- *Corresponding Author:
- S. A. Shah
Department of Botany, Bacha Khan University Charsadda
E-mail: sajjadbiotec@gmail.com
| Date of Received | 09 July 2019 |
| Date of Revised | 25 December 2019 |
| Date of Accepted | 24 January 2020 |
| Indian J Pharm Sci 2020;82(2):374-378 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
The current research work was about a suitable eco-friendly process to produce gold nanoparticles using ethanol extract of Digera muricata. Completion of synthesis, size and morphology of nanoparticles and various functional groups present were confirmed using various techniques. Formation and physical appearance of gold nanoparticles was confirmed through the presence of dark purple colour. Ultraviolet/Vis spectra give a sharp narrow peak at the λmax specified for gold nanoparticle. Scanning electron microscopy indicated nanoparticles were spherical in shape, fully dispersed in reaction medium with 40±5 nm size. X-ray diffraction analysis revealed that synthesized nanoparticles were crystalline with an average particle size of 33 nm. The presence of bio-organic compound was detected by Fourier-transform infrared spectroscopy. Antibacterial potential of the synthesized gold nanoparticles of Digera muricata extract was evaluated in comparison to crude ethanol extract. The gold nanoparticles exhibited greater antibacterial potential as compared to crude ethanol extract of Digera muricata against different drug-resistant bacteria including Vibrio cholera, Staphylococcus pyrogen, Klebsiella, Citrobacter and Enterobacter. The results indicated that the gold nanoparticles could be used as a promising antibacterial agents.
Keywords
Digera muricata, Ethanol extract, Gold nanoparticles, Characterization, Drug resistant bacteria
Nanotechnology, a field of science and technology at molecular level controls and exploits the structure of matter on a large scale below 100 nanometers[1]. It is based on its ability of controlling metals into their nano size, which eventually change the physical, chemical and optical properties of metals. This is simply because of the nanoparticles (NPs) greater surface area per weight than larger particles, which make them more reactive as compare to other molecules[2]. On the base of size, distribution and morphology, NPs have new or improved properties. Green synthesis of metal NPs is devoid of consuming high energy, pressure and temperature. It is a useful and eco-friendly method to synthesize NPs on a large scale that attracted researchers throughout the world to find out the potential of different herbs. On the other hand, the chemical synthesis of NPs might result in poisonous chemicals adsorbed on their surface which might have unfavorable effects in medical applications[3].
Gold NPs (Au-NPs) have found use in diagnostic and drug delivery areas[4]. Au-NPs are synthesized from leaves, bark, stem, roots of herbal plants[5]. Au-NPs, also called gold colloids, have attracted attention for the fabrication of smart sensing due to their exclusive properties in devices in biomedical sciences as diagnostic tools in multi-disciplinary research fields[6]. In recent years in vitro approaches have been developed using plant extracts to bio reduce metal ions to synthesize NPs. Digera muricata species belongs to family Amaranthaceae. It is also known as Digera arvensis, Digera alternifolia and false amaranth or Digera angustifolia. All these are the synonyms of D. muricata. It is herbaceous plant grown annually (fig. 1). The whole plant especially its leaves and stems are useful due to their curative effects. It used for the inflammation, urination, as refrigerant and in sexual anomaly[7,8]. D. muricata is used in kidney disorder in folk medicine. The whole plant is used for digestive system as well as in urinary disorders. The extract of this plant was used in biliousness[9]. A decoction of leaves is given once in a day as kidney stone treatment[10]. Leaf paste is used in prevention of pus formation. The crushed plant is used as a mild astringent in bowel complaints[11]. The whole plant extract is also used to improve blood content[12]. D. muricata has flavonoids and been used as an anticancer drug[13]. Keeping in view of the above literature, the aim of current work is the biosynthesis of Au-NPs and evaluation of antibacterial activity against various drug-resistant bacterial strains. For the synthesis of Au-NPs and antibacterial assay, auric chloride (AuHCl4) was purchased from (E. Merck, Darmstadt, Germany), D. muricata extract, ethanol, nanopure water are used throughout the experiment. Bacterial strains used were Enterobacter, Vibrio cholera, Klebsiella pneumonia, Streptococcus pyrogen and Citrobactor. The plant was collected from the local fields of District Charsadda, Pakistan. After collection the plant material was washed 3 times using distilled water to remove all sand and dust particles. The plant was the cut into pieces and shade dried for 10-15 d. The dried plant was powdered in a mechanical grinder to a fine powder. Extract was prepared by soaking 500 g of powdered plant in 1500 ml of ethanol with shaking on an electric shaker at room temperature for 24 h. The extract was filtered through a Whatman filter paper No. 1 and the filtrate was stored in the refrigerator at 4º for further studies.
