- Corresponding Author:
- M. Amir
DDepartment of Pharmaceutical Chemistry, Faculty of Pharmacy, Hamdard University, Hamdard Nagar, New Delhi - 110 062, India
E-mail: mamir_s2003@yahoo.co.in
| Date of Submission | 23 March 2006 |
| Date of Revision | 5 February 2007 |
| Date of Acceptance | 3 May 2007 |
| Indian J. Pharm. Sci., 2007, 69 (3): 337-343 |
Abstract
Pyrimidine nucleus exhibited remarkable pharmacological activities. Literature indicates that compounds having pyrimidine nucleus have wide range of therapeutic uses that include antiinflammatory, antibacterial, anticancer, antiviral, antiHIV, antimalarial, antihypertensive, sedatives and hypnotics, anticonvulsant and antihistaminic. The present review provides a broad view of the antiinflammatory activity possessed by compounds having a pyrimidine nucleus.
In medicinal chemistry pyrimidine derivatives have been very well known for their therapeutic applications. The presence of a pyrimidine base in thymine, cytosine and uracil, which are the essential binding blocks of nucleic acids, DNA and RNA is one possible reason for their activity. The literature indicated that compounds having pyrimidine nucleus possess broad range of biological activities, like 5-fluorouracil as anticancer; idoxuridine and trifluoridine as antiviral; zidovudine and stavudine as antiHIV; trimethoprim, sulphamethiazine and sulphadiazine as antibacterial; sulphadoxin as antimalarial and antibacterial; minoxidil and prazosin as antihypertensive; barbiturates eg. phenobarbitone as sedative, hypnotics and anticonvulsant; propylthiouracil as antithyroid; thionzylamine as H1-antihistamine; and toxoflavin and fervennuline as antibiotics.
As a result of remarkable pharmacological efficiency of pyrimidine derivatives, intensive research has been focused on antiinflammatory activity of pyrimidine nucleus. Recently two PCT international applications have been filed for 2-thiopyrimidine derivatives possessing potent activity against inflammation and immune disorders [1,2]. The present review highlights the antiinflammatory activity of pyrimidine and its derivatives.
Pirisino et al. [3] have studied 2-phenylpyrazolo-4- ethyl-4,7-dihydro[1,5-a]pyrimidine-7-one (1, FPP028) for its analgesic, antipyretic and antiinflammatory activities. The antiinflammatory properties of FPP028 were evaluated by carrageenan-induced paw oedema and cotton pellet-induced granuloma methods and found to possess the activity similar to indomethacin, phenylbutazone and isoxicam. Similarly FPP028 was shown to possess analgesic and antipyretic activities comparable to the former drugs. At the same time ulcerogenic activity of FPP028 was studied in restraint-stressed rats, demonstrated a gastro protective effect since the number of gastric lesions induced either by stress or phenylbutazone treatment was decreased.
Modica et al. [4] synthesized some new thiadiazolothienopyrimidinones and tested them for antiinflammatory and analgesic activities and found encouraging results. Cenicola et al. [5] evaluated some imidazolo[1,2-c]pyrimidines (2) for antiinflammatory, analgesic and antipyretic activities. Antiinflammatory activity was studied by carrageenan-induced paw oedema in rats and found to show activity comparable to indomethacin. The compounds showed lower analgesic and antipyretic activities. The compounds were also studied for ulcerogenic property and the inhibitory activity on the MDA production.
Nargund et al. [6] reported the synthesis of few substituted 2-mercapto-3-(N-alkyl) pyrimido[5,4- c]cinnolin-4-(3H)-ones (3) and screened them for antiinflammatory and antimicrobial activities. The antiinflammatory activity was done by carrageenan induced paw oedema method and it was found that the most active compound showed 47.6% decreased in oedema in comparison to reduction in oedema of 48.8% by phenylbutazone.The compounds showed moderate to good antimicrobial activity against various gram positive and gram negative bactreria.
