- Corresponding Author:
- R. M. Singh
Department of Pharmaceutics, Jamia Hamdard University, New Delhi-110 062
E-mail: raman19662002@gmail.com
| Date of Submission | 04 September 2014 |
| Date of Revision | 26 January 2015 |
| Date of Acceptance | 20 November 2015 |
| Indian J Pharm Sci 2015;77(6):723-728 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
Abstract
A simple, rapid, reliable, robust and optimized reversed phase high performance liquid chromatographic method for simultaneous estimation of doxycycline hyclate and curcumin was successfully developed and validated as per International Conference on Harmonization guidelines. The objective was achieved in terms of well separated peaks within 10 min on a Waters Sunfire C8 column with dimensions of 250×4.6 mm, particle size 5.0 μm using mobile phase consisting of 30 volumes of potassium dihydrogen phosphate buffer (50 mM) adjusted to pH 6.5±0.1 with triethylamine and 70 volumes of methanol at flow rate of 0.85 ml/min. The column effluents were monitored at 400 nm maintained at ambient column temperature (28o). The developed method was found linear over the concentration range of 200-700 μg/ml for doxycycline hyclate and 8-28 μg/ml for curcumin, the detection and quantitation limit was found to be 26.063 and 78.97 μg/ml for doxycycline hyclate; 0.795 and 2.13 μg/ml for curcumin, respectively. The developed method was optimized using Minitab software version 16 to meet the current quality by design requirements. The method validation was done for linearity, range, detection and quantitation limit, accuracy, precision, specificity, system suitability testing, and robustness.
Keywords
ICH, quality by design, relative standard deviation, periodontitis, doxycycline hyclate, curcumin.
Doxycycline hyclate is a broad spectrum antibiotic used in the treatment of infections associated with humans and animals[1,2]. Recently, doxycycline hyclate has been employed for the treatment of periodontal infections and associated risk conditions such as respiratory problems, diabetes mellitus, cardiovascular risks, and foetopahtic changes in gestation period, alveolar bone loss[3-7].
Curcumin a plant derived dietary spice is known to possess antiinflammatory activity[8]. A fixed dose combination of these drugs can be used for the treatment of periodontal infections due to the antibacterial and antiinflammatory activity of doxycycline hyclate and curcumin.
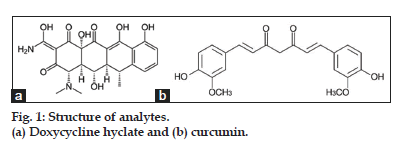
Chemically, doxycycline hyclate (fig. 1a) is C22H24N2O8.Hcl (C2H5OH)1/2 (H2O)1/2 with molecular mass 512.94 g/mol, is the hydrochloride hemiethanol hemihydrate of doxycycline[9]. The drug is chemically S-4-(dimethylamine)-1,4,4a,5,5a,6,11,12a-octahydro- 3,5,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-2- napthacenecarboxamide hydrochloride monohydrate combined with ethyl alcohol and is a yellow coloured hygroscopic powder in physical appearance and must be stored in an airtight container, protected from light[10].
Curcumin (fig. 1b) a pharmacologically active turmeric extract chemically known as 1,7-Bis(4- hydroxy-3-methoxyphenyl)hepta-1,6-diene-3,5-dione with molecular formula and molecular weight to be C21H20O6 and 369.38, respectively, is known for its antioxidant, antiinflammatory, antimicrobial, antiparasite and anticancer activity[11].
A liquid chromatographic method for estimation of drugs individually is a choice of various pharmacopoeias[10,12,13]. Since, no methods for their simultaneous estimation is available in the literature. Therefore we propose a simple, economic, effective, robust and validated method for the estimation of both the drugs in combination, which can also be used for the routine analysis of the two drugs.
The dose of doxycycline hyclate was chosen as per the USFDA’s prescribing information on Atridox. Dose of curcumin was selected based on Curnext oral gel intended for gum treatment from Abbott Healthcare Pvt. Ltd., which is available at a dose of 0.002% w/w (10 mg/50 g). Therefore, doxycycline hyclate at a dose of 50 mg and curcumin at a dose of 2 mg can be formulated as microparticles.