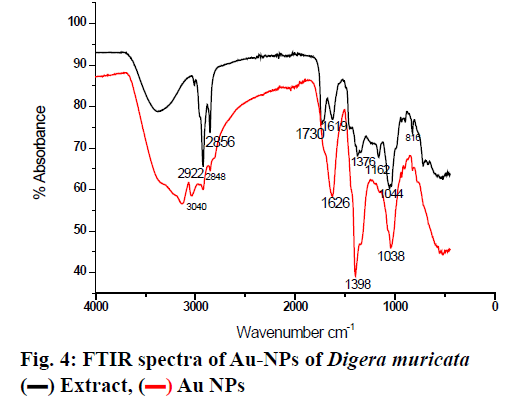
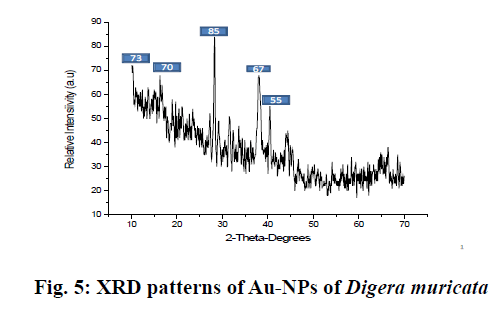
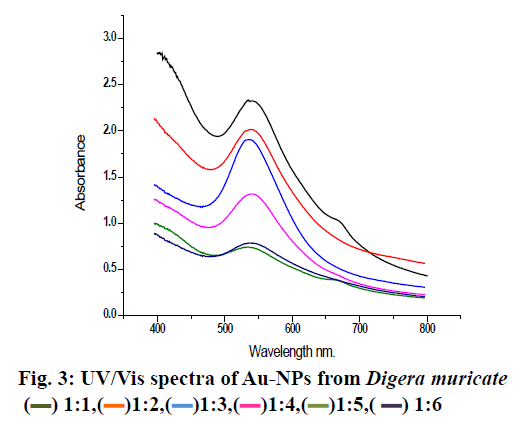
Basically, 0.6 g semi solid extracts was dissolved in 100 ml distill water for further analysis. The stock solutions of different concentration were prepared and stored at 4º. Seven hundred parts per million of the sample was mixed with 1 mM aqueous solution of AuHCl4 in different ratios by volume. Ten millilitres of plant extract was mixed with 10, 20, 30, 40, 50 and 60 ml of aqueous AuHCl4 solution to make 1:1, 1:2, 1:3, 1:4, 1:5 and 1:6, respectively for synthesising Au-NPs and these mixtures were stirred for 3 h till the color of solution completely changed to dark purple. To confirm the Au ion reduction, UV/Vis spectra of the prepared solutions were recorded against distilled water while ethanol was used as the blank. At a resolution of 1 nm (300-800 nm) the spectral analysis was completed on a double beam UV/Vis spectrophotometer (PerkinElmer) set at a resolution of 4 cm-1 in the range of 4000-0 cm-1. KBr pellets were prepared with these samples and Fourier-transform infrared spectroscopy (FTIR) analysis was completed using a thin KBr disc compressed using a hydraulic pellet press. FTIR spectra were run at the Chemistry Department, Bacha Khan University Charsadda, Pakistan. X-ray diffraction (XRD) was used for detection of crystalline nature and phase purity of the synthesized Au-NPs. XRD was set at a current of 30 mA with CuKα 1 radiation and a voltage of 40kV. Morphology, size and electron diffraction pattern of the synthesized Au-NPs were determined using scanning electron microscopy (SEM) at a voltage 15 kV, respectively. Streptococcus pyrogen, Klebsiella pneumonia, Enterobacter, Vibrio cholera and Citrobacter were cultured in Luria broth medium as a suspension culture in 250 ml flasks containing 1.25 g/50 ml in a shaker incubator set 150 rpm at 30° overnight. After reaching best growth all strains were stored at 4° in an incubator. Bactericidal activity of the plant extract and Au-NPs was carried out on 5 human pathogenic bacteria, Enterobacter, Streptococcus pyrogen, Klebsiella pneumonia, Vibrio cholera and Citrobacter using the agar well diffusion method. Nutrient agar was prepared by dissolving 10 g/500 ml along with 2.5 g agar. Medium was autoclaved at 121° for 20 min and then poured into sterilized Petri dishes, allowed to solidify for 1.5 h in a laminar flow under UV light. On sterilized agar plates fresh overnight culture of inoculums (200 μl) were spread and wells were made with the help of a stainless steel cork borer. Test solution of each ratio of Au-NPs at a concentration of 50 μl/ well and the plant extract were added to each well of agar plates along with the control and incubated at 37º for 24h. Distilled water used as a positive control. The diameter of the inhibition zone formed around the well were measured and the antibacterial activity was calculated using the standard procedure[14]. The extract of D. muricata was used as a reducing agent for the preparation of Au-NPs, which were characterized using various methods. The prepared Au-NPs were analyzed using UV, FT-IR, XRD and SEM. Further the prepared Au-NPs were evaluated for antibacterial activity. After exposure to D. muricata extract reduction of metal ion and the appearance of dark purple color indicated the production of Au-NPs. The change in color occurred due to the surface plasmon resonance