- *Corresponding Author:
- M. Nagoor Meeran
Department of Chemistry, Vivekanandha College of Arts and Sciences for Women
Tiruchengode-637 303
E-mail: nagoorjmc@rediffmail.com
| Date of Submission | 06 October 2016 |
| Date of Revision | 19 January 2017 |
| Date of Acceptance | 05 June 2017 |
| Indian J Pharm Sci 2017;79(4):641-645 |
Abstract
Isatin is synthetically a versatile substrate that can be used for the synthesis of large variety of heterocyclic compounds. 5-substituted isatins were treated with 2-amino-5-chlorobenzo phenone to form Schiff’s bases. Spirothiozolidin-4-ones and 5’-methyl-spiro-4-thiazolidiones were synthesized Schiff’s bases of isatins by treating with thioglycolic acid and thiolactic acid, respectively in the presence of 1,4-dioxane and anhydrous zinc chloride. The structures of the synthesized compounds were characterized by elemental and spectral analysis. These compounds were screened for their antioxidant activity by 2,2-diphenyl-1-picrylhydrazyl method. Compound A02 found to possess potent antioxidant activity compared to the standards ascorbic acid and butylated hydroxytoluene.
Keywords
5-substituted isatins, 2-amino-5-chlorobenzophenone, antioxidant activity
Heterocyclic chemistry is of great importance to the medicinal chemists because of their utility in medicine. Large numbers of heterocyclic compounds are being used as therapeutic agents. Isatin (1-H-indole-2,3- dione) and its derivatives possess diverse biological and pharmacological activities and are widely used as a starting material for the synthesis of a broad range of heterocyclic compound substrates for drug synthesis [1]. Isatin is a heterocyclic compound and derivatives of isatin possess biological activities such as antimicrobial [2], antibacterial, antifungal [3], antiviral [4], antiHIV [5], anticancer [6,7], antiproliferative [8], antioxidant activity [9]. Spirocyclic systems containing one common carbon atom to two rings are structurally interesting [10] and naturally occurring substances being known for their wide range of biological activities [11,12]. 4-thiazolidinone also possesses various important biological activities such as anticancer, antiviral, anticonvulsant, cardiovascular, antiinflammatory, analgesic activity, antidiabetic, antimicrobial, antihyperlipidemic, antituberculosis, antiparasitic, antiarthritic, antidiarrhoeal and antioxidant activities [13]. Free radicals can oxidize biomolecules viz. nucleic acids, proteins, lipids, DNA, lead to tissue damage and can initiate degenerative diseases. Oxidative damage plays a significantly pathological role in human diseases such as cancer, emphysema, cirrhosis, atherosclerosis and arthritis [14,15]. Almost all organisms are protected to some extent against free radicals such as peroxide, hydroperoxide and lipid peroxyl damage by enzymes such as superoxide dismutase and catalase or compounds such as ascorbic acid (AA), tocopherols, phenolic acids, polyphenols, flavonoids and glutathione [16]. However, antioxidant supplements or dietary antioxidants may be sources of protection that the body needs to protect against the damaging effects of free radicals [17]. Presently synthetic antioxidants are widely used because they are effective and cheaper than natural antioxidants. By observing the importance of the above said problem, the isatin-based Schiff base and spiro isatin derivatives of spirothiozolidin- 4-ones and 5'-methyl-spiro-4-thiazolidiones from 5-substituted isatin and 2-benzoyl-4-chloroaniline were synthesized following the steps given in Figures 1 and 2. The purity of the compounds was monitored by thin layer chromatography (TLC) and the structures of the products were confirmed by elemental and spectral analysis. All the newly synthesized compounds were screened for their antioxidant activity by 2,2-diphenyl- 1-picrylhydrazyl (DPPH) free radical scavenging assay method.
All the chemicals used in this study were purchased from Sigma-Aldrich Chemicals and used without further purification. The compounds (A01-E03) were synthesized by reported method [18]. The melting points were determined by open capillary tube method and are uncorrected. The purity of compounds was confirmed by TLC using silica gel coated glass plates as the stationary phase and with suitable mobile phase.
