- *Corresponding Author:
- M. S. Nagarsenker
Department of Pharmaceutics, Bombay College of Pharmacy, Kalina, Santacruz (East), Mumbai - 400 098, India
E-mail: mangal_na9511@yahoo.co.in
| Date of Submission | 3 February 2006 |
| Date of Revision | 30 January 2007 |
| Date of Acceptance | 16 May 2007 |
| Indian J Pharm Sci, 2007, 69 (3): 370-377 |
Abstract
Celecoxib surface solid dispersions were prepared by various techniques using different ratios of drug and carriers. Infra-red spectroscopy and differential scanning calorimetry showed no significant change in solid state of celecoxib in the surface solid dispersions and physical mixtures of celecoxib and carriers. Surface solid dispersions exhibited superior dissolution profiles and improved antiinflammatory activity in rat paw oedema model. The increase in the dissolution rate and consequent enhancement of antiinflammatory effect in rats of celecoxib were attributed to reduced particle size of celecoxib deposited on the surface of carrier and enhanced wettability of the drug particles brought about by the carrier. Improved dissolution characteristics were retained when compressed into tablets.
Keywords
Celecoxib, superdisintegrants, surface solid dispersion, antiinfl ammatory activity.
Celecoxib (CXB) is an anti inflammatory, analgesic, and antipyretic drug, used in the treatment of rheumatoid arthritis, osteoarthritis [1]. The aqueous solubility of CXB is 3 to 7 μg/ml when determined at pH 7.0 at 40º. Peak plasma concentration is reported three hours after oral dosing. CXB is evenly distributed in vivo and has a volume of distribution of 455±166 l in humans [2]. This larger volume of distribution and low aqueous solubility may be related to the lipophilic nature of CXB and be reflective of low bioavailability [1].
Different approaches are reported to improve the solubility of CXB. Use of cosolvents like ethanol, ethylene glycol, propylene glycol and polyethylene glycol has been tried [3]. Formulation of surface solid dispersion (SSD) using a hydrophilic carrier could aid in improvement of aqueous solubility and the bioavailability of the CXB. Cyclodextrins have been extensively used to improve the solubility of poorly water soluble drugs. Inclusion complexation of CXB with β-cyclodextrin and hydroxypropyl-β-cyclodextrin enhanced its aqueous solubility [4,5]. SSDs of CXB have been formulated with polymers such as polyvinyl pyrrolidone, polyethylene glycol, and polyvinyl pyrrolidone-vinyl acetate copolymer by fusion technique to improve its solubility [6]. Preparation of SSD, a technique that provides deposition of the drug on the surface of certain materials, can alter the dissolution characteristics of drug. Deposition of drug on the surface of an inert carrier leads to reduction in the particle size of drug, thereby providing faster rate of dissolution. Various hydrophilic materials with high surface area can be utilized to deposit the drug on their surface. The selection of carrier and method of preparation are critical factors influencing the properties of the drug incorporated in the SSD. Dissolution rate of griseofulvin was increased by depositing it on the surface of disintgrants such as Primogel, starch, Nymcel [7]. In other study, nifedipine was deposited on the surface of superdisintegrants such as Ac-Di-Sol, Kollidon and Explotab to increase its dissolution [8]. Solvent deposition technique was used to enhance the dissolution rate and antiinflammatory effect of piroxicam [9].
The objective of the present study was to formulate CXB dispersions to improve the aqueous solubility and dissolution rate of CXB to facilitate faster onset of action. In the present study, SSDs of CXB with superdisintegrants were prepared, characterized using differential scanning calorimetry (DSC) and infrared spectroscopy and evaluated for in vivo advantage in rat paw edema model. Different ratios of drug: polymers and different methods of preparation were tried and feasibility of formulating surface SSDs into tablet and capsules was also investigated.
Materials and Methods
CXB was a gift sample from Cheminor Drugs Ltd. Hydrabad, India. Croscarmellose sodium, Ac-Di-Sol, FMC Corp., USA (CS) was obtained as a free sample from Khandelwal Lab Mumbai, India, Crospovidone, Kollidon CL, BASF Germany, (CP), sodium starch glycolate, (PrimogelTM, Avebe UK Inc.) (SG), colloidal silicon dioxide (Cab-O-Sil, Cabot, Germany), talc IP were kindly donated by Sun Pharmaceutical Industries Ltd., Mumbai. All other reagents and solvents used were of analytical grade. Female Wistar rats were procured from Bharat Serums and Vaccines Ltd., Mumbai, India.
