- Corresponding Author:
- V. K. Mourya
Department of Pharmaceutics, Shri Bhagwan College of Pharmacy, N-6, CIDCO, Aurangabad-431 003
E-mail: pavanbrathi@gmail.com
| Date of Submission | 13 April 2010 |
| Date of Revision | 6 May 2011 |
| Date of Acceptance | 17 May 2011 |
| Indian J. Pharm. Sci., 2011, 73 (3): 315-319 |
Abstract
The extended Hildebrand solubility parameter approach is used to estimate the solubility of satranidazole in binary solvent systems. The solubility of satranidazole in various dioxane-water mixtures was analyzed in terms of solutesolvent interactions using a modified version of Hildebrand-Scatchard treatment for regular solutions. The solubility of satranidazole in the binary solvent, dioxane-water shows a bell-shaped profile with a solubility maximum well above the ideal solubility of the drug. This is attributed to solvation of the drug with the dioxane-water mixture, and indicates that the solute-solvent interaction energy is larger than the geometric mean (δ1δ2) of regular solution theory. The new approach provides an accurate prediction of solubility once the interaction energy is obtained. In this case, the energy term is regressed against a polynomial in δ1 of the binary mixture. A quartic expression of W in terms of solvent solubility parameter was found for predicting the solubility of satranidazole in dioxane-water mixtures. The method has potential usefulness in preformulation and formulation studies during which solubility prediction is important for drug design.
Keywords
Dioxane, extended Hildebrand solubility approach, ideal solubility, interaction energy, regular solution theory, satranidazole, solubility parameter
Solubility data on drugs and pharmaceutical adjuncts in mixed solvents have wide applications in the drug sciences. Knowledge of interaction forces between solutes and solvents are of considerable theoretical and practical interest throughout the physical and biological sciences [1]. The theory of solution is one of the most challenging branch of physical chemistry. The Hildebrand-Scatchard theory of regular solution is the pioneer approach in this field, used to estimate solubility only for relatively non-polar drugs in nonpolar solvents [2]. An irregular solution is one in which self-association of solute or solvent, solvation of the solute by the solvent molecules, or complexation of two or more solute species are involved [3]. Polar systems exhibit irregular solution behaviour and are commonly encountered in pharmacy. The extended Hildebrand solubility approach (EHSA), a modification of the Hildebrand-Scatchard equation, permits calculation of the solubility of polar and non polar solutes in solvents ranging from non polar hydrocarbons to highly polar solvents such as water, ethanol, and glycols [4]. The solubility parameters of solute and solvent were introduced to explain the behaviour of regular and irregular solutions [5]. The extended Hildebrand solubility parameter approach has been developed to reproduce the solubility of drugs and other solids in the binary solvent systems [6].
The Hildebrand-Scatchard Eqn. for solubility
of solids in a regular solution may be written
as [7], 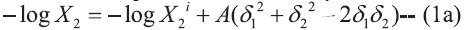 and
and 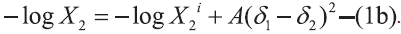
The extended Hildebrand Eqn for the solubility of
solids in an irregular solution may be written as [8]: 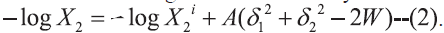 From
the geometric mean:
From
the geometric mean: 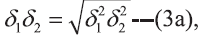 where,
in pharmaceutical solutions, the square root of
geometric mean of δ12 and δ22, that is δ1δ2= (δ12δ22)1/2, is too restrictive and ordinarily provides a poor fit to
experimental data in irregular solutions. The assumption
that the geometric mean of two geometric parameters
δ1δ2 (eqn 1a) can be replaced by a less restrictive term
W (eqn 2), interaction energy parameter, which is
allowed to take on values as required to yield correct
mole fraction solubilities, X2 as [9],
where,
in pharmaceutical solutions, the square root of
geometric mean of δ12 and δ22, that is δ1δ2= (δ12δ22)1/2, is too restrictive and ordinarily provides a poor fit to
experimental data in irregular solutions. The assumption
that the geometric mean of two geometric parameters
δ1δ2 (eqn 1a) can be replaced by a less restrictive term
W (eqn 2), interaction energy parameter, which is
allowed to take on values as required to yield correct
mole fraction solubilities, X2 as [9], 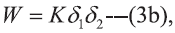 Where, K is the proportionality factor relating
interaction energy (W) to the geometric mean of
solubility parameter (δ).