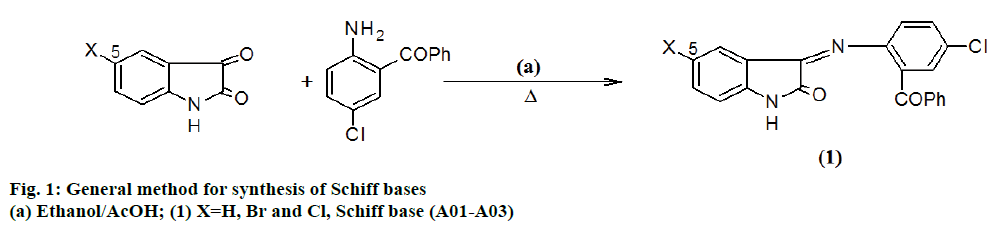
Schiff bases (A01-A03) were synthesized by following method. An equimolar (0.01 mol) mixture of 2-amino- 5-chlorobenzophenone and 5-substituted indole-2,3- dione was dissolved in ethanol (20 ml) with few drops of acetic acid. It is reacted under refluxing conditions. After the completion of reaction the content was cooled and kept overnight. The separated product was filtered and recrystallized using ethanol.
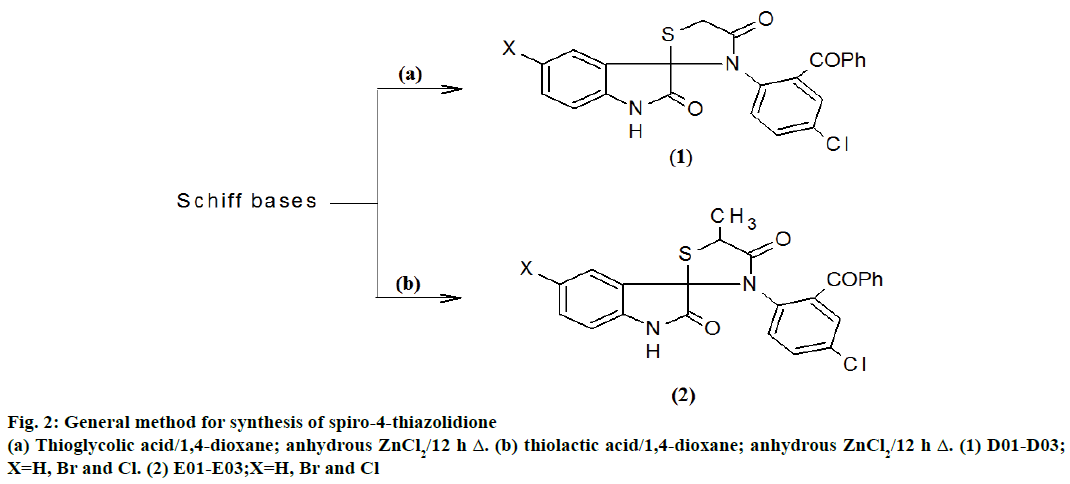
5-substituted indol-2,3-dione based spiro-4- thiazolidiones (D01-D03) were synthesized by following method. A mixture of Schiff bases (A01) (0.01 mol) and thioglycolic acid (0.01 mol) was refluxed with 1,4-dioxane for 12 h in the presence of zinc chloride. The completion of reaction was monitored by TLC. After completion, reaction mixture was poured in ice cold water. The product formed was isolated, washed with water and recrystallized from ethanol to give compound D01. Similarly other compounds of D02 and D03 are synthesized from A02 and A03 by the same procedure.
5-substituted indol-2,3-dione based 5’-methyl-spiro- 4-thiazolidiones (E01-E03) were synthesized by following process. A mixture of Schiff bases (A01) (0.01 mol) and thiolactic acid (0.01 mol) was refluxed with 1,4-dioxane for 12 h in the presence of zinc chloride. The completion of reaction was monitored by TLC. After completion, reaction mixture was poured in ice cold water. The product formed was isolated washed with water and recrystallized from ethanol to give compound E01. Similarly, other compounds of E02 and E03 were synthesized from A02 and A03 by the same procedure.
