- *Corresponding Author:
- S. B. Wankhede
University department of pharmaceutical sciences, Nagpur university, Nagpur - 440 033, india
E-mail: sagar2277@rediffmail.com
| Date of Submission | 7 November 2005 |
| Date of Revision | 26 July 2006 |
| Date of Acceptance | 11 April 2007 |
| Indian J Pharm Sci, 2007, 69 (2): 298-300 |
Abstract
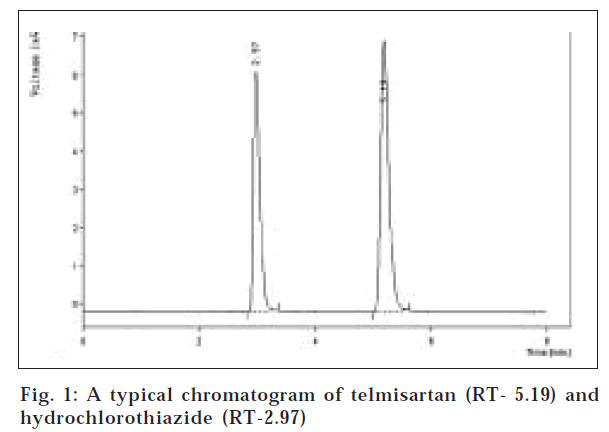
The present work describes a validated reverse phase high performance liquid chromatographic method for simultaneous estimation of telmisartan and hydrochlorothiazide in tablet formulation. Chromatography was performed on a ODS Hypersil C18 (25 cm×4.6 mm I.D) column from thermo in isocratic mode with mobile phase containing acetonitrile:0.05 M KH2PO4 pH 3.0 (60:40). The flow rate was 1.0 ml/min and the eluent was monitored at 271 nm. The selected chromatographic conditions were found to effectively separate telmisartan (RT- 5.19 min) and hydrochlorothiazide (RT- 2.97 min). Linearity for telmisartan and hydrochlorothiazide were found in the range of 4.1-20.48 µg/ml and 1.28-6.4 µg/ml, respectively. The proposed method was found to be accurate, precise, reproducible and specific and can be used for simultaneous analysis of these drugs in tablet formulation.
Telmisartan is 41-[1,41-dimethyl-21-propyl-[2,61-bi-1H-benzimidazole]-11-yl)methyl][1,11-biphenyl]-2-carboxylic acid and hydrochlorothiazide is 6-chloro-3,4-dihydro-2H-1,2,4-benzothiadiazine-7-sulphonamide-1,1-dioxide. Telmisartan is a new angiotensin II receptor antagonist for the treatment of essential hypertension usually given in combination with hydrochlorothiazide. The combination is useful in the treatment of mild to moderate hypertension, well tolerated with a lower incidence of cough than ACE inhibitors. The marketed tablet formulation contains telmisartan and hydrochlorothiazide in the ratio of 40:12.5 mg. Literature survey revealed linear sweep polarography [1], parallel catalytic hydrogen wave method [2], and HPLC [3] method for estimation of telmisartan alone in pharmaceutical preparations. Hydrochlorothiazide in combination with other drugs is reported to be estimated by spectrophotometric [4,5], HPLC [6,7] methods. The analytical method for simultaneous determination of telmisartan and hydrochlorothiazide in pharmaceutical formulation has not been reported so far. The present work describes a validated reverse phase HPLC method for simultaneous determination of these drugs in tablets.
An isocratic HPLC system (Shimadzu) consisting of LC10AT liquid pump, Rheodyne injector (2E, 7725; 20μl-loop), SPD-10A UV/Vis detector, an ODS Hypersil C-18 RP column (25 cm×4.6 mm ID), 25 μl Hamilton injecting syringe and window based single channel software was used. Pure drug samples of telmisartan and hydrochlorothiazide were procured from Aristo Pharmaceuticals Ltd., Raisen. The tablet formulations Telma-H (Glenmark) and Telpress-H (Nicolas Piramal) with a labeled claim of 12.5 mg telmisartan and 40 mg hydrochlorothiazide were obtained from local drug stores. The solvents acetonitrile and methanol used in the investigation were of HPLC grade (Merck). Potassium dihydrogen phosphate (0.05 M) was prepared by dissolving 6.8045 g in 1000 ml of double distilled water and the pH was adjusted to 3.0 by using orthophosphoric acid. Buffer (400 ml) and acetonitrile (600 ml) were mixed and filtered through 45 μ filter paper and sonicated.
Accurately weighed quantity of telmisartan (25.6 mg) and hydrochlorothiazide (8 mg) was transferred to a standard 50 ml volumetric flask, dissolved and diluted to the mark with methanol. The so prepared stock solution was further diluted with mobile phase to get a final concentration range of 4.10-20.48 μg/ml of telmisartan and 1.28-6.4 μg/ml of hydrochlorothiazide. Plotting a graph of peak area vs. concentration allowed the checking of linearity of detector response.
