- *Corresponding Author:
- H. Wang
Department of Gynecology and Obstetrics, Beijing Tongren Hospital, Capital Medical University, Beijing, China
E-mail:quant20110123@163.com
| This article was originally published in a special issue, “Biomedical research applications in Pharmaceutical Sciences” |
| Indian J Pharm Sci 2020:82(2)Spl issue3;1-7 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
To investigate the correlation between high-risk human papillomavirus infection and the expression of ER, PR, P53 and Ki67 in patients with different grades of cervical intraepithelial neoplasia. A retrospective study was conducted in which from June 2016 to June 2018, 140 specimens of cervical intraepithelial neoplasia were collected from the pathology department of the Beijing Tongren Hospital that included 40 specimens of cervical intraepithelial neoplasia1, 50 specimens of cervical intraepithelial neoplasia2 and 50 specimens of cervical intraepithelial neoplasia1. The expression of ER, PR, p53 and Ki67 were determined by immunohistochemistry. High-risk human papillomavirus infections were detected by polymerase chain reaction fluorescence quantification and were given the correlation analysis. Results of the 140 specimens indicated the rates of human papillomavirus16 and human papillomavirus18 in cervical intraepithelial neoplasia1 specimens were 27.5 and 25.0 % respectively, in cervical intraepithelial neoplasia2 specimens were 64.0 and 60.0 %, respectively and in cervical intraepithelial neoplasia3 specimens were 90.0 and 92.0 % respectively, the difference were statistically significant (p<0.05). There was no significant correlation between human papillomavirus 16 and human papillomavirus 18 positive rate and patient age, tissue differentiation, and tumour size. With the increasing cervical intraepithelial neoplasia grade, the rate of ER, PR, p53 and Ki67 expression in specimens was also increased significantly (p<0.05). Pearson correlation analysis showed there were positive correlation between the rates of human papillomavirus 16 and human papillomavirus 18 and the rates of ER, PR, p53 and Ki67 expression (p<0.05). In conclusion with increasing cervical intraepithelial neoplasia level, rate of high-risk human papillomavirus infection increased along with increased rate of expression of ER, PR, p53 and Ki67.
Keywords
Cervical intraepithelial neoplasia, HPV infection, correlation, p53, Ki67g
General cervical cancer is one of the common malignant tumors in women. It is developed from cervical squamous epithelial lesions. New cases in China account for about 1/3 of the world’s morbidity rate and it ranks second in the world for a long time[1,2]. There are many factors that cause cervical cancer and are closely related to human papillomavirus (HPV) infection. HPV DNA can be detected in more than 99 % of cervical cancer tissues[3]. HPV is a type of cyclic DNA virus that can specifically infect the skin and mucous membranes of humans. According to the relationship between HPV subtypes and tumorigenesis, HPV can be divided into low and medium and high risk types. High-risk HPV includes HPV 16, 18, 31 and 33. It can participate in cervical squamous epithelial lesions and cervical cancer to varying degrees[4,5]. Modern studies have shown that the development of cervical squamous epithelial lesions is a multi-step process that develops from normal to inflammatory to tumors, accompanied by multiple genetic changes. In this process, it is associated with changes in various genes such as all oncogenes, tumor suppressor genes and growth factor genes[6,7]. Estrogen receptor (ER) and progesterone receptor (PR) are present on the surface of hormone target cells and are mainly distributed in targets such as uterus and cervix. These can specifically bind to corresponding hormones and play a role in regulating the occurrence and development of cervical squamous epithelial lesions[8,9]. Ki67 is a relatively positive sign of nuclear proliferation. It is also an important reference for predicting the development of cervical squamous intraepithelial neoplasia (CIN) and cervical cancer. Its expression can reflect the biological behaviour of tumor cells[10,11]. The p53 gene is an important tumor suppressor gene located in human chromosome 17p 13.1. Its encoding product p53 protein plays an important role in cell division and differentiation. The normal expression of p53 protein can induce apoptosis and cause cell cycle block. p53 protein mutations can lead to cell transformation and over-proliferation and tumor behavior[12,13]. This paper specifically discusses the expression of ER, PR, p53, Ki67 and the correlation of HPV infection in patients with different levels of cervical squamous epithelial lesions. It is hoped to provide evidence for the pathogenesis of cervical squamous epithelial lesions. Further clarify the relationship between ER, PR, p53, Ki67 protein expression and HPV infection.
