- *Corresponding Author:
- Sharma N
Dr. Reddy?s Laboratories Ltd., IPDO, Bachupally, Hyderabad‑500 090, India
E-mail: nitish2k4@gmail.com
| Date of Received : | 29 January 2013 |
| Date of Revised : | 24 January 2013 |
| Date of Accepted : | 01 July 2012 |
| Indian J Pharm Sci 2013;75(1):76-82 |
Abstract
The objective of current investigation was to study the degradation behaviour of eberconazole nitrate and mometasone furoate under different International Conference on harmonisation recommended stress condition using reverse phase high performance liquid chromatographic method and to establish validated stability-indicating high performance liquid chromatographic method to determine purity of eberconazole nitrate and mometasone furoate in presence of its impurities, forced degradation products and placebo in pharmaceutical dosage forms. The method was developed using Hypersil BDS, C18, 150Χ4.6 mm, 5 μ as stationary phase with mobile phase containing a gradient mixture of solvent A and B. 0.01 M phosphate buffer with 0.1% triethyl amine, adjusted pH 7.0 with phosphoric acid was used as buffer. Buffer pH 7.0 was used as solvent A and methanol:acetonitrile in 150:850 v/v ratios were used as solvent B. The eluted compounds were monitored at 240 nm. The run time was 50 min. The developed method was validated as per international conference on harmonization guidelines with respect to specificity, linearity, limit of detection, limit of quantification, accuracy, precision and robustness.
Keywords
Cream formulation, eberconazole nitrate, mometasone furoate, related substances, validated
Eberconazole nitrate and mometasone furoate in cream formulation (topical antifungal and antiparasites) is the combination of eberconazole nitrate a topical imidazole derivative, which has shown high potency against dermatophytes and yeasts (several species of Candida, Malassezia) in vitro and in experimental models. Eberconazole nitrate is described chemically as: (+/‑)‑1‑(2,4‑dichloro‑10, 11‑dihydro‑5H‑dibenzo [a, d] cyclohepten‑5‑yl) imidazole. Its empirical formula is C18H14Cl2N2 and molecular weight is 329.22.
Mometasone furoate is a synthetic corticosteroid with antiinflammatory activity. Chemically, mometasone furoate is 9α,21‑dichloro‑11β, 17‑dihydroxy‑16α‑ methylpregna‑1,4‑diene‑3, 20‑dione17‑(2‑furoate). Its molecular formula is C27H30CI2O6 and molecular weight is 521.40. Each 1 g of cream contains 1% w/v eberconazole nitrate and 0.1% w/v mometasone furoate.
Methods are available for determination of mometasone furoate and its degradation in human plasma [1‑3]. Mometasone furoate assay by UV detection methods are also available [4‑7]. Supercritical fluid chromatography detection methods are also available for mometasone furoate [8]. So far to our present knowledge, no validated stability indicating analytical high performance liquid chromatography (HPLC) method was available in literature for mometasone furoate and eberconazole nitrate and its impurities (fig. 1) in cream formulation. Attempts were made to develop a stability indicating LC method for the related substance determination of eberconazole nitrate and mometasone furoate in cream formulation. This paper deals with the validation of the developed method for the accurate quantification of eberconazole nitrate and mometasone furoate impurities in cream formulation.
