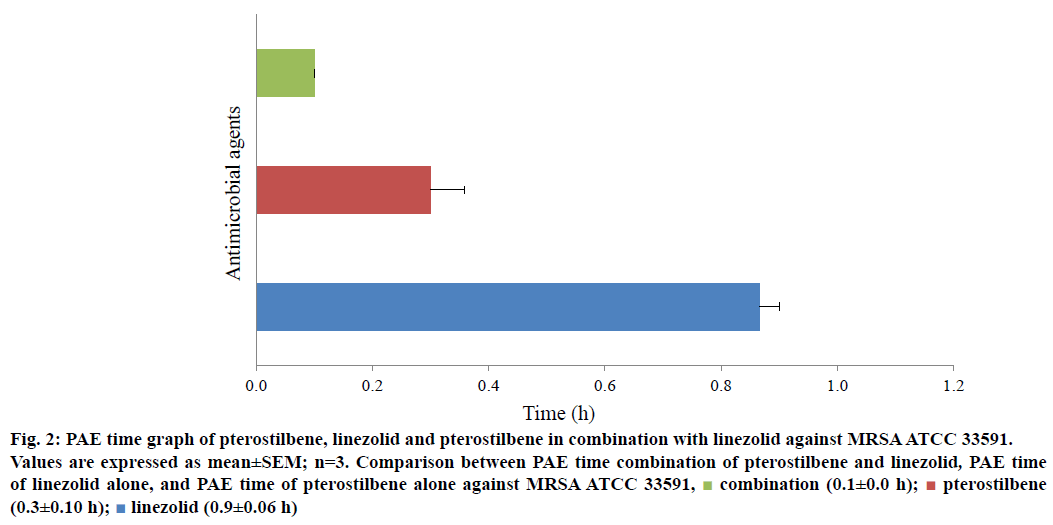
Figures 1 and 2 illustrated the comparison of PAE time of pterostilbene alone, linezolid alone and their combination against MRSA ATCC 43300 and 33591, respectively. The duration of PAE obtained for pterostilbene, linezolid and pterostilbene-linezolid combination against MRSA ATCC 43300 were 3.6±0.15 h, 1.8±0.31 h and 0.9±0.00 h, respectively (Figure 1). It was observed that PAE duration time for pterostilbene alone was the longest, followed by linezolid alone whereas the combination treatment has the shortest suppression time of bacterial regrowth. However, pterostilbene exhibited shorter PAE time (0.3±0.1 h) when exposed singly to MRSA ATCC 33591 compared to linezolid (0.9±0.06 h) as shown in Figure 2.
Figure 1: PAE time graph of pterostilbene, linezolid and pterostilbene in combination with linezolid against MRSA ATCC 43300.
Values are expressed as mean±SEM; n=3. Comparison between PAE time combination of pterostilbene and linezolid, PAE time of linezolid alone, and PAE time of pterostilbene alone against MRSA ATCC 43300.  Combination (0.9±0.0 h);
Combination (0.9±0.0 h);  peterostilbene (3.6±0.15 h);
peterostilbene (3.6±0.15 h);  linezolid (1.8±0.31 h)
linezolid (1.8±0.31 h)
Figure 2: PAE time graph of pterostilbene, linezolid and pterostilbene in combination with linezolid against MRSA ATCC 33591.
Values are expressed as mean±SEM; n=3. Comparison between PAE time combination of pterostilbene and linezolid, PAE time of linezolid alone, and PAE time of pterostilbene alone against MRSA ATCC 33591,  combination (0.1±0.0 h);
combination (0.1±0.0 h);  peterostilbene (0.3±0.10 h);
peterostilbene (0.3±0.10 h);  linezolid (0.9±0.06 h)
linezolid (0.9±0.06 h)
The present finding demonstrated that pterostilbene showed equal anti-MRSA potency against MRSA ATCC 43300 and 33591, which is in accordance with[22] both MRSA strains were equally susceptible towards the effect of pterostilbene. However, this is in contrast with previous study on other stilbenoids that showed less potency against MRSA ATCC 33591 with higher MIC of ��-viniferin and johorenol A against this strain[27]. Pterostilbene, resveratrol, ��-viniferin and johorenol A are formed in the flavonoid biosynthesis pathway belonging to the phenylpropanoid family[28]. Resveratrol, on the other hand was reported to possess antimicrobial activity against grapevine pathogens[29]. Thus, pterostilbene could be an alternative treatment to overcome the problem of bacterial infection as it was known that selected phytochemicals have the ability to combat Escherichia coli and S. aureus and their biofilms[30].
