- *Corresponding Author:
- Y. Zhen
Department of Orthopaedics, Children’s Hospital of Soochow University, Suzhou Industrial Park, No.92 Zhongnan Street, Suzhou, Jiangsu Province, China
E-mail: yunfangfzhen_suda@163.com
| This article was originally published in a special issue, |
| "Clinical and Experimental Studies on Drug and Intervention Repurposing in China" |
| Indian J Pharm Sci 2019:81(4)spl issue1;150-155 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
The objective of this study was to investigate the role of AMPK/P53-CyPD signaling pathway in osteoblast apoptosis induced by dexamethasone and to confirm that the activation of adenosine monophosphate-activated protein kinase signaling pathway can delay the progression of osteonecrosis of the femoral head and promote the regeneration of osteoblasts. In this study, 24 C57BL/6J 8-week old mice were selected as research subjects, which were randomly divided into the model group, the blank group and the intervention group, with 8 mice in each group. The model group was given subcutaneous injection of dexamethasone, the blank group was given subcutaneous injection of the same dose of normal saline, and the intervention group was given intraperitoneal injection of cyclophilin D (CyPD) inhibitor and P53 inhibitor on the basis of the model group. Eight weeks after the experiment, all mice were sacrificed, femoral head samples were taken for nuclear magnetic resonance and micro CT scanning, and the femoral head tissue samples were made for H and E staining to analyze the effect of inhibiting the AMPK/P53-CyPD signaling pathway on the femoral head structure in mice. Then, the AMPK α-expressing osteoblast cell line was constructed by exogenously adding the AMPK activator acadesine to the femoral head tissue of the model group. The cell survival cycle and apoptosis of osteoblasts were observed. The results showed that firstly, the morphology of the femoral head in the model group changed, the bone trabecula became thinner, the continuity was destroyed and the subchondral bone was partially necrotic. The morphology of the femoral head in the intervention group was significantly changed, the bone density decreased and the bone marrow cavity increased significantly and the structure of the normal femoral head was lost. The number of bone cells in the lacuna was significantly decreased. Secondly, after the exogenous addition of acadesine, the AMPK signaling pathway was activated, the apoptosis was gradually improved and the formation of osteoblasts was significantly increased. It could be concluded that the AMPK/P53-CyPD signaling pathway plays an important role in osteoblast apoptosis induced by dexamethasone, which can inhibit osteoblast apoptosis, delay the process of femoral head osteonecrosis, and promote the recovery of femoral head osteonecrosis.
Keywords
AMPK signalling pathway, mitochondrial P53-CyPD, femoral head osteonecrosis, dexamethasone, osteoblast
Osteonecrosis of the femoral head (ONFH) is a common orthopaedic disease caused by a variety of causes of apoptosis of bone cells and abnormal metabolism of fat cells, resulting in partial or complete ischemic necrosis of the femoral head[1]. ONFH usually causes changes in bone morphology, which causes femoral head collapse and osteoarthritis. Therefore, femoral head osteonecrosis is also the main cause of hip joint damage[2]. The pathogenesis of ONFH is still unclear. Trauma, alcoholism, and hormones may cause ONFH. The femoral head osteonecrosis caused by hormones is the most common non-traumatic cause, which is more common in young people.
Application of large doses of hormonal drugs in the short term, such as intra-articular injection of glucocorticoids can cause bone cell apoptosis due to a series of pathological changes, leading to the occurrence of ONFH[3]. In the early stage of the disease, the lesions mostly occur in the subchondral bone. As the disease progresses further, the cartilage collapses in the load-bearing area of the femoral head occurs, and finally the hip osteoarthritis. In the 1930s, studies reported that high-dose and continuous use of hormonal drugs may have an effect on bone tissue[4]. Since then, more and more cases have suggested that there is a certain relationship between hormones and ONFH. However, due to individual differences and the different courses of treatment and dosage of different hormones, the degree of osteonecrosis varies.