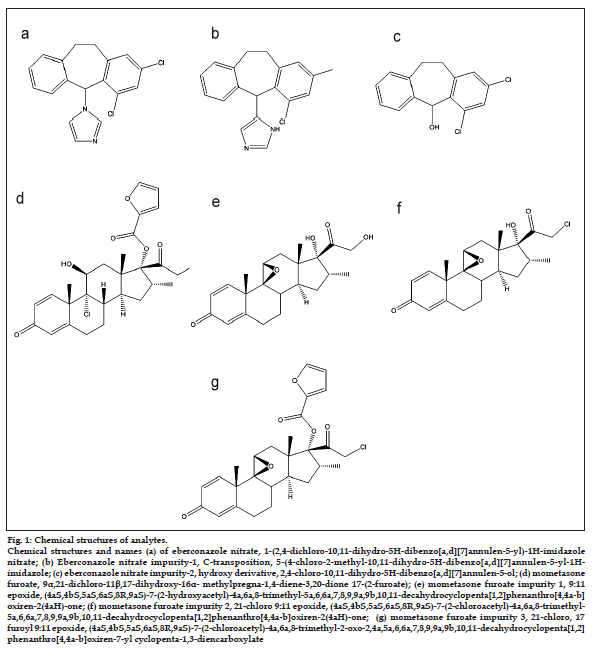
Figure 1: Chemical structures of analytes. Chemical structures and names (a) of eberconazole nitrate, 1-(2,4-dichloro-10,11-dihydro-5H-dibenzo [a,d] [7] annulen-5-yl)-1H-imidazole nitrate; (b) Eberconazole nitrate impurity‑1, C‑transposition, 5-(4-chloro-2-methyl-10,11-dihydro-5H-dibenzo [a,d] [7] annulen-5-yl-1Himidazole; (c) eberconazole nitrate impurity‑2, hydroxy derivative, 2,4-chloro-10,11-dihydro-5H-dibenzo [a,d] [7]annulen-5-ol; (d) mometasone furoate, 9?,21-dichloro-11?,17-dihydroxy-16?- methylpregna-1,4-diene-3,20-dione 17-(2-furoate); (e) mometasone furoate impurity 1, 9:11 epoxide, (4aS,4bS,5aS,6aS,8R,9aS)-7-(2-hydroxyacetyl)-4a,6a,8-trimethyl-5a,6,6a,7,8,9,9a,9b,10,11-decahydrocyclopenta [1,2] phenanthro [4,4a-b] oxiren-2(4aH)-one; (f) mometasone furoate impurity 2, 21‑chloro 9:11 epoxide, (4aS,4bS,5aS,6aS,8R,9aS)-7-(2-chloroacetyl)-4a,6a,8-trimethyl- 5a,6,6a,7,8,9,9a,9b,10,11-decahydrocyclopenta [1,2] phenanthro [4,4a-b]oxiren-2(4aH)-one; (g) mometasone furoate impurity 3, 21‑chloro, 17 furoyl 9:11 epoxide, (4aS,4bS,5aS,6aS,8R,9aS)-7-(2-chloroacetyl)-4a,6a,8-trimethyl-2-oxo-2,4a,5a,6,6a,7,8,9,9a,9b,10,11-decahydrocyclopenta [1,2] phenanthro [4,4a-b] oxiren-7-yl cyclopenta-1,3-diencarboxylate
Materials and Methods
Cream, eberconazole nitrate and mometasone furoate working standard and impurities were supplied by Dr. Reddy’s Laboratories Limited, Hyderabad, India. Deionised water was prepared using a Milli‑Q plus water purification system from Millipore (Bedford, MA, USA). The HPLC grade acetonitrile, methanol, triethylamine, analytical grade K2HPO4 and ortho phosphoric acid were purchased from Merck, Mumbai, India.
The Waters HPLC photodiode array (PDA) 2996 system (Waters Corporation, Milford, MA, USA) used consists of a quaternary solvent manager, a sample manager and a PDA detector. The output signal was monitored and processed using empowers 2 software. The specificity study was conducted by using heating oven (MACK Pharmatech, Hyderabad, India), and water baths equipped with Milli Volt controller (Julabo, Seelbach, Germany) were used for hydrolysis studies.
Photo stability studies were carried out in a photo stability chamber Sanyo, Leicestershire, UK. The pH of the solutions was measured by a pH meter (Mettler‑Toledo, Switzerland). All samples were centrifuged by Multifuge, Thermo Scientific, USA.
Chromatographic conditions
The method was developed using Hypersil BDS column, 150×4.6 mm, 5 µm, as stationary phase. 0.01 M phosphate buffer with 0.1% triethyl amine, adjusted pH 7.0 with phosphoric acid was used as buffer. Buffer pH 7.0 was used as solvent A and methanol and acetonitrile in 150:850 v/v ratios were used as solvent B. The gradient programme (T (min)/%B) was set as 0/35, 9/40, 15/50, 21/35, 24/50, 33/50, 36/60, 42/65, 46/35 and 50/35, respectively. The mobile phase pumped at 1.2 ml/min. The eluted compounds were monitored at 240 nm. The column temperature was maintained at 25°. The sample injection volume was 40 μl.