of metal nanoparticles. Usually reduction of gold ions to Au-NPs a time taking process. After the addition of plant extract rapid color change occurred and the concentration of color was directly proportional to stirring and uniform heat. After continuous stirring (80 rpm) for 3 h the color deepens to purple indicated the formation of Au-NPs and uniform heat (40º) stopped the particles from aggregation as shown in fig. 2. Previous studies suggested that the change in color occurred on completion of the reaction. In a previous study, the reaction mixture of 10 ml AuHCl4 and 2 ml of extract had changed color from light yellow to stable violet color on continuous stirring for 10 min[15]. It was also reported that the solution color first changed into brownish pink and with rising incubation time from 30 min to 48 h changed to pink. The study also reported that reaction was complete in 48 h but in the present study the reaction tokk only 3 h for completion. It could be concluded that due to surface plasmon resonance, which is due to collective oscillation of electrons. It is reported previously that by adding 5 ml plant extract to 3 mM aqueous solution of AuHCl4 and stirring it continuously at room temperature the color of solution gradually changes from light pink to characteristic dark ruby red when the reaction was completed[16]. Similarly a reaction mixture of 10 ml AuHCl4 and 2 ml extract changed colour from light yellow to stable violet on continuous stirring for 10 min[17]. The Au-NPs formation was confirmed by UV/Vis spectra analysis. The first clue of Au-NP formation was conversion of pale yellow color into dark purple. Various concentrations of Au-NPs and ethanol extract of the plant were used for optimization. Each ratio gave different absorbance peaks. The maximum absorbance peak was recorded at 1:1 at wavelength at 535 nm, which is then followed by 1:2, 1:3 and 1:4, respectively. At 1:5 and 1:6 ratios the peak became broader and absorbance decreased with large particle size (fig. 3). Narrow sharp peaks represented that the synthesized NPs were small, spherical in shape and polydisperse with no aggregation. The plasmon band frequency decreased as the size of NPs increased and the band shifted to longer wavelength. D. muricata extract was accountable for the synthesis of NPs and played a major role in the production. Natural product extracts acted as reducing agent for the generation of metal NPs. Previous finding suggested that the absorbance range for Au-NPs is 500-600 nm. In a reported study on biosynthesis of Au-NPs using Azimat etracantha leave extract showed absorbance band was between 535-550 nm[18]. Similarly Au-NPs were prepared with the help of Azadirachta indica (Neem) leaf extract, which showed absorbance peak at 560nm[19]. Literature review confirmed that the spectral analysis in the present study was consistent with previous findings. FTIR analysis was used to characterize the NPs and observed chemical changes in the functional groups of D. muricata extract, which interacted with Au ions as shown in fig. 4. The D. muricata extract exhibited many peaks of absorption, which revealed that the plant has a variety of compounds present in it. Spectra of plant extract and Au-NPs revealed bands, 2922 and 3040, 2856 and 2848, 1730 and 1626, 1619 and 1398 and 1376 and 1038 cm-1. The peaks at 1162, 1044, and 816 cm-1 found in the plant extract were absent in the NPs. This indicated that