AMPK is an adenylate-activated protein kinase, and the catabolic pathway also occurs when the AMPK signalling pathway is activated. Studies have found that by activating the AMPK signalling pathway, mouse embryonic osteoblastic precursor (MC3T3-E1) cells can be induced to differentiate, thereby stimulating the proliferation and differentiation of osteoblasts[5]. Therefore, regulation of the AMPK signaling pathway can affect the differentiation of osteoblasts. In addition, P53 gene also has certain effects on apoptosis, regulation and metabolism in cells, and P53 protein can play a crucial role in regulating the differentiation and development of osteoblasts through various signalling pathways[6]. Based on the above theory, this study constructed a model of dexamethasone-induced ONFH in mice, and analysed the effect of inhibiting AMPK/ P53-CyPD signalling pathway on apoptosis of bone cells; and then the mouse AMPK with high α expression osteoblast cell line was constructed by exogenously adding AMPK activator acadesine (5-aminoimidazole- 4-carboxamide riboside, AICAR) and the effect of activating AMPK/P53-CyPD signalling pathway on osteoblast apoptosis induced by dexamethasone was analysed.
A total of 24 healthy 8 w old C57BL/6J mice of SPF grade were selected, weighing 20.8±4.3 g, and housed under standard laboratory conditions, the temperature was controlled at 22~24°, and the humidity was controlled at 30~40 %. All mice were randomly divided into 3 groups, the model group, the blank group and the intervention group, with 8 mice in each group. The model group received subcutaneous injection of 20 mg/kg dexamethasone was, once a day for 8 w. The blank group received subcutaneous injection of same volume of physiological saline once a day for 8 w. The intervention group was given dexamethasone+CyPD inhibitor (cyclosporine A, CsA)+P53 inhibitor (PTFα). CsA was injected intraperitoneally at 10 mg/kg, and PTFα was intraperitoneally injected at 2 mg/kg, once a day for 8 w. All mice were given intramuscular injection of gentamicin sulphate to prevent infection during the experiment, 50 000 U/time, once a week. After successful model construction, all mice were fed with standard mouse feed, and given free access to drink water. The position of the drinker was raised, allowing the mice to be in a standing position while drinking water, increasing the load on the femoral head to promote the establishment of a ONFH model. The mental state, diet, weight, activity and urination of the mice were closely observed during modelling.
Eight weeks after the experiment, all mice were sacrificed by cervical dislocation. One side of the femoral head was taken out and immersed in 10 ml of a 10 % formalin in a centrifuge tube, and stored in a refrigerator. Micro CT scan (80 kV; 500 μA; resolution 10 μm) was performed on the femoral head samples of mice to obtain continuous scan images. After the scan, the bone trabecula and bone marrow cavity, the bone volume and total volume (BV/TV) scores, thickness and number of bone trabecula, bone area to bone volume ratio (BS/BV) and other results were quantitatively analysed by computer software.
Preparation of specimens of ONFH, firstly, the mouse femoral head after micro CT scanning was placed in a centrifuge tube for decalcification, which was considered to be successful when a fine needle can penetrate the bone tissue without resistance. Secondly, the femoral head specimens were dehydrated in ethanol at a concentration gradient of 70, 85, 90 %, and finally treated with 100 % ethanol. Thirdly, xylene was used to hyalinize the femoral head sample. Fourthly, the transparent specimen was immersed in the paraffin liquid. Fifthly, after paraffin embedding, the femoral head tissue of the mouse was sectioned along the coronal plane of the femoral neck at a thickness of 4 μm, and separated after warm water bath. Sixthly, the section was placed at the center of the glass slide and then placed in the oven for bake. After that, it was stored at room temperature.
Hematoxylin and eosin (HE) staining of rat femoral head tissue was performed after dewaxing. The above sample was sliced and baked, then put into xylene and dewaxed for about 15 min. Secondly, it were placed in 100, 95, 85 % alcohol in sequence and dehydrated for 3 min. Thirdly, hematoxylin was used for staining for 3~5 min, and then the section was washed with running water. Fourthly, the section was differentiated in 1 % hydrochloric acid alcohol and put into the warm water returned to blue and then counterstained with ponceaux acid fuchsin for 3 min. Fifthly, after dehydration in alcohol, it was soaked in xylene, sealed with neutral gum and dried.
The general condition of each group of mice, including body weight, activity, diet was observed during the experiment. The results of nuclear magnetic resonance and micro CT scan of each group of mice were used to evaluate the model of femoral head osteonecrosis in mice. HE staining results of femoral head tissue of each group of mice was observed, focusing on the structural changes of bone trabecula. Osteoblast growth, cell necrosis and apoptosis after activation of AMPK pathway in mouse model group was tested using an ELISA assay.