Preparation of stock solutions
A mixture of buffer pH 7.0 (0.01 M phosphate buffer with 0.1% triethyl amine, adjusted pH 7.0 with phosphoric acid) and methanol in 10:90 v/v was used as diluent to prepare solutions. A stock solution of eberconazole nitrate (1 mg/ml) and mometasone furoate (1 mg/ml) was prepared by dissolving appropriate amount of drug in diluent.
Standard solutions of 10 and 1 µg/ml were prepared from the above stock solution for the related substance determination. A stock solution of impurity (mixture of imp‑1, imp‑2 for eberconazole nitrate at 0.1 mg/ml and imp‑1, imp‑2 and imp‑3 for Mometasone furoate) at 0.1 mg/ml was also prepared in the diluent.
System suitability solution prepared by mixing (1 mg/ml) eberconazole nitrate with 10 µg/ml impurities and 0.1 mg/ml mometasone furoate with 1 µg/ml impurities from above impurity stock.
Preparation of sample solution
Transferred 1 g of sample containing 10 mg eberconazole nitrate and 1 mg of mometasone furoate in to a 10 ml volumetric flask, dissolved in 7 ml of diluent and the mixture was sonicated for about 30 min and volume was made with diluent. The solution was filtered through 0.45 µm mdi TG filter.
Forced degradation studies
Forced degradation studies were performed at 1000 µg/ml for eberconazole nitrate and 100 µg/ml for mometasone furoate on cream formulation to provide an indication of the stability indicating property and specificity of proposed method. Peak purity test was carried out for the eberconazole nitrate and mometasone furoate peak by using PDA detector on stress. Transferred 1 g of sample containing 100 mg eberconazole nitrate and 10 mg of mometasone furoate in to a 10 ml volumetric flask, dissolved in 7 ml of diluent then add 1 ml reagent and mixed. The flask was placed at desired temperature in a water‑bath for required time, After degradation, the flasks were removed and placed on bench top to attain the laboratory temperature, added 1 ml neutralised reagent (only for acid and base hydrolysis) and finally made up to the volume with diluent and mixed well.
Placebo (without actives, contains only excipient), placebo for eberconazole nitrate (containing mometasone furoate with excipient), and placebo for mometasone furoate (containing eberconazole nitrate with excipient) were prepared in similar way to identify degradation pathways.
Method validation
The proposed method was validated as per international conference on harmonization guidelines [9‑11].
System suitability
System suitability was determined before sample analysis from system suitability solution; Similarity factor from two continuous standard injections and United States Pharmacopoeia (USP) tailing factor for mometasone furoate peak and eberconazole nitrate peak from standard solution.
Specificity
Specificity is the ability of the method to measure the analyte response in the presence of its potential impurities and excipients. Placebo interference was evaluated by analysing the placebo prepared as per test method. Peak purity test was carried out for the eberconazole nitrate and mometasone furoate peaks by using PDA detector in stress samples.
Precision
The precision of the related substances method was checked by injecting six individual preparations of eberconazole nitrate (1 mg/ml) and mometasone furoate (0.1 mg/ml) spiked with 0.20% of imp‑1, imp‑2 for eberconazole nitrate and imp‑1, imp‑2 and imp‑3 for mometasone furoate with respect to respective analyte concentration. Percentage relative standard deviation (%RSD) of area for each impurity was calculated.
The intermediate precision of the method was also evaluated using different analyst and different instrument in the same laboratory.
Limits of detection and quantification
The limits of detection (LOD) and limits of quantification (LOQ) for impurities and analyte (with respect to unknown impurities) were determined at a signal‑to‑noise ratio of 3:1 and 10:1, respectively, by injecting a series of dilute solutions with known concentrations. Precision and accuracy studies were also carried out at the LOQ level by injecting six individual preparations of eberconazole nitrate and it’s known impurities (imp‑1, imp‑2) and mometasone furoate and it’s known impurities (imp‑1, imp‑2 and imp‑3) and calculating the %RSD of the area.