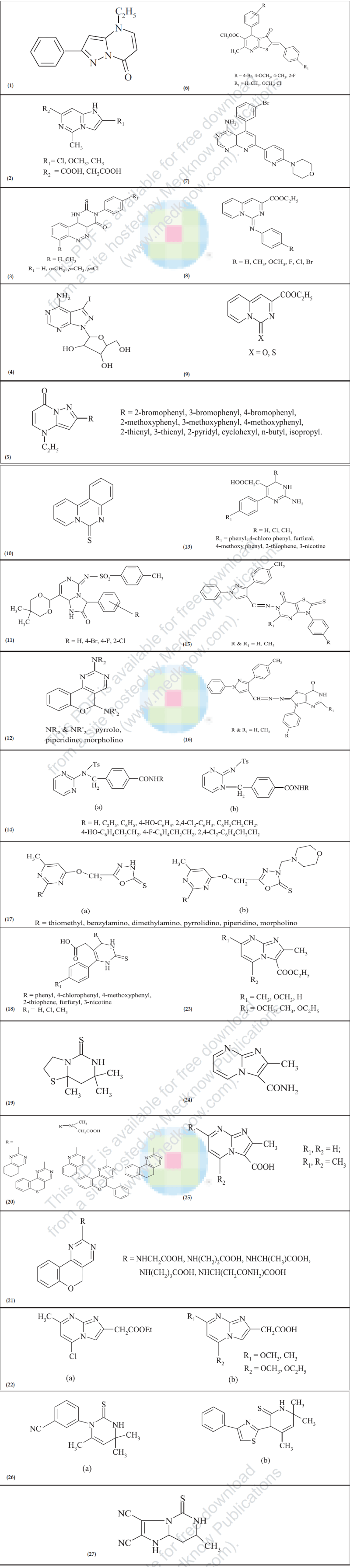
Several pyrazolo[3,4-d]pyrimidine derivatives were synthesized as potential inhibitor of adenosine kinase by Cottam et al [7 ] .One of the componds (4) was found to display good antiinflammatory activity at a dose of 30 mg/kg, p.o. when evaluated in vivo in rat pleurisy inflammation model.
Another series of pyrazolo[1,5-a]pyrimidin-7-one (5) were synthesized as a structural modification of FPP028 (1) at 2-position by Bruni et al8. All the synthesized compounds were evaluated for antiinflammatory activity by carrageenan-induced paw oedema method. A significant increase in antiinflammatory activity was observed when phenyl group at 2-position of FPP028 was replaced by 2- thienyl and 2-pyridyl group. Whereas compounds with alkyl and saturated ring systems showed reduction in activity, substituents at 4-position of the phenyl ring completely abolished antiinflammatory activity.
Tozkoparan et al [9] synthesized sixteen new 2- benzylidene-7-methyl-3-oxo-5-(substituted phenyl)-2,3- dihydro-5H-thiazolo[3,2-a]pyrimidine-6-carboxylic acid esters(6) by reaction of 1,2,3,4-tetrahydropyrimidine-2- thiones with chloroacetic acid and aromatic aldehydes in a single step. All the synthesized compounds were screened for antiinflammatory activity (100 mg/kg p.o. dose) by carrageenan-induced paw oedema method and compared with indomethacin. None of the compounds showed noticeable antiinflammatory activity except few with moderate activity.
Adenosine (ADO) is an endogenous homeostatic inhibitory neuromodulator that reduces cellular excitability at sites of tissue injury and inflammation. Inhibition of adenosine kinase (AK), primary metabolic enzyme for ADO, selectively increases ADO concentration at sites of tissue trauma and enhances the analgesic and antiinflammatory actions of ADO. Lee et al [10 ]synthesized and studied some 6-substituted pyridopyrimidine analogues as potential AK inhibitors, led to the identification of 4-amino-5-(3-bromophenyl)- 7-(6-morpholinopyridin-3-yl) pyrido[2,3-d] pyrimidine (7,ABT-702), a novel and potent non-nucleoside AK inhibitor with oral activity in animal models of pain and inflammation. The ABT-702 was further studied in detail by Boyle et al. [11] to evaluate its potential utility in chronic inflammation. The compound was tested in rat adjuvant arthritis model and a significant inhibition of arthritis as determined by paw volume was observed. In addition, histologic and radiographic evidence of bone and cartilage destruction was significantly decreased.