Materials and Methods
From June 2016 to June 2018, 140 CIN specimens preserved in the department of pathology of the Beijing Tongren Hospital were selected. Inclusion criteria were, complete clinical and pathological data; pathological diagnosis of cervical squamous intraepithelial lesions at different levels; all patients were not treated with radiotherapy, chemotherapy or hormone therapy before operation, 20-80 y old. Exclusion criteria were past history of cervical cancer, immunodeficiency disorders, absence of clinical and pathological data, minimum age of 26 y old, maximum age of 78 y old, average ages is 47.49±3.39 y, the age of 90 cases is over 40 y old. CIN grading was as follows, 40 cases in grade 1, 50 cases in grade 2 and 50 cases in grade 3; 20 cases in histological differentiation were poorly differentiated, 30 cases in moderate differentiation and 90 cases in high differentiation.
Mouse antihuman p53 monoclonal antibody, mouse antihuman PR monoclonal antibody, mouse antihuman Ki67 monoclonal antibody and mouse antihuman ER monoclonal antibody were purchased from Santa Cruz Biotechnology Company; immunohistochemical universal staining kit, PBS buffer, DAB staining kit were purchased from Beijing Zhongshan Biotechnology Co., Ltd. All tissue specimens were sectioned continuously in wax with a thickness of 2 microns. Xylene and ethanol were used for transparency and dehydration. The tissue sections were stained with hematoxylin for 15-30 s and sealed with neutral gum. The results were observed under an optical microscope. Positive control group was set up in each section, positive control group was cervical cancer tissue, and negative control group was PBS instead of primary antibody. Each slice was randomly observed in 5 high power microscopic fields. ER and PR were positive for brown granules in the nucleus. Ki67 was mainly positive in the nucleus. The positive signals of p53 staining were brown and yellow and were strictly located in the nucleus. The pathologists made immunohistochemical grading diagnosis of cervical squamous intraepithelial lesions according to the combined grading criteria. The negative staining intensity was 0, weak but stronger than negative control was 1, the clear staining was 2 and the strong staining was 3; the number of positive cells <10 % was 0, 10- 30 % was 1, 31-60 % was 2, and > 60 % was 3. The above two scores were added up: 0-1 was (-, negative), 2 was (+, weak positive), 3-4 was (+++, medium positive), 5-6 was (+++++, strong positive), and (+++++++) was positive.
The specimens were rinsed with sterile water and fixed with 95 % ethanol for 15 min. The cervical secretions of the specimens were dissolved with lysate. Sterile ophthalmic shears were used to cut the tissue to about 1 mm 3 sizes, add 0.25 % trypsin and digest it in 37° water bath. The tissue suspension was filtered by 200 mesh stainless steel mesh and then transferred into the test tube. After 5 min of centrifugation, the supernatant was discarded. Cell precipitation was washed 2-3 times with Hanks solution to obtain single cell suspension. Fully automatic nucleic acid extractor produced by Jiangsu Shuoshi Biotechnology Co., Ltd. was used to operate according to the scheduled scheme. Specific primers of HPV16 and HPV18 were designed and amplified according to NCBIBLAST sequence. The forward sequence of HPV-16 primers was 5’-GAATCCATGCTTATGATAA-3’, the reverse sequence was 5’-GATETGCAACAAGACATACAT-3’, the forward sequence of HPV18 primers was 5’-CACGGEGAECCTAAGCTACC-3’ and the reverse sequence was 5’-TGCAGEACGCAGCTAAGCTACC-3’. The cycling conditions of PCR fluorescence method were 95 for 5 min, 55 for 30 s, 72 for 1 min, 40 cycles and 72 for 10 min. The products of PCR digestion were sent to Shanghai Biotechnology Service Co., Ltd. for DNA sequencing and compared with NCBI gene sequence.