Understanding the future requirements International Conference on Harmonization (ICH) proposed guidelines elaborating quality by design (QbD), quality by review, and risk assessment, viz ICH Q8, Q9 and Q10. Quality by design has become a new concept for developing and analyzing of quality pharmaceutical products. It is an essential part of modern approach to develop and analyze pharmaceutical product with quality attributes, QbD proves to be major solution to build quality in products but a major challenge to implement at an industrial scale[14]. Concept of quality by design lies in generating a design space, control strategy[15]. The design space helps in finding the design points, which are capable of producing results as per requirements and set conditions with possibilities of reproducibility, accuracy, and repeatability. Several mathematical models are used to analyze mathematical data to generate a design space.
Materials and Methods
Doxycycline hyclate working standard and bulk drug was obtained from M/s Gracure Pharmaceuticals Pvt. Ltd., India and G. D. Laboratories Pvt. ltd., India, respectively. Curcumin was obtained as a gift sample from Chromadex, Germany. Tablets of doxycycline hyclate 100 mg and Curenext gel 10 mg (curcuma longa extract) were purchased from local pharmacy store. HPLC grade methanol (Merck, Mumbai, India), AR grade potassium dihydrogen orthophosphate (Himedia, Mumbai, India), GR grade triethlyamine (Merck, Mumbai, India) and deionized Mili Q water (Milipore, Bedford, MA) was used to prepare mobile phase and diluent.
HPLC analysis was performed on a Thermo Scientific Dionex Ultimate 3000 UHPLC+ focused system equipped with Ultimate 3000 RS binary gradient pump, Ultimate 3000 RS autosampler, Ultimate 3000 RS column compartment and Ultimate 3000 RS Diode array detector and Chromeleon software.
Selection of wavelength
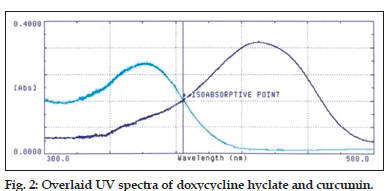
UV/Vis scan of both the drugs was done and a wavelength max (λmax) of 360.0 nm and 425.0 nm for doxycycline hyclate and curcumin, respectively was observed. The overlaid scan for both the drugs showed the isobestic point to be 384.5 nm (fig. 2). With these observations 400.0 nm was selected for the estimation of the two drugs.
Preparation of standard and samples solutions
For the preparation of standard solution, 10 mg of the standard doxycycline hyclate and 10 mg of curcumin was weighed and transferred into two separate clean dried 10 ml volumetric flasks, dissolved in suitable amount of diluent (phosphate buffer pH 6.5 and methanol in a ratio of 20:80 v/v) and mixed with the aid of ultrasound for 5 min. The volume was made upto 10.0 ml to obtain a stock solution of concentrations 1000 μg/ml each. An aliquot equivalent to 5 ml doxycycline hyclate and 0.2 ml curcumin was pipetted out in a clean dried 10 ml volumetric flask, diluted up to the mark to get a solution with concentrations 500 and 20 μg/ml for doxycycline hyclate and curcumin, respectively.
For the preparation of sample solution, twenty tablets of doxycycline hyclate were weighed, crushed and a quantity of tablet powder equivalent to 50 mg of doxycycline hyclate was transferred into a 50 ml clean dried volumetric flask and dissolved in suitable amount of diluent with the aid of ultrasound for 5 min and make up the volume. Curenext Gel equivalent to 5 mg curcumin was dispersed in a clean dried 10 ml volumetric flask, and mixed with the aid of ultrasonication for 5 min, make up the volume up to 10 ml with diluent and the solution was centrifuged at 1000 rpm for 5 min. Two milliliter of this solution was diluted to 10 ml with diluent. Further, dilute 2 ml of this solution and 5 mL of doxycycline hyclate solution to 10 ml with diluent. The solution was filtered through 0.2 μm filter and analyzed.