Linearity
Linearity test solutions for the related substance method were prepared by diluting stock solutions to the required concentrations. The solutions were prepared at five concentration levels from LOQ to 200% of the specification level 0.2%. Correlation coefficient, value for the slope, Y‑intercept and % bias of the calibration curve was calculated.
Accuracy
The accuracy study of all impurities were carried out in triplicate at LOQ, 50, 100, 125 and 150% of the target concentration level 0.2%. The percentages of recoveries for impurities were calculated.
Robustness
To determine the robustness of the developed method, experimental conditions were deliberately altered and system suitability parameters were recorded. The variable evaluated in the study was pH of the mobile phase (+0.2), column temperature (+5°), flow rate (+0.2).
Solution stability
The solution stability of eberconazole nitrate and mometasone furoate and its impurities in the related substance method was carried out by leaving spiked sample solutions in tightly capped volumetric flasks at room temperature for 24 h. Content of eberconazole nitrate imp‑1, imp‑2 and mometasone furoate imp‑1, imp‑2, imp‑3 determined for 24 h interval up to the study period.
Results and Discussion
The effect of organic modifier and stationary phase was studied for separation of critical closely eluting mometasone furoate impurity C‑transposition peak and impurity please correct to 21‑chloro‑17- fluoro‑9,11-epoxide peaks from mometasone furoate peak and hydroxy derivative impurity and eberconazole nitrate peak. Initial trials has been taken on Hypersil ODS 150×4.6 mm 5 µm column for method development with gradient elution with solvents a and b but in above condition resolution in between mometasone furoate impurity C‑transposition peak peaks was less than 1.5. Mobile phase buffer pH trials have been taken at pH 2.5, 4.5 and 7.0 on Hypersil BDS 150×4.6 mm 5 µm columns. Ion pair like triethyl amine was used for better resolution.
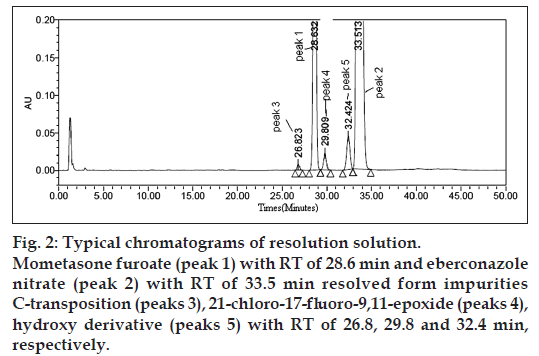
System suitability parameters were measured so as to verify the system, method and column performance. As seen from this data, the acceptable system suitability parameters would be: Relative retention time of each impurity should be comparable, tailing factor for eberconazole nitrate, mometasone furoate in standard solution is not more than 2.0, and resolution between all peaks should be more than 1.5. Spiked chromatogram of impurity/degradation products with eberconazole nitrate, mometasone furoate is presented in figs. 2 and 3.
Figure 2: Typical chromatograms of resolution solution. Mometasone furoate (peak 1) with RT of 28.6 min and eberconazole nitrate (peak 2) with RT of 33.5 min resolved form impurities C‑transposition (peaks 3), 21‑chloro‑17‑fluoro-9,11-epoxide (peaks 4), hydroxy derivative (peaks 5) with RT of 26.8, 29.8 and 32.4 min, respectively.
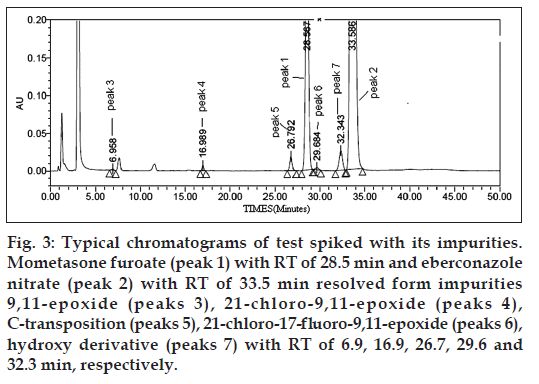
Figure 3: Typical chromatograms of test spiked with its impurities. Mometasone furoate (peak 1) with RT of 28.5 min and eberconazole nitrate (peak 2) with RT of 33.5 min resolved form impurities 9,11-epoxide (peaks 3), 21‑chloro‑9,11-epoxide (peaks 4), C‑transposition (peaks 5), 21‑chloro‑17‑fluoro-9,11-epoxide (peaks 6), hydroxy derivative (peaks 7) with RT of 6.9, 16.9, 26.7, 29.6 and 32.3 min, respectively.
