- *Corresponding Author:
- Jain S
Shri Ravishankar College of Pharmacy, Bypass Road, Bhanpura, Bhopal - 462 010, India
E-mail: jainsurendra@rediffmail.com
| Date of Received : | 22 March 2006 |
| Date of Revised : | 17 July 2007 |
| Date of Accepted : | 25 December 2007 |
| Indian J. Pharm. Sci., 2007, 69 (6): 850-852 |
Abstract
The hepatoprotective activity of the ethanol extract of the leaf of Ptrospermum acerifolium was investigated in rats for carbon tetrachloride induced hepatotoxicity. Hepatotoxicity was induced in male Wistar rats by intraperitoneal injection of carbon tetrachloride (0.1 ml/kg/d p.o. for 14 d). Ethanol extract of P. acerifolium leaves were administered to the experimental rats (25 mg/kg/d p.o. for 14d). The Hepatoprotective effect of these extracts was evaluated by liver function biochemical parameters (total bilirubin, serum protein, alanine aminotransaminase, asparatate aminotransaminase and alkaline phosphates activites) and histopathological studies of liver. In ethanol extract-treated animals, the toxicity effect of carbon tetrachloride was controlled significantly by restoration of the levels of serum bilirubin and enzymes as compared to the normal and standard drug silymarin-treated groups. Histology of liver sections of the animals treated with the extracts showed the presence of normal hepatic cords, absence of necrosis and fatty infiltration which further evidence the hepatoprotective activity.
Keywords
Ptrospermum acerifolium, hepatotoxicity, hepatoprotective activity
Ptrospermum acerifolium (Sterculiaceae) is well distributed in India and found particularly in Sub – Himalayan tract outer Himalayan valley and hills up to 4000 ft [1] . The plant is commonly known as kanakchampa, karnikara, muchukunda and matsakanda. According to the Ayurvedic text it is used for haemostatic, anti-inflammatory, heamicaradian pain, ear pain, small pox, leucorrhoea [2]. The presence of flavonoid constituent’s luteolin-7β- glucuronide, luteolin-7β-D-glucoside, kampferol-3-OD- galactoside, friedeline, barurenol in the leaf [3,4]. It is well documented that the compounds are strong antioxidants and can be used for hepatoprotective activity. In view of this, the present study was aimed at evaluating the hepatoprotective activity of the leaves of P. acerifolium against carbon tetrachloride (CCl4)-induced hepatotoxicity in rats.
The leaves of P. acerifolium were collected from Bhopal, Madhya Pradesh. The plant authenticated by comparing with the herbarium voucher specimen deposited at CDRI, Lucknow. The material was air dried under shade, powdered mechanically and stored in airtight containers. About 1 kg of the powdered material was subjected to Soxhlation. It was refluxed with 90% ethanol for 72h in batches of 250 g each [3]. The extracts were pooled together and concentrated in vacuum using rotary flash evaporator. The acute toxicity studies were carried out as per stair case method [5]. The Animal Ethics Committee of the institution approved the study protocol (CPCSEA No. 870/03/C/CPCSEA).
Fifty male Wistar rats (150–200 g) were divided into five groups of 10 each and were administered with aliquot doses of extracts orally (100,150, 200, 250 and 300 mg/kg). Mortality was not noticed up to 200 mg/kg, whereas 100% mortality was noticed in the dose of 300 mg/kg. The LD50 of extracts was found to be 250 mg/kg body weight. One-tenth of this dose was selected as the therapeutic dose for the evaluation [6].
Four groups of animals containing six each were used for the study. The animals from Group I served as the control and received the vehicle 1% w/v gum tragacanth at a dose of 1ml kg/d of p.o. for 14 d. Groups II-V received the vehicle 1% ml/kg/d i.p. of CCl4 for 14 d [7]. The standard drug silymarin was administered to Group III animals in the dose 100 mg/kg/day p.o. for 14 d. While, Groups IV Animals were treated with ethanol extract of P. acerifolium in the dose of 25 mg/kg/d p.o. (as per acute toxicity studies) for 14 d respectively. The silymarin and the extracts were administered concomitantly to the respective groups of animals.
All the animals were killed on day 14 under light ether anesthesia. The blood sample were collected separately by carotid bleeding into sterilized dry centrifuge tube and allowed to coagulate for 30 min at 37°. The clear serum was separated at 2500G for 10 min and biochemical investigations were carried out assess liver function viz., total bilirubin [8], total protein [9], serum transaminases10 and serum alkaline phosphates [11].
