- *Corresponding Author:
- S. T. Prajapati
Shri Sarvajanik Pharmacy College, Nr. Arvind Baugh, Mehsana - 384 001, India
E-mail: stprajapati@gmail.com
| Date of Submission | 13 March 2006 |
| Date of Revision | 15 March 2007 |
| Date of Acceptance | 30 May 2007 |
| Indian J. Pharm. Sci., 2007, 69 (3): 427-430 |
Abstract
Carbamazepine a dibenzapine derivative with structural resembling to the tricyclic antidepressant, it is used to control some types of seizures in the treatment of epilepsy. It is also used to relieve pain due to trigeminal neuralgia. One of the major problems with this drug is its very low solubility in biological fluids, which results into poor bioavailability after oral administration. Hence present study was carried out to enhance dissolution properties of carbamazepine. Physical mixtures and solid dispersions of carbamazepine were prepared to enhance its water solubility. Physical mixtures and solid dispersions of carbamazepine were prepared by using polyvinyl pyrrolidone K-30, polyethylene glycol 4000 and polyethylene glycol 6000 as water-soluble carrier at various proportion (1:0.1, 1:0.2, 1:0.4, 1:0.6, 1:0.8, by weight) by employing solvent evaporation method. The drug release profile was studied according to USP XXIII monograph in 1% sodium lauryl sulphate solution. It was found that the dissolution rate and the dissolution parameters of the drug from the physical mixture as well as solid dispersion were higher than those of the intact drug. The degree of the dissolution rate enhancement depended on the nature and the amount of the carrier, i.e., the higher amount of the carrier used, the higher dissolution rate was obtained except for polyvinyl pyrrolidone K-30 and PEG 4000 solid dispersions. Among carrier studied solid dispersion of Carbamazepine: PVP K-30 at 1:0.2 (drug:carrier ratio) gave highest dissolution. The increase in dissolution rate of the drug may be due to increase wettability, hydrophilic nature of the carrier and also possibility due to reduction in drug crystalinity.
Carbamazepine, a dibenzapine derivative with structure resembling the tricyclic antidepressans, is used to control some types of seizures in the treatment of epilepsy. It is also used to relieve pain due to trigeminal neuralgia (tic douloureux). It should not be used for other more common aches or pains. One of the major problems with this drug is its very low solubility in biological fluids and its biological half-life between 18 to 65 h that results into poor bioavailability after oral administration [1,2]. It shows erratic dissolution profile in gastric and intestinal fluid due to its poor water solubility. Rate of absorption and/or extent of bioavailability for such insoluble drug are controlled by rate of dissolution in gastrointestinal fluids. The peak plasma concentration (Cmax) and the time taken to reach Cmax (tmax) depend upon extent and rate of dissolution of drug respectively. The effort to improve the dissolution and solubility of a poorly water-soluble drug remains one of the most challenging tasks in drug development. Several methods have been introduced to overcome this problem like solid dispersions, complexation, Zydis technology, and by the use of hydrophilic carriers.
Solid dispersion, which was introduced in the early 1970s [3], is essentially a multicomponent system, having drug dispersed in and around hydrophilic carrier(s). Solid dispersion technique has been used for a wide variety of poorly aqueous soluble drugs such as nimesulide [4], ketoprofen [5], tenoxicam [6], nifedipine [7], nimodipine [8], ursodeoxycholic acid [9] and albendazole [10]. Various hydrophilic carriers, such as polyethylene glycols [11], polyvinylpyrrolidone [12], hydroxypropylmethylcellulose [13], gums [8], sugar [14], mannitol [15] and urea [9] have been investigated for improvement of dissolution characteristics and bioavailability of poorly aqueous-soluble drugs. Solid dispersion can be prepared by various methods such as solvent evaporation and melting method. Solid dispersion technique has been extensively used to increase the solubility of a poorly water-soluble drug. According to this method, a drug is thoroughly dispersed in a water-soluble carrier by suitable method of preparation. The mechanism by which the solubility and the dissolution rate of the drug is increased includes: firstly, the particle size of a drug is reduced to submicron size or to molecular size in the case where solid solution is obtained. The particle size reduction generally increases the rate of dissolution; secondly, the drug is changed from crystalline to amorphous form, the high energetic state which is highly soluble; finally, the wettability of the drug particle is improved by the dissolved carrier [16-18]. In the present investigation, solvent evaporation method was employed for the preparation of carbamazepine solid dispersions. The carriers used were polyvinyl pyrrolidone K-30 (PVP K-30), polyethylene glycol 4000 (PEG 4000) and polyethylene glycol 6000 (PEG 6000). The samples were prepared at various drug-tocarrier weight ratios.