Hydration capacity of carriers
A sample of disintegrants weighing 2 g was placed in 100 ml centrifuge tube. Water, 40 ml was added; tube was stoppered and shaken vigorously to suspend the sample thoroughly. The suspension was allowed to stand for 10 min and then subjected for centrifugation at 1000×g for 15 min. The supernatant liquid was carefully decanted and the tube was inverted to allow draining. The tube with sediment was then restoppered and contents were weighed [10]. The hydration capacity was calculated using equation, hydration capacity=(weight of the tube with sediment)─(weight of the empty tube)/weight of the sample on dry basis.
Preparation of SSD by various methods
SSDs of the drug and hydrophilic carriers were prepared in ratios of 9:1, 4:1, 2:1, 1:1, 1:4 and 1:9 of drug:carrier. Two techniques used for preparation were co-evaporation; co-grinding. Physical mixtures were formulated for comparison. In co-evaporation, an accurately weighed quantity of CXB was dissolved in a minimum amount of ethanol in which hydrophilic carrier was suspended. The suspension was stirred using a magnetic stirrer continuously at room temperature until all the solvent evaporated. In cogrinding technique, CXB was triturated with minimum quantity of ethanol in a glass mortar until it dissolved. The carrier was then added and suspension was triturated rapidly at room temperature until the solvent evaporated. Physical mixtures were formulated by mixing drug and carrier in geometric proportion using spatula without applying pressure. The SSDs were prepared in triplicate and sieved through BSS # 85 (180 μ) and stored over anhydrous calcium chloride in a desiccator until further evaluation. The selected formulae are given in Table 1.
| SSD codes | Component | Ratio | Method |
|---|---|---|---|
| CSG – 11 CE | CXB: SG | 0.042361111 | Co evaporation method |
| CSG – 14 CE | CXB: SG | 0.044444444 | Co evaporation method |
| CSG – 19 CE | CXB: SG | 0.047916667 | Co evaporation method |