Where, K is the proportionality factor relating
interaction energy (W) to the geometric mean of
solubility parameter (δ).
In Eqns 1 and 2, X2 and X2i are the mole fraction solubility and ideal mole fraction solubility of the solute respectively. The terms δ1 and δ2 are the solubility parameters for the solvent and solute respectively. The geometric mean, δ1δ2, provides a reasonable estimate of solvent-solute interaction in regular (ordinarily nonpolar) mixtures, whereas W or Kδ1δ2 is required to express solubility’s in nonregular systems (irregular solutions) of drugs in associating mixed solvents.
The term A in Eqns 1 and 2 is defined as [10]:
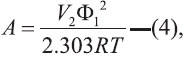
where, V2 is the molar volume of the solute taken as
a supercooled liquid at solution temperature, R is the universal gas constant, T is the absolute temperature,
298.2 K, of the experiment and Φ1, the volume
fraction of the solvent, is [11]: 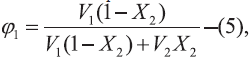 where, V1 is the molar volume of the solvent at 25º.
where, V1 is the molar volume of the solvent at 25º.
Satranidazole, [1-methylsulphonyl-3-(1-methyl-5- nitro-2-imidazolyl)-2-imidazolidinone], a potent broad spectrum antiprotozoal, is very sparingly soluble in water (0.01mg/ml) [12,13]. It is not official in I.P., U.S.P. and B.P till date [14,15].
A perusal to the structure of satranidazole (fig. 1) indicates that the molecule is highly aromatic and the functional groups may not contribute much to the aqueous solubility. The poor aqueous solubility and wettability of satranidazole give rise to difficulties in pharmaceutical formulations meant for oral or parenteral use, which may lead to variation in bioavailability [16]. Therefore, it is necessary to explore the solubility of satranidazole in water-dioxane binary mixture.
Dioxane is a very interesting cosolvent to study the interrelation between drug solubility and medium polarity because it is completely non-aqueous water miscible solvent [17]. Water-dioxane binary mixtures are strongly non ideal and can act in the solute-solvation process via hydrophobic interactions and preferential solvation because, water-dioxane mixtures cover a wide range of Hildebrand solubility parameters from 10.00 (Cal/cm3)0.5 (pure dioxane) to 23.4 (Cal/cm3)0.5 (pure water). Thus, satranidazole is an ideal candidate for the study of solubility behaviour in mixtures of dioxane and water. The present investigation pertains to the utility of EHSA in relation to the satranidazole solubility in the solvent pair dioxane-water binary solvents.
Satranidazole, obtained as gift sample from Alkem Laboratories Ltd., Baddi, India, was purified by recrystallization process. The solvent used for recrystallization of satranidazole was acetone. 1,4-Dioxane and acetone were obtained as gift sample from E. Merck, Ltd.; Mumbai, India and Qualigens Fine Chemicals, Mumbai, India, respectively. Throughout the study double distilled water was used for experimental purpose. All chemicals and reagents used in the study were of analytical grade and used as such. Double beam UV/Vis spectrophotometer, Shimadzu model 1601 with spectral bandwidth of 2 nm, wavelength accuracy ±0.5 nm and a pair of 10 mm matched quartz cells was used to measure absorbance of the resulting solutions. Citizen balance, CX-100, was used for weighing of Satranidazole.