these functional groups interacted with Au ion and were responsible for reduction of Au3+ to Auº. The band at 2922 and 2856 cm-1 corresponded to asymmetric stretching of C-H groups while at 816 was due to C-H bending. The C=O stretch present at 1376 cm-1while C-C stretch present at 1619, 1162 and 1044 cm-1. The band at 1730 represented N-H stretching. The FT-IR spectrum of Au-NPs revealed shift in the C-H groups from 2922 to 3040 cm-1 and from 2856 to 2848 cm-1. N-H stretches shifted from 1730 to 1626cm-1, C-C stretching shifted from 1619 to 1398 cm-1, C=O stretching at 1370 was shifted in Au-NPs to 1038 cm-1. Through XRD analysis the exact nature and size of Au-NPs were studied. The strong and narrow diffraction peaks indicated that the product is crystalline in nature as shown in fig. 5. The XRD analysis showed peaks at 2θ=85°, 73°, 70°, 67° and 55° can be indexed to the (111), (200), (220) and (311), respectively. The formation of nanoparticles was confirmed by the broad Bragg’s peaks. By using simple aqueous reduction method it is possible to obtain monodispersed Au-NPs with controllable size and uniform shape. The mean size of Au-NPs was calculated by the Debye-Scherrer’s Eqn by determining the width of the (111) Bragg’s reflection[20]. The XRD peaks were assigned to diffraction from the (111), (200), (220), (311), (222) planes of face centered cubic (fcc) crystal structure. The peak corresponding to (111) was the more intense than the other planes suggesting that (111) was the predominant orientation and the synthesized Au-NPs were crystalline in nature. Similar results were reported previously on Au-NPs synthesis[21]. It was impossible to form anisotropic Au-NPs because of the symmetry breaking in fcc metals. Face centered cubic metals have tendency to nucleate and grow into cloned and multiple cloned particles with their surfaces bounded by the lowest-energy (111) facets on the nanometer scale[22]. It could be concluded from the SEM images that the size of the particles is 40-55 nm using image j software. The size was not in good agreement with the size as calculated from XRD graph using Debby Sherrers formula. Therefore, it was concluded that there was agglomeration between the nanoparticles as shown in fig. 6. The large grain size is due to the aggregation of particles. The overall morphology of synthesized nanoparticles is spherical in shape. In this study, the antibacterial activity of the plant extract and different ratios of Au-NPs prepared were studied against Enterobacter, Vibrio cholera, Klebsiella pneumonia, Streptococcus pyrogen and Citrobactor. Results obtained showed that ethanol extract of D. muricata as well as Au-NPs showed antibacterial activity. The results of antibacterial screening is presented in Tables 1 and 2. The maximum inhibition shown by 1:1 ratio against Citrobacter was 15 mm and 1:3 ratio against C. vibrio was also 15 mm while minimum inhibition was produced by 1:5 ratio against C. vibrio 6 mm and 1:6 against Citrobacter was 5 mm. The plant extract and the Au-NPs showed antibacterial activity against bacterial strains tested at the specified concentrations. A previous study reported antibacterial activity of Au-NPs synthesized with Caesalpinia pulcherrima extracts[23]. The Au-NPs found to be more effective against human pathogenic bacteria species as compared to the crude extract. These Au-NPsappear to have the potential to be used against bacterial infections.
