- *Corresponding Author:
- V. K. Rao
Chemotherapy Group and 1Chemical Carcinogenesis Group, Khanolkar Shodhika, Advanced Centre for Treatment, Research and Education in Cancer (ACTREC), Tata Memorial Centre, Kharghar, Navi Mumbai-410 208, India.
E-mail: krao@actrec.res.in
| Date of Submission | 22 June 2005 |
| Date of Revision | 24 October 2005 |
| Date of Acceptance | 20 July 2006 |
| Indian J Pharm Sci,2006, 68 (4): 465-469 |
Abstract
In the present study, we examined antiproliferative activity of quercetin in vitro and in vivo . Antiproliferative activity was demonstrated against MCF7 cell line in a dose and time dependent manner with IC50 value found to be 10 µg/ml. Further quercetin arrested MCF7 cells in G2/M phase of cell cycle in a dose and time dependent way. MCF-7 cells exposed to quercetin beta cyclodextrin complex showed reduced cell survival. Quercetin has found to act as an antiangiogenic molecule with concentrations up to 10 µg but had damaging effect on chick embryo chorioallantoic membrane at 25 µg. In animal studies quercetin inhibited tumor growth by more than 58% in mice grafted with mammary carcinoma and prolonged survival period of sarcoma 180 bearing mice by 2.3 times, respectively. We also evaluated whether quercetin enhances the therapeutic effect of mitomycin C, especially on mammary tumor growth. These studies indicated that quercetin markedly enhances the ability of mitomycin C to inhibit tumorigenicity in mammary adenocarcinoma. These effects are mediated in part by the often poorly vasalarized and hypoxic regions of tumors.
Introduction
Flavonoids comprise an important group of naturally occurring, bioactive polyphenolics, ubiquitous in plants of higher generation [1]. Recent interests on flavonoids have largely focused on two different aspects. The first concerns their various biological activities [2], the second relates to their anticarcinogenic properties [3]. The dietary bioflavonoid has been shown to inhibit carcinogen induced mammary, colonic oral and lung neoplasia in rodents and DNA adducts in human hepatoma cells [3]. The anti carcinogenic property of flavonoids is most frequently attributed to their antioxidant activity. They can act as antioxidants in several ways: chelating metals and serving as reducing agents, scavenging reactive oxygen species, breaking oxidative chain reactions, quenching the formation of singlet oxygen and protecting vitamin C from oxidation [4].
Flavonoids seems to be strong among in vitro antiproliferative agents against colorectal, ovarian, lymphoid and breast cancer cells [5-7]. Furthermore they have been shown to induce chromatin condensation and apoptosis in some cancer cells [8]. Although the exact mechanisms of flavonoid induced cytotoxicity are not very clear, the impact of disruption of signaling events was an important factor in the occurrence of apoptosis. Abnormal cell signal transduction arising from protein tyrosine kinases has been implicated in the initiation and progression of a variety of human cancers. Over the past two decades efforts were focused to develop compounds that can selectively modulate the abnormal signaling pathways [9]. In the past five years enormous progress has been made in developing tyrosine kinase inhibiting compounds [9]. Among compounds of known structure, quercetin (QC) deserves special attention as a protein tyrosine kinase inhibitor [10].
Preliminary studies carried by us earlier on the flavonoid quercetin appear promising [11-13]. To get more information and to understand growth inhibitory effect we evaluated the responsiveness of cancer cell lines to quercetin utilizing human breast cancer cell line (MCF7) and S180 ascites tumor as well as mammary adenocarcinoma grown in mice. Most cancers of breast are initially responsive to chemotherapy, but the duration of remission is often shorts and tumor becomes refractory to chemotherapy [7]. Flavonoids in view of their dual properties like antioxidant/prooxidant and estrogenic/antiestrogenic with the likelihood of over coming multidrug resistance (MDR) are worth trying against human breast carcinoma. Mostly natural products have tumor suppressive ability against ascites form of sarcomas. Hence the present investigation was undertaken on these tumor systems. We report here the anti tumor activity of QC, its efficacy when administered along with standard cytotoxic agent mitomycin C (MMC) and finally the therapeutic effects observed with its cyclodextrin inclusion complex.