| CSG – 11 GR | CXB: SG | 0.042361111 | Grinding method |
| CSG – 14 GR | CXB: SG | 0.044444444 | Grinding method |
| CSG – 19 GR | CXB: SG | 0.047916667 | Grinding method |
| CSG – 21 GR | CXB: SG | 0.084027778 | Grinding method |
| CSG – 41 GR | CXB: SG | 0.167361111 | Grinding method |
| CSG – 91 GR | CXB: SG | 0.375694444 | Grinding method |
| CCP – 41 GR | CXB: CP | 0.167361111 | Grinding method |
| CCS – 41 GR | CXB: CS | 0.042361111 | Grinding method |
| CSG – 11 PM | CXB: SG | 0.042361111 | Physical mixing |
| CSG – 14 PM | CXB: SG | 0.044444444 | Physical mixing |
| CSG – 19 PM | CXB: SG | 0.047916667 | Physical mixing |
| CSG – 21 PM | CXB: SG | 0.084027778 | Physical mixing |
| CSG – 41 PM | CXB: SG | 0.167361111 | Physical mixing |
| CSG – 91 PM | CXB: SG | 0.375694444 | Physical mixing |
Table 1: Compositions And Codes of Different Ssds
Characterization of SSDs
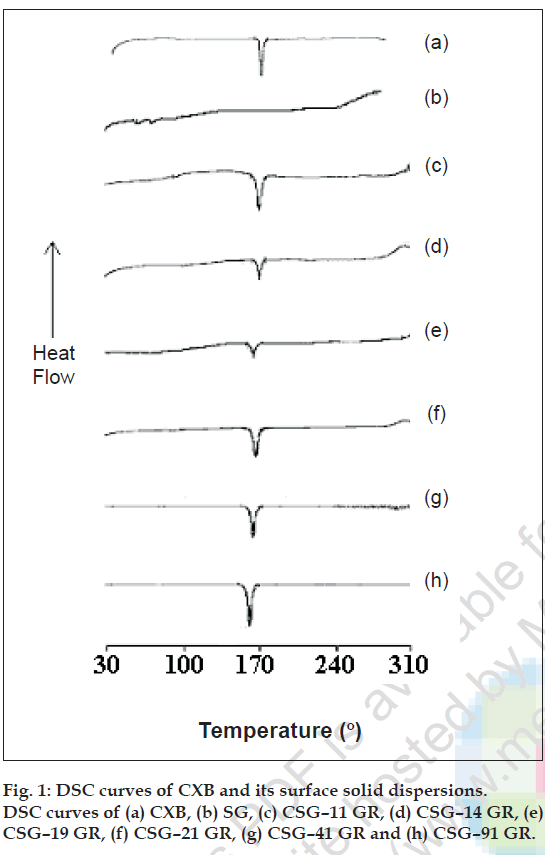
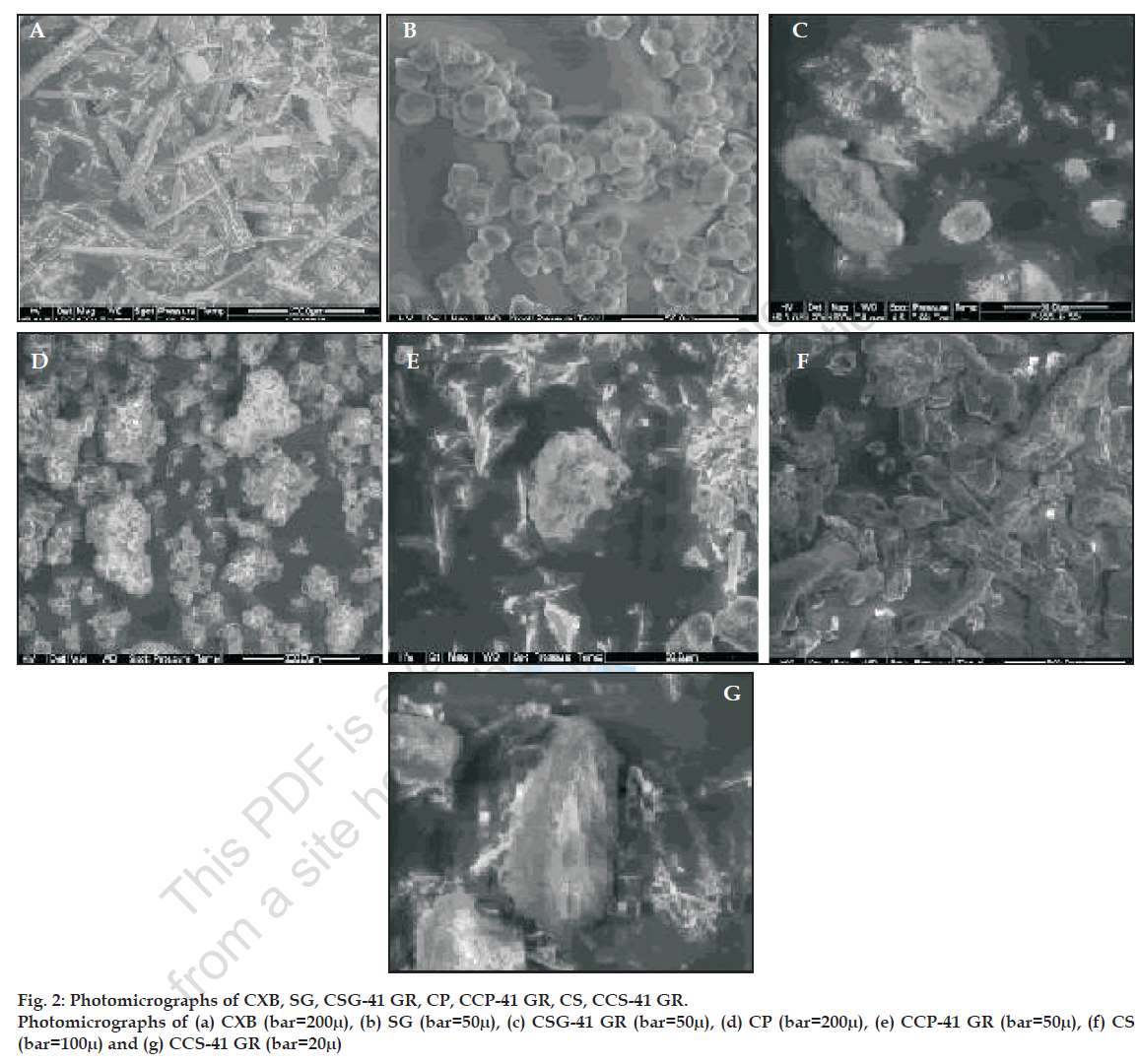
SSDs were characterized by infrared (IR) spectroscopy, differential scanning calorimetry (DSC), scanning electron microscopy (SEM) and optical microscopy. Infrared spectra of powder CXB and its SSDs were recorded on a Jasco FT/IR 5300 infrared spectrophotometer, by using KBr disc method. The scanning range was 450 to 4000 cm-1 and the resolution was 4 cm-1. DSC curves of the CXB, SG, and their SSDs were recorded using Shimadzu DT-40 Thermal analyzer. The samples were heated from 30º to 300º at a rate of 10º/ min in an open pan using alumina as a reference material. DSC thermograms of CXB, excipients and SSDs are given in fig. 1 Scanning electron microscopy of CXB, SG, CP, CS and their respective SSDs were done by Phillips FEI Quanta 200. The samples were fixed on SEM- stub using double sided adhesive tape. The photomicrographs are given in fig. 2. The particle size distribution of selected samples was studied by optical microscopy. Dimensions of 300-500 particles were measured.