The solubility of satranidazole was determined in binary solvent mixtures of dioxane and water. Double distilled water was used to prepare mixtures with dioxane in concentrations of 0-100% by volume of dioxane. About 10 ml of dioxane, water, or binary solvent was introduced into screw-capped vials containing an excess amount of satranidazole. After being sealed with several turns of electrical tape, the vials were submerged in water at 25±0.4° and were shaken at 150 rpm for 24 h in a constant-temperature bath. Preliminary studies showed that this time period was sufficient to ensure saturation at 25°.
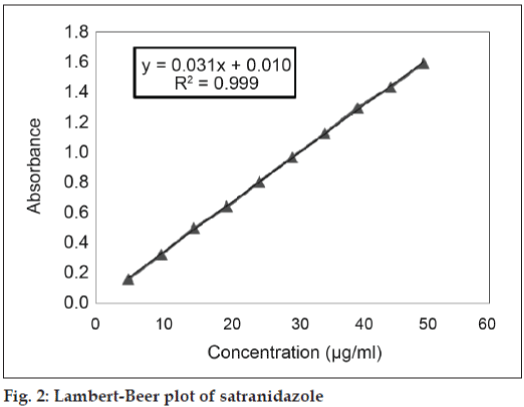
After equilibrium was attained, vials were removed for analysis. Firstly, solutions were filtered through Whatman filter paper (No. 41). After appropriate dilutions with double distilled water, the solutions were analyzed by using a spectrophotometer set at the wavelength of maximum absorption of the satranidazole (λmax-319.80 nm). Calibration graph of satranidazole in solvent blend was previously established with very high degree of correlation coefficient (R2) 0.9997, slope 0.0318 and negligible intercept (0.0101) as shown in fig. 2. The working concentration range was from 5 to 50 μg/ml. The solubility of the satranidazole was determined at least three times for each solvent, and the average value was taken. The densities of the solvent mixtures and the filtrates of the saturated solutions of satranidazole were determined in triplicate at 25±0.4° using 10-ml specific gravity bottle.
The solubility parameters of the solvents were obtained from the literature [18]. The solubility parameter of satranidazole was calculated by the method of Fedor [19,20], which was confirmed by solubility analysis in dioxane-water blend.
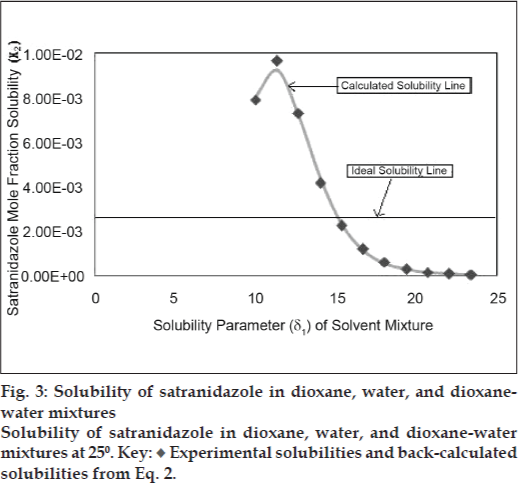
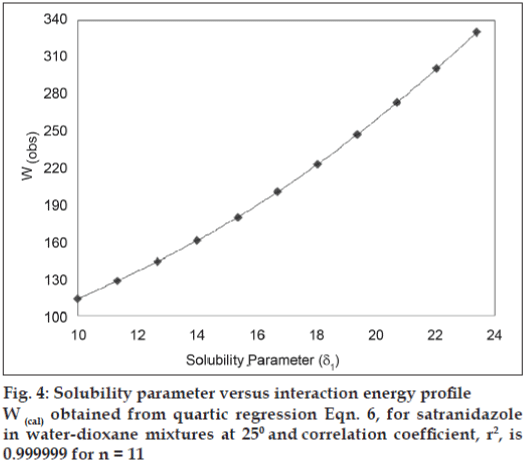
Experimental data of mole fraction solubility of satranidazole in dioxane-water are plotted against solubility parameters of solvent blend (fig. 3) exhibit a maxima at δ1= 11.34 (peak solubility = 0.0096347 mol/l). The observed solubility is comparatively higher than the ideal solubility (X2i= 0.00245614 mol/l). According to the regular solution theory, solubility cannot exceed ideal solubility. However, in non regular solutions, peak solubility may depart from ideal solubility due to solute solvent interactions. This abnormal behavior has been dealt with the theoretical replacement of mean geometric solubility parameters (δ1δ2) term with the interaction energy term (W). To relate these two variables, a fourth power polynomial (quartic expression) has been developed to back calculate the value of Wcal. For Dioxane-water system, the polynomial has following values: Wcal= 21.70104+10.372731 δ1-0.401841 δ12 +0.032507δ13 -0.000435δ14, (n=11, R2= 0.9999) -- (6)
These polynomials are used successfully for the calculation of W, at any value of solubility parameter (δ1), which was subsequently employed to calculate mole fraction solubility of solute in a solvent blend using backward regression. Representative data along with validation parameters are summarized in Table 1. Wcal values are indicating significant interaction of satranidazole and solvent molecules at the peak of solubility profile.