Materials and Methods
Quercetin (purity 98%) was purchased from SD Fine Chemicals, Mumbai. All other chemicals were obtained from Gibco Research Laboratories and Sigma (St. Louis, MO, USA). Quercetin was dissolved in dimethyl sulphoxide to make stock solutions.
Cell culture and cytotoxicity assay
Human breast carcinoma MCF7 cells were grown in monolayer and propagated in RPMI 1640 medium supplemented with 10% heat inactivated fetal bovine serum (FBS) and antibiotics (100 μg/ml Penicillin G, Streptomycin C) in a humidified atmosphere of 5% CO2 at 37°. To maintain exponential growth, the cultures were divided every third day by dilution to a concentration of 1×105 cells/ml. To 2 ml of the complete cell culture medium QC was added in increasing concentrations (5, 10, 25, 50, 100 μg/ml). All these solutions were added to exponentially growing MCF7 cells seeded at a density of 4×105 cells/ml. Cyclodextrin solutions were subsequently added. DMSO (0.1%) was used as control. Cell numbers were measured by counting trypan blue excluding cells using hemocytometer when the cells were removed after 72 h exposure. Results were calculated as viable cells in treated cultures relative to control. IC50, the drug concentration causing cell toxicity by 50% following a 72 h exposure was used as a parameter for cytotoxicity [11].
Flow cytometric measurement of cell cycle phase distribution
The preparation of samples for measurement of the cell-cycle distribution was performed accordingly to a method reported earlier [11]. Briefly, cultures were centrifuged for 5 min at 800 g and the cells (1×106) were suspended in 50 μl of citrate buffer. Next 100 μl of RNase A (0.1 mg/ml) and 100 μl of PI (500 μg/ml) were added to the cell suspension. The cells were stored at 4°. The samples were measured in a FACS scan unit and the cell cycle distribution was analyzed using cellfit software package (Becton Dickinson).
MTT assay
Cell viability was determined by the tetrazolium dye method as described earlier [7] with minor modification. MCF7 tumor cells were cultured in a multiwell plate (aliquots of approximately 2.5×103 cells per well) for 24 h in a standard incubator at 37°. Different concentrations of QC were used in a volume of 20 μl/2 ml. Cell survival rate was determined by cell proliferation assay [4 additional hours incubation with 0.13 ml MTT (5 mg/ml)] based on the living cell capacity of reducing tetrazolium salts into formazan derivatives. Relative viable cell number (absorbance at 540 nm) was determined. The 50% cytotoxic concentration (CC50) was determined from the dose response curve.
Chick embryo chorioallantoic membrane (CAM) assays
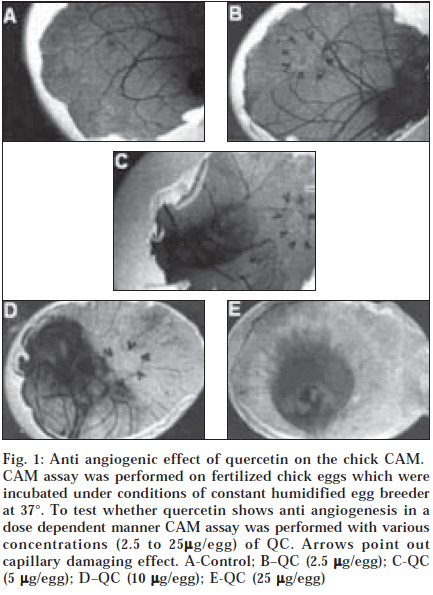
Angiostatic activity (inhibition of new capillary growth) was determined using the CAM assay, which has been described previously [14]. Fertile white Leghorn eggs were incubated for five to six days in the shell in a 97% air/ 3% CO2 atmosphere at 37° before dosing with test compounds. Test compounds, prepared in lipid suspension (10% chmyristoyl phosphatidylcholine, DMPC) were mixed with agarose to make final concentration of 1 mg/ ml test compound in 2.5% lipid and 1% agarose. The compound suspension was applied to the CAM of 5-6 day old embryos in the form of small (10 μl) dose beads containing 2.5 to 25 μg of test compound. After 48 h, angiostatic activity was evaluated by observing the inhibition of new blood vessel growth around the dose beads and the frequency of response was determined for each dose group. Each compound was tested in at least two separate experiments consisting of 10 fertile eggs per test group and an average percent response was determined for each group. Each experiment included a positive control group of embryos treated with 10 μg/ dose of the angiostatic steroid, tetrahydrocastisone (THF) and a negative control group which received agarose pellets with only the lipid carrier.