Molina et al. [12] synthesized a number of pyrido[1,2- c]pyrimidines (8-10) on solid phase using the iminophosphorane methodology and tested for effects on leukocyte function in vitro and antiinflammatory activity. The compounds inhibited the generation of PGE2 by COX-2 in RAW 264.7 macrophages stimulated with lipopolysaccharide. In addition few compounds also showed antiinflammatory activity in the carrageenan-induced paw oedema in rats at a dose of 20 mg/kg p.o. where they inhibited PGE2 levels in inflamed paw without affecting the content of eicosanoids in stomach.
Vidal et al. [13 ]have studied the effects of some hexahydroimidazo[1,2-c]pyrimidine derivatives (11) on leukocyte functions in vitro and screened for antiinflammatory activity in two models of inflammation. The compound with R=F showed significant inhibition of paw swelling with reduced PGE2 levels in paw homogenates.
Bruno et al. [14] reported the synthesis of some new 2,5- cycloamino-5H(1) benzopyrano[4,3-d]pyrimidines (12) and screened them for antiinflammatory, analgesic and antipyretic activities and in vitro antiplatelet activity on guinea pig platelet rich plasma. All the compounds failed to exhibit antiinflammatory, analgesic and antipyretic activities at 100 mg/kg p.o. dose but they showed an interesting antiplatelet activity.
Bahekar et al. [15] reported the synthesis of some [2- amino-6-(4-substituted aryl)-4-(4-substituted phenyl)- 1, 6-dihydropyrimidin-5-yl] acetic acid derivatives (13) and evaluated for antiinflammatory activity by carrageenan-induced paw oedema method using diclofenac sodium as standard drug. All the compounds showed reduction in paw oedema but only few of them showed remarkable antiinflammatory activity.
Neutrophills play a crucial role in initiating and maintaining inflammatory processes in the joints. Besides, it has been reported that 5-lipoxygenase inhibition may be beneficial in some inflammatory diseases. In view of this fact Ferri et al. [16] synthesized some 2-tosylamino (14a) and 2-tosyliminopyrimidine (14b) derivatives and studied their interference with some leukocyte functions and 5-LO activity. The study demonstrated that all the compounds inhibited cell free 5-LO activity and reduced activation of neutrophills, which may have relevance for the modulation of the inflammatory response.
Bekhit et al. [17] reported the synthesis of two series of 1H-pyrazolyl derivatives of thiazolo[4, 5-d]pyrimidines (15,16) and screened for in vivo antiinflammatory activity in two different bioassays namely, cotton pallet-induced granuloma and carrageenan-induced paw oedema in rats. The most active compounds were examined for in vitro inhibitory activity on human COX-1 and COX-2 enzymes. In addition, ulcerogenic effect and acute toxicity (LD50) values of these compounds were also determined. Some of the tested compounds showed remarkable antiinflammatory activity with lesser ulcerogenicity.
Jakubkiene et al. [18 ]reported the synthesis of some 5-(6-methyl-2-substituted-4-pyrimidinyloxymethyl)- 2,3-dihydro-1,3,4-oxadiazole-2-thiones (17a) and their 3-morpholinomethyl derivatives (17b) and evaluated them for antiinflammatory activity by carrageenan and bentonite-induced paw oedema in rats. Most of the tested compounds were found to be active and some of them were more active than acetylsalicylic acid.
Bahekar et.al. [19] synthesized a series of [4,6-(substituted aryl-2-thioxo-1,2,3,4-tetrahydro-pyrimidin-5-yl]acetic acid (18) by the base catalyzed condensation of β-aroylpropionic acid and thiourea with aldehyde in ethanol and screened them for antiinflammatory activity using diclofenac sodium as standard drug. Some of the tested compounds showed moderate activity.