The results are expressed as mean±SEM of six animals from each group. The data were evaluated by one–way ANOVA followed by Turkey’s multiple comparison test. P values ≤0.01 were considered statistically significant (Table 1).
| Group (n) | Bilrubin (mg%) | Total protein (mg/ml) | AST (IU/l) | ALT (IU/I) | ALP (IU/I) | |
|---|---|---|---|---|---|---|
| Control | 0.48±0.031 | 9.24±0.13 | 51.06±0.61 | 147.90±0.36 | 172.61±2.57 | |
| CCl4+treated* | 2.21±0.13 | 6.08±0.33 | 1342.31±28.28 | 2154.45±42.55 | 403.10±18.11 | |
| CCl4+silymarin** | 0.49±0.01 | 8.73±0.01 | 88.54±0.42 | 204.05±0.92 | 182.67±0.52 | |
| CCl4+ethanol extract** | 0.66±0.04 | 8.16±0.02 | 144.50±0.62 | 224.20±0.86 | 191.27±1.32 | |
| One way | F | 161.46 | 56.82 | 1894.8 | 2085.6 | 151.45 |
| ANOVA | d.f. | 4,25 | 4,25 | 4,25 | 4,25 | 4,25 |
| P | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 | |
Values are expressed as mean±SEM; n=6 rats in each group; *P<0.01 compared to control group; **P<0.01 compared to CCl4-treated group
Table 1: Effect of p. Acerifolium leaves extracts on ccl4 induced hepatotoxicity in rats.
After draining the blood liver samples were excised, washed normal saline and processed separately for histological observations. Initially, the materials were fixed in 10% buffered neutral formalin for 48h and then bovine solution for 6 h. Paraffin sections were taken at 5mm thickness. Processed in alcohol-xylene series and were stained with alum hematoxylin and eosin [12]. The sections were examined microscopically for histopathological changes.
The administration of CCl4 to the animals resulted in a marked increase in total bilirubin, serum amino transaminases (AST and ALT) and serum alkaline phosphates activities. However, the serum total protein level was decreased. The toxic effect of CCl4 was controlled in the animals treated with the standard drug silymarin. Among the extract–treated groups, significant hepatoprotective activity was observed in those treated with ethanol extract.
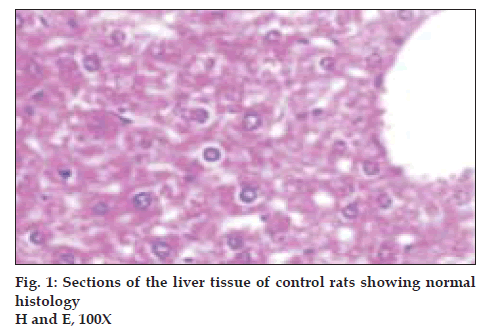
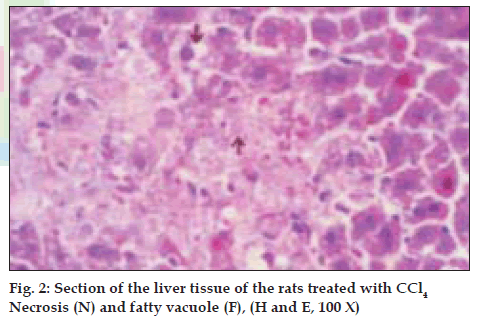
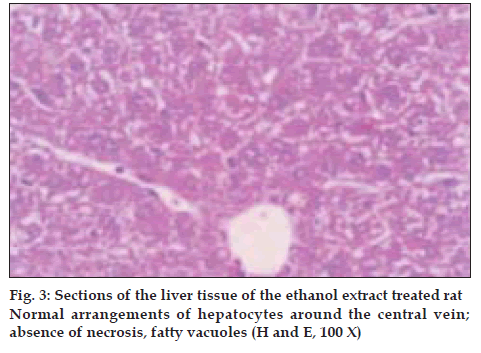
Histological profile of the control animals showed normal hepatocytes (fig. 1) Group II animals exhibited intense centrilobular necrosis (N), vacuolization (F) and macrovesicular fatty change (fig. 2). The sections of liver taken from the animals treated with standard drug silymarin showed hepatic architecture, which was similar to that of control. The animals treated with ethanol extract exhibited significant liver protection against the toxicant as evident by presence of normal hepatic cords, absence of necrosis and lesser fatty infiltration (fig. 3).
CCl4 has been used as a tool induced hepatotoxicity in experimental animals [13,14]. This toxic chemical causes peroxidative degradation in the adipose tissue resulting in fatty infiltration of the hepatocytes. The increase in the levels of serum bilirubin reflects the depth of jaundice; and the increase in transaminases and alkaline phosphates are indications of cellular leakage and loss of functional integrity of the cell membrane [15].