Preparation of forced degradation samples
The samples were subjected to different stress conditions viz acid, base, peroxide, thermal, and photolytic stress conditions separately and the solutions were analyzed using HPLC. Table 1 summarize the procedure to prepare the sample solutions for forced degradation studies.
Method optimization
The identified method was optimized understanding the need of approaching quality by design as per the ICH Q8 guideline. The concept of QbD was mentioned in the ICH Q8 guideline, which states that “quality cannot be tested into products, i.e., quality should be built in by design”[14]. In practice, the ideal QbD-based pharmaceutical development effort will involve a systematic method relating mechanistic understanding of input material attributes and process parameters to drug product critical quality attributes. Such a development effort is accomplished through the use of multivariate experiments involving modern process controls enabling process understanding[16]. These parameters are analyzed and a design space is generated. Understanding the design space for a pharmaceutical process generally involves the identification of critical attributes for the input materials, the process, and the final product[15].
Method validation
The developed RP-HPLC method was validated to confirm that it was suitable for its intended purpose as described in ICH Q2 (R1) guidelines[17]. ICH Q6A guidelines explicitly require forced decomposition studies to be conducted under a variety of conditions and separation of the pure drug from its degradation products for stability-indicating assay methods[18]. UVdegradation was carried out according to ICH (Q1B) guidelines[19]. The described method was extensively validated in terms of specificity, linearity, accuracy, precision, detection limit, quantitation limit, robustness and system suitability.
Results and Discussion
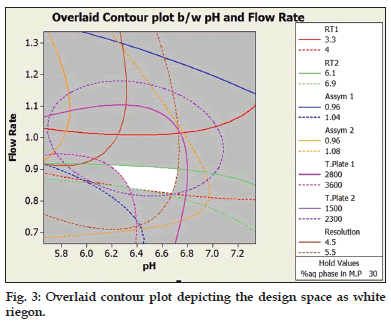
The developed method was optimized using statistical software Minitab. Three factors viz flow rate, percentage of buffer in the mobile phase and pH of the buffer were found to have an effect on the peak area, retention time, asymmetry and resolution between both the drugs. As depicted in fig. 3 the white region, shows the design space from which the different available combinations for the chromatographic separations gives results in the desired ranges with respect to retention time, asymmetry, and theoretical plates for both the drugs and resolution between both the drugs.
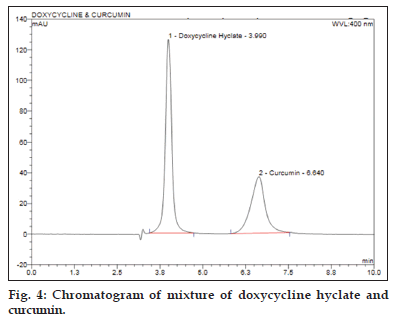
The optimized chromatographic method consisted of isocratic separation achieved on waters sunfire C8 column with dimensions 250×4.6 mm and particle size 5.0 μm using mobile phase consisting of 30 volumes of potassium dihydrogen phosphate buffer (50 mM) adjusted to pH 6.5±0.1 with triethylamine and 70 volumes of methanol at flow rate of 0.85 ml/min with injection volume of 10 μL. The column effluents were monitored at 400 nm maintained at ambient column temperature (28o). The chromatogram recorded by running the chromatographic separation at the optimized condition is depicted in fig. 4.
| Stress condition | Quantity of drug (mg) | Volume of stress agent | Analyze after 24 h | |||
|---|---|---|---|---|---|---|
| Doxycycline | Curcumin | Volume of | Volume of | |||
| hyclate | neutralization agent | diluent (ml) | ||||
| Acid-induced degradation | 10 | 10 | 50 ml (0.1 N HCl) | 50 ml (0.1 NaOH) | 100 | |
| Base-induced degradation | 10 | 10 | 50 ml (0.1 N NaOH) | 50 ml (0.1 N HCl) | 100 | |
| Photolytic-induced degradation | 10 | 10 | 5 ml (3% w/v H2O2) | 5 ml (3% sodium | 195 | |
| Thermal-induced degradation | 5 | 5 | Kept in oven at 80° | - | 200 | |
| UV-induced degradation | 5 | 5 | Exposed to UV-radiation at 320–400 nm metabisulphite) | - | 200 | |
Table 1: Method Of Preparing Forced Degradation Samples.