Sodhi et al. [20] reported the synthesis of various 2-thiopyrimidine derivatives by an efficient one pot reaction of functionalized amines with either 4-isothiocyanato-4-methyl-2-pentanone or 3-isothiocyanaotobutanal. The compounds were screened for antiinflammatory, analgesic and protein kinase inhibitory activities. Compound 7,7,8atrimethylhexahydrothiazolo[ 3,2-c]pyrimidine-5-thione (19) showed good antiinflammatory (37.4% at 100 mg/kg p.o.) and analgesic (75% at 100 mg/kg p.o.) activities. Rest of the compounds showed moderate activity.
Bruno et al. [21] synthesized two different series Nmethyl- N-pyrimidin-2-yl glycine (20) and N-5H-[1] benzopyrano[4,3-d]pyrimidin-2-yl substituted amino acids (21) and tested for antiinflammatory activity. All the compounds showed significant antiinflammatory activity.
Sacchi et al. [22] synthesized a series of imidizo[1,2- a]pyrimidine 2-carboxylic acid and 2-acetic acid analogs (22a-b) and tested them for antiinflammatory activity. Almost all the carboxylic acid derivative showed a remarkable antiinflammatory activity.
Abignente et al. [23] synthesized of a group of imidazo[1,2-a]pyrimidine-2-carboxylic esters, acids and amides. All the synthesized compounds were tested in vivo for antiinflammatory and analgesic activities as well as for ulcerogenic action. Some of them showed antiinflammatory action in the rat paw oedema, while almost all compounds displayed significant analgesic activity in the acetic acid writhing test. All new compounds were found to be lacking in inhibitory activity on cyclooxygenase in vitro.
Laneli et al. [24 ]reported the synthesis and antiinflammatory activity of some new 2- methylimidazo[1,2-a]pyrimidine-3-carboxylic esters, acids and amides. Antiinflammatory activity was done by carrageenan-induced rat paw oedema method. Structure shown in figure 23-25 displayed maximum antiinflammatory activity.
Sondhi et al. [25] synthesized some mono, bi and tricyclic pyrimidine derivatives and evaluated them for antiinflammatory and analgesic activity using carrageenan-induced paw oedema assay, and compounds presented in figure 26a-b and 27 exhibited good antiinflammatory activity that is, 27.9, 34.5 and 34.3% at 50 mg/kg p.o. respectively. Analgesic activity evaluation was carried out using phenylquinone writhing assay.
Conclusion
Although pyrimidine and its derivatives are reported to have diverse pharmacological activities but their role, as antiinflammatory agent has been well defined. There is still scope for more research work to be done in this field to find a novel antiinflammatory agent. This study would be useful for the researchers working in this field.
References
- Belema,M.,Bunker,A.,Nguyen,V.,Beaulieu,F.,Ouellet,C.,Marinier,A.,Roy,S.,Yung,X.,Qiu,Y.,Zhang,Y.,Martel,and Zuci,C.,PCT Int. Appl. WO 2003084.,ThroughChem. Abstr.,2003,139,337987x.
- Bonnert,R.V.,Cage,P.A.,Hunt,S.F.,Walters,I.J.S. and Austin,R.P.,PCT Int. Appl. WO 2003024966.,throughChem. Abstr.,2003,138,271701v.
- Pirisino,R.,Bainchini,F.,Banchelli,G.,Ignesti,G. and Ramondi,L.,Pharmacol. Res. Comm.,1996,18,241.
- Modica,M.,Santagati,M.,Santagati,A.,Cutuli,V.,Mangano,N. and Caruso,A.,Pharmazie,2000,55,500.
- Cenicola,M.L.,Donnoli,D.,Stella,L.,Paola,C.D.,Constantino,M.,Anignente,E.,Arena,F.,Luraschi,E. and Saturnino,C.,Pharmacol. Res.,1990,22,80.
- Nargund,L.V.G.,Badiger,V.V. and Yarnal,S.M.,J. Pharm. Sci.,1992,81,365.