Peak tailing was observed to be around 1 for both eberconazole and mometasone in standard preparations. Similarity factor for standard (between two preparations) was observed within limit (Eberconazole: 1.00, Mometasone: 1.02). Resolution between 21‑chloro‑17‑fluoro-9,11-epoxide and mometasone furoate peak and resolution between hydroxy derivative and eberconazole peak was found 2.1 and 2.0, respectively.
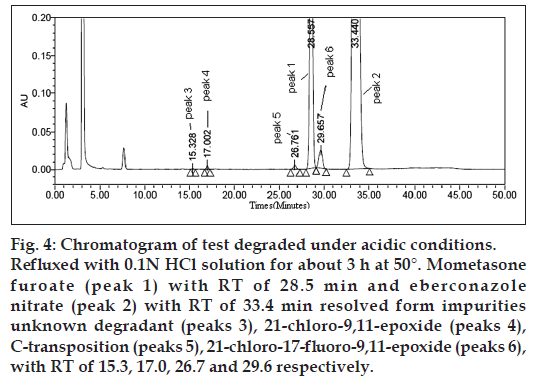
All forced degradation samples were analysed at an initial concentration of eberconazole nitrate and mometasone furoate with HPLC conditions using PDA detector to ensure the homogeneity and purity of both drugs. No peak detected due to placebo at the retention time of actives and their impurities peaks. Significant degradation of eberconazole nitrate and mometasone furoate was observed in heat (80° for 5 h), photolytic UV light (200 watt h/square meter), sun light (1.2 Million Lux h), oxidative (3% H2O2 at room temperature for 3 h), acid (0.1 N HCl at 50° for 3 h) and base (0.1 N NaOH at 25° for 30 min) conditions leading to the formation of impurities (% degradation should be >0.1% and <20%). % degradation values presented in Table 1 and degradation chromatograms presented in figs. 4-8.
| Stress condition impurity name | Acid Base Peroxide Heat Light | ||||
|---|---|---|---|---|---|
| (%) | (%) | (%) | (%) | (%) | |
| Eberconazole nitrate | |||||
| C‑Transposition | 0.18 | 0.21 | 0.2 | 0.18 | 0.3 |
| Hydroxy derivative | 0 | 0.22 | 0.3 | 0.51 | 1.42 |
| Maximum unknown 1 | 0.02 | 0.07 | 0.07 | 0.03 | 0.3 |
| Maximum unknown 2 | 0 | 0.07 | 0.03 | 0 | 0.2 |
| % degradation | 0.2 | 0.7 | 0.6 | 0.7 | 2.2 |
| Mometasonefuroate | |||||
| 9:11 epoxide | 0 | 0.02 | 0.03 | 0 | 0.3 |
| 21‑chloro 9:11 epoxide | 0.1 | 0.58 | 0.04 | 0.03 | 0.1 |
| 21‑chloro, 17 fluoro 9:11 epoxide 1.00 | 3 | 0 | 0.15 | 0 | |
| Maximum unknown 1 | 0.02 | 0.11 | 0.04 | 0.32 | 0.15 |
| Maximum unknown 2 | 0 | 0.1 | 0.04 | 0 | 0.18 |
| % degradation | 1.2 | 3.8 | 0.2 | 0.5 | 0.8 |
Note: Impurity name unknown shown maximum two unknown impurity
Table 1: Degradation Eberconazole Nitrate and Mometasone Furoate.
Figure 4: Chromatogram of test degraded under acidic conditions. Refluxed with 0.1N HCl solution for about 3 h at 50°. Mometasone furoate (peak 1) with RT of 28.5 min and eberconazole nitrate (peak 2) with RT of 33.4 min resolved form impurities unknown degradant (peaks 3), 21‑chloro‑9,11-epoxide (peaks 4), C‑transposition (peaks 5), 21‑chloro‑17‑fluoro-9,11-epoxide (peaks 6), with RT of 15.3, 17.0, 26.7 and 29.6 respectively.