Animal studies
All animal experiments were performed in compliance with the regulations for animal treatment after obtaining the approval of the Institutional Animal Ethics Committee
Sarcoma-180 ascites tumor
Swiss mice of either sex, 4-6 weeks old were kept at the Institute’s Animal Care Facility under standard conditions (12 h light/12 h dark cycle, humidity at 50±15%, temperature 22±2° and 12 air changes/h). For the purpose of Sarcoma-180 (S-180) experiments, mice were split into four groups of six mice each after inoculation of 5×106cells/ml, tumor cells which were freshly obtained from the National Centre for Cell Sciences, Pune. Physiological saline containing 0.5% DMSO, 1.0% Tween 80 were used for drug administration. Drug doses were administered from day 1 to 7 through intraperitoneal (i.p.) route. The control groups received an equal volume of vehicle (0.2 ml) during the treatment period. Animals were weighed and the number of deaths was recorded daily. Antitumor activity was determined by the basis of median survival time (MST) of treated (T) and control (C) groups and expressed as percent T/C value. A T/C percentage value ≥125 is considered as indicative of activity [15].
Mammary carcinoma model
C3H/J mice, 4-6 weeks old, initially weighing 18-20 g were obtained from the Institutes Animal House. Tumors were induced in 24 mice by subcutaneous injection of 0.2 ml PBS containing 3×107 tumor cells, on the right flank of mice. The animals were then divided into four groups, one control and three experimental groups consisting of six mice per group. Drugs were administered when mammary tumors become palpable (day 1). Length and width of the tumors were measured by calipers twice weekly up to day 30 and the tumor volume (TV) was calculated according to the formula, TV=(length×width2)/2. Then relative tumor volume (RTV) was calculated as: RTV=TVn/TVo, where TVn is the TV at day n and TVo is the TV at day 0. Body weight of the mice was measured twice weekly up to day 30 to monitor treatment-related toxic effects [15]. Combined activity was evaluated by comparing the antitumor efficacy of combination treatment of mitomycin C (MMC) and QC with that of MMC treatment alone.
Results and Discussion
Medicinal plants contain pharmacological substances including flavonoids and their extracts have been therapeutically administered for cancer therapy. Flavonoids are po1yphenolic compounds that occur ubiquitously in foods and beverages of plant orgin [1]. A broad spectrum of enzymes have been inhibited by polyphenolic QC including EGF receptor tyrosine kinase [10] and protein kinase C10 and glutathione reductase [4]. Although the exact mechanisms of quercetin induced cytotoxicity are not very clear, it has been reported that the inhibition of EGF receptor kinase by QC was an important factor in the occurrence of apoptosis of colorectal tumor cells [8].