- Cottam,H.B.,Wasson,D.B.,Shih,H.C.,Raychaudhary,A.,Pasquale,G.D. and Carson,D.A.,J. Med. Chem.,1993,36,3424.
- Bruni,F.,Costanzo,A.,Selleri,S.,Guerrini,G.,Fantozzi,R.,Pirisino,R. and Brunelleschi,S.,J. Pharm. Sci.,1993,82,480.
- Tozkoparan,B.,Ertan,M.,Kelicen,P. and Demirdamar,R.,Il Farmaco,1999,54,588.
- Lee,C.H.,Jiang,M.,Cowart,M.,Gfesser,G.,Perner,R.,Kim,K.H.,Gu,Y.G.,Williams,M.,Jarvis,M.F.,Kowaluk,E.A.,Stewart,A.O. and Bhagwat,S.S.,J. Med. Chem.,2001,44,2133.
- Boyle,D.L.,Kowluk,E.A.,Jarvis,M.F.,Lee,C.H.,Bhagwat,S.S.,Williams,M. and Firestein,G.S.,J. Pharmacol. Exp. Ther.,2001,296,495.
- Molina,P.,Aller,E.,Lorengo,A.,Cremadis,P.L.,Rioja,I.,Ubeda,A.,Terencio,M.C. and Alcaraz,M.J.,J. Med. Chem.,2001,44,1011.
- Vidal,A.,Ferrandiz,M.L.,Ubeda,A.,Alarcon,A.A.,Arques,J.S. and Alcaraz,M.J.,J. Pharm. Pharmacol.,2001,53,1379.
- Bruno,O.,Brullo,C.,Ranise,A.,Schenone,S.,Bondavalls,S.,Barocelli,E.,Ballabeni,V.,Chiavarini,M.,Tognolini,M. and Impicciatore,M.,Bioorg. Med. Chem. Lett.,2001,11,1397.
- Bahekar,S.S. and Shinde,D.B.,ActaPharm.,2003,53,223.
- Ferri,P.F.,Ubeda,A.,Guillen,I.,Lasri J.,Rosende,M.E.G.,Akssir,M,and Arques,J.S.,Eur. J. Med. Chem.,2003,38,289.
- Bekhit,A.A,Fahmy,H.T.Y.,Rostom,S.A.F.,and Baraka,A.M.,Eur.J. Med. Chem.,2003,38,27.
- Jakubkiene,V.,Burbuliene,M.M.,Mekuskiene,G.,Udrenaite,E.,Gaidelis,P. and Vainilavicius,P.,Il Farmaco,2003,58,323.
- Bahekar,S.S. and Shinde,D.B.,Bioorg. Med. Chem. Lett.,2004,14,1733.
- Sondhi,S.M.,Goyal,R.N.,Lahoti,A.M.,Singh,N.,Shukla,R. and Raghubir,R.,Bioorg. Med. Chem.,2005,13,3185.
- Bruno,O.,Schenone,S.,Ranise,A.,Bondavalli,F.,Filippelli,W.,Falcone,G.,Motola,G. and Mazzeo,F.,Il Farmaco,1999,54,95.
- Sacchi,A.,Laneri,S.,Arena,F.,Luraschi,E.,Abignente,E.,D'Amico,M.,Berrino,L. and Rossi,F.,Eur. J. Med. Chem.,1997,32,677.
- Abignente,E.,Sacchi,A.,Laneri,S.,Rossi,F.,D'Amico,M.,Berrino,L.,Calderaro,V. and Parrillo,C.,Eur. J. Med. Chem.,1994,29,279.
- Laneri,S.,Sacchi,A.,Gallitelli,M.,Arena,F.,Luraschi,E.,Abignente,E.,Filippelli,L. and Rossi,F.,Eur. J. Med. Chem.,1998,33,163.
- Sondhi,S.M.,Singh,N.,Johar,M. and Kumar,A.,Bioorg. Med. Chem.,2005,13,6158.