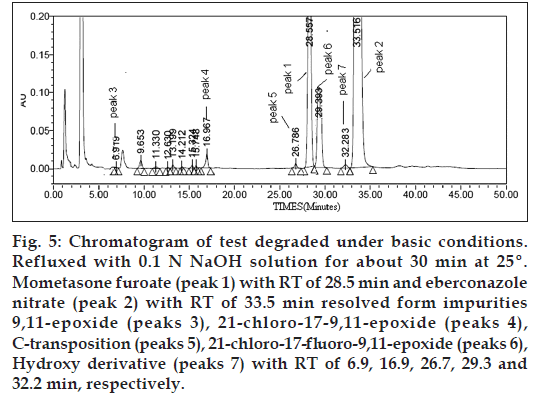
Figure 5: Chromatogram of test degraded under basic conditions. Refluxed with 0.1 N NaOH solution for about 30 min at 25º. Mometasone furoate (peak 1) with RT of 28.5 min and eberconazole nitrate (peak 2) with RT of 33.5 min resolved form impurities 9,11-epoxide (peaks 3), 21‑chloro‑17‑9,11-epoxide (peaks 4), C‑transposition (peaks 5), 21‑chloro‑17‑fluoro-9,11-epoxide (peaks 6), Hydroxy derivative (peaks 7) with RT of 6.9, 16.9, 26.7, 29.3 and 32.2 min, respectively.
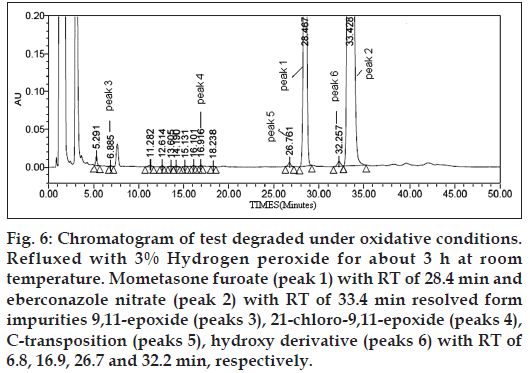
Figure 6:Chromatogram of test degraded under oxidative conditions. Refluxed with 3% Hydrogen peroxide for about 3 h at room temperature. Mometasone furoate (peak 1) with RT of 28.4 min and eberconazole nitrate (peak 2) with RT of 33.4 min resolved form impurities 9,11-epoxide (peaks 3), 21‑chloro‑9,11-epoxide (peaks 4), C‑transposition (peaks 5), hydroxy derivative (peaks 6) with RT of 6.8, 16.9, 26.7 and 32.2 min, respectively.
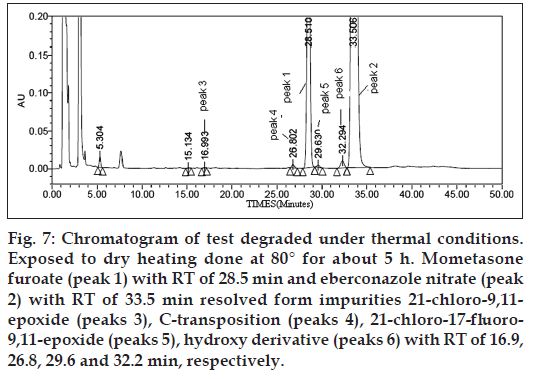
Figure 7: Chromatogram of test degraded under thermal conditions. Exposed to dry heating done at 80° for about 5 h. Mometasone furoate (peak 1) with RT of 28.5 min and eberconazole nitrate (peak 2) with RT of 33.5 min resolved form impurities 21‑chloro‑9,11-epoxide (peaks 3), C‑transposition (peaks 4), 21‑chloro‑17‑fluoro-9,11-epoxide (peaks 5), hydroxy derivative (peaks 6) with RT of 16.9, 26.8, 29.6 and 32.2 min, respectively.