All the synthesized compounds were screened for antioxidant activity using the DPPH assay method [19]. Antioxidant data of all the test samples was summarized in Table 1. The DPPH assay method depended on the reduction of purple DPPH to a yellow colored diphenyl picrylhydrazine and the remaining DPPH, which showed maximum absorption at 517 nm was measured. About 2 ml of various concentrations of the synthesized compounds or standards were added to 2 ml of DPPH solution (0.1 mM, 2 ml). After 20 min of incubation at 37° in the dark, the absorbance was recorded at 517 nm. AA and BHA (3-t-butyl-4-hydroxy anisole) was used as positive controls. The formula used for percent inhibition is as follows; %inhibition=(blank OD– sample OD/blank OD)×100.
| Compound | Concentration (µg/ml) | IC50 | ||||
|---|---|---|---|---|---|---|
| 20 | 40 | 60 | 80 | 100 | ||
| A01 | 30.18 | 49.28 | 61.8 | 76.4 | 87.4 | 44.46 |
| A02 | 36.4 | 51.3 | 65.8 | 81.2 | 93.4 | 38.31 |
| A03 | 12.8 | 26.4 | 41.2 | 58.8 | 70.8 | 70.78 |
| D01 | 22.4 | 38.6 | 52.4 | 65.1 | 76.4 | 58.58 |
| D02 | 12.2 | 26.4 | 48.2 | 65.1 | 79.4 | 64.3 |
| D03 | 18.2 | 28.1 | 54.2 | 72.4 | 89.9 | 57.3 |
| E01 | 26.5 | 54.1 | 69.7 | 74.8 | 91.2 | 42.36 |
| E02 | 9.8 | 17.4 | 63.8 | 78.4 | 89.7 | 58.35 |
| E03 | 19.4 | 37.1 | 56.2 | 71.7 | 83.4 | 55.62 |
| BHT | 54.6 | 64.2 | 72.4 | 81.9 | 94.2 | 11.59 |
| AA | 58.1 | 64.8 | 75.1 | 87.7 | 98.2 | 8.06 |
Table 1: Antioxidant activity of standard and synthesized compounds
Schiff bases were obtained by Figure 1 and 4-thiazolidiones derivatives were obtained by Figure 2. The required starting material Schiff base (A01-A03) were synthesized from 5-substituted indole-2,3-dione, which was further on treatment with thioglycolic acid and thiolactic acid yielded the spirothiozolidin-4-ones (D01-D03) and 5'-methyl-spiro-4-thiazolidiones (E01-E03).
3- [(2-benzoyl-4-chlorophenyl)imino]-1,3-dihydro- 2H-indol-2-one (A01), m.p.: 212°; IR (KBr) λmax in cm-1: 3418 (N-H str), 3190 (Ar C-H str), 1728 (Ar C=O str), 1612 (HN-C=O str), 1581 (C=N), 1535 (Ar C=C str), 1324 (C-N); 1H-NMR (DMSO-d6, 400 MHz) δ: 11.05 (s, 1H) 7.61 - 7.59 (d, J=7.6, 1H), 7.58 - 7.54 (t, J=8.4, 1H), 7.51-7.39 (m, 5H), 7.34-7.31 (dd, J=9.2, 2.4, 1H) 7.22-7.20 (d, J=7.6, 1H), 7.19- 7.18 (d, J=2.4, 1H), 7.08-7.05 (t, J=7.2, 1H), 6.93-6.90 (d, J=2.4, 1H); 13C-NMR (DMSO-d6, 100 MHz) δ: 196.8 (C=O), 184.3 (H-N-C=O), 159.3 (C=N), 146.9 (C8), 138.4 (C7'), 137.6 (C15), 132.4 (C18), 131.08 (C11), 130.2 (C9), 129.2 (C10), 129.0 (C16 and C20), 128.8 (C17 and C19), 128.6 (C12), 124.9 (C13), 124.2 (C4), 121.2 (C5), 119 (C6), 117.5 (C3'), 111.6 (C7); MS (EI): m/z 360 [M+]; Anal. Found: C, 69.64; H, 3.69; N, 7.80; Cl, 9.80 (%). Calc. for (C21H13ClN2O2): C, 69.84; H, 3.60; N, 7.76; Cl, 9.83.