Table 1 demonstrates the response of MCF7 cells to QC and its complex when the cells were exposed for 72 h. Trypan blue-excluding cells were counted using a hemocyometer. The 50% inhibitory concentration (IC50) of QC against human breast cancer cell line was 10 μg/ ml. The inclusion complex of QC induced significant dose-dependant inhibition in cell growth. Using 10 μg/ml as an IC50 concentration, MCF-7 cells were exposed for different time intervals. At 24, 48 and 72 h there was corresponding decreased in cell viability (62.7, 46.4 and 36.2, respectively). The proliferation pattern of MCF7 cells exposed to QC was also confirmed by 3-[4,5dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT) assay. MTT is oxidized by cells with intact mitochondria and yields a purple product that can be solubilized with DMSO. Polyphenolic QC has been shown to act as antioxidant and prooxidant [4]. There is a possibility that it may affect the cytotoxic assay, which is based on the redox conversion of tetrazolium salt. The cytotoxic concentrations (CC50) were found to be higher for QC and its complexes (data not shown).
| Concentration | Viability (%) | ||
|---|---|---|---|
| (µg/ml) | β -cyclodextrin | QC | QC-complex |
| 0 | 100.00 | 100±0 | 100±0 |
| 2.5 | 100.00 | 94.5±2.8 | 99.0±1.8 |
| 5.0 | 100.00 | 84.4±4.3 | 92.0±2.2 |
| 10.0 | 98.5 | 36.2±3.2 | 80.0±3.0 |
| 25.0 | 95.3 | 30.8±4.6 | 38.0±2.6 |
| 50.0 | - | 20.6±6.2 | 32.0±1.7 |
Cells were treated with various concentrations of QC and the inclusion complex. The number of viable cells was determined by trypan blue dye exclusion assay. The Values are the mean ±SE of three independent experiments
Table 1: Comparative Cytotoxicity Of Qc And Its Complex
The percentages of cells in each cell cycle phase at 72 h after addition of QC are summarized in Table 2. An increase in S phase cells was observed with increase in QC concentration up to 10 μg/ml followed by a decrease coupled with a reduction in G1 phase cells. The results also demonstrated that QC inhibited the proliferation of MCF7 cells via arrest of the cell cycle at the G2/M phase. It could be associated with disturbance of cell division due to effect of Qc on DNA as damage to DNA is known to arrest cells at G2/M phase. On the basis of these results it can be concluded that mechanism of QC action at the cell level is complex. Nonetheless flavonoids are a class of clinically attractive drugs for the treatment of breast cancer due to their ability to counteract estradiol and growth factor-induced growth stimulation. Further studies are required to evaluate the potency of different inclusion complexes of QC namely hydroxypropyl and methyl ßcyclodextrins in comparison with that of QC on the growth and proliferation of this cell line.
| Concentration | Cells (%) | Apoptosis | ||
|---|---|---|---|---|
| (µg/ml) | G0/G1 | S | G 2/M | |
| 0 | 83.84 | 5.4 | 12.65 | |
| 2.5 | 74.87 | 8.8 | 18.24 | |
| 5.0 | 70.76 | 15.26 | 17.55 | 0.36 |
| 10.0 | 30.25 | 45.40 | 23.48 | 2.37 |
| 25.0 | 40.23 | 25.33 | 34.82 | 1.62 |
| 50.0 | 48.35 | 24.17 | 22.47 | 5.48 |
Cell cycle distribution of MCF-7 cells after 72 h incubation with different concentration of QC (2.5-50 μg/ml) was analyzed. The percentage of cells in the different cell cycle phases present as determined by flow cytometry.
Table 2: Effects Of Qc On Cells Cycle Parameters Of Human Breast Cancer Cell Line Mcf-7
Tumors are believed to develop their vasculature mainly by angiogenesis, a process whereby new vessels develop from preexisting ones [16]. During the early phase of tumor formation, malignant cells have been observed to co-opt normal vessels of the organ in which the tumor arises, prior to new vessel formation [16]. Recent studies have demonstrated that inhibitors of angiogenesis increase the uptake of chemotherapeutic drugs into a tumour [17]. Moreover, it has been shown that the combination of antiangiogenic therapy with chemotherapy leads to better antitumor and antimetastatic effects as compared to either of these therapies applied alone [17]. The results of studies performed in various experimental mouse models have shown that antiangiogenesis therapy did not cause drug resistance, a major problem significantly limiting antitumor chemotherapy [17]. We examined the ability of QC to inhibit the vasculature and growth of mammary carcinoma implanted into C3H/J mice. Although it is a potent antiangiogenic agent as shown by CAM assay (fig.1), QC failed to inhibit the vascularity, reduce the perfusion or alter the growth of the carcinoma to significant extent (data not shown). The antiangiogenic property is also shown by other workers [18], which we intend to exploit in our further studies, which are being conducted with cytotoxins.
