- Corresponding Author:
- P. Ghosh
Natural Product and Polymer Chemistry Laboratory, Department of Chemistry
E-mail: pizy12@yahoo.com
| Date of Submission | 26 March 2010 |
| Date of Revision | 14 February 2011 |
| Date of Acceptance | 15 March 2011 |
| Indian J Pharm Sci, 2011, 73 (1): 231-233 |
Abstract
Two triterpenoids, taraxerone and tricadenic acid A were isolated from the methanol extract of the outer bark of Schleichera oleosa available in Darjeeling foothills. A preliminary study on their antimicrobial activities was also performed against some fungal and bacterial species. The structure of these compounds was determined by means of chemical characterisation and IR, NMR spectral data.
Keywords
Antimicrobial, Schleichera oleosa, taraxerone, tricadenic acid A, triterpene
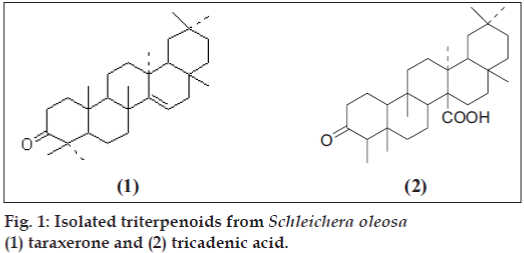
Darjeeling foothills have many plants that are used as folklore medicines by the local people. Schleichera oleosa (Lou.) Oken, commonly known as Ceylon Oak belongs to the family of sapindaceae [1] occurs naturally at the foot hills of Darjeeling and is used in traditional medicine for several indications from the time immemorial. It is generally used as analgesic, antibiotic and against dysentery [1]. S. oleosa is a well known medicinal plant [2] in the Teak forest of East Java and is also used in West Bengal in India, as a commercial host plant for obtaining sticklac for production of shellac [2]. In parts of southern India, S. oleosa is a prominent bee plant for nectar [2] and is used for production of animal feedstock. The oil obtained from its seed, called Kusum oil or Macassar oil is traditionally used for the cure of itch, acne, burns, other skin troubles, rheumatism (external massage), hair dressing and for promoting hair growth [3]. Very recently Mohapatra and Sahoo have reported the use of its bark along with water to treat menorrhoea [4]. Previous studies have revealed several bioactive triterpenoids including betulinic acid and betulin. In an ongoing search for bioactive triterpenoids from S. oleosa, the methanol extract was selected for further investigation. This is the first report of the existence of taraxerone (1) and tricadenic acid A (2) (fig. 1) in the outer barks of the above plant.
All the melting points were determined by open capillary method and are uncorrected. The NMR spectra were recorded in CDCl3 solutions at ambient temperature on a Bruker Avance 300 MHz-FT NMR spectrometer using 5 mm BBO probe. The chemical shift d are given in ppm related to tetramethylsilane (TMS) as internal standard. The coupling constant (J) are reported in Hz. The IR spectra were recorded in Shimadzu FT-IR spectrophotometer in KBr discs.
Fresh outer bark of S. oleosa was collected from Sukna belt of foothills of Darjeeling in an early summer. The plants collected were shade dried at room temperature and mechanically reduced to coarse powder. The prepared powdered leaves were then used for further studies.
The powdered plant material (1000 g) was extracted with methanol in a Soxhlet apparatus for 72 h. The solvent was recovered at reduced pressure that yielded a deep brown gummy residue (200 g). This crude methanol extract of the plant was then fractionated over a column of silica gel of 60-120 mesh using petroleum ether and ethyl acetate with increasing concentration as eluent.