3- [(2-benzoyl-4-chlorophenyl)imino]-5-bromo-1,3- dihydro-2H-indol-2-one (A02), m.p.: 220°; IR (KBr) λmax in cm-1: 3417 (N-H str), 3101 (Ar C-H str), 1705 (Ar C=O str), 1612 (N-H-C=O str), 1584 (C=N), 1535 (Ar C=C str), 1356 (C-N); 1H-NMR (DMSO-d6, 400 MHz) δ: 11.13 (s, 1H) 7.74-7.71 (dd, J=8.4 Hz, 2.4 Hz, 1H), 7.65-7.64 (d, J=7.6 Hz, 1H), 7.48-7.39 (m, 5H), 7.33-7.30 (dd, J=8.8 Hz, 2.4 Hz, 1H) 7.17 (d, J=2.4 Hz, 1H), 6.91-6.89 (d, J=8.4 Hz, 1H), 6.87-6.85 (d, J=8.4 Hz, 1H); 13C-NMR (DMSO-d6, 100 MHz) δ: 196.7 (C=O), 183.1 (H-N-C=O), 158.9 (C=N), 146.9 (C8), 145.8 (C7'), 137.6 (C15), 132.4 (C18), 131.08 (C6), 131.03 (C12), 130.2 (C9), 129.2 (C10), 129.0 (C16 and C20), 128.8 (C17 and C19), 128.5 (C11), 127.8 (C4), 124.9 (C13), 119.6 (C3'), 117.6 (C5), 114.7 (C7); MS (EI): m/z 439 [M+]; Anal. Found: C, 57.34; H, 2.76; N, 6.32; Cl, 8.02; Br, 18.31 (%). Calc. for (C21H12BrClN2O2): C, 57.31; H, 2.72; N, 6.36; Cl, 8.06; Br, 18.17.
3- [(2-benzoyl-4-chlorophenyl)imino]-5-chloro-1,3- dihydro-2H-indol-2-one (A03), m.p.: 219°; IR (KBr) λmax in cm-1: 3419 (N-H str), 3186 (Ar C-H str), 1735 (Ar C=O str), 1612 (N-H-C=O str) 1592 (C=N), 1534 (Ar C=C str), 1356 (C-N); 1H-NMR (DMSO-d6, 400 MHz) δ: 11.39 (s, 1H) 7.75-7.23 (dd, J=8.8 Hz, 2.4 Hz, 1H), 7.67-7.66 (d, J=2.4 Hz, 1H), 7.54-7.39 (m, 5H), 7.34-7.31 (dd, J=8.8 Hz, 2.4 Hz, 1H) 7.20 (d, J=2.4 Hz, 1H), 6.94-6.92 (d, J=8.8 Hz, 1H), 6.73-6.71 (d, J=8.8 Hz, 1H); 13C-NMR (DMSO-d6, 100 MHz) δ: 196.7 (C=O), 183.3 (HN-C=O), 159.1 (C=N), 146.9 (C8), 144.8 (C7'), 137.6 (C15), 132.4 (C18), 131.0 (C12), 130.2 (C9), 130.0 (C6), 129.2 (C10), 129.0 (C16 and C20), 128.8 (C17 and C19), 128.6 (C11), 126.3 (C5), 124.9 (C13), 124.4 (C4), 119.4 (C3'), 114.4 (C7); MS (EI): m/z 395 [M+]; Anal. Found: C, 63.71; H, 3.05; N, 7.10; Cl, 17.98 (%). Calc. for (C21H12Cl2N2O2): C, 63.75; H, 3.03; N, 7.08; Cl, 17.94.