Fig. 1: Anti angiogenic effect of quercetin on the chick CAM. CAM assay was performed on fertilized chick eggs which were incubated under conditions of constant humidified egg breeder at 37°. To test whether quercetin shows anti angiogenesis in a dose dependent manner CAM assay was performed with various concentrations (2.5 to 25µg/egg) of QC. Arrows point out capillary damaging effect. A-Control; B–QC (2.5 µg/egg); C-QC (5 µg/egg); D–QC (10 µg/egg); E-QC (25 µg/egg)
Our data further demonstrated that QC causes prolongation of survival period of S180 tumor bearing mice. The effect at 100 mg/kg was greater than those observed at lower doses (Table 3).
| Group | Dose | Av. body wt. | Median | T/C (%) |
|---|---|---|---|---|
| (mg/kg) | difference on day | survival time (MST) | ||
| 5 (in gm) | (Days) | |||
| Control | 0 | +2.0 | 14.0 | 100 |
| Q C | 25 | +1.8 | 14.0 | 100 |
| 50 | +0.9 | 14.5 | 103 | |
| 100 | +0.1 | 32.5* | 232 |
Swiss mice were inoculated with 5×l06 S-180 ascitic tumor cells i.p. on day 0. Treatment with various doses of QC was started after 24 h by i.p. administrations and continued for 7 days. *P<0.001 - significant from control.
Table 3: Effect Of Qc On Survival Of S-180 Ascitis Tumour Bearing Mice
Advanced cervical cancer (ACC) is incurable with standard therapeutic modalities. Therefore, the identification of active agents/or combinations is of critical importance in this disease. Cisplatin (CDDP) is one of the most widely used agents in patients with ACC, producing about 15-20% objective responses. These responses are usually partial and of short duration. Since QC shows promising activity in combination with CDDP in S180 ascites tumor model (unpublished work) we suggest QC should be associated with CDDP as it has major relevance in this disease. Moreover combining an inhibitor of PI3K like QC with a chemotherapeutic agent such as taxol is an effective way to control the growth of ovarian carcinoma with fewer side effects than either agent alone.
Cancer cells present in solid tumors are often surrounded by stressful microenvironments, such as glucose starvation, low pH, hypoxia and other nutrient deprivation [19]. These physiological stress conditions are sources of cellular drug resistance and undergo glucose starvation and hypoxia thereby become resistant to multiple drugs, including the topoII- directed drug etoposide and doxorubicin [19,20]. Studies have suggested that tumor hypoxia could be exploited by the use of bioreductive agents, which preferentially kill hypoxic cells [20]. QC by causing vasoconstriction in tumor capillaries induces sufficient tumor hypoxia necessary for activation of mitomycin C (MMC) which exhibits toxicity under hypoxic condition. We evaluated the combination effect of bioreductive agent MMC with QC in the treatment of transplantable mammary carcinoma tumor. QC could significantly enhance the sensitivity of MMC to mammary carcinoma (Table 4).