Statistical method:
SPSS22.00 software was selected to analyse the measurement data and counting data of this study. The measurement data describing the normal distribution were expressed by mean±standard deviation. The counting data were expressed as frequency. The comparison was t-test, chi-square test and variance analysis. Pearson correlation analysis was used for correlation analysis, and the test level was a=0.05.
Results and Discussion
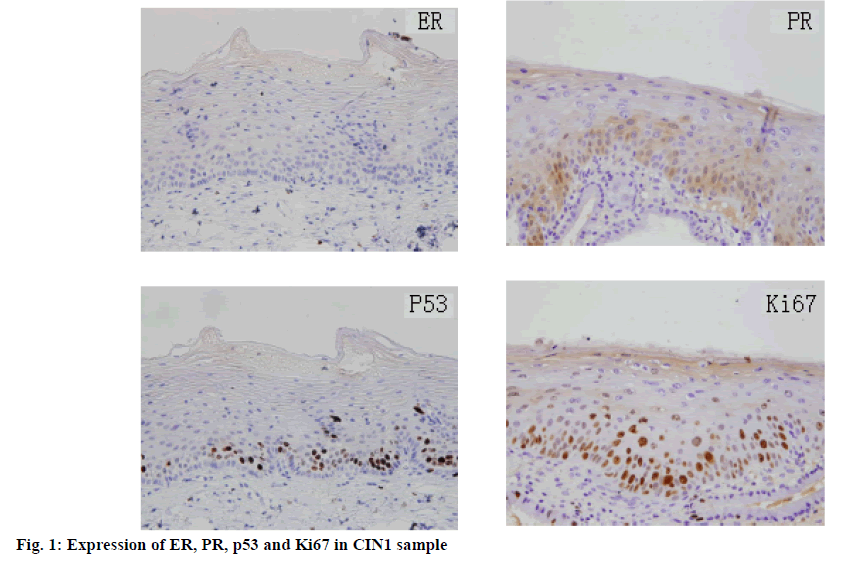
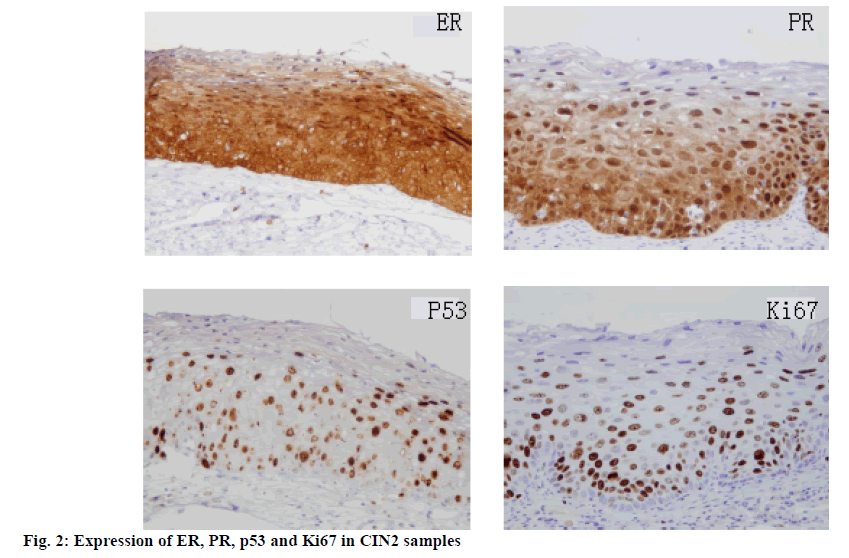
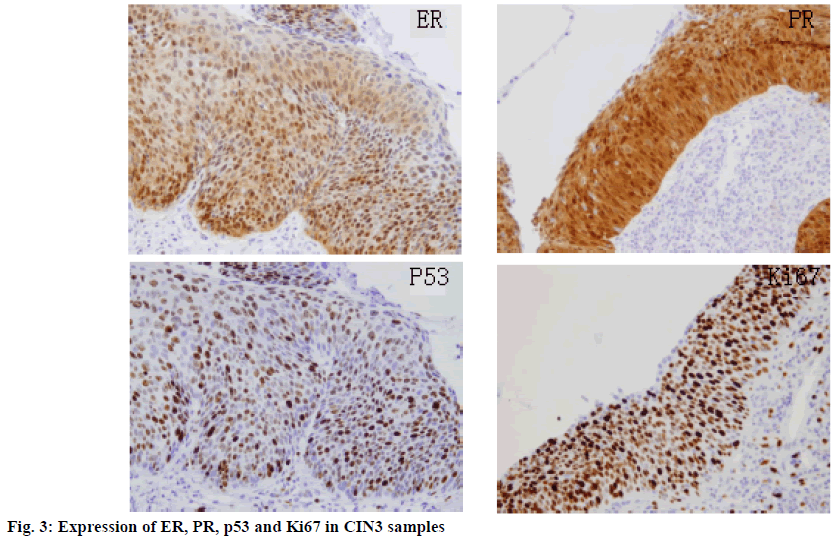
In 140 samples, the incidence rates of HPV16 and HPV18 were 27.5 and 25.0 % in LSIL, 64.0 and 60.0 % in HSILa, 90.0 and 92.0 % in HSILb, respectively. The difference was statistically significant (p<0.05; Table 1). The positive rates of HPV16 and HPV18 were not significantly correlated with age and tissue differentiation (Table 2). The rates of ER, PR, p53 and Ki67 expression in specimens were also significantly increased with the increase of CIN grading (p<0.05; Table 3, figures 1, 2 and 3). Pearson correlation analysis showed that the incidence rates of HPV16 and HPV18 were positively correlated with the rates of expression of ER, PR, P53 and Ki67 (p<0.05; Table 4).
Table 1: Comparison of High-Risk HPV Incidence Rates in Cervical Squamous Intraepithelial Lesions of Different Grades
| Group | Case (n) | HPV16 Positive | Positive rate | HPV18 Positive | Positive rate |
|---|---|---|---|---|---|
| LSIL | 40 | 11 | 27.5 % | 10 | 25.0 % |
| HSILa | 50 | 32 | 64.0 % | 30 | 60.0 % |
| HSILb | 50 | 45 | 90.0 % | 46 | 92.0 % |
| F | 37.224 | 42.169 | |||
| P | 0.000 | 0.000 | |||
| Total | 88 | 62.9 % | 86 | 61.4 % |
Table 2: Correlation between High-Risk HPV Incidence Rate and Clinical Features of Cervical Squamous Intraepithelial Lesions
| Clinical features | Case | HPV16 Positive rate (n=88) | F or 2 | P | HPV18 Positive rate (n=86) | F or 2 | P |
|---|---|---|---|---|---|---|---|
| Age≧40 | 90 | 80 (88.9 %) | 73.144 | 0.000 | 78 (86.7 %) | 67.745 | 0.000 |
| <60 | 50 | 8 (16.0 %) | 8 (16.0 %) | ||||
| Histological differentiation-High differentiated | 90 | 55 (61.1 %) | 12.336 | 0.002 | 53 (58.9 %) | 14.549 | 0.001 |
| Moderately differentiated | 30 | 14 (46.7 %) | 15 (50.0 %) | ||||
| Poorly differentiated | 20 | 19 (95.0 %) | 20 (100.0 %) |
Table 3: Rates of ER, PR, P53 and Ki67 Expression in Cervical Squamous Intraepithelial Lesions of Different Grades
| Group | Case (n) | ER positive rate | PR positive rate | p53 positive rate | Ki67 positive rate |
|---|---|---|---|---|---|
| LSIL | 40 | 16(40.0 %) | 15(37.5 %) | 17(42.5 %) | 16(40.0 %) |