3 ' - ( 2 - b e n z o y l - 4 - c h l o r o p h e n y l ) - 4 ' H - spiro [indole-3,2'- [1,3]thiazolidine]-2,4'(1H)-dione (D01), m.p.: 203°; IR (KBr) λmax in cm-1: 3417 (N-H str), 3062 (Ar C-H str), 2924 (Ali C-H Str), 1705 (C=O str), 1680 (thio C=O), 1612 (HN-C=O str), 736 (C-S str); 1H-NMR (DMSO-d6, 400 MHz) δ: 11.57 (s, 1H), 7.74 - 7.70 (t, J=7.2 Hz, 1H), 7.64-7.63 (d, J=7.6 Hz, 1H), 7.51-7.40 (m, 5H), 7.33-7.30 (dd, J=8.8 Hz, 2.4 Hz, 1H), 7.28-7.26 (d, J=7.6 Hz, 1H), 7.19-7.18 (d, J=2.4 Hz, 1H), 7.08-7.05 (t, J=7.6 Hz, 1H), 6.92-6.90 (d, J=9.2 Hz, 1H), 3.86 (s, 2H); 13C-NMR (DMSO-d6, 100 MHz) δ: 196.8 (C=O), 184.2 (HNC= O), 174.0 (-S-HN-C=O), 141.6 (C7'), 137.9 (C8), 136.6 (C15), 132.4 (C18), 131.6 (C12), 130.3 (C10), 129.5 (C16 and C20), 129.3 (C6), 129.2 (C11), 129.0 (C9), 128.8 (C17 and C19), 125.5 (C5), 125.4 (C4), 124.8 (C3'), 122.9 (C13), 110.4 (C7), 77.7 (Spiro-C3), 35.75 (CH2); MS (EI): m/z 434 [M+]; Anal. Found: C, 63.52; H, 3.48; N, 6.44; Cl, 8.12; S, 7.36 (%). Calc. for (C23H15ClN2O3S): C, 63.46; H, 3.44; N, 6.43; Cl, 8.15; S, 7.37 (%).
3 ' - (2-benzoyl-4-chlorophenyl) -5-bromo- 4 ' H-spiro [indole-3,2'- [1,3]thiazolidine]-2,4'(1H) dione (D02), m.p.: 201°; IR (KBr) λmax in cm-1: 3417 (N-H Str), 3062 (Ar C-H Str), 2962 (Ali C-H Str), 1697 (C=O str), 1671 (thio C=O), 1606 (HN-C=O str), 737 (C-S str); 1H-NMR (DMSO-d6, 400 MHz) δ: 11.67 (s, 1H), 7.63-7.60 (dd, J=8.4 Hz, 2.4 Hz, 1H), 7.59 (d, J=2.4 Hz, 1H), 7.44-7.35 (m, 5H), 7.34-7.31 (dd, J= 8.8 Hz, 2.4 Hz, 1H), 7.19-7.18 (d, J=2.4 Hz, 1H), 6.99-6.97 (d, J=8.8 Hz, 1H), 6.93-6.90 (d, J=8.8 Hz, 1H), 3.69 (s, 2H); 13C-NMR (DMSO-d6, 100 MHz) δ: 196.7 (C=O), 184.0 (HN-C=O), 174.1 (-S-HN-C=O), 140.5 (C7’), 137.9 (C8), 136.3 (C15), 132.4 (C18), 131.6 (C12), 130.3 (C10), 129.3 (C16 and C20), 131.2 (C6), 129.2 (C11), 129.0 (C9), 128.8 (C17 and C19), 125.3 (C3’), 123.6 (C4), 122.9 (C13), 116.9 (C5), 111.4 (C7), 77.7 (Spiro-C3), 34.7 (CH2); MS (EI): m/z 513 [M+]; Anal. Found: C, 53.77; H, 2.75; N, 5.45; Cl, 6.92; Br, 15.71; S, 6.24 (%). Calc. for (C23H14ClBrN2O3S): C, 53.71; H, 2.72; N, 5.44; Cl, 6.90; Br, 15.55, S, 6.23 (%).