The linearity of the method was determined at six concentration levels 200-700 μg/ml for doxycycline hyclate and 8-28 μg/ml for curcumin, respectively. The linearity plots were constructed for concentration of doxycycline hyclate and curcumin versus area as shown in Table 2. The developed analytical method was found linear over a concentration range of 200-700 μg/ml and 8-28 μg/ml for doxycycline hyclate and curcumin, respectively.
LOD refers to the minimum amount of drug, which can be detected using the analytical method, which has been developed and LOQ refers to the minimum amount of drug, which is quantified by the analytical method. These can be calculated using simple formula as shown in equation (1) and (2). LOD=3.3*(σ/S)… Eqn 1 and LOQ=10*(σ/S)… Eqn 2.
Where, ‘σ’ represents residual standard deviation from linearity regression data and ‘S’ represents slope from linearity plot. The LOD and LOQ values were found to be 26.063 and 78.979 μg/ml for doxycycline hyclate and 0.705 and 2.137 μg/ml for curcumin, respectively as described in Table 2.
The accuracy of the method was evaluated in triplicate by analysis of concentration levels i.e. 50, 100 and 150% of the target concentration 250 and 10 μg/ml for doxycycline hyclate and curcumin, respectively. The percentage recoveries were calculated and found to be 94.6-96.7% and 97.8-103.14% for doxycycline hyclate and curcumin, respectively. Table 3 summarize the recovery values for the analysis.
Precision was estimated with respect to both repeatability (intra-assay) and intermediate (interday) precision. The test was performed for three concentration levels 300, 500 and 700 μg/ml for doxycycline hyclate and 12, 20 and 28 μg/ml for curcumin. Intraday precision was assessed by analysis of the sample solution in triplicates and inter-day precision was assessed by analysis of the same sample solution in triplicates for three successive days. Table 3 summarize the calculated content.
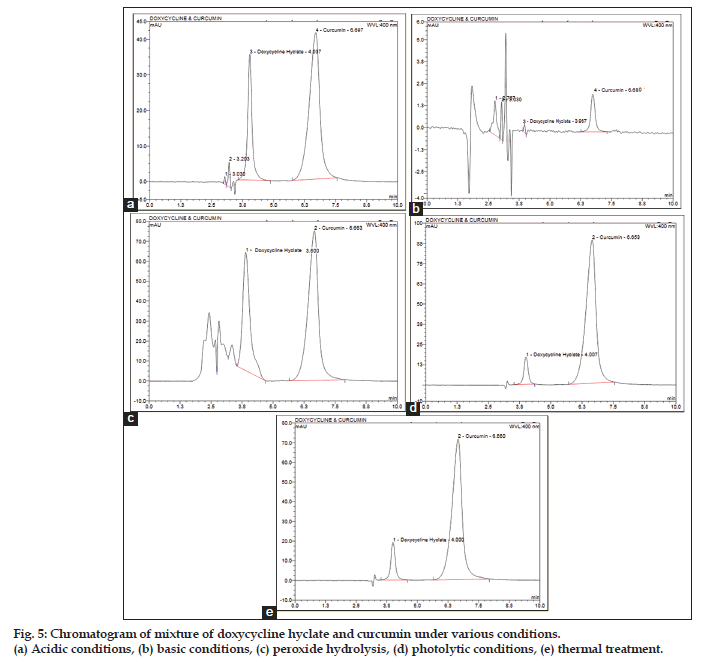
Specificity of the method was also evaluated by injecting the stressed solutions of doxycycline hyclate and curcumin. The proposed method efficiently resolved both the drugs from the degradants, doxycycline hyclate (Rt=4.001) and curcumin (Rt=6.653). Thermal and photolytic stressed solution showed no change in the chromatograms whereas acid, base and oxidation induced samples show degradant peaks not interfering the principal peaks (fig. 5).