| Water: Dioxane(%v/v) | Solubility(g/ml) | δ1(Cal/cm3)0.5 | V1 | Density of blend | Mol. Wt of blend | X2(obs) | W(obs) |
|---|---|---|---|---|---|---|---|
| 100:0 | 0.0005821 | 23.40 | 18.00 | 0.9980 | 18.00 | 3.6317E-05 | 330.45 |
| 90:10 | 0.0009112 | 22.06 | 24.77 | 1.0014 | 25.01 | 7.8740E-05 | 300.96 |
| 80:20 | 0.0012872 | 20.72 | 31.54 | 1.0048 | 32.02 | 1.4198E-04 | 273.03 |
| 70:30 | 0.0020104 | 19.38 | 38.31 | 1.0082 | 39.03 | 2.6954E-04 | 246.97 |
| 60:40 | 0.0039050 | 18.04 | 45.08 | 1.0116 | 46.04 | 6.1647E-04 | 222.93 |
| 50:50 | 0.0064722 | 16.70 | 51.85 | 1.0150 | 53.06 | 1.1757E-03 | 200.45 |
| 40:60 | 0.0109196 | 15.36 | 58.62 | 1.0184 | 60.07 | 2.2456E-03 | 179.77 |
| 30:70 | 0.0180788 | 14.02 | 65.39 | 1.0218 | 67.08 | 4.1594E-03 | 160.85 |
| 20:80 | 0.0286006 | 12.68 | 72.16 | 1.0252 | 74.09 | 7.2968E-03 | 143.66 |
| 10:90 | 0.0344943 | 11.34 | 78.93 | 1.0286 | 81.10 | 9.6347E-03 | 127.92 |
| 0:100 | 0.0262503 | 10.00 | 85.70 | 1.0320 | 88.11 | 7.8876E-03 | 113.38 |
δ1= Solubility parameter of solvent blend, V1= Molar volume of the solvent blend
Table 1: Molar observed solubility and calculation parameters of satranidazole in dioxanewater mixtures
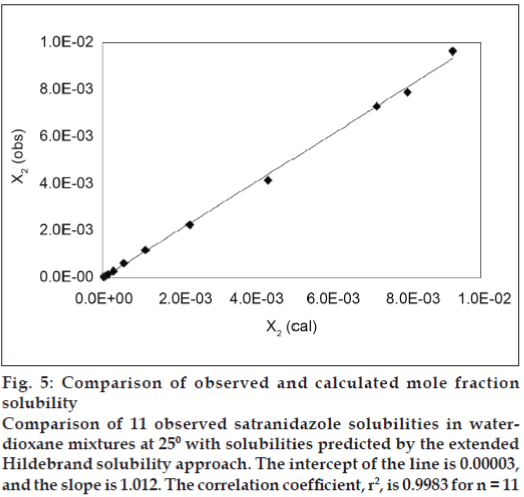
Observed solubility data was subjected to the evaluation of interaction energy. Experimental values of interaction energy (Wobs) were regressed against solubility parameter to obtain Wcal (fig. 4), which was then used to back calculate the mole fraction solubility (X2cal). A mathematical model is proposed for individual system as fourth power polynomial. Validation of this equation has been done by comparing experimentally obtained and calculated values of mole fraction solubility by estimating residuals and percent difference (Table 2). The Extended Hildebrand Approach applied to the solubility data of satranidazole in waterdioxane mixtures, which reproduces the satranidazole solubility within the accuracy ordinarily achieved in such measurements. The predictive capability of the model for satranidazole is represented in fig.5, which indicates a very high degree of correlation coefficient (R2) 0.9983 and negligible intercept (-0.00003) equal to zero.