Materials and Methods
Carbamazepine was obtained as a gift sample from Torrent Research Centre, Bhat village; Gandhinagar. The carrier used, PEG 4000; PEG 6000 and PVP K- 30 were from S. D. Fine Chem. Limited. Biosar. All other materials used were of pharmaceutical grade.
Preparation of physical mixtures
Accurately weighed amount of carbamazepine and either carrier at various drug-to-carrier weight ratios were thoroughly blended in glass mortar for 5 min. The composition of various batches is shown in Table 1. The products were kept in desiccator for further study.
| Batches for Physical mixture | Carbamazepine (mg) | PVP K-30 (mg) | PEG 4000 (mg) | PEG 6000 (mg) | Drug: Carrier ratio | Batches for solid dispersions |
|---|---|---|---|---|---|---|
| A1 | 200 | 20 | - | - | 01:00.1 | P1 |
| A2 | 200 | 40 | - | - | 01:00.2 | P2 |
| A3 | 200 | 80 | - | - | 01:00.4 | P3 |
| A4 | 200 | 120 | - | - | 01:00.6 | P4 |
| A5 | 200 | 160 | - | - | 01:00.8 | P5 |
| B1 | 200 | - | 20 | - | 01:00.1 | E1 |
| B2 | 200 | - | 40 | - | 01:00.2 | E2 |
| B3 | 200 | - | 80 | - | 01:00.4 | E3 |
| B4 | 200 | - | 120 | - | 01:00.6 | E4 |
| B5 | 200 | - | 160 | - | 01:00.8 | E5 |
| C1 | 200 | - | - | 20 | 01:00.1 | G1 |
| C2 | 200 | - | - | 40 | 01:00.2 | G2 |
| C3 | 200 | - | - | 80 | 01:00.4 | G3 |
| C4 | 200 | - | - | 120 | 01:00.6 | G4 |
| C5 | 200 | - | - | 160 | 01:00.8 | G5 |
Compositions given for one batch
Table 1: Composition of batches containing carbamazepine and carriers
Preparation of solid dispersions
The solid dispersions of carbamazepine and either carrier at various drug-to-carrier weight ratios were prepared by solvent evaporation method. The 4 g of Carbamazepine was dissolved in 40 ml of isopropranol in a beaker and carrier was added and mixed to dissolve at 40° on a hot plate. Isopropranol was evaporated at room temperature for 1 h and the resulting semi-wet mass was passed through 60 #. The granules were dried at room temperature for 1 h and further dried at 65° overnight in hot air oven. The products were kept in desiccator for further study. Compositions of solid dispersions were same as used in physical mixtures is shown in Table 1.
In vitro dissolution studies [19]
The dissolution of the samples was studied, using dissolution apparatus I (USP XXIV) basket method. The dissolution medium was 900 ml of 1% w/v sodium lauryl sulphate solution, maintained at 37±0.5°. The stirring speed was 75 rpm. Accurately weighed samples (physical mixtures and solid dispersion granules), equivalent to 200 mg of carbamazepine were filled in ‘0’ size hard gelatin capsule by hand filling method and placed in basked of dissolution apparatus. A 5.0 ml sample solution was withdrawn at appropriate time intervals and filtered through 0.45 µm millipore filter. An equal volume of fresh dissolution medium was immediately replaced. The concentration of carbamazepine at each sampling time was analyzed spectrophotometrically at 288 nm. The experiments were performed in triplicate. With help of standard curve equation concentration were found using absorbance values.