The system suitability tests represented an integral part of the method validation and were used to ensure retention time, peak area, peak asymmetry, number of theoretical plates and resolution using replicate injection of a standard solution. A standard solution with concentrations 500 and 20 μg/ml for doxycycline hyclate and curcumin, respectively. The responses were analyzed and the results are summarized in the Table 3.
| Parameter (units) | Doxycycline hyclate | Curcumin |
|---|---|---|
| Linearity (µg/ml) | 200–700 | 8–28 |
| r±SD | 0.999±0.0001 | 0.999±0.0001 |
| Slope±SD | 0.061±0.0004 | 1.005±0.003 |
| Intercept±SD | 2.62±0.113 | 1.78±0.003 |
| LOD (µg/ml) | 26.06 | 0.702 |
| LOQ (µg/ml) | 78.97 | 2.31 |
Table 2: Regression Data For Caliberation Curves.
It determines the reproducibility of the test result with small and deliberate variations in the method parameters. The experiment was carried out by producing slight variations in the percentage of buffer in mobile phase (±1.5%), Flow Rate (±2.5%), and pH of the buffer (±3%). The effectiveness of the deliberate variations were checked on retention time of peak. Table 3 summarize the retention time and relative standard deviations for the test. The proposed method was successfully applied for the estimation of prepared test solutions. The assay results are compiled in the Table 3.
| Recovery study | Doxycycline hyclate | Curcumin | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Percentage±SD | %RSD | Percentage±SD | %RSD | |||||||||||
| Levels | ||||||||||||||
| 50% level | 96.78±0.48 | 0.49 | 97.86±0.63 | 0.64 | ||||||||||
| 100% level | 95.51±0.51 | 0.53 | 103.14±0.34 | 0.33 | ||||||||||
| 150% level | 95.00±0.57 | 0.57 | 101.62±1.03 | 1.01 | ||||||||||
| Robustness test | Doxycycline hyclate | Curcumin | ||||||||||||
| RT±SD | %RSD | RT±SD | RSD | |||||||||||
| Parameter | ||||||||||||||
| Flow rate | 4.034±0.064 | 1.59 | 6.73±0.112 | 1.66 | ||||||||||
| (±3% variation) | ||||||||||||||
| % buffer in mobile phase | 4.018±0.016 | 0.33 | 6.74±0.110 | 1.61 | ||||||||||
| (±1.5% variation) | ||||||||||||||
| pH of the buffer | 4.02±0.026 | 0.56 | 6.75±0.066 | 0.98 | ||||||||||
| (±3% variation) | ||||||||||||||
| System suitability | Doxycycline hyclate | Curcumin | ||||||||||||
| criteria (n=6) | ||||||||||||||
| RT±SD | %RSD | RT±SD | %RSD | |||||||||||
| Parameters | ||||||||||||||
| RT | 4.013±0.002 | 0.06 | 6.715±0.005 | 0.08 | ||||||||||
| Resolution between | 4.52 | |||||||||||||
| the peaks | ||||||||||||||
| Theoretical plates | 2953 | 1433 | ||||||||||||
| (average) | ||||||||||||||
| Tailing factor | 0.96 | 0.91 | ||||||||||||
| (asymmetry) (average) | ||||||||||||||
| Precision (n=3) | Intraday | Interday | ||||||||||||
| precisiona | precisionb | |||||||||||||
| Concentration | Area±SD | %RSD | Area±SD | %RSD | ||||||||||
| Doxycycline | 300 | 15.36±0.11 | 0.69 | 15.27±0.07 | 0.51 | |||||||||
| hyclate | 500 | 27.79±0.18 | 0.18 | 27.7±0.35 | 1.27 | |||||||||
| 700 | 39.92±0.09 | 0.09 | 39.39±0.62 | 1.59 | ||||||||||
| Curcumin | 12 | 10.50±0.16 | 1.12 | 10.47±0.14 | 1.39 | |||||||||
| 20 | 18.44±0.08 | 0.46 | 18.34±0.20 | 1.12 | ||||||||||
| 28 | 26.78±0.46 | 1.01 | 26.72±0.33 | 1.24 | ||||||||||
| Doxycycline hyclate | Curcumin | |||||||||||||
| Average (%)±SD | %RSD | Average (%)±SD | %RSD | |||||||||||
| 91.98±0.43 | 0.46 | 108.6±0.001 | 0.01 | |||||||||||
Table 3: Validation Summary And Assay.