| HSILa | 50 | 35(70.0 %) | 34(68.0 %) | 37(74.0 %) | 41(82.0 %) |
| HSILb | 50 | 48(96.0 %) | 48(96.0 %) | 50(100.0 %) | 49(98.0 %) |
| F | 33.670 | 35.797 | 38.466 | 42.326 | |
| P | 0.000 | 0.000 | 0.000 | 0.000 |
Table 4: The Expression of ER, PR, P53 and Ki67 in Patients with Different Grades of Cervical Squamous Intraepithelial Lesions and Their Correlation with High-Risk HPV Infection
| Index | ER | PR | p53 | Ki67 |
|---|---|---|---|---|
| HPV16-r | 0.563 | 0.610 | 0.544 | 0.395 |
| P | 0.000 | 0.000 | 0.000 | 0.013 |
| HPV18-r | 0.433 | 0.594 | 0.388 | 0.410 |
| P | 0.012 | 0.000 | 0.016 | 0.010 |
Cervical squamous intraepithelial lesions are malignant tumors that seriously threaten women’s health. The etiology of cervical squamous intraepithelial lesions has not been fully understood. Relevant studies have shown that the incidence of cervical squamous intraepithelial lesions is related to such factors as early marriage, sexual disorder, prolificacy, economic status, premature sexual life, economic status and race. Persistent infection of high-risk human papillomavirus (HPV) is also an important factor[14-16].
HPV is a common pathogen of female genital tract infection. So far, more than 200 kinds of HPV have been found. Epidemiological investigation showed that HPV16 accounted for about 50 % of cervical squamous cell carcinoma and HPV18 accounted for about 15 %[17]. The results showed that the incidence rates of HPV16 and HPV18 were 27.5 and 25.0 % in LSIL, 64.0 and 60.0 % in HSILa, 90.0 and 92.0 % in HSILb, respectively. These rates of HPV16 and HPV18 were not significantly correlated with age and histological differentiation of patients (p<0.05). Leading role in change, Current studies have shown that overexpression of E6 and E7 proteins in HPV promotes the release of p53 and pRB proteins and immortalizes host cells, leading to cervical epithelial neoplasia and canceration[18].
Although HPV is a necessary condition for the occurrence of cervical squamous intraepithelial lesions, HPV infection does not always lead to the occurrence of cervical squamous intraepithelial lesions. The majority of HPV-infected people are subclinical or latent viral infections[19]. HPV infection alone is not enough to cause cervical squamous intraepithelial lesions or cervical cancer. The development from HPV infection to cervical squamous intraepithelial lesions depends on the synergistic effect of other factors[20]. Current studies have shown that the occurrence of cervical squamous intraepithelial lesions is a complex process involving multiple stages and factors. It