Results and Discussion
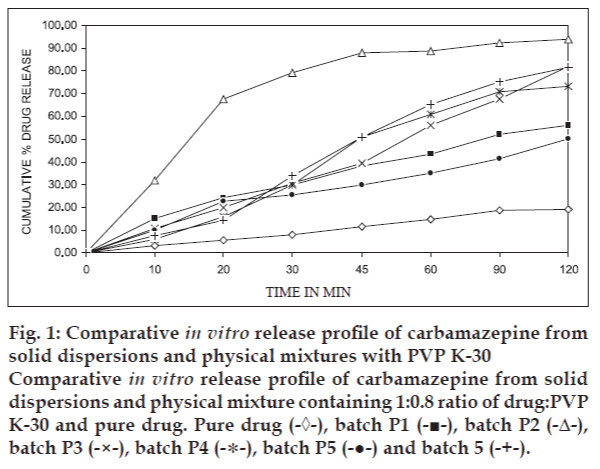
The dissolution rate of pure carbamazepine was very poor and during 120 min a maximum about 19.30% of the drug was released. The reason for the poor dissolution of pure drug could be poor wettability and/or agglomeration or particles size. It was found that the dissolution rate of the drug increased according to increasing amount of hydrophilic carriers in physical mixture batches. This was due to the increase in solubility of drug by the presence of hydrophilic carrier surrounding the drug particles. fig. 1 shows comparative release profile of various solid dispersions of carbamazepine with PVP K-30, physical mixture containing 1:0.8 ratio of drug: PVP K-30 and pure drug. From release profile it can be seen that dissolution of carbamazepine in solid dispersions increase with increase in PVP K-30 up to 1:0.2 ratio of drug: PVP K-30. This increase in the dissolution rate may due to increase in drug wettability, solubilization of drug by carriers. After this particular ratio with further increase in the amount of PVP K-30, the dissolution was decreased. The decrease in dissolution may be due to binding effect of PVP K-30. It was found that the drug release from physical mixture is greater than that of the pure drug and slower than that of solid dispersions. From the results, it was conclude that the dissolution rate of carbamazepine increased by preparing solid dispersion with PVP K-30.
Fig. 1: Comparative in vitro release profile of carbamazepine from solid dispersions and physical mixtures with PVP K-30 Comparative in vitro release profile of carbamazepine from solid dispersions and physical mixture containing 1:0.8 ratio of drug:PVP K-30 and pure drug. Pure drug (-◊-), batch P1 (-■-), batch P2 (-Δ-), batch P3 (-×-), batch P4 (-∗-), batch P5 (-●-) and batch 5 (-+-).
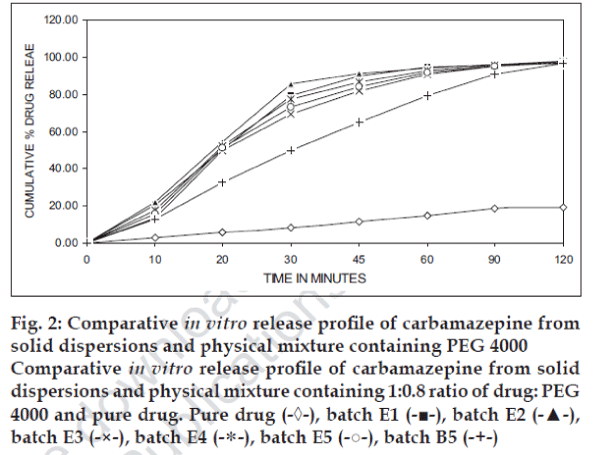
Fig. 2 shows the comparative release profile of solid dispersions of Carbamazepine with PEG 4000, physical mixture batch containing physical mixture containing 1:0.8 ratio of drug: PEG 4000 and pure drug. Release profile shows that dissolution of carbamazepine increase with increase in PEG 4000 up to 1:0.2 ratio of drug: PEG 4000. This increase in the dissolution rate may due to increase in drug wettability, solubilization of drug by carriers. After 1:0.2 ratio of drug: PEG 4000 increase in carrier ratio showed no significant change in dissolution rate of drug that may be due to localized effect of carrier. The drug release from physical mixture is greater than that of the pure drug and slower than that of solid dispersions. From the results, it was concluded that dissolution rate of Carbamazepine increased by preparing solid dispersion with PEG 4000.
Fig. 2: Comparative in vitro release profile of carbamazepine from solid dispersions and physical mixture containing PEG 4000 Comparative in vitro release profile of carbamazepine from solid dispersions and physical mixture containing 1:0.8 ratio of drug: PEG 4000 and pure drug. Pure drug (-◊-), batch E1 (-■-), batch E2 (-▲-), batch E3 (-×-), batch E4 (-∗-), batch E5 (-○-), batch B5 (-+-)
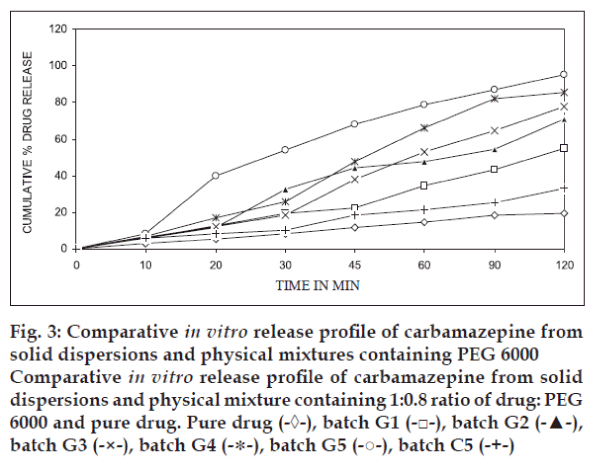
Fig. 3 shows the comparative release profile of various solid dispersions of Carbamazepine with PEG 6000, physical mixture batch containing 1:0.8 ratio of drug: PEG 6000 and pure drug. Dissolution of carbamazepine increases with increase in PEG 6000 up to 1:0.8 ratio of drug: PEG 6000. This increase in the dissolution rate may due to increase in drug wettability, solubilization of drug by carriers. It was concluded that the drug release from the physical mixture is greater than that of the pure drug and slower than that of solid dispersions. From the results, it was conclude that the dissolution rate of carbamazepine increased by preparing solid dispersion with PEG 6000.