Figure 5: Comparison of observed and calculated mole fraction solubility Comparison of 11 observed satranidazole solubilities in waterdioxane mixtures at 250 with solubilities predicted by the extended Hildebrand solubility approach. The intercept of the line is 0.00003, and the slope is 1.012. The correlation coefficient, r2, is 0.9983 for n = 11
| W(obs) | W(cal) | X2(obs) | X2(cal) | logγ2/A(obs) | logγ2/A(cal) | Residual | Percentdifference |
|---|---|---|---|---|---|---|---|
| 330.447760 | 330.457908 | 3.6317E-05 | 3.6612E-05 | 16.396580 | 16.376285 | -8.0988E-03 | -8.10E-01 |
| 300.959157 | 300.911658 | 7.8740E-05 | 7.5824E-05 | 14.457387 | 14.552385 | 3.7032E-02 | 3.70E+00 |
| 273.034580 | 273.083225 | 1.4198E-04 | 1.4757E-04 | 12.981340 | 12.884050 | -3.9377E-02 | -3.94E+00 |
| 246.968191 | 247.040724 | 2.6954E-04 | 2.8550E-04 | 11.380118 | 11.235053 | -5.9205E-02 | -5.92E+00 |
| 222.925499 | 222.818600 | 6.1647E-04 | 5.6651E-04 | 9.322702 | 9.536501 | 8.1035E-02 | 8.10E+00 |
| 200.450067 | 200.417633 | 1.1757E-03 | 1.1460E-03 | 7.721966 | 7.786834 | 2.5208E-02 | 2.52E+00 |
| 179.769542 | 179.804938 | 2.2456E-03 | 2.3086E-03 | 6.122616 | 6.051824 | -2.8046E-02 | -2.80E+00 |
| 160.846625 | 160.913961 | 4.1594E-03 | 4.3814E-03 | 4.599250 | 4.464578 | -5.3374E-02 | -5.34E+00 |
| 143.657648 | 143.644482 | 7.2968E-03 | 7.2243E-03 | 3.199204 | 3.225536 | 9.9401E-03 | 9.94E-01 |
| 127.918366 | 127.862615 | 9.6347E-03 | 9.2394E-03 | 2.490968 | 2.602470 | 4.1024E-02 | 4.10E+00 |
| 113.375305 | 113.400807 | 7.8876E-03 | 8.0423E-03 | 2.981490 | 2.930486 | -1.9622E-02 | -1.96E+00 |
Table 2: Comparisons of observed and calculated mole fraction solubilities of satranidazole in dioxane-water mixtures at 25°
On the basis of validation parameters, it can be expressed that the behavior of non regular solution can be quantified more precisely using EHSA. The procedure can be explored further to predict the solubility of satranidazole in pure water or dioxane and in any water-dioxane mixtures. Simultaneously, this tool may become useful in optimization problems of clear solution formulations. Thus the method has potential usefulness in preformulation and formulation studies during which solubility prediction is important for drug design.
Acknowledgements
Authors wish to express their gratitude to M/S Alkem Laboratories Limited, Baddi for providing gift sample of Satranidazole. Authors are also thankful to E. Merck, Ltd.; Mumbai, India and Qualigens Fine Chemicals, Mumbai, India for providing gift sample of 1,4-Dioxane and Acetone respectively.