In the present study, five different fungal pathogens (Colletotrichum camelliae, Fusarium equisiti, Alternaria alternata, Curvularia eragrostidis, Colletotrichum gloeosporioides) were used for in vitro antifungal assay [5,6]. Antibacterial assay were performed against four bacterial pathogens (Escherichia coli, Bacillus subtilis, Staphylococcus aureus, Enterobactor). Suitable strains of these organisms were procured from the microbiology laboratory of our institute. MICs (Minimum inhibitory concentration) of the triterpenoids against bacterial and fungal pathogens have been presented in Table 1 and 2, respectively. The antifungal and antibacterial media used are as follows. For nutrient agar (NA) 28 g of media (HiMedia) was suspended in 1000 ml of distilled water according to the manufacturer’s protocol. It was boiled to dissolve the medium completely at sterilized by autoclaving at 15 lbs pressure (121º for 15 min). The nutrient agar contained peptic digest of animal tissue (5 g), sodium chloride (5 g), beef extract (1.5 g), yeast extract (1.5 g), agar (15 g) and distilled water (1000 ml). pH was adjusted to 7.2. For preparation of PDA (potato-dextrose-agar) peeled potato was cut into small pieces and boiled in required volume of distilled water. The mixture was filtered through muslin cloth and the extract was mixed with dextrose and agar. The resultant mixture was heated in order to dissolve. Finally the media was sterilized at 15 lbs (121º for 15 min). Composition of the media was peeled potato (400 g), dextrose (20 g), agar (20 g) and distilled water (1000 ml). pH was adjusted to 6.0. DMSO (dimethyl sulfoxide) was used as solvent to prepare different concentrations of the triterpenoids. Solvent control (DMSO) was also maintained throughout the experiment. All experiments were performed in Petri dishes and were incubated at 37º for 48 h. The required media (either PDA or NA) was poured in a Petri dish and allowed for solidification. After solidification wells or cups were made by inserting a cork borer in the media. The number of wells were made according to the requirement of the experiment. Fungal spores were suspended on the PDA media before well or cup formation. Test solution (100 ml/ well) was poured in the well or cup. Additionally slide germination method was also used for determination of antifungal activity [5,6] (Table 3). We compared the antifungal activities of these compounds with that of Bavistan and antibacterial activity with that of ampicillin, a b-lactam antibiotic.
| Compounds | MIC in mg/ml against different bacterial strains | |||
|---|---|---|---|---|
| EC | BS | SA | EB | |
| 1 | 100 | <100 | 100 | 100 |
| 2 | 150 | 100 | 200 | 100 |
| Ampicillin | 128 | 64 | 64 | 128 |
BS: Bacillus. subtilis, EC: Escherichia coli, SA: Staphylococcus. aureus, EB: Enterobactor, MIC: Minimum inhibitory concentration.
TABLE 1: MIC OF ISOLATED COMPOUNDS AGAINST DIFFERENT BACTERIA
| Compounds | MIC in mg/ml against different fungal strains | ||||
|---|---|---|---|---|---|
| CG | FE | CE | AA | CC | |
| 1 | <5 | 20 | 40 | 10 | <5 |
| 2 | 4.87 | 19.5 | 40 | 19.5 | 39 |
| Bavistan | 3.5 | 3.5 | 3.7 | 4.0 | 4.2 |
CG: Colletotrichum gloeosporioides, FE: Fusarium equisiti CE: Curvularia eragrostidis, AA: Alternaria alternata, CC: Colletotrichum camelliae.
TABLE 2: MIC OF ISOLATED COMPOUNDS AGAINST DIFFERENT FUNGI
| Fungal pathogen | Taraxerone | Tricadenic acid | ||||
|---|---|---|---|---|---|---|
| PGa | PI | ALb(mm) | PGa | PI | ALb(mm) | |
| CC | 00 | 100 | 00 | 05 | 95 | 4.5 |
| FE | 00 | 100 | 00 | 00 | 100 | 00 |
| AA | 00 | 100 | 00 | 00 | 100 | 00 |
| CG | 00 | 100 | 00 | 00 | 100 | 00 |
| CE | 05 | 96 | 6.0 | 00 | 100 | 00 |
CG: Colletotrichum gloeosporioides, FE: Fusarium equisiti, CE: Curvularia eragrostidis, AA: Alternaria alternata, CC: Colletotrichum camelliae. PG: Percent germination, PI: Percent inhibition, AL: Average germ tube length, aBased on 200 spores, bBased on 25 germ tubes.