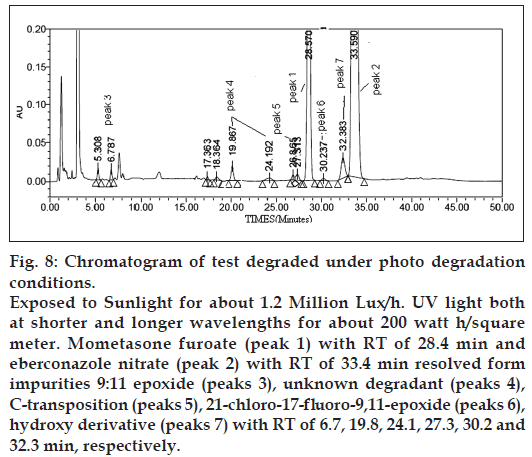
Figure 8: Chromatogram of test degraded under photo degradation conditions. Exposed to Sunlight for about 1.2 Million Lux/h. UV light both at shorter and longer wavelengths for about 200 watt h/square meter. Mometasone furoate (peak 1) with RT of 28.4 min and eberconazole nitrate (peak 2) with RT of 33.4 min resolved form impurities 9:11 epoxide (peaks 3), unknown degradant (peaks 4), C‑transposition (peaks 5), 21‑chloro‑17‑fluoro-9,11-epoxide (peaks 6), hydroxy derivative (peaks 7) with RT of 6.7, 19.8, 24.1, 27.3, 30.2 and 32.3 min, respectively.
The %RSD for the area of eberconazole nitrate imp‑1 and imp‑2 and mometasone furoate imp‑1, imp‑2, imp‑3 in related substances method precision was found less than 10% (should be less than 15.0%) conforming good precision of the method. %RSD values presented in Table 3.
The determination of limit of detection and limit of quantification of all the impurities namely eberconazole nitrate, imp‑1 and imp‑2 and mometasone furoate, imp‑1, imp‑2, imp‑3 are reported in Table 3. The precision at the LOQ concentrations for eberconazole nitrate and imp‑1 and imp‑2 and mometasone furoate imp‑1, imp‑2, imp‑3 were found below 10% (should be less than 15.0%).
Linear calibration plot for the related substance method was obtained over the calibration ranges tested, i.e. LOQ to 200% for impurity (eberconazole nitrate, imp‑1 and imp‑2 and mometasone furoate, imp‑1, imp‑2, imp‑3). The correlation coefficient obtained was greater than 0.997 (Table 2). The above result show that an excellent correlation existed between the peak area and the concentration. The % bias also calculated for all related compounds and main analytes and found less than 5%.
| LOD | LOQ | Slope | Intercept | Correlation | Precision | Precision LOQ | |
|---|---|---|---|---|---|---|---|
| (μg/ml) | (μg/ml) | (m) | (c) | coefficient | (%RSD) | (%RSD) | |
| C‑Transposition | 0.033 | 0.1 | 31,853.10 | 3279.9 | 1 | 1 | 10.3 |
| Hydroxy derivative | 0.008 | 0.024 | 87,378.30 | 1944.4 | 1 | 0.6 | 4.8 |
| Eberconazole nitrate | 0.007 | 0.02 | 47,148.80 | 1920.7 | 0.999 | 0.6 | 6.4 |
| 9:11 Epoxide | 0.007 | 0.022 | 70,640.80 | 1053.3 | 0.999 | 1.3 | 9.7 |
| 21‑chloro 9:11 Epoxide | 0.022 | 0.066 | 58,209.30 | 891.8 | 0.999 | 1.3 | 4 |
| 21‑chloro, 17 fluoro 9:11 Epoxide | 0.012 | 0.036 | 95,436.80 | 275.6 | 0.999 | 2.3 | 4.2 |
| Mometasonefuroate | 0.012 | 0.035 | 100,732.90 | 430.8 | 1 | 3.4 | 1.6 |
LOD=Limits of detection, LOQ=Limits of quantification, RSD=Relative standard deviation
Table 2: Regression and precision data. Eberconazole nitrate and mometasone furoate.