3'-(2-benzoyl-4-chlorophenyl)-5-chloro-4' H-spiro [indole-3,2'- [1,3]thiazolidine]-2,4'(1H)- dione (D03), m.p.: 218°; IR (KBr) λmax in cm-1: 3201 (N-H str), 3062 (Ar C-H str), 2962 (Ali C-H str), 1696 (C=O str), 1671 (thio C=O), 1607 (HN-C=O str), 733 (C-S str); 1H-NMR (DMSO-d6, 400 MHz) δ: 11.07 (s, 1H), 7.76 (d, J=2.4 Hz, 1H), 7.71-7.69(dd, J=8.8 Hz, 2.4 Hz, 1H), 7.48-7.36 (m, 5H), 7.32-7.30 (dd, J=8.8 Hz, 2.4 Hz, 1H), 7.19-7.18 (d, J=2.4 Hz, 1H), 7.0 (d, J=8.8 Hz, 1H), 6.86-6.84 (d, J=8.8 Hz, 1H), 3.76 (s, 2H); 13C-NMR (DMSO-d6, 100 MHz) δ: 196.5 (C=O), 184.1 (HN-C=O), 174.0 (-S-HN-C=O), 141.8 (C7'), 137.9 (C8), 136.3 (C15), 132.4 (C18), 131.6 (C12), 130.3 (C10), 129.1 (C16 and C20), 127.8 (C6), 129.2 (C11), 129.02 (C9), 129.05 (C17 and C19), 124.8 (C3’), 123.09 (C4), 129.9 (C13), 129.01 (C5), 111.9 (C7), 78.07 (Spiro-C3), 35.8 (CH2); MS (EI): m/z 469 [M+]; Anal. Found: C, 58.86; H, 2.91; N, 5.97; Cl, 15.10; S, 6.83 (%). Calc. for (C23H14Cl2N2O3S): C, 58.80; H, 2.98; N, 5.96; Cl, 15.11; S, 6.83 (%).
3 ' - [ 4 - c h l o r o - 2 - ( p h e n y l c a r b o n y l ) p h e n y l ] - 5'-methyl-4'H-spiro [indole-3,2'- [1,3]thiazolidine]-2, 4'(1H)-dione (E01), m.p.: 234°; IR (KBr) λmax in cm-1: 3209 (N-H str), 3055 (Ar C-H Str), 2924, 2877 (Ali C-H Str), 1705 (C=O str), 1672 (thio C=O), 1604 (HN-C=O str), 728 (C-S str); 1H-NMR (DMSO-d6, 400 MHz) δ: 11.56 (s, 1H), 7.73-7.69 (t, J=8.4 Hz, 1H), 7.64-7.62 (d, J=7.6 Hz, 1H), 7.52-7.37 (m, 5H), 7.33-7.30 (dd, J= 8.8 Hz, 2.4 Hz, 1H), 7.29-7.25 (d, J=7.6 Hz, 1H), 7.18-7.17 (d, J=2.4 Hz, 1H), 7.07-7.03 (t, J=7.6 Hz, 1H), 6.93-6.91 (d, J=8.8 Hz, 1H), 3.87-3.81 (q, J=7.2 Hz, 1H), 1.38-1.37 (d, J=7.2 Hz, 3H); 13C-NMR (DMSO-d6, 100 MHz) δ: 196.7 (C=O), 184.2 (HN-C=O), 178.3 (-S-HN-C=O), 140.4 (C7’), 137.8 (C8), 136.3 (C15), 132.4 (C18), 131.6 (C12), 131.2 (C6), 130.2 (C10), 129.0 (C9), 129.5 (C11), 129.4 (C16 and C20), 128.8 (C17 and C19), 124.3 (C3’), 123.7 (C4), 123.0 (C13), 117.0 (C5), 111.4 (C7), 80.2 (Spiro-C3), 49.1 (CH), 19.2 (CH3); MS (EI): m/z 448 [M+]; Anal. Found: C, 64.21; H, 3.82; N, 6.24; Cl, 7.92; S, 7.14 (%). Calc. for (C24H17ClN2O3S): C, 64.15; H, 3.78; N, 6.23; Cl, 7.90; S, 7.14 (%).