The study was performed using SPSS 20.0 statistical software. The data were expressed as mean±standard deviation and the data were verified with homogeneity test of variance. If p>0.05, the variance is equal, and the comparison between the 2 means is analysed using one-way analysis of variance; if p<0.05, the variance is not uniform, then the one-way analysis of variance is corrected by Welch; the multivariate analysis is performed by LSD method for comparison between two pairs, p<0.05 is considered as statistically different.
In general, the mouse model of early ONFH is evaluated by imaging and pathological examination. Imaging examinations include X-ray, nuclear magnetic resonance and micro CT and so on. However, X-rays have a poor effect on displaying early changes in the femoral head of mice, and have limited diagnostic value for early ischemic necrosis of femoral head, so magnetic resonance imaging and micro CT were adopted as the means of modelling evaluation in this study, which can help clearly observe the bone trabecula and surrounding structures.
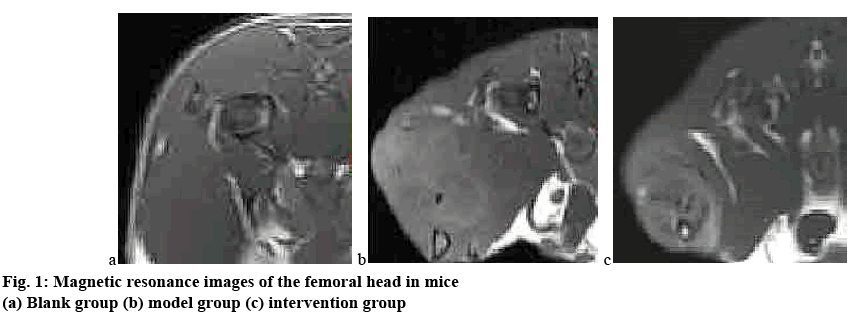
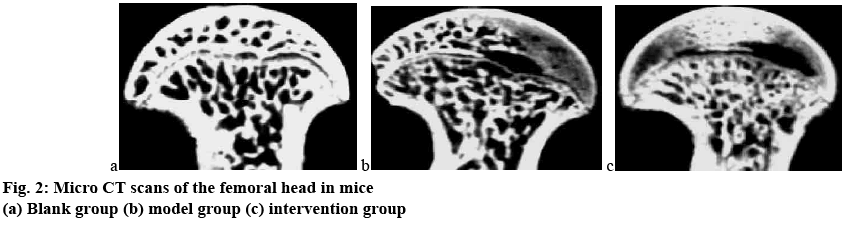
By observing the general condition of the mice, it was found that in the modelling process of this experiment, the gait of the blank group was normal and the comprehensive condition was good; while in the model group and the intervention group, hair loss occurred between 4 w and 5 w, the body weight was slightly reduced, and the activity was reduced; at 8 w, the hair loss of the model group and the intervention group was aggravated, the food intake was reduced, and the mice in the model group showed mild lameness, and the mice in the intervention group were severely paralyzed, and most of the mice were unable to walk. Micro CT is an important method for the early diagnosis of femoral head osteonecrosis under the action of hormones. It has high sensitivity and accuracy, so it was used in this study as an evaluation method for the establishment of a model of femoral head osteonecrosis in mice. Figs. 1 and 2 show the results of magnetic resonance and micro CT scan of the femoral head after 8 w of experiment in all 3 groups of mice.
It can be clearly observed from figs. 1 and 2 that the femoral head of the blank group had normal structure, regular shape, regular bone trabecula arrangement. There was no abnormal signal in the femoral head, and the hip joint was normal. In the model group, the morphology of the femoral head was changed. The patchy irregular signal was seen in the femoral head. The bone trabecula became thinner, the structure was loose and sparse, the subchondral bone was partially necrotic, and the bone marrow cavity was formed; the structure of hip joint was normal. In the intervention group, the morphology of the femoral head was significantly changed, the bone density decreased, the cavity of the bone marrow cavity increased significantly, and the malunion of bone trabecula fracture occurred in the weight-bearing area, and the normal femoral head tissue structure was lost. The difference between the model group and the intervention group was statistically significant (p<0.05).