The FIC index value of >1 but <4 between pterostilbene and linezolid indicated that this interaction was indifference despite a marked reduction of MIC value of linezolid by fourfold against MRSA strains. This is also in line with the results reported by Zhang et al.[31] that there was a significant reduction in the MIC value of antibiotic which showed both indifference and additive effects, when used in combination. According to Sopirala et al.[32] if the MIC of an antibiotic changed within a one-fold dilution, the result was considered as insignificant but because the decreased in the MIC of linezolid in this study was four-fold, the interaction could not possibly be indifference. However, the current FIC data was supported by our previous literature[23] that pterostilbene lacked interaction with linezolid despite the change in the MIC value of linezolid against both MRSA strains.
The PAE time of linezolid at 10×MIC in this study was within acceptable range of PAE parameters of between 0.7 h and 1.3 h after exposure of clinical strains of MRSA to linezolid at 1×MIC and 4×MIC[33]. Pterostilbene appeared to reduce the PAE time of linezolid by half suggesting that it antagonized the persistent post-antimicrobial effect of linezolid against 43300 strain. Linezolid, on the other hand, reduced the time taken for bacterial regrowth after removal of pterostilbene by 4 times which indicates that the interaction between the 2 agents was antagonism. As far as ATCC 43300 was concerned, pterostilbene demonstrated a prolonged PAE time after exposure to this MRSA strain as singly treatment. This is supported by a study on Quercus infectoria gall extract which showed longer PAE value compared to vancomycin as a standard antibiotic[34] against MRSA ATCC 33591. However, pterostilbene exhibited shorter PAE time when exposed singly to MRSA ATCC 33591 compared to linezolid despite the same pattern of combinational PAE observed. In other words, pterostilbene still seemed to reduce the PAE time of linezolid meaning that it antagonized the effect of linezolid in sustaining its PAE. Linezolid, on the other hand, also reduced the time taken for bacterial regrowth after removal of pterostilbene by three-fold which confirms that the interaction between these 2 agents was antagonism. In short, pterostilbene was not capable of prolonging the duration of PAE time of linezolid in both MRSA ATCC strains. This defined pattern of antagonism in PAE study did not correlate with indifference effect from the FIC result. This contradicts with[35] that the antimicrobial combination used affected the PAE either as additive or an indifferent manner against MRSA. However, there is no report on the PAE of pterostilbene to date. Enhancement of prolongation of PAE against S. aureus was primarily dependent on the ability of each individual drug to induce a PAE since the final PAE was a rough mathematical sum of the individual PAE of both agents[35].
In the current study, it was noted that when only one of the agents induced a PAE, the final result was similar to the PAE of that particular drug depicted as indifference. Pterostilbene did not increase the PAE of a standard antibiotic, despite exhibiting a PAE itself. Pterostilbene was unique in that it showed lasting suppression of bacterial regrowth, but when employed with linezolid, the combination effect reduced its PAE. Based on the data in this study, we can postulate that pterostilbene could possibly have the same mechanism of action as that of linezolid as proposed in our previous finding[23] that pterostilbene could target the cell wall different from where oxacillin acts. Further studies to confirm that pterostilbene targets the bacterial protein different from the penicillin-binding protein where oxacillin acts are clearly needed to validate this hypothesis.
As conclusion, in spite of the indifference effect portrayed between pterostilbene and linezolid from FIC index value, PAE analysis confirmed antagonism interaction as pterostilbene was shown to decrease the duration of PAE time of linezolid against both the MRSA ATCC 43300 and 33591. This finding concluded that as a phytoalexin stilbenoid, pterostilbene was not capable of prolonging the duration of PAE time of linezolid in order to produce advantageous optimal antimicrobial dosing intervals in combination therapy against MRSA infection. The significance of the present study highlighted that pterostilbene and linezolid demonstrated a more favourable timing of dosage when these agents were used singly rather than in combination.