| Plant extract | Control | Streptococcus pyrogen | Vibrio cholera | Klebsiella | Enterobacter | Citrobacter |
|---|---|---|---|---|---|---|
| 50µl | 0.0 | 10 | 11 | 9 | 12 | 7 |
Table 1: Antibacterial Effect of Plant Extract
| Zones of inhibition (mm) | |||||
|---|---|---|---|---|---|
| Test samples | Streptococcus pyrogen | Vibrio cholera | Klebsiella | Enterobacter | Citrobacter |
| 1:1 | 13 | 13 | 13 | 14 | 15 |
| 1:2 | 10 | 11 | 12 | 13 | 13 |
| 1:3 | 11 | 15 | 9 | 13 | 10 |
| 1:4 | 12 | 12 | 11 | 12 | 10 |
| 1:5 | 7 | 6 | 10 | 8 | 7 |
| 1:6 | 10 | 8 | 7 | 9 | 5 |
| Control | _ | _ | _ | _ | _ |
Table 2: Zones Inhibition Produced by Different Ratios of AU-NPS
Conflict of Interest
Authors declared that there is no conflict of interest.
References
- Rathjen D, Read L. Nanotechnology. Enabling technologies for Australian innovative industries. Paper prepared by an independent working group for the Prime Minister’s Science, Engineering and Innovation Council, PMSEIC. 2005.
- Veerapandian M, Yun K. Functionalization of biomolecules on nanoparticles: specialized for antibacterial applications. Appl Microbiol Biotechnol 2011;90(5):1655-67.
- Rao ML, Savithramma N. Biological synthesis of silver nanoparticles using Svensonia hyderabadensis leaf extract and evaluation of their antimicrobial efficacy.Int J Pharm Sci Rev Res 2011;3(3):1117.
- Song JY, Jang HK, Kim BS. Biological synthesis of gold nanoparticles using Magnolia kobus and Diopyros kaki leaf extracts. Process Biochem 2009;44(10):1133-8.
- Daniel MC, Astruc D. Gold nanoparticles: assembly, supramolecular chemistry, quantum-size-related properties, and applications toward biology, catalysis, and nanotechnology. Chem Reviews 2004;104(1):293-346.
- Ahmed KBA, Subramanian S, Sivasubramanian A, Veerappan G, Veerappan A.Preparation of gold nanoparticles using Salicornia brachiata plant extract and evaluation of catalytic and antibacterial activity.Spectrochim Acta A Mol Biomol Spectrosc 2014;130:54-8.
- Sheth AK, Mitaliya K, Joshi S. The herbs of Ayurveda. Vol-1. Published, Edited and distributed by Ashok Sheth. 2005.
- Kirtikar KR, Basu BD. Indian medicinal plants. 2nd ed. Bio-Green Books; 1918.
- Sharma N, Vijayvergia R. A Review on Digera muricata (L.) Mart-a great versatile medicinal plant. Int J Pharm Sci Rev Res 2013;20(1):114-9.
- Hariharan A, Begum TN, Ilyas MH, Jahangir HS, Kumpati P, Mathew S, et al. Synthesis of plant mediated gold nanoparticles using Azima tetracantha Lam. leaves extract and evaluation of their antimicrobial activities. Pharmacog J 2016;(8):5.