In vitro release study
SSDs equivalent to 75 mg of CXB were subjected for drug release study in 600 ml simulated intestinal fluid (pH 6.8, SIF) without pancreatin but with 0.5% w/v tween 80 using USP type 2 apparatus (Electrolab, India) rotating at 50 rpm. Samples were withdrawn at predetermined time intervals and analyzed spectrophotometrically using Shimadzu 160A UV/Vis spectrophotometer at 258.2 nm. Drug release from selected physical mixtures was also studied for comparison.
Pharmacodynamic evaluation in rats
Animal study protocol was approved by the Institutional Animals Ethics Committee, IAEC/ CPCSEA Mumbai. The antiinflammatory activity was assessed on female Wistar rats (200-250 g). The animals were housed into groups of six and maintained on a standard diet with free access to water. Animals were fasted overnight before experiment. SSDs (equivalent to 5 mg/kg) or plain drug (5 mg/kg) were administered orally as aqueous suspensions containing 0.25% carboxymethyl cellulose (CMC). Control group was administered an aqueous dispersion of 0.25% CMC. Edema was induced by injecting 0.1 ml lambda carrageenan (1% w/v) into the plantar tissue of the hind paw. The volume of the treated paw was measured with an Ugo Basile 7140 plethysmometer, after 1, 2, 3, 4, 5 and 6 h after administration of carrageenan. The percent inhibition of edema was calculated [11].
Development of solid dosage form
Different formulae of tablets (Table 2) containing SSDs equivalent to 100 mg of drug were prepared to evaluate the effect of compression on the behavior of SSD. The blend was compressed using round shaped standard concave punches on 8-station single rotary machine (Cadmach, India) to obtain hardness of 4-5 Kp and 4.6±0.2 mm thicknesses. SSD (CSG-41 GR) equivalent to 100 mg of CXB was filled in capsule size 1 (CAP 1) and were compared with capsules of size 1 filled with physical mixture (CSG-41 PM) equivalent to 100 mg of CXB for comparison which was coded as CAP2. Tablets and the capsules were subjected to dissolution studies using simulated intestinal fluid containing 0.5% w/v tween 80 as a dissolution medium.
| Ingredients (mg) | CST 1(mg) | CST 2(mg) | CST 3(mg) | CST 4(mg) | CST 5(mg) |
|---|---|---|---|---|---|
| CSG – 41 GR | 133.5 | 126 | - | - | - |
| CCP – 41 GR | - | - | - | 134 | - |
| CCS – 41 GR | - | - | - | - | 131 |
| CSG – 41 PM | - | - | 135.5 | - | |
| Avicel PH102 | 114 | 112 | 113.5 | 116.5 | |
| Directly Compressible lactose |
- | 121.5 | - | - | - |
| Talc (1%) | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 |
Total tablet weight = 250 mg
Table 2: Formulae of Different Tablet Dosage Forms
Results and Discussion
Hydration capacity test is indicative of extent of solid liquid interactions or swelling power [Kornblum 1973]. In this test the material under test is wetted by water and evaluated for water holding ability. Hydration capacity of CS was found to be 11.0 which is superior to SG (9.18) and CP (3.71). Sample of CS exhibited quick water uptake in 10 min. As rapid water uptake is one of the requirements for inducing the disintegration, CS was anticipated to display superior performance.