5-bromo-3'- [4-chloro-2-(phenylcarbonyl)phenyl]- 5'-methyl-4'H-spiro [indole-3,2'- [1,3] thiazolidine]- 2,4'(1H)-dione (E02), m.p.: 225°; IR (KBr) λmax in cm-1: 3417 (N-H Str), 3062 (Ar C-H Str), 2924, 2877 (Ali C-H Str), 1705 (C=O str), 1661 (thio C=O), 1612(N-C=O str), 740 (C-S str); 1H-NMR (DMSO-d6, 400 MHz) δ: 11.59 (s, 1H), 7.69 - 7.66 (dd, J=8.4 Hz, 2.4 Hz,1H), 7.64-7.63 (d, J=2.4 Hz, 1H), 7.49-7.37 (m, 5H), 7.35-7.32 (dd, J= 8.8 Hz, 2.4 Hz, 1H), 7.18- 7.17 (d, J=2.4 Hz, 1H), 6.93-6.91 (d, J=8.8 Hz, 1H), 3.61-3.55 (q, J=7.2 Hz, 1H), 1.35-1.33 (d, J=7.6 Hz, 3H); 13C-NMR (DMSO-d6, 100 MHz) δ: 196.7 (C=O), 184.2 (HN-C=O), 178.3 (-S-HN-C=O), 140.4 (C7’), 137.8 (C8), 136.3 (C15), 132.4 (C18), 131.6 (C12), 131.2 (C6), 130.2 (C10), 129.0 (C9), 129.5 (C11), 129.4 (C16 and C20), 128.8 (C17 and C19), 124.3 (C3’), 123.7 (C4), 123.0 (C13), 117.0 (C5), 111.4 (C7), 80.2 (Spiro-C3), 49.1 (CH), 19.2 (CH3); MS (EI): m/z 527 [M+]; Anal. Found: C, 54.61; H, 3.06; N, 5.31; Cl, 6.76; Br, 15.29; S, 6.07 (%). Calc. for (C24H16ClBrN2O3S): C, 54.56; H, 3.03; N, 5.30; Cl, 6.76; Br, 15.18; S, 6.07 (%).
5-chloro-3'- [4-chloro-2-(phenylcarbonyl)phenyl]- 5'-methyl-4'H-spiro [indole-3,2'- [1,3]thiazolidine]- 2,4'(1H)-dione (E03), m.p.: 212°; IR (KBr) λmax in cm-1: 3417 (N-H Str), 3062 (Ar C-H Str), 2924, 2877 (Ali C-H Str), 1705 (C=O str), 1664 (thio C=O), 1612 (HN-C=O str), 731 (C-S str); 1H-NMR (DMSO-d6, 400 MHz) δ: 11.05 (s, 1H), 7.64-7.61 (dd, J=8.4 Hz, 2.4 Hz,1H), 7.59-7.52 (d, J=2.4 Hz, 1H), 7.42-7.35 (m, 5H), 7.32-7.29 (dd, J= 8.8 Hz, 2.4 Hz, 1H), 7.19-7.18 (d, J=2.4 Hz, 1H), 6.91-6.89 (d, J=8.8 Hz, 1H), 6.84-6.82 (d, J=8.8 Hz, 1H), 3.49-3.44 (q, J=6.8 Hz, 1H), 1.32-1.31 (d, J=7.6 Hz, 3H); 13C-NMR (DMSO-d6, 100 MHz) δ: 196.82 (C=O), 184.17 (HN-C=O), 178.15 (-S-HN-C=O), 141.82 (C7’), 137.88 (C8), 136.36 (C15), 132.42 (C18), 131.61 (C12), 130.23 (C10), 129.54 (C11), 129.42 (C16 and C20), 129.37 (C5), 129.02 (C9), 128.84 (C17 and C19), 127.84 (C6), 123.99 (C3’), 123.04 (C13), 122.85 (C4), 111.87 (C7), 79.87 (Spiro-C3), 49.18 (CH), 18.93 (CH3); MS (EI): m/z 483 [M+]; Anal. Found: C, 59.64; H, 3.34; N, 5.80; Cl, 14.67 S, 6.63 (%). Calc. for (C24H16Cl2N2O3S): C, 59.58; H, 3.31; N, 5.79; Cl, 14.65; S, 6.63 (%).
The results of free radical scavenging activity of methanol solutions of compounds at different concentrations are shown in Table 1. It is evident from these results that free radical scavenging activity of these compounds was concentration-dependent. The IC50 values calculated (Table 1) indicated that none of the synthesized compounds were as active as the two standards AA and BHA as evidenced by the fact that the most active compound among the synthesized compounds is A02, which gave an IC50 value of 38.3 μg/ml, while AA and BHA gave 8.08 and 11.59 μg/ml, respectively. Finally it can be concluded that new isatin derivatives of Schiff bases, spiro-4- thiazolidiones, 5’-methyl-spiro-4-thiazolidiones have been successfully synthesized and evaluated for their antioxidant activity. Maximum free radical scavenging activity was found in compound A02.