- Rahman MA, Begum K, Rashid ME, Harun-ur-Rashid M. Medicinal plant diversity in the flora of Bangladesh and their conser. Plant Archives 2012;12(2):1023-35.
- Shah A, Marwat SK, Gohar F, Khan A, Bhatti KH, Amin M, et al. Ethnobotanical study of medicinal plants of semi-tribal area of Makerwal & Gulla Khel (lying between Khyber Pakhtunkhwa and Punjab Provinces), Pakistan. Am J Plant Sci2013;4(1):98.
- Usmani S, Arshad H, Farooqui AHA, Mohammed A, Sahabjada S, Mohammed A, et al. Anti-proliferative Activity of Crude Extract and Fractions Obtained from Digera muricata on HeLa Cell Lines of Human Cervix and A549 Cell Lines of Human Lung. Pharmacog J 2014;(6): 32-8.
- Jayaseelan C, Rahuman AA, Kirthi AV, Marimuthu S, Santhoshkumar T, Bagavan A, et al. Novel microbial route to synthesize ZnO nanoparticles using Aeromonas hydrophila and their activity against pathogenic bacteria and fungi. Spectrochim. Acta A: Mol. Biomol. Spectrosc2012;90:78-84.
- Dorosti N, Jamshidi F. Plant-mediated gold nanoparticles by Dracocephalum kotschyi as anticholinesterase agent: Synthesis, characterization, and evaluation of anticancer and antibacterial activity. J Appl Biomed 2016;14(3):235-45.
- Rajeshkumar S, Malarkodi C, Gnanajobitha G, Paulkumar K, Vanaja M, Kannan C, et al. Seaweed-mediated synthesis of gold nanoparticles using Turbinaria conoides and its characterization. J Nanostructure Chem 2013;3(1):44.
- Dorosti N, Jamshidi F. Plant-mediated gold nanoparticles by Dracocephalum kotschyi as anticholinesterase agent: synthesis, characterization, and evaluation of anticancer and antibacterial activity. J Appl Biomed 2016; 14(3):235-45.
- Hariharan A, Begum TN, Ilyas MH, Jahangir HS, Kumpati P, Mathew S, Govindaraju A, Qadri I.Synthesis of plant mediated gold nanoparticles using Azima tetracantha Lam. leaves extract and evaluation of their antimicrobial activities. Pharmacog J 2016;8(5).
- Shankar SS, Rai A, Ahmad A, Sastry M. Rapid synthesis of Au, Ag, and bimetallic Au core–Ag shell nanoparticles using Neem (Azadirachta indica) leaf broth. Colloid Interfac Sci 2004;275(2):496-502.
- Borchert H, Shevchenko EV, Robert A, Mekis I, Kornowski A, Grübel G, et al.Determination of nanocrystal sizes: a comparison of TEM, SAXS, and XRD studies of highly monodisperse CoPt3 particles. Langmuir 2005;21(5):1931-6.
- Aromal SA, Philip D. Benincasa hispida seed mediated green synthesis of gold nanoparticles and its optical nonlinearity.Physica E Low Dimens Syst Nanostruct 2012;44(7-8):1329-34.
- Goubet N, Portalès H, Yan C, Arfaoui I, Albouy PA, Mermet A, et al. Simultaneous growths of gold colloidal crystals. J Am Chem Soc 2012;134(8):3714-3719.
- Nagaraj B, Divya TK, Malakar B. Phytosynthesis of gold nanoparticles using Caesalpinia pulcherrima (peacock flower) flower extract and evaluation of their antimicrobial activities. Dig J Nanomater Bios 2012;7(3):899-905.


 ) 1:1,(
) 1:1,( )1:2,(
)1:2,( )1:3,(
)1:3,( )1:4,(
)1:4,( )1:5,(▬) 1:6
)1:5,(▬) 1:6