IR spectroscopy was used to study the possible interaction between CXB and the carriers in the SSD. The FT IR spectra of all drug-carrier SSDs did not differ from that of CXB and displayed absorption bands as in IR spectra of CXB, in particular the characteristic sulfonamide-stretching band of CXB (1165 cm-1) was unchanged showing no evidence of significant interaction between drug and carrier (data not shown). The DSC thermograms of CXB, SG, its SSDs and physical mixtures are given in fig.1. The DSC thermogram of CXB exhibits sharp melting endotherm at 165.4º with onset at 159.8º and recovery at 172.9º. SG does not exhibit a melting endotherm as would be observed for crystalline materials, suggesting amorphous nature. In all the SSDs prepared with SG, melting endotherm of CXB was seen in the temperature range of 159.8º to172.9º suggesting that drug crystallinity was retained in the SSDs. Scanning electron photomicrographs of CXB showed plate shaped crystals. SEM studies on carriers showed SG particles moderately spherical shaped, CP as aggregates of irregular shaped particles and rod shaped CS particles. The photomicrographs of CSG-41 GR showed very small crystals of CXB uniformly deposited on the surface of SG. The CCS-41 GR also showed uniform deposition of small crystals on the surface of CS. However the CCP-41 GR had shown non uniform deposition of bigger crystals of drug on the surface of CP and frequent occurrence of free drug crystal in the sample. The arithmetic mean particle dimensions of CXB, carriers and selected dispersions are given in the Table 3. The particle size of SG was found to be smaller and uniform as compared to crospovidone and croscarmellose so also mean particle size of CSG-41 GR was the smallest in the dispersions studied.
| Sample | Arithmetic mean diameter (µm) |
|---|---|
| CXB | 177 |
| Sodium starch glycolate (SG) | 11.36 |
| Crospovidone (CP) | 17.03 |
| Croscarmellose (CS) | 21.7 |
| CSG-41 GR | 93 |
| CCP-41 GR | 108 |
| CCS-41 GR | 142.7 |
Table 3: Mean Particle Size of Cxb, Carriers And Ssds
CXB has low wettability and poor solubility in SIF pH 6.8 hence tween 80 (0.5% w/v) was incorporated in the dissolution medium as a wetting agent. Two parameters that is amount of drug dissolved at 15 min (DP15) and dissolution efficiency at 60 min (DE60) were computed from in vitro release profile. CXB due to its hydrophobic nature exhibited tendency to form large aggregates and floated on the surface of dissolution medium. The aggregation caused reduction in effective surface area of drug particles available for dissolution, resulting in 52% of DP15 and ~ 64% of DE60 (Table 4).
| Sample | DP15 | DE60 |
|---|---|---|
| CXB | 52.28±1.2 | 63.74 |
| CSG-11 GR | 88.13±0.47 | 82.53 |
| CSG-14 GR | 90.64±0.2 | 83.3 |
| CSG-19 GR | 87.58±2.4 | 81.38 |
| CSG-21 GR | 92.12±2.1 | 87.92 |
| CSG-41 GR | 93.65±2.3 | 91.07 |
| CSG-91 GR | 82.40±1.84 | 82.59 |
| CCP-41 GR | 77.67±1.09 | 79.85 |
| CCS-41 GR | 88.88±0.9 | 84.82 |
| CSG-11 PM | 66.93±3.28 | 67.75 |
| CSG-14 PM | 64.84±3.43 | 66.89 |
| CSG-19 PM | 78.83±2.27 | 78.05 |
| CSG-21 PM | 68.11±3.17 | 69.97 |
| CSG-41 PM | 67.60±1.99 | 67.97 |
| CSG-91 PM | 67.47±4.26 | 67.28 |
Table 4: Dissolution Parameters of Ssds
SSDs formulated using various carriers and different methods of preparation showed marked enhancement in the dissolution of drug as compared to plain drug powder and is clearly evident from the DP15 and DE60 values of pure drug and its SSDs, represented in Table 4. SSDs prepared using co-evaporation technique resulted in significant improvement in dissolution profile of the drug. CSG11-CE formulated with drug: SG ratio 1:1 showed DP15 (57.1±3.33%) comparable to 52% of pure drug powder. The increase in SG content in CSG14-CE and CSG19-CE resulted in marked increase in DP15 up to 86.22% and 94.16% respectively (data not shown in Table 4). This could be due to partial amorphization and deposition of the drug on the surface of carrier during the evaporation of solvent. However, during preparation of SSDs by co-evaporation method, agitation provided by magnetic stirrer was insufficient to prevent the settling of insoluble SG particles during the evaporation of solvent and precipitation of drug. There was non uniformity in distribution of the drug on the surface of carrier. Hence use of the method was discontinued and SSDs were prepared in mortar by co-grinding method.