Acknowledgement
The authors are grateful to SIF, VIT University, Vellore, Tamil Nadu, India for carrying out the NMR spectral analysis.
Financial assistance
None.
Conflict of interests
None declared.
References
- De Silva JFM, Garden SJ, Pinto AC. The chemistry of isatins: a review from 1975 to 1999. J Braz Chem Soc 2001;12:273-324.
- Panwar H, Verma RS, Srivastave VK, Kumar A. Synthesis of some substituted azetidinonyl and thiazolidinonyl-1,3,4-thiadiazino[6,5-b]indoles as prospective antimicrobial agents. Indian J Chem 2006;45B:2099-104.
- Jarrahpour A, Khalili D, De Clercq E, Salmi C, Brunel JM. Synthesis, antibacterial, antifungal and antiviral activity evaluation of some new bis-Schiff bases of isatin and their derivatives. Molecules 2007;12:1720-30.
- Zhang HM, Dai H, Hanson PJ, Li H, Guo H, Ye X, et al. Antiviral activity of an isatin derivative via induction of PERK-Nrf2-mediated suppression of cap-independent translation. ACS Chem Biol 2014;9(4):1015-24.
- Selvam P, Murugesh N, Chandramohan M, Debyser Z, Witvrouw M. Design, synthesis and anti-HIV activity of novel isatine-sulphonamides. Indian J Pharm Sci 2008;70(6):779-82.
- Prasad RK, Narsinghani T, Sharma R. QSAR analysis of novel N-alkyl substituted isatins derivatives as anticancer agents. J Chem Pharm Res 2009;1(1):199-206.
- Teng YO, Zhao HY, Wang J, Liu H, Gao ML, Zhou Y, et al. Synthesis and anti-cancer activity evaluation of 5-(2-carboxyethenyl)-isatin derivatives. Eur J Med Chem 2016;112:145-56.
- Aboul-Fadl T, Radwan AA, Attia MI, Al-Dhfyan A, Abdel-Aziz HA. Schiff bases of indoline-2,3-dione (isatin) with potential antiproliferative activity. Chem Cent J 2012;6:49.
- Prakash CR, Raja S, Saravanan G, Dinesh Kumar P, Panneer Selvam T. Synthesis and evaluation of antioxidant activities of some novel isatin derivatives and analogs. Asian J Res Pharm Sci 2011;1:140-3.
- Sannigrahi M. Stereocontrolled synthesis of spirocyclics. Tetrahedron 1999;55:9007-71.
- James DM, Kunze HB, Faulkner DJ. Two new brominated tyrosine derivatives from the sponge Druinella (Psammaplysilla) purpurea. J Nat Prod 1991;54:1137-40.
- Kobayashi J, Tsuda M, Agemi K, Shigemori H, Ishibashi M, Sasaki T, et al. Purealidins E-G, new bromotyrosine alkaloids from the Okinawan marine sponge Psammaplysilla purea. Chem Inform 1991;47:6617-22.
- Tripathi AC, Gupta SJ, Fatima GN, Sonar PK, Verma A, Saraf SK. 4-Thiazolidinones: The advances continue. Eur J Med Chem 2014;72:52-77.
- Halliwell B, Gutteridge JM. Oxygen toxicity, oxygen radicals, transition metals and disease. Biochem J 1984;219:1-14.
- Maxwell SR.Prospects for the use of antioxidants therapies. Drugs 1995;49:345-61.
- Niki E, Shimaski H, Mino M. Antioxidantism-free radical and biological defense. Tokyo: Gakkai Syuppn Center; 1994. p. 3-6.
- Prior RL, Cao G. Variability in dietary antioxidant related natural product supplements: The need for methods of standardization. J Am Nutraceutical Assoc 1999;2:46-56.
- Mashelkar UC, Rane DM. Synthesis of some isatin based novel spiroheterocycles and their biological activity studies. Indian J Chem 2005;44B:1937-9.
- Kumar RS, Rajkapoor B, Perumal P. Antioxidant activities of Indigofera cassioides Rottl. Ex. DC. using various in vitro assay models. Asian Pac J Trop Biomed 2012;2(4):256-61.