is also a disorder of the balance between cell proliferation, differentiation and apoptosis. ER and PR are located in the nucleus and mainly exist in the tissue cells of female reproductive system. The binding of receptors and hormones can induce gene activation and regulate cell proliferation and apoptosis[21]. Current studies have shown that estrogen can induce ER and PR production at the level of DNA replication and transcription during endometrial cell proliferation, while progesterone has antagonistic effect on estrogen, which can make dysplasia cells mature and down-regulate at the level of transcription and post-transcription[22]. Some studies have shown that ER and PR are most expressed in normal endometrium, but less expressed in cancer. ER and PR may participate in abnormal proliferation and inhibit normal cell apoptosis[23]. p53 gene abnormality is closely related to the carcinogenesis of cervical epithelial cells. p53 gene mutation exists in cervical squamous intraepithelial lesions. As an important tumor suppressor gene, p53 inactivation plays an important role in the occurrence and development of cervical squamous intraepithelial lesions. High-risk HPV E6 proto-oncogene coding protein can bind to p53 protein and degrade it. Wildtype p53 can participate in the regulation of cell growth, development and differentiation through p53- dependent or p53-independent pathways[24]. Current studies have also shown that p53 can prevent cells from entering S phase from G1 phase, thereby inhibiting cell proliferation. Overexpression of p53 has been proved to be significantly related to the initiation of HPHPV transformation infection[25,26]. Ki67 is a definite marker of nuclear proliferation at present. Its half-life is short. It can be quickly interpreted as a marker to detect the proliferation activity of cancer cells after it is detached from cell cycle. HPV infection can accelerate cell proliferation and increase the number of Ki67 positive nuclei, and the number of Ki67 positive nuclei increases with the grade of cervical lesions[27,28]. This study showed that the positive rates of ER, PR, p53 and Ki67 expression in cervical squamous intraepithelial lesions increased significantly with the grade of cervical squamous intraepithelial lesions (p<0.05).These results indicate that ER, PR, p53 and Ki67 are of great value in predicting the occurrence and development of cervical squamous intraepithelial lesions and have guiding significance for the treatment and prognosis of cervical squamous intraepithelial lesions.