The percentage recovery of eberconazole nitrate and mometasone furoate and respective impurities in cream varied from 90 to 110% at LOQ, 50, 100, 125 and 150% levels of target 0.2% of respective analyte target concentrations. The LC chromatogram of spiked sample at 0.2% level of all five impurities in cream sample solution is shown in fig. 3. % Recovery values for impurities are presented in Table 3.
| Name of impurity | (%) | LOQ | 50% | 100% | 125% | 150% |
|---|---|---|---|---|---|---|
| C‑Transposition | Recovery | 93.1 | 106.1 | 106.4 | 110.3 | 109.9 |
| Hydroxy derivative | Recovery | 108.2 | 108.9 | 108 | 107.7 | 106.5 |
| Eberconazole nitrate | recovery | 92.2 | 95.3 | 93.4 | 96.7 | 96.2 |
| 9:11 epoxide | Recovery | 102.9 | 105.8 | 102.9 | 99.6 | 103.1 |
| 21‑chloro 9:11 epoxide | Recovery | 99.2 | 96 | 96.3 | 99 | 91.5 |
| 21‑chloro, 17 fluoro | Recovery | 95.7 | 99.6 | 100.2 | 100 | 101.1 |
| 9:11 epoxide | ||||||
| Mometasonefuroate | Recovery | 105.7 | 105.9 | 108.8 | 106.4 | 108.2 |
LOQ=Limits of quantification
Table 3: Evaluation of Accuracy Eberconazole Nitrate and Mometasone Furoate.
In all the deliberate varied chromatographic conditions (flow rate, column temperature and pH of mobile phase buffer), the resolution between critical pairs of peaks were greater than 1.5, illustrating the robustness of the method.
No significant changes were observed in the content of impurities namely impurity‑1 and imp‑2 of eberconazole nitrate and imp‑1, imp‑2, imp‑3 of mometasone furoate during solution stability experiments. The solution stability experiment data confirms that the sample solutions used during the related substances determination were stable for 24 h.
The gradient RP‑HPLC method developed for eberconazole nitrate and mometasone furoate related substances in pharmaceutical dosage forms is found precise, accurate, linear, robust, rugged and specific. Method is stability‑indicating and can be used for routine analysis of production samples and to check the stability of samples.
Acknowledgements
The authors wish to thank the management of Dr. Reddy’s Laboratories Ltd. for supporting this work.
References
- Teng XW, Foe K, Brown KF, Cutler DJ, Davies NM. High‑performance liquid chromatographic analysis of mometasonefuroate and its degradation products: Application to in vitro degradation studies. J Pharm Biomed Anal 2001;26:313‑9.
- Sahasranaman S, Tang Y, Biniasz D, Hochhaus G. A sensitive liquid chromatography‑tandem mass spectrometry method for the quantification of mometasonefuroate in human plasma. J Chromatogr B AnalytTechnol Biomed Life Sci 2005;819:175‑9.
- Davies NM. Degradation and metabolism of mometasonefuroate in humans: Influence of reversible, sequential metabolism, and ionic strength. J Pharm Sci 2004;93:2877‑80.
- Shaikh S, Muneera MS, Thusleem OA, Tahir M, Kondaguli AV. A simple RP‑HPLC method for the simultaneous quantitation of chlorocresol, mometasonefuroate, and fusidic acid in creams.J ChromatogrSci 2009;47:178‑83.
- Zhu J, Coscolluella C. Chromatographic assay of pharmaceutical compounds under column overloading. J Chromatogr B Biomed SciAppl 2000;741:55‑65.
- Vairale A, Sherikar AV, Bindu VH, Sivaswaroop P. Determination of mometasonefuroate by HPLC in topical preparations:Validation. Trade SciInc 2009;8:1.
- Kulkarni AA, Nanda RK, Ranjane MN, Ranjane PN. Simultaneous estimation of Nadifloxacin and MometasoneFuroate topical cream by HPTLC method. Der PharmaChem 2010;2:25‑30.
- Wang Z, Zhang H, Liu O, Donovan B. Development of an orthogonal method for mometasonefuroate impurity analysis using supercritical fluid chromatography. J Chromatogr A 2011;1218:2311‑9.
- ICH. International Conference on Harmonization Q2 (R1), Validation of Analytical Procedures: Text and Methodology; 2005.
- ICH. International Conference on Harmonization Q1A (R2), Stability testing of new drug substances and products; 2000.
- ICH. International Conference on Harmonization Q1B (R2), photo stability testing on new drug substances and products; 1996.