The SSDs formulated by co grinding technique also showed improved dissolution profile as compared to plain drug powder (Table 4). The improvement in the dissolution rate of CXB could be attributed to uniform deposition of the drug particulates on the surface of hydrophilic carrier- SG and drug amorphization as evident from SEM (fig. 2). In addition, it was expected that the SG would absorb large amount of water when exposed to dissolution medium and swell as observed in hydration capacity test. This would result in wetting of small drug particulates deposited on the surface of SG. The swelling of SG would cause deaggregation of clusters of small drug particles and facilitate their dissolution. It was observed that amount of SG as low as 10% was sufficient to physically associate with drug particles due to relatively spherical shape of SG particles and provide better drug dissolution. Physical mixtures of drug with SG in the same ratio as in SSDs were formulated for comparison. Physical mixtures (drug to carrier ratios ranging from 9:1 to 1:9) also improved the dissolution rate of drug and DP15 values were found to be in the range of 65% - 79% but there was no improvement in the extent of drug release as indicated by DE60 values. (Table 5). This could be due to hydrophilic nature of SG, which rendered the mixture hydrophilic and improved the wettability. Dissolution profile of drug CSG-41 GR was the best with highest values of DP15 and DE60 among the SDDs. Therefore SSD with other carriers were prepared using drug: carrier ratio of 4:1. SDDs formulated with CP and CS, CCP-41 GR and CCS-41 GR, respectively, also showed better results as evident from higher values of DP15 and DE60 than that of pure drug. The improved dissolution could also be due to reduction in particle size of the drug, its deposition on the surface of carrier and improved hydrophillicity.
| Sample | DP30 | DE120 |
|---|---|---|
| CST 1 | 79.60±2.06 | 59.42 |
| CST 2 | 59.85±1.68 | 50.69 |
| CST 3 | 66.92±1.29 | 53.02 |
| CST 4 | 45.00±0.67 | 46.44 |
| CST 5 | 56.45±2.24 | 51.3 |
DP30 = Percent drug dissolved in 30 min. DE120: Dissolution efficiency at t = 120 min. Each value is the average of three determinations (coefficient of variation CV < 2.5%)
Table 5: Dissolution Parameters of Tablets
When SSDs formulated with different carriers were compared for release rate, CSG-41 GR was found to be superior compared to other carriers. This could be attributed to superior hydration capacity of SG and uniform deposition of very small drug particles on the relatively spherical shaped SG particles (fig. 2). The hydration capacity of croscarmellose was also found to be higher but the mean particle size of croscarmellose and its dispersion (CCS-41 GR) was bigger (Table 3) than SG and its dispersion (CSG-41 GR), which would have reduced the dissolution rate. Crospovidone showed very low hydration capacity as compared to SG and CS, which is correlated with lower values of DP15 and DE60. SEM studies revealed non uniform deposition of bigger crystals of drug on the surface of crospovidone and frequent occurrence of bigger crystals of drug without association with carrier in CCP-41 GR sample. All these factors resulted in lower release rate and dissolution efficiency compared to CSG-41 GR and CCS-41 GR.
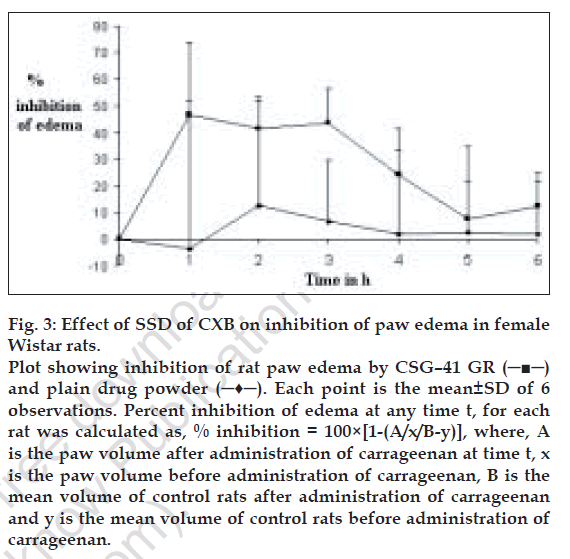
At a dose level of 5 mg/kg, the mean values of the % inhibition of rat paw edema produced by CSG-41 GR were higher at most of the time points as compared to those produced by plain drug. The higher values of inhibition at 1 hr indicates faster onset of action of the drug when administered as a SSD (fig. 3). The plain drug showed very low values of inhibition of edema at a dose level of 5 mg/kg. Limited aqueous solubility of CXB must have affected its availability and resulted in low values of inhibition of edema in rats treated with pure drug. The t test indicated significant difference at 3 hrs in inhibition of edema between groups treated with drug and SSD at 5% level of significance. Lower standard deviation values in case of response to CSG-41 GR in comparison with that for standard are indicative of better reproducibility of drug action when given as a SSD.