Cervical squamous intraepithelial lesions are the result of interaction between environmental factors and genetic factors. Cell atypia is prone to occur on the basis of abnormal gene expression and viral infection[29]. Especially HPV infection is not the only pathogenic factor of cervical squamous intraepithelial lesions. The functional changes of oncogenes and tumor suppressor genes also play a key role in the occurrence and development of cervical squamous intraepithelial lesions[30]. In 140 samples, Pearson correlation analysis showed that the positive rates of HPV16 and HPV18 were positively correlated with the positive rates of ER, PR, p53 and Ki67 (p<0.05). Current studies have shown that E6 proteins of HPV16 and HPV18 can bind to wild-type p53 proteins, and with the participation of E6-related proteins, promote the degradation of p53 proteins, eventually lead to the reduction of p53-induced G1 phase arrest in cell cycle, thus completing DNA synthesis and promoting cell proliferation[31,32]. HPV infection also destroys normal sex hormone function, which weakens the antiestrogen effect of progesterone in endometrial lesions and increases the malignant transformation of cells[33]. HPV can induce the cell cycle activity of Ki67 positive cells. With the increase of Ki67 expression level, the number of HPV copies also increased significantly[34]. This study apparently has some shortcomings, the number of experimental cases is relatively small, relatively limited, and there is no healthy population, there can be some research bias, will be in-depth analysis in the next step.
In conclusion, with the increase of the grade of cervical squamous intraepithelial lesions, the rate of high-risk HPV infection increased, along with the rate of ER, PR, p53 and Ki67 expression increased, demonstrating a positive correlation.
Conflict of interest:
The authors declare that they have no conflict of interest.
References
- Gopalani SV, Janitz AE, Campbell JE. Trends in cervical cancer incidence and mortality in Oklahoma and the United States, 1999-2013. Cancer Epidemiol 2018;31;140-5.
- Elhalawani H, Lin TA, Volpe S, Mohamed AS, White AL, Zafereo J, et al. Machine Learning Applications in Head and Neck Radiation Oncology: Lessons From Open-Source Radiomics Challenges. Front Oncol 2018;17:294.
- Wu H, Zhang J. Chitosan-based zinc oxide nanoparticle for enhanced anticancer effect in cervical cancer: A physicochemical and biological perspective. Saudi Pharm J 2018;26:205-10.
- LeConte BA, Szaniszlo P, Fennewald SM, Lou DI, Qiu S, Chen NW, et al. Differences in the viral genome between HPV-positive cervical and oropharyngeal cancer. PLoS One 2018;13:e0203403.