References
- Martin A, Newburger J, Adjei A. Extended Hildebrand Solubility Approach: Solubility of Theophylline in Polar Binary Solvents. J Pharm Sci 1980;69:487-91.
- Martin A, WU PL. Extended Hildebrand Solubility Approach: Hydroxybenzoic Acid in Mixtures of Dioxane and Water. J Pharm Sci 1980;72:587-92.
- Thimmasetty J, Subramanyam CVS, Vishwanath BA, SateshSabu PR. Solubility Parameter Estimation of Celecoxib by Current Methods. Asian J Res Chem 2009;2:188-95.
- Subramanyam CVS, Sreenivasa RM, Venkata RJ, Gundu RP. Irregular solution behaviour of paracetamol in binary solvents. Int J Pharm 1992;78:17-24.
- SatheshBabu PR, Subrahmanyam CVS, Thimmasetty J, Manavalan R, Valliappan K. Extended Hansen’s Solubility Approach: Meloxicam in Individual Solvents. Pak J Pharm Sci 2007;20:311-6.
- Martin A, Carstensen J. Extended solubility approach: Solubility parameters for crystalline compounds. J Pharm Sci 1981;70:170-2.
- Martin A, WU PL, Velasquez T. Extended Hildebrand Solubility Approach: Sulfonamides in Binary and Ternary Solvents. J Pharm Sci 1985;74:277-82.
- Martin A, Miralles MJ. Extended Hildebrand Solubility Approach: Solubility of tolbutamide, acetohexamide and sulfisomidine in binary solvent mixtures. J Pharm Sci 2006;71:439-42.
- Mourya VK, Yadav SK, Saini TR. Solubility Studies of Metronidazole in Binary Solvent Blends. Indian J Pharm Sci 1997;59:200-2.
- Martin A, WU PL, Beerbower A. Expanded Solubility Parameter Approach I: Napthalene and Benzoic Acid in Individual Solvents. J Pharm Sci 1984;73:179-88.
- Herrador MA, Gonzalez AG. Solubility prediction of caffeine in aqueous N, N-dimethyl formamide mixtures using the Extended Hildebrand Solubility Approach. Int J Pharm 1997;156:239-44.
- Shinde SR, Bhoir SI, Pawar NS, Yadav SB, Bhagwat AM. Simultaneous Estimation of Satranidazole and Ofloxacin in Tablet Dosage Form by High Performance Liquid Chromatography. E-J Chem 2010;7:198-202.
- Derle D, Magar M. Studies on the Preparation, Characterization and Solubility of β-Cyclodextrin- Satranidazole Inclusion Complexes. Indian J Pharm Educ Res 2006;40:232-6.
- Burange P, Arote S, Damle M, Godge R. A Validated RP-HPLC Method for Simultaneous Estimation of Ofloxacin and Satranidazole from Tablets. J Pharm Sci 2008;7:70-2.
- Wankhede SB, Nanda RK, Prakash A, Chitlange SS. Simultaneous Spectrophotometric Estimation of Ofloxacin and Satranidazole in Tablet Dosage Form. J Pharm Sci 2008;7:92-4.
- Derle D, Magar M. Studies on the Preparation, Characterization and Solubility of β-Cyclodextrin- Satranidazole Inclusion Complexes. Indian J Pharm Educ Res 2006;40:232-6.
- Spiegel AJ, Noseworthy MM. Use of non-aqueous solvents in parenteral products. J Pharm Sci 1963;52:917-27.
- Patrick JS. Martin’s Physical Pharmacy and Pharmaceutical Sciences, 5th ed. Philadelphia: Lippincott Williams and Wilkins; 2006. p. 245-6, 595-6.
- Barton AF. CRC Handbook of Solubility Parameters and other Cohesion Parameters, 2nd ed. New York: CRC Press; 1983. p. 7-59, 157-85.
- Greenhalgh DJ, Williams AC, Timmins P, York P. Solubility Parameters as Predictors of Miscibility in Solid Dispersions. J Pharm Sci 1999;88:1182-90.