Figure 3: Effect of SSD of CXB on inhibition of paw edema in female
Wistar rats.
Plot showing inhibition of rat paw edema by CSG–41 GR (─■─)
and plain drug powder (─♦─). Each point is the mean±SD of 6
observations. Percent inhibition of edema at any time t, for each
rat was calculated as, % inhibition = 100×[1-(A/x/B-y)], where, A
is the paw volume after administration of carrageenan at time t, x
is the paw volume before administration of carrageenan, B is the
mean volume of control rats after administration of carrageenan
and y is the mean volume of control rats before administration of carrageenan.
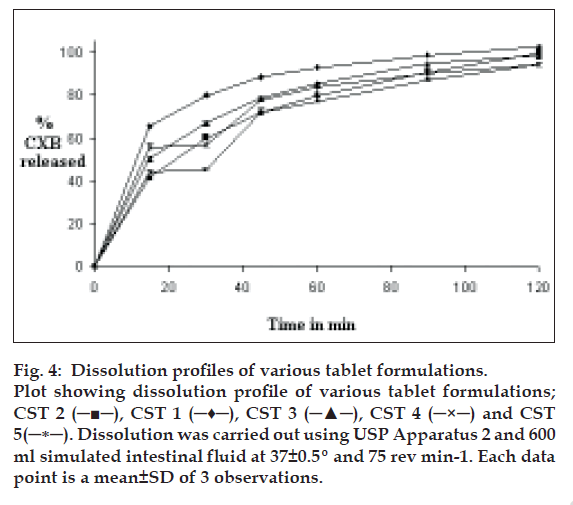
The nature and the properties of the excipients and dosage form influence the performance of SSD. Although SSDs of CXB were able to provide quick dissolution, its dissolution properties could be affected by the presence of diluents. Drug release pattern was studied after SSDs were mixed with two diluents and compressed. Disintegration of the tablets required 2 to 3 min that delayed the release of drug from tablets compared to SSD. DP30, amount of drug released in 30 min and DE120, dissolution efficiency, were computed from drug release profile. The data on dissolution parameters is given in Table 5 and the drug release profiles are shown in fig. 4 The tablets of formula CST 1 prepared using CSG-41 GR and microcrystalline cellulose as a diluent, released 79.60% in 30 min and had DE120 ~ 59%. The release of drug was slower than that of dispersion due to the additional disintegration step required prior to the dissolution of the drug. The formula CST 2 tablets in which CSG-41 GR was compressed using with lactose as a diluent had lower values of both DP30 and DE120. Faster release from CST 1 tablets in comparison with CST 2 tablets was attributed to swelling of microcrystalline cellulose in presence of water and its ability to hold high amount of water. This resulted in better wetting of drug particles in addition to that of wetting provided by SG and faster dissolution of the drug. It was observed that CST 2 tablets did not disintegrate by bursting in the dissolution medium but eroded slowly.
Figure 4: Dissolution profiles of various tablet formulations. Plot showing dissolution profile of various tablet formulations; CST 2 (─■─), CST 1 (─♦─), CST 3 (─▲─), CST 4 (─×─) and CST 5(─∗─). Dissolution was carried out using USP Apparatus 2 and 600 ml simulated intestinal fluid at 37±0.5º and 75 rev min-1. Each data point is a mean±SD of 3 observations.
Formulation CST 3, prepared by compressing CSG-41 PM was used for comparison. The DP30 and DE120 were found to be 67% and 53%. This was lower than CST 1 tablets but higher than CST 2 tablets. The higher release of CST 3 tablets than CST 2 tablets could be due to fast disintegration of the tablets and better wetting attributed to higher water holding capacity of microcrystalline cellulose than lactose.