Administration of Ethanol extract of P. acerifolium leaf extract showed significant hepatoprotective activity, which was comparable with the standard drug silymarin. The effect was more pronounced with ethanol extract. Many phytochemical reports revealed that the ethanol extract of the plant was found to contain higher concentrations of flavonoids [16]. The qualitative phytochemical investigations on the P. acerifolium also showed positive for flavonoids by ferric chloride, alkaline reagent and Shinoda tests. Further, it has been reported that flavonoid constituents of the plant possess antioxidant properties [17] and was found to be useful in the treatment of liver damage [18]. The administration of hepatoprotective drugs may induce the hepatocytes to resist the toxic effect of CCl4. The results indicate that the ethanol extract of P. acerifolium has significant hepatoprotective activity. This may be probably due to the flavonoids. The earlier investigators have screened the hepatoprotective of the flavonoids compound, rutin, isolated from Artemisia scoparia, which is also claimed to have free radical scavenging and antilipid peroxidant activities against CCl4-induced hepatic toxicity. The isolation and characterization of the flavonoids from the leaves of P. acerifolium and screening of the pharmacological action against the liver damage is being investigated. In most of the developed and developing countries, the incidence of viral hepatitis is more so, the investigation for an efficient hepatoprotective drug from the natural resource is an urgent necessity.
Acknowledgements
The authors are thankful to RKDF Principal, College of Pharmacy, Bhopal, Dr. S. L. Bodhankar, Head, Department of Pharmacology, Poona College of Pharmacy, Pune and Dr. (Mrs.) Suneela S. Dhaneshwar, Assistant Professor, Department of Pharmaceutical Chemistry, Poona College of Pharmacy, Pune for their valuable help and support in planning and carrying out statistical analysis of the biological data.
References
- Rizvi SA, Sultan T. Phytochemical studies of the flowers of Ptrospermumacerifolium. Phytochemistry 1972;:856-8.
- Chopra RN, Nayar SL, Chopra IC. Glossary of Indian Medicinal Plant. New Delhi: CSIR; 1956.
- Gunase GR, Subramanian SS. Flavonoids of three Ptrospermum. Indian J Pharm Sci 1979;72-3.
- Rastogi RP, Mehrotra BN. Compendium of Indian Medicinal Plant, Vol-2, Lucknow: CDRI; 1991. p. 571-53.
- Ghosh MN. Fundamentals of experimental pharmacology. 2nd ed. Kolkatta: Scientific Book Agency; 1984.
- Jalalpure SS, Patil MB, Prakash NS, Hemalata K, Manvi FV. Hepatoprotective activity of fruits of Piper longum Linn. Indian J Pharm Sci 2003;65:360-6.
- Jaiprakash B, Aland R, Karadi RV, Savadi RV, Hukkeri VI. Hepatoprotective activity of fruit pulp of Balanitesaegyptiaca. Indian Drugs 2003;40:296-7.
- Mallory HT, Evelyn, EA. The determination of bilirubin with photoelectric colorimeter. J BiolChem 1937;119:481-5.
- Kingsley SR, Frankkel SJ. The determination of serum total protein albumin and globulin by the biuret reaction. J BiolChem 1939;128:131-7.
- Reitman S, Frankel A. A Colorimetric method for the determination of serum glutamic oxoloacetic and glutamic pyruvic transaminase. Am J ClinPathol 1957;28:56-63.
- Bessey, OA, Lowery DH, Brock MJ. A method for the rapid determination of alkaline phosphatase with five cubic meters of serum. J BiolChem 1964;164:321-9.
- Galighter AE, Koyloff EN. Essential of practical micro technique. Philadelphia: Lea and Febiger; 1971.
- Recnagel RO. Carbon tetrachloride hepatotoxicity status quo and future prospects. Trends PharmacolSci 1983;4:129-31.
- Okuno H, Hazama H, Muraze T, ShiozakiSomeshima YT. Drug metabolizing activity in rats with chronic liver injury, induced by carbon tetrachloride relationship with the hydroxyproline content in liver. Jpn J Pharmacol 1986;413:63-71.
- SarswatB,Visen PK, Patnaik GK, Dhawan BN. Anticholestic effect of picroliv, active hepatoprotective principle of Picrorhizakurrooa, against carbon tetrachloride-induced cholestatis. Indian J ExpBiol 1993;31:316-8.
- Harboni JB. Phytochemical Methods. 2nd ed. 1979.
- Hesham R, Seedi EL, Nishiyama S. Chemistry of Bioflavonoids. Indian J Pharm Educ 2002;36:191-4.
- Khalid H, Janbaz KH, Sheikh AS, Anwar HG. Protective effect of rutin on paracetamol and CCl4 induced hepatotoxicity in rodents. Fitoterapia 2002;73:557-63.