| Tumor volume (mm3) | ||||
|---|---|---|---|---|
| Days | Tumor | Tumor + QC | Tumor + MMC | Tumor + QC + MMC |
| control | (100 mg/kg) | (2.5 mg/kg) | (100 + 2.5 mg/kg) | |
| 1 | 7.69 | 0.5 | 15.3 | 20.2 |
| 5 | 34.5 | 11.8 | 48.8 | 93.1 |
| 9 | 274.1 | 119.6 | 35.9 | 27.1 |
| 13 | 2023.3 | 665.5 | 19.6 | 36.3 |
| 16 | 2128.7 | 728.7 | 46.4 | 28.6 |
| 19 | 3212.9 | 947.0 | 1042.2 | 517.9 |
| 21 | 3867.9 | 1180.1 | 1149.4 | 738 |
Mice were inoculated s.c. into the right flank with 3×l07 mammary adenocarcinoma cells. When tumors were measurable, mice were assigned into four groups tumor control, tumor + QC, tumor + MMC and tumor + QC + MMC. Treatment was given on days 1, 5, 9, 13. Caliper measurements of tumor diameter were performed to estimate tumor volume.
Table 4: Effect Of Qc And Mitomycin C (Mmc) Combined Treatment On Mammary Tumor Growth
In summary our results suggest that QC exerts antiproliferative effect on MCF- 7 cells and antiangiogenic properties as assessed by using CAM model. Further investigations are needed to confirm that a selective inhibition of mammary adenocarcinoma can be achieved in vivo. Because QC is already approved for use in Human (in leukemic blasts), clinical trials with cyclodextrin complex could be undertaken readily. The natural polyphenolic QC has definite potential to become a chemotherapeutic agent for human cancer.
References
- Wang, H.K., Expert Opin. Invest. Drugs, 2000, 9, 2103.
- Ren, W., Qiao, Z., Wang, H., Zhu, L. and Zhang, L., Med. Res. Rev., 2003, 23, 519.
- Siess, M.H., Le Bon, A. M., Canivenc-Lavier, M.C. and Suschetet, M., Biofactors, 2000, 12, 193.
- Silva, M.M., Santos, M.R., Caroco, G., Rocha, R., Justino, G. and Mira, L., Free Radic. Res., 2002, 36, 1219.
- Liesveld, J.L., Abboud, C.N., Lu, C., McNair, C., Menon, A., Smith, A., Rosell, K. and Rapoport, A.P., Leuk. Res., 2003, 27, 517.
- Molnar, J., Beladi, I., Domonkos K., Foldeak, S., Boda, K. and Veckenstedt, A., Neoplasma, 1981, 28, 11.
- Rodgers, E.H. and Grant, M.H., Chem, Biol. Interact., 1998, 116, 213.
- Richter, M., Ebermann, R. and Marian, B., Nutr. Cancer, 1999, 34, 88.
- Bhise, S.B., Nalawade, A.D. and Wadhawa, H., Indian J. Biochem. Biophys.,2004, 41, 273.
- Srivastava, A.K., Biochem. Biophys. Res.Commun., 1985, 131, 1.
- Indap, M.A., Bhosale, S.C., Miranda, M. and Rao, S.G.A., Indian Drugs, 1998, 35, 128.
- Indap, M.A., Bhosale, S.C., Vavia, P.R. and Tayade, P.T., Indian Drugs, 1998, 35, 545.
- Indap, M.A., Bhosale, S.C., Vavia, P.R. and Tayade, P.T., Indian J. Pharm. Sci., 2002, 64, 349.
- Ribatti, D., Gualandris, A., Bastaki, M., Vacca, A., Iurlaro, M., Roncali, L. and Presta, M., J. Vasc. Res., 1997, 34, 455.
- Geran, R.I., Greenberg, W.H., MacDonald, M.M., Schumcher, A.M. and Abbot, B.J., Cancer Chemother. Rep., 1972, 3, 1.
- Risau, W., Nature, 1997, 386, 671.
- Hampton, T., J. Amer. Med. Assoc., 2005, 293, 1051.
- Tan, W.F., Lin, L.P., Li, M.H., Zhang, Y.X., Tong, Y.G., Xiao, D. and Ding, J., Eur. J. Pharmacol., 2003, 459, 255.
- Brown, J.M. and Giaccia, A.J., Cancer Res., 1998, 58, 1408.
- Martin Brown, J., Cancer Res., 1999, 59, 5863.