It can be confirmed that dexamethasone, when used in large doses would destroy the bone structure, change the blood coagulation state, cause vascular disease and bone tissue ischemia, and eventually lead to necrosis of the femoral head. Therefore, the mouse ONFH model can be successfully constructed by subcutaneous injection of dexamethasone. Both AMPK and P53-CyPD signalling pathways are involved in the regulation of osteoblast growth, development, and apoptosis, and play an important role in responding to cytokine responses. In accordance with previous clinical trials and animal experiments, cyclosporin A and PTFα can reduce bone mineral density and bone mineralization. In this study, exogenous addition of cyclosporine A and PTFα to inhibit AMPK and P53-CyPD signalling pathways along dexamethasone administration can accelerate the apoptosis of mouse osteoblasts and accelerate the process of ONFH. Therefore, it can be concluded that AMPK and P53- CyPD signalling pathway play an important role in the apoptosis of osteoblasts.
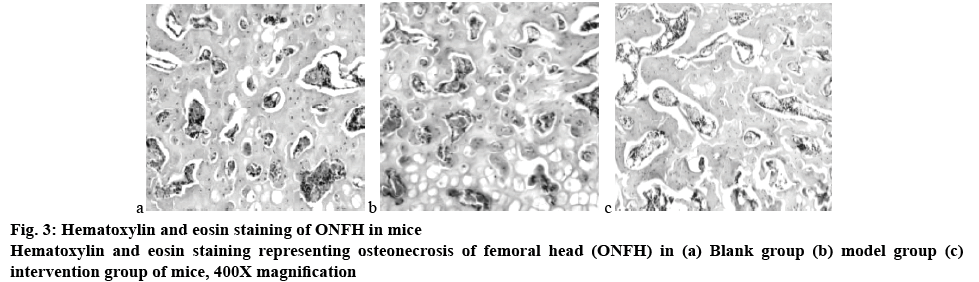
The results of pathological examination are the gold standard for the diagnosis of osteonecrosis. The main pathological marker of dexamethasoneinduced avascular necrosis of the femoral head is the disappearance of bone cells in the lacuna. It can be seen from fig. 3 that in this study, the bone trabeculae of the blank group were intact and arranged in an orderly manner; the bone trabecula of the model group was significantly thinner and sparse, and some of the bone trabeculae were continuously damaged during the fracture process. The bone trabecula structure of the mice in the intervention group was obviously disordered, the cells were arranged irregularly, and the dense cortex-like structure appeared, and the number of bone cells in the lacuna decreased significantly. The difference between the model group and the intervention group was statistically significant (p<0.05). It can be concluded that dexamethasone had a certain destructive effect on the structure of bone trabecula and together with the inhibition of AMPK and P53-CyPD signal transduction pathway can accelerate the process of bone tissue necrosis, and the mouse model of ONFH was constructed in cooperation with dexamethasone.
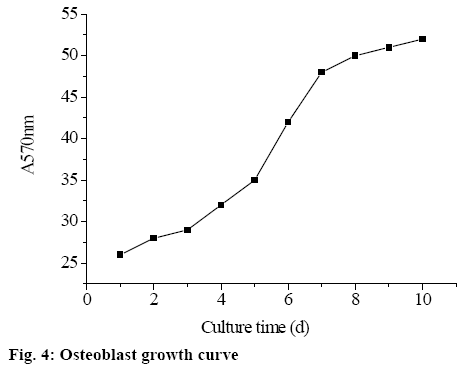
In this study, AMPK/P53-CyPD signalling pathway was activated by exogenously adding AMPK activator AICAR to mouse model tissue of dexamethasoneinduced ONFH (model group), and the role of AMPK/ P53-CyPD signalling pathway in inhibiting apoptosis of mouse osteoblasts was analysed. The study found that after exogenous addition of AICAR, osteoblasts grew slowly in the first 3 d. From the fourth day, the growth rate of osteoblasts increased significantly, suggesting that the cells entered the logarithmic growth phase, of which on the 4th to 7th d, the trend of logarithmic growth was observed. On the 8th d, the growth of osteoblasts again became flat, suggesting that cell growth entered the plateau. The osteoblast growth curve is shown in fig. 4.
The absorbance after ELISA can indirectly indicate the apoptosis of osteoblasts. The higher the absorbance, the higher the apoptotic rate of osteoblasts. The results showed that the mice cells in the model group had a higher degree of apoptosis; while after the exogenous addition of AMPK activator AICAR, the apoptosis was gradually improved. Thus, in the process of combating apoptosis, the AMPK signalling pathway can be activated by growth factors and certain extracellular signals, regulating cell proliferation and differentiation. In this experiment, it was found that after the exogenous addition of AICAR, the AMPK signalling pathway was activated, which can delay the appearance of osteoblast aging, reduce inflammation, inhibit vascular endothelial cell apoptosis, and improve the bone density of the femoral head of mice and has a protective effect on the apoptosis of osteoblasts.