Acknowledgements
This project is funded by the Malaysian Government under the Fundamental Research Grant Scheme FRGS /2/2014/SG05/UKM/02/3.
Conflict of interest
The authors have declared that they have no competing interests.
Financial support and sponsorship
Nil.
References
- Adwan G, Mhanna M. Synergistic effects of plant extracts and antibiotics on Staphylococcus aureusstrains isolated from clinical specimens.Middle East J Sci Res 2008;3:134-39.
- Stavri M, Piddock LJV, Gibbons S. Bacterial efflux pumps inhibitors from natural sources. J Antimicrob Chemother 2007;59:1247-60.
- Lau QY, Tan YYF, Goh CCY, Lee DJQ, Ng FM, Ong EHQ, et al. An FDA-drug library screen for compounds with bioactivities against methicillin-resistant Staphylococcus aureus(MRSA). Antibiotics2015;4:424-34.
- Shorr AF, Combes A, Kollef MH, Chastre J. Methicillin-resistant Staphylococcus aureusprolongs intensive care unit stay in ventilator-associated pneumonia, despite initially appropriate antibiotic therapy. Crit Care Med2006;34:700-6.
- Cosgrove SE, Qi Y, Kaye KS, Harbarth S, Karchmer AW, Carmeli Y. The impact of methicillin resistance in Staphylococcus aureus bacteremia on patient outcomes: mortality, length of stay, and hospital charges. Infect Control Hosp Epidemiol 2005;26:166-74.
- Harada Y, Chong Y, Shimono N, Miyake N, Uchida Y, Kadowaki M, et al.Nosocomial spread of methicillin-resistant Staphylococcus aureuswith b-lactam-inducible arbekacin resistance. J Med Microbiol2014;63:710-14.
- Bozdogan B, Esel D, Whitener C, Browne FA, Appelbaum PC. Antibacterial susceptibility of a vancomycin-resistant Staphylococcus aureusstrain isolated at the Hershey Medical Center. J Antimicrob Chemother 2003;52:864-68.
- Morales G, Picazo JJ, Baos E, Candel FJ, Arribi A, Peláez B, et al.Resistance to linezolid is mediated by the cfr gene in the first report of an outbreak, linezolid-resistant Staphylococcus aureus. Clin Infect Dis2010;50:821-25.
- Liu C, Bayer A, Cosgrove SE, Daum RS, Fridkin SK, Gorwitz RJ, et al.Clinical practice guidelines by the infectious diseases society of America for the treatment of methicilin-resistant Staphylococcus aureusinfections in adults and children: executive summary. Clin Infect Dis2011;52:285-92.
- Svahn SK, Göransson U, El-Seedi H, Bohlin L, Larsson DGJ, Olsen B, et al.Antimicrobial activity of filamentous fungi isolated from highly antibiotic-contaminated river sediment. Infect Ecol Epidemiol 2012;2:11591-96.
- Rybak MJ, McGrath BJ. Combination antimicrobial therapy for bacterial infections. Guidelines for the clinician. Drugs 1996;52:390-405.
- Marr KA, Boeckh M, Carter RA, Kim HW, Corey L. Combination antifungal therapy for invasive aspergillosis. Clin Infect Dis2004;39:797-802.
- Hemaiswarya S, Kruthiventi AK, Doble M. Synergism between natural products and antibiotics against infectious diseases. Phytomedicine2008;15:639-52.
- Moyo B,Mukanganyama S. Antibacterial effects of Cissus welwitschii and Triumfetta welwitschiiextracts against Escherichia coliand Bacillus cereus. Int J Bacteriol 2015;162028:10.
- Langcake P, Cornford CA, Pryce RJ. Identification of pterostilbene as a phytoalexin from Vitis viniferaleaves. Phytochemistry 1979;18:1025-27.
- Manickam M, Ramanathan M, Jahomi MA, Chansouria JP, Ray AB. Antihyperglycemic activity of phenolics from Pterocarpus marsupium. J Nat Prod 1997;60:609-10.
- Pezet R, Pont V. Identification of pterostilbene in grape berries of Vitis vinifera. Plant Physiol Biochem 1988;26:603-7.