- Lopez-Charcas O, Espinosa AM, Alfaro A, Herrera-Carrillo Z, Ramirez-Cordero BE, Cortes-Reynosa P, et al. The invasiveness of human cervical cancer associated to the function of Na(V)1.6 channels is mediated by MMP-2 activity. Sci Rep 2018;8:12995.
- Gavioli CF, Festa Neto C, Tyring SK, Silva LL, Oliveira WR. High-risk mucosal HPV types associated with squamous cell carcinoma on the nose tip in an immune competent young man. An Bras Dermatol 2018;93:716-18.
- Best AL, Logan RG, Vázquez-Otero C, Fung W, Chee V, Thompson EL, et al. Application of a Health Literacy Framework to Explore Patients' Knowledge of the Link between HPV and Cancer. J Health Commun 2018;23:695-702.
- Dawkins JC, Lewis GK, Toy EP. Cervical cancer complicating pelvic organ prolapse, and use of a pessary to restore anatomy for optimal radiation: A case report. Gynecol Oncol Rep 2018;26:14-6.
- Pankaj S, Nazneen S, Kumari S, Kumari A, Kumari A, Kumari J, et al. Comparison of conventional Pap smear and liquid-based cytology: A study of cervical cancer screening at a tertiary care center in Bihar. Indian J Cancer 2018;55:80-83.
- Subramaniam N, Thankappan K, Anand A, Balasubramanian D, Iyer S. Implementing American Joint Committee on Cancer 8th edition for head-and-neck cancer in India: Context, feasibility, and practicality. Indian J Cancer 2018;55:4-8.
- O'Connor M, O'Leary E, Waller J, Gallagher P, Martin CM, O'Leary JJ, et al. Socio-economic variations in anticipated adverse reactions to testing HPV positive: Implications for the introduction of primary HPV-based cervical screening. Prev Med 2018;23:90-6.
- Belglaiaa E, Souho T, Badaoui L, Segondy M, Prétet JL, Guenat D, et al. Awareness of cervical cancer among women attending an HIV treatment center: a cross-sectional study from Morocco. BMJ Open 2018;8:e020343.
- Liu J, Lv D, Wang H, Zou J, Chen F, Yang H. Recombinant adenovirus-p53 enhances the therapeutic effect of surgery and chemo radiotherapy combination in hypo pharyngeal squamous cell carcinomas patients. Medicine 2018;97:e12193.
- Palicelli A, Barbieri P, Mariani N, Re P, Galla S, Sorrentino R, et al. Unicystic high-grade intraductal carcinoma of the parotid gland: cytological and histological description with clinic-pathologic review of the literature. APMIS 2018;126:771-6.
- Dai Y, Wang L, Li D. Effectiveness of novel folate receptor-mediated staining solution detection (FRD) for cervical cancer screening. Medicine 2018;97:e11868.
- Costa AP, Gonçalves AK, Machado PR, de Souza LB, Sarmento A, Cobucci RN, et al. Immune Response to Human Papillomavirus One Year after Prophylactic Vaccination with AS04-Adjuvanted HPV-16/18 Vaccine: HPV-Specific IgG and IgA Antibodies in the Circulation and the Cervix. Asian Pac J Cancer Prev 2018;19:2313-7.
- Lee H, Kim D, Kiang PN, Cooley ME, Shi L, Thiem L, et al. Awareness, knowledge, social norms, and vaccination intentions among Khmer mother-daughter pairs. Ethn Health 2018;8:1-13.
- Bisi-Onyemaechi AI, Chikani UN, Nduagubam O. Reducing incidence of cervical cancer: knowledge and attitudes of caregivers in Nigerian city to human papilloma virus vaccination. Infect Agent Cancer 2018;17:29.
- Raulf N, Lucarelli P, Thavaraj S, Brown S, Vicencio JM, Sauter T, et al. Annexin A1 regulates EGFR activity and alters EGFR-containing tumour-derived exosomes in head and neck cancers. Eur J Cancer 2018;102:52-68.