Fig. 3: Comparative in vitro release profile of carbamazepine from solid dispersions and physical mixtures containing PEG 6000 Comparative in vitro release profile of carbamazepine from solid dispersions and physical mixture containing 1:0.8 ratio of drug: PEG 6000 and pure drug. Pure drug (-◊-), batch G1 (-□-), batch G2 (-▲-), batch G3 (-×-), batch G4 (-∗-), batch G5 (-○-), batch C5 (-+-)
Finally from the above study it was concluded that the solid dispersion technique had been shown as a successful approach to improve the dissolution rate of carbamazepine. The nature and the amount of the carrier used played an important role in the enhancement of the dissolution rate. The increase in the dissolution rate would provide the rapid onset of action after the drug is taken orally.
Acknowledgements
Authors thank Torrent Research Centre, Bhat village, Gandhinagar for providing gift samples of carbamazepine USP.
References
- Reynolds, J.E.F., Eds., In; Martindale; The Extra Pharmacopoeia, 29 thEdn., The Royal Pharmaceutical Society of Great Britain, London, 1993, 295.
- McNaman, J.O., In: Hardman, J.G., Limbird, L.E., Molinoff, P.B. and Ruddon, R.W. Eds., The Pharmacological Basis of Therapeutics: 9 thEdn. McGraw-Hill, New York, 1996, 46.
- Chiou, W.L. and Riegelman, S., J. Pharm. Sci ., 1971, 60, 1281.
- Babu, G.V., Kumar, N.R., Himasankar, K., Seshasayana, A. and Murthy, K.V., Drug Develop. Ind. Pharm., 2003, 29, 855.
- Rogers, J.A. and Anderson, A.J., Pharm. ActaHelv ., 1982, 57, 276.
- El-Gazayerly, O.N., Drug Develop. Ind. Pharm ., 2000, 26, 925.
- Vippagunta, S.R., Maul, K.A., Tallavajhala, S. and Grant, D.J.W., Int. J. Pharm ., 2002, 236, 111.
- Murali Mohan Babu, G.V., Prasad, C.H.D.S. and Ramana Murthy, K.V., Int. J. Pharm., 2002, 234, 1.
- Okonogi, S., Yonemochi, E., Oguchi, T., Puttipipatkhachorn, S. and Yamamoto, K., Drug Develop. Ind. Pharm ., 1997, 23, 1115.
- Torrado S., Torrado, S., Torrado, J. and Cadorniga, R., Int. J. Pharm ., 1996, 140, 247.
- Margarit, M.V., Rodrýguez, I.C. and Cerezo, A.. Int. J. Pharm ., 1994, 108, 101.
- Yagi, N., Terashima, Y., Kenmotsu, H., Sekikawa, H. and Takada, M., Chem. Pharm. Bull., (Tokyo). 1996, 44, 241.
- Kushida, I., Ichikawa, M. and Asakawa, N ., J. Pharm. Sci ., 2002, 91, 258.
- Danjo, K., Nakata, T. and Otsuka, A., Chem. Pharm. Bull ., (Tokyo). 1997, 45, 1840.
- Arias, M.J., Ginιs, J.M., Moyano, J.R., Pιrez-Martνnez, J.I. and Rabasco, A.M., Int. J. Pharm ., 1995, 123, 25.
- Ford, J.L., PharmaceuticaActaHelvae ., 1986, 61, 69.
- Serajuddin, T.M., J. Pharm. Sci., 1999, 88, 1058.
- Leuner, C. and Dressman, J., Eur. J. Pharm. Biopharm ., 2000, 50, 47.
- The United States Pharmacopoeia, XXIII, National Formulary XVIII, Asian edition, US Pharmacopoeial Convention, Inc., Rockville, MD, 1995, 267.