Tablets of formulation CST 4 were found to release 45.00% in 30 min. This was significantly less than that released by CST 1 tablets. This is in line with hydration capacity of CP, which was lower than that of SG. Another possible reason could be non uniform deposition of bigger crystals of drug on the surface of CP and larger mean particle size of CCP-41 GR (Table 3) in comparison with mean particle size of CSG–41 GR. The dissolution parameters (DP30 and DE120) for CST 5 tablets were found to be 56.45% and 51%, which were inferior to that of CST 1 tablet. Though the swelling capacity of CS was found to be better than SG, particle size studies showed that CSG- 41 GR has smaller mean particle size than CCS-41 GR, (Table 3) which has affected drug release.
The release parameters of capsule dosage forms are given in Table 6. DP15 values for CAP 1 and CAP 2 formulation were 36.79% and 45.89%, respectively. After disintegration, CAP 2 showed better release profile than CAP 1. Drug release was apparently higher with CAP 2 formulation in 15 min, but not statistically different at 5% level of significance. But after 15 min, the CAP 1 formulation was showing higher drug release than CAP2 formulation at all time points. The difference in the release profile is statistically different at 5% level of significance at 30, 45, 60 and 90 min confirming improved release properties of SSD.
| Sample | DP15 | DP30 | DE120 |
|---|---|---|---|
| CAP 1 | 36.79±7.18 | 79.84±2.06 | 57.94 |
| CAP 2 | 45.89±3.36 | 71.78±1.68 | 54.2 |
Table 6: Dissolution Parameters of Capsules
In conclusion, SSDs of CXB with various superdisintegrants prepared by co-grinding techniques showed significantly higher drug dissolution in comparison with powder drug and physical mixtures having same drug to carrier ratio. IR and DSC studies showed no evidence of interaction between the drug and the carrier. SEM studies revealed deposition of the small drug particulates on the surface of carriers. in vivo studies of the SSDs in rats showed higher percentage inhibition of edema in the rats than that achieved with drug. This is attributed to improved bioavailability due to enhancement in rate and extent of drug release when drug was administered as SSD using SG as carrier. Compression of SSD into tablets resulted in slower drug release. Release profiles of tablets were affected by the excipients. Capsules filled with SSD exhibited enhanced release compared to the capsules filled with physical mixture having the same drug to carrier ratio as in the dispersion.
Acknowledgements
The authors wish to acknowledge Indian Institute of Technology for SEM studies.
References
- Davies NM, McLachlan AJ, Day RO, Williams KM. Clinical pharmacokinetics an pharmacodynamics of celecoxib: A selective cyclo-oxygenase-2 inhibitor. Clin Pharmacokinet 2000;38:225-42.
- Paulson SK, Vaughn MB, Jessen SM, Lawal Y, Gresk CJ, Yan B, et al. Pharmacokinetics of celecoxib after oral administration in dogs and humans: Effect of food and site of absorption. J Pharmacol Exp Ther 2001;297:638-45.
- Seedher N, Bhatia S. Solubility enhancement of Cox-2 inhibitors using various solvent systems. AAPS Pharm SciTech 2003;4:E33.
- Reddy MN, Rehana T, Ramakrishna S, Chowdary KP, Diwan PV. β-cyclodextrin complexes of celecoxib: Molecular- modeling, characterization, and dissolution studies. AAPS Pharm Sci 2004;6:E7.
- Nagarsenker MS, Joshi MS. Celecoxib-cyclodextrin systems: Characterization and evaluation of in vitro and in vivo advantage. Drug Develop Ind Pharm 2005;31:169-78.
- Devi VK, Vijayalakhmi P, Avinash M. Preformulation Studies on Celecoxib with a view to improve bioavailability. Indian J Pharm Sci 2003;65:542-545.
- Law SL, Chiang CH. Improving dissolution rate of Griseofulvin by deposition on disintegrants. Drug Develop Ind Pharm 1990;16:137-47.
- Yen SY, Chen CR, Ling MT, Chen L. Investigation of dissolution enhancement of Nifedipine by deposition on the superdiintegrants. Drug Develop Ind Pharm 1997;23:313-7.
- Barzegar JM, Maleki N, Garjani A, Khandar AA, Haji M, Hosseinloo M, et al. Enhancement of dissolution rate and antiinßammatory effects of Piroxicam using solvent deposition technique. Drug Develop Ind Pharm 2002;28:681-6.
- Kornblum SS, Stoopak SB. A new tablet disintegrating agent: Cross linked polyvinyl pyrrolidone. J Pharm Sci 1973;62:43-9.
- Vogel HG, editor. Anti inflammatory activity: Paw edema. Drug discovery and evaluation: pharmacological assays. 2nd ed. Germany; Springer-Verlag Publishers: 1997. p. 759-62.