Acknowledgements
We are grateful to M/s Gracure laboratories ltd. (New Delhi, India) and M/s Chromadex (Germany) for supplying standard gift samples of doxycycline hyclate and curcumin, respectively. Special thanks to Indian Pharmacopoeia Commission, Raj Nagar (Ghaziabad), India for providing instrumental facilities.
References
- Yasin A, Jefferies TM. Analysis of tetracycline antibiotics and their common impurities by high-performance liquid chromatography using a polymeric column. J Pharm Biomed Anal 1988;6:867-73.
- Fiori J, Grassigli G, Filippi P, Gotti R, Cavrini V. HPLC-DAD and LC-ESI-MS analysis of doxycycline and related impurities in doxipan mix, a medicated premix for incorporation in medicated feedstuff. J Pharm Biomed Anal 2005;37:979-85.
- Scannapieco FA. Role of oral bacteria in respiratory infection. J Periodontol 1999;70:793-802.
- Grossi SG, Genco RJ. Periodontal disease and diabetes mellitus: A two-way relationship. Ann Periodontol 1998;3:51-61.
- Buhlin K, Gustafsson A, Pockley AG, Frostegård J, Klinge B. Risk factors for cardiovascular disease in patients with periodontitis. Eur Heart J 2003;24:2099-107.
- López NJ, Smith PC, Gutierrez J. Higher risk of preterm birth and low birth weight in women with periodontal disease. J Dent Res 2002;81:58-63.
- Grossi SG, Genco RJ, Machtei EE, Ho AW, Koch G, Dunford R, et al. Assessment of risk for periodontal disease. II. Risk indicators for alveolar bone loss. J Periodontol 1995;66:23-9.
- Basnet P, Skalko-Basnet N. Curcumin: An anti-inflammatory molecule from a curry spice on the path to cancer treatment. Molecules 2011;16:4567-98.
- Ramesh PJ, Basavaiah K, Tharpa K, Vinay KB, Revanasiddappa HD. Development and Validation of RP-HPLC method for the determination of doxycycline hyclate in spiked human urine and pharmaceuticals. J Per ClinClin Res 2010;4:101-7.
- British Pharmacopoeia. Vol. 1. London: HMSO Publication; 2012. p. 771-2.
- Jayaprakasha GK, Jagan Mohan Rao L, Sakariah KK. Improved HPLC method for the determination of curcumin, demethoxycurcumin, and bisdemethoxycurcumin. J Agric Food Chem 2002;50:3668-72.
- United States Pharmacopoeia/National Formulary. 35th ed., Vol. II, Rockville, MD: Pharmacopoeial Convention; 2012. p. 2987-9.
- Indian Pharmacopoeia. Vol. I. New Delhi: Controller of Publication, Govt. of India, Ministry of Health and Family Welfare; 2010. p. 1257-9.
- Patil AS, PetheAM. Quality by Design (QbD): A new concept for development of quality pharmaceuticals. Int J QualAssur 2013;4:13-9.
- Vogt FG, Kord AS. Development of quality-by-design analytical methods. J Pharm Sci 2011;100:797-812.
- Drenne JK. Quality by Design – What does it really mean J Pharm Innov 2007;2:65-6.
- ICH. Q2(R1), Harmonized Tripartite Guidelines, Validation of Analytical Procedures: Test and Methodology. Geneva, Switzerland: International Conference on Harmonization; 2005.
- ICH. Q6A, Harmonized Tripartite Guidelines, Specifications: Test Procedures and Acceptance Criteria for New Drug Substance and New Drug Products: Chemical Substances. Geneva, Switzerland: International Conference on Harmonization; 1999.
- ICH. Q1B, Harmonized Tripartite Guidelines, Stability Testing: Photostability Testing of New Drug Substances and Products. Geneva, Switzerland: International Conference on Harmonization; 1996.