- Shen C, Chen F, Wang H, Li G, Yu C, Wang X, et al. The Pinx1 Gene Downregulates Telomerase and Inhibits Proliferation of CD133+ Cancer Stem Cells Isolated from a Nasopharyngeal Carcinoma Cell Line by Regulating Trfs and Mad1/C-Myc/p53 Pathways. Cell Physiol Biochem 2018;49:282-94.
- Owusu-Afriyie O, Owiredu WK, Owusu-Danquah K, Komarck C, Foltin SK, Larsen-Reindorf R, et al. Expression of immunohistochemical markers in non-oropharyngeal head and neck squamous cell carcinoma in Ghana. PLoS One 2018;13:e0202790.
- Tamura RE, de Luna IV, Lana MG, Strauss BE. Improving adenoviral vectors and strategies for prostate cancer gene therapy. Clinics 2018,73:e476.
- Qin G, Li P, Xue Z. Triptolide induces protective autophagy and apoptosis in human cervical cancer cells by down regulating Akt/mTOR activation. Oncol Lett 2018;16:3929-34.
- Mikami Y, Fujii S, Kohashi KI, Yamada Y, Moriyama M, Kawano S, et al. Low-grade myofibroblastic sarcoma arising in the tip of the tongue with intravascular invasion: A case report. Oncol Lett 2018;16:3889-94.
- Skah S, Richartz N, Duthil E, Gilljam KM, Bindesbøll C, Naderi EH, et al. cAMP-mediated autophagy inhibits DNA damage-induced death of leukemia cells independent of p53. Oncotarget 2018;9:30434-49.
- Wilkie MD, Lau AS, Vlatkovic N, Jones TM, Boyd MT. Metabolic signature of squamous cell carcinoma of the head and neck: Consequences of TP53 mutation and therapeutic perspectives. Oral Oncol 2018;8:1-10.
- Wang M, Xiao C, Ni P, Yu JJ, Wang XW, Sun H. Correlation of Betel Quid with Oral Cancer from 1998 to 2017: A Study Based on Bibliometric Analysis. Chin Med J 2018;131:1975-82.
- Lv Q, Wu K, Liu F, Wu W, Chen Y, Zhang W. Interleukin‑17A and heparanase promote angiogenesis and cell proliferation and invasion in cervical cancer. Int J Oncol 2018;53:1809-17.
- Hafsi H, Dillon MT, Barker HE, Kyula JN, Schick U, Paget JT, et al. Combined ATR and DNA-PK Inhibition Radiosensitizes Tumor Cells Independently of Their p53 Status. Front Oncol 2018;13:245-8.
- Palicelli A, Barbieri P, Mariani N, Re P, Galla S, Sorrentino R, et al. Unicystic high-grade intraductal carcinoma of the parotid gland: cytological and histological description with clinic-pathologic review of the literature. APMIS 2018;126:771-6.
- Mohamed H, Aro K, Jouhi L, Mäkitie A, Remes S, Haglund C, et al. Expression of hormone receptors in oropharyngeal squamous cell carcinoma. Eur Arch Otorhinolaryngol 2018;275:1289-1300.
- Li Z, Wu Q, Song J, Zhang Y, Zhu S, Sun S. Risk of Second Primary Female Genital Malignancies in Women with Breast Cancer: a SEER Analysis. Horm Cancer 2018;9:197-204.
- Liu H, Zhang Y, Chang J, Liu Z, Tang N. Differential expression of neuroendocrine markers, TTF-1, p53, and Ki-67 in cervical and pulmonary small cell carcinoma. Medicine 2018;97:e11604.
- Sengupta D, Guha U, Mitra S, Ghosh S, Bhattacharjee S, Sengupta M. Meta-Analysis of Polymorphic Variants Conferring Genetic Risk to Cervical Cancer in Indian Women Supports CYP1A1 as an Important Associated Locus. Asian Pac J Cancer Prev 2018;19:2071-81.