- Roupe KA, Remsberg CM, Yáñez JA, Davies NM. Pharmacometrics of stilbenes: seguing towards the clinic. Curr Clin Pharmacol 2006;1:81-101.
- Rimando AM, Cuendet M, Desmarchelier C, Mehta RG, Pezzuto J, Duke SO. Cancer chemopreventive and antioxidant activities of pterostilbene, a naturally occurring analogue of resveratrol. J Agric Food Chem 2002;50:3453-57.
- Pan MH, Chiou YS, Chen WJ, Wang JM, Badmaev V, Ho CT. Pterostilbene inhibited tumor invasion via suppressing multiple signal transduction pathways in human hepatocellular carcinoma cells. Carcinogenesis 2009;30:1234-42.
- Pan MH, Chang YH, Tsai ML, Lai CS, Ho SY, Badmaev V,et al. Pterostilbene suppressed lipopolysaccharide induced up-expression of iNOS and COX-2 in murine macrophages. J Agric Food Chem 2008;56:7502-9.
- Li DD, Zhao LX, Mylonakis E, Hu GH, Zou Y, Huang TK, et al.In vitroand in vivoactivities of pterostilbene against Candida albicansbiofilms. Antimicrob Agents Chemother 2014;58:2344-55.
- Ishak SF, Ghazali AR, Mohamad Zin N, Basri DF. Pterostilbene enhanced anti-methicillin resistant Staphylococcus aureus(MRSA) activity of oxacillin. Am J Infect Dis 2016;12:1-10.
- Mackay ML, Milne K, Gould IM. Comparison of methods for assessing synergistic antibiotic interactions. Int J Antimicrob Agents2000;15:125-29.
- Bharadwaj R, Vidya A, Dewan B, Pal A. An in vitrostudy to evaluate the synergistic activity of norfloxacin and metronidazole. Indian J Pharm Sci 2003;35:220-26.
- Craig WA, Gudmundsson S. Postantibiotic effect. 4th ed. Baltimore Md, editor. USA: The Williams and Wilkins;1996.
- Basri DF, Xian LW, Abdul Shukor NI, Latip J. Bacteriostatic antimicrobial combination: antagonistic interaction between epsilon-viniferin and vancomycin against methicillin-resistantStaphylococcus aureus. BioMed Res Int 2014;461756:8.
- Chong J, Poutaraud A, Hugueny P. Metabolism and roles of stilbene in plants. Plant Sci 2009;177:143-55.
- Chalal M, Klinguer A, Echairi A, Meunier P, Vervandier-Fasseur D, Adrian M. Antimicrobial activity of resveratrol analogues. Molecules 2014;19:7679-88.
- Monte J, Abreu AC, Borges A, Simões LC, Simões M. Antimicrobial activity of selected phytochemicals against Escherichia coliand Staphylococcus aureus and their biofilms. Pathogens 2014;3:473-98.
- Zhang Y, Chen F, Sun E, Ma R, Qu C, Ma L. In vitroantibacterial activity of combinations of fosfomycin, minocycline and polymyxin B on pan-drug-resistant Acinetobacter baumannii.Exp Ther Med 2013;5:1737-39.
- Sopirala MM, Mangino JE, Gebreyes WA, Biller B, Bannerman T, Balada-Llasat JM, et al. Synergy testing by E-test, microdilution checkerboard, and time-kill methods for pan-drug-resistant Acinetobacter baumannii. Antimicrob Agents Chemother 2010;54:4678-83.
- Hosgor-Limoncu M, Ermertcan S, Tasli H, Aydemir S.The post-antibiotic effects of linezolid against Gram-positive pathogens. Saudi Med J 2007;28:551-54.
- Amman V, Basri DF, Huyop F. Determination of the post-antibiotic effect (PAE) of combinations of extracts from galls of Quercus infectoriawith vancomycin against methicillin-resistant Staphylococcus aureus (MRSA). Afr J Biotechnol 2011;10:18274-78.
- Gudmundsson S, Erlendsdóttir H, Gottfredsson M, Gudmundsson A. The post antibiotic effect induced by antimicrobial combinations. Scand J Infect Dis Suppl 1990;74:80-93.