TABLE 3: ANTIFUNGAL PROPERTIES OF TRITERPENOIDS AGAINST FIVE FUNGAL PATHOGENS BY SPORE GERMINATION BIOASSAY AFTER 48 H OF INCUBATION [5]
Five components were separated of which we found taraxerone (1) and tricadenic acid A (2) along with three others reported earlier and the structures of both the compounds were determined based on their chemical and spectral data (IR, UV, and NMR) as discussed below. These two compounds were evaluated for their antifungal and antibacterial activities. Compound (1) was isolated as white crystals (CHCl3+MeOH) of m.p. 239-242º. It showed IR absorption spectra at 3047.3, 2924.8, 2851.6, 1708.7, 1375.2 and 955.2 cm-1. 1H NMR spectra showed eight tertiary methyl’s between 0.82 to 1.21 ppm and at 5.51 (dd, 1H, J = 8, 3.2 Hz) for the olefinic proton at C-15. All other peaks are for the presence of saturated methylene group. 13C NMR spectra showed peaks at 40.65 (C-1), 33.1 (C-2), 210.1 (C-3), 47.72 (C-4), 55.8 (C-5), 33.59 (C-6), 34.18 (C-7), 37.83 (C-8), 48.8 (C-9), 38.49 (C-10), 35.12 (C-11), 36.69 (C-12), 37.6 (C-13), 157.7 (C-14), 117.0 (C-15), 33.49 (C-16), 33.89 (C-17), 48.88 (C-18), 41.4 (C-19), 29.5 (C-20), 36.26 (C-21), 27.29 (C-22), 28.1 (C-23), 15.60 (C-24), 14.26 (C-25), 29.58 (C-26), 30.0 (C-27), 29.88 (C-28), 33.23 (C-29) and 21.4 (C-30) ppm and was found identical with already reported compound.
Compound (2) was isolated as white crystals (CHCl3+MeOH) of m.p. 294o, [a]D= +25.0o. IR spectra exhibited peaks at 3300 cm-1 for -OH group and at 1685 cm-1 for the carboxylic acid group, corresponding methyl ester has the m.p. 200-201o. 1H spectra showed the presence of six tertiary methyl groups that appear as singlet’s at d 0.81, 0.85 (d, 3H J = 6 Hz), 0.93, 0.99, 1.11, 1.20 and a secondary methyl group that appears as a doublet at 0.74 (d, 3H, J = 7 Hz); 3.76 (s, 3H) for -COOCH3 group.
Both the compounds showed prominent antimicrobial activities against the tested fungal and bacterial pathogens as evidenced from the Tables 1, 2 and 3. Compound 1 showed better activity against all the microorganisms than compound 2 and its activity is comparable to that of Ampicillin against E. coli and Enterobactor. The activity of compound 1 is nearly comparable to that of Bavistan, when it was tested against Colletotrichum gloeosporioides and Colletotrichum camelliae. Finally it can be concluded that the present study will be extremely helpful to enrich the present knowledge about different types of triterpenoids and also help the researchers to design newer generation of drugs based on such information about triterpenoids.
Acknowledgements
The authors thank UGC, New Delhi, India for financial support to carry out the work.
References
- Rout SD, Panda T, Mishra N. Ethno-medical plants used to cure different diseases by tribal of Mayurbhanj district of North Orissa. Ethno-Med 2009;3:27-6.
- Pettit GR, Numata A, Cragg GM, Herald DL, Takada T, Iwamoto C, etal. Isolation and Structures of Schleicherastatins 1-7 and Schleicheols1 and 2 from the Teak Forest Medicinal tree Schleichera oleosa. J Nat Prod 2000;63:72-5.
- Palanuvej C, Vipunngeun N. Fatty acid constituents of Schleicheraoleosa (Lour) Oken seed oil. J Health Res 2008;22:203-2.
- Mahaptma SP, Sahoo HP. An ethano medico botanical study of Bolangi, Orissa, India: Native plant remedies against Gynaecological diseases. Ethanobot Leafl 2008;12:846-4.
- Suleman P, Al-Musallam A, Menezes CA. The Effect of Biofungicide mycostop on Ceratosystis radicicola, the Causal Agent of Black Scorch on Date Palm. Biocontrol 2002;47:207-16.
- Saha D, Dasgupta S, Saha A. Antifungal activity of some plant extracts against fungal pathogen of tea (Camellia sinensis). Pharma Biol 2005;43:87-5.