Ischemic osteonecrosis is a pathological process accompanied by a decrease in blood supply to the bone, an increase in intra-osseous pressure, especially ONFH[7,8]. It is the result of a combination of traumatic and non-traumatic factors, and is the compensatory response of femoral head. With the collapse of osteonecrosis, it indicates the development and deterioration of the disease, and also determines that ONFH usually leads to osteonecrosis of the hip joint. Therefore, in the long run, total hip replacement arthroplasty is an inevitable choice. According to the survey, about 8 to 15 % of patients with ONFH in developed countries choose total hip replacement arthroplasty.
In recent years, with the changes in people's lifestyles and the widespread use of hormones, one of the serious consequences is the ischemic ONFH and its incidence is increasing year by year. High-dose, continuous use of hormonal drugs may have an effect on bone tissue, which may lead to the apoptosis of osteoblasts, fat cells, in the femoral head, thereby causing a series of pathological changes such as tissue ischemia. By regulating AMPK and P53-CyPD signalling pathway, it has certain influence on hormone-induced osteoblast apoptosis. In this paper, this was used as an entry point to further analyse the mechanism of activation of AMPK/P53-CyPD signalling pathway to inhibit apoptosis and promote cell formation.
In this study, the commonly used hormone dexamethasone was used to induce this condition. When used in large doses and continuously, it will destroy the bone structure, change the blood coagulation state, cause vascular disease and bone tissue ischemia, and eventually lead to ONFH. By exogenously adding cyclosporine A and PTFα to inhibit AMPK and P53-CyPD signalling pathways based on the administration of dexamethasone, it can accelerate the apoptosis of mouse osteoblasts and accelerate the process of ONFH. Apoptosis was gradually improved by exogenously adding AMPK activator AICAR to dexamethasone-induced ONFH. Therefore, it was concluded that AMPK and P53-CyPD signalling pathway play an important role in osteoblast apoptosis induced by dexamethasone, which not only can inhibit osteoblast apoptosis, delay the process of femoral head osteonecrosis, but also promote osteoblast production, thereby can promote the recovery of ONFH.
Acknowledgements
This work was supported by Jiangsu Science and Technology Office (Grant No: BE2016674).
References
- Kim SY, Jeong HC, Hong SK, Lee MO, Cho SJ, Cha HJ. Quercetin induced ROS production triggers mitochondrial cell death of human embryonic stem cells. Oncotarget 2017;8(39):64964-73.
- Tang XF, Liu HY, Wu L, Li MH, Li SP, Xu HB. Ginseng Rh2 protects endometrial cells from oxygen glucose deprivation/re-oxygenation. Oncotarget 2017;8(62):105703-13.
- Yoshida GJ. Therapeutic strategies of drug repositioning targeting autophagy to induce cancer cell death: from pathophysiology to treatment. J Hematol Oncol 2017;10(1):67.
- Troncone M, Cargnelli SM, Villani LA, Isfahanian N, Broadfield LA, Zychla L, et al. Targeting metabolism and AMP-activated kinase with metformin to sensitize non-small cell lung cancer (NSCLC) to cytotoxic therapy: translational biology and rationale for current clinical trials. Oncotarget 2017;8(34):57733.
- de Almeida AJPO, Ribeiro TP, de Medeiros IA. Aging: Molecular Pathways and Implications on the Cardiovascular System. Oxid Med Cell Longev 2017;2017:7941563.
- Mogavero A, Maiorana MV, Zanutto S, Varinelli L, Bozzi F, Belfiore A, et al. Metformin transiently inhibits colorectal cancer cell proliferation as a result of either AMPK activation or increased ROS production. Sci Rep 2017;7(1):15992.
- Kamiya N, Yamaguchi R, Aruwajoye O, Adapala NS, Kim HK. Development of a mouse model of ischemic osteonecrosis. Clin Orthop Relat Res 2015;473(4):1486-98.
- Kamiya N, Kuroyanagi G, Aruwajoye O, Kim HKW. IL6 receptor blockade preserves articular cartilage and increases bone volume following ischemic osteonecrosis in immature mice. Osteoarthritis Cartilage 2019;27(2):326-35.