Assay was performed on two different marketed formulations. Twenty tablets were weighed and powdered. Accurately weighed quantity of tablet powder equivalent to about 25 mg of telmisartan was transferred to 50.0 ml volumetric flask, added 25 ml of methanol and shaken for ten minutes, the volume was then adjusted to mark with methanol and mixed. The solution was filtered through whatman filter paper no. 41 and the filtrate was then appropriately diluted with mobile phase to get a final concentration of 8 μg/ml of telmisartan and 2.5 μg/ml of hydrochlorothiazide. Twenty micro liters of sample preparation was injected into injector of liquid chromatograph and chromatography was performed on ODS Hypersil C-18 RP column (25 cm×4.6 mm I.D) using a mixture of acetonitrile:0.05 M KH2MPO4 (60:40 v/v) as mobile phase. The eluent was monitored at 271 nm. A typical chromatogram of telmisartan and hydrochlorothiazide is shown in fig. 1. Each sample was injected and analysed in triplicate. From the mean peak area of telmisartan and hydrochlorothiazide the amount of drug in tablet was computed (Table 1).
| Tablet formulation | Label claim (mg/tablet) | Amount of drug estimated* (mg/tablet) | % of label claim*±SD | |||
|---|---|---|---|---|---|---|
| TEL | HTZ | TEL | HTZ | TEL | HTZ | |
| Telma-H | 40 | 12.5 | 39.66 | 12.498 | 99.15 | 99.98 |
| ± 0.89 | ±0.88 | |||||
| Telpress-H | 40 | 12.5 | 40.472 | 12.312 | 101.18 | 98.5 |
| ±0.85 | ±0.37 | |||||
*mean of five observations
Table 1: Results of analysis of tablet formulation
To study the accuracy, precision and reproducibility of the proposed method recovery experiments were carried out. To an accurately weighed quantity of pre-analyzed tablet powder, standard telmisartan and hydrochlorothiazide (in solution form) were added at four different levels (about 10, 15, 20 and 25% of labeled claim). The amount of drug recovered by the proposed was found out. The results of recovery studies are shown in Table 2. Specificity studies were carried out by deliberately degrading the tablet sample. The stress conditions applied were acidic (0.1 M HCl), alkaline (0.1 M NaOH) and oxidizing condition (3% H2O2) at 50° for 24 h. Also heat (60°) and UV-exposure for 24 h was studied. After exposing the samples to above mentioned stress conditions for 24 h, the samples were diluted, filtered and analyzed in similar manner as described under assay of marketed formulation. The results obtained under different stress conditions are as follows:acidic (telmisartan- 99.63% and hydrochlorothiazide-98.24%), in alkaline condition (0.1 N NaOH) the peak area of telmisartan and hydrochlorothiazide is found to increase by ten folds and five folds, respectively. Oxidizing (telmisartan- 101.48% and hydrochlorothiazide 99.60%), heat (60° ) (telmisartan- 97.43% and hydrochlorothiazide- 102.80%) and uv-exposure (telmisartan- 98.15% and hydrochlorothiazide- 98.96%).
| Amount of drug added (mg) | Amount of drug recovered (mg) | Mean percent recovery* | |||
|---|---|---|---|---|---|
| TEL | HTZ | TEL | HTZ | TEL | HTZ |
| 9.5 | 2.76 | 9.53 | 2.77 | 100.32 | 100.36 |
| 7.71 | 2.14 | 7.64 | 2.11 | 99.09 | 98.6 |
| 5.97 | 1.51 | 5.91 | 1.49 | 99 | 98.68 |
| 3.93 | 0.89 | 3.91 | 0.88 | 99.49 | 98.88 |
| Mean percent recovery* | SD | 99.48 | 99.13 | ||
| ± 0.60 | ± 0.83 | ||||
*mean of four observations
Table 2: Results of recovery studies
The proposed chromatographic system was found suitable for effective separation and quantitation of telmisartan (RT-5.19 min) and hydrochlorothiazide (2.97 min). The system suitability parameters are as follows: resolution-9.620, capacity factor- 4.21 for telmisartan and 1.98 for hydrochlorothiazide, tailing factor- 1.647 for telmisartan and 1.917 for hydrochlorothiazide, Number of theoretical plates/column- 3420 for telmisartan and 6410 for hydrochlorothiazide. The plot of peak area vs. concentration was found to be linear in the range of 4.10-20.48 μg/ml for telmisartan and 1.28-6.4 μg/ml for hydrochlorothiazide (correlation coefficient for calibration curve of both telmisartan and hydrochlorothiazide, r= 0.99). The mean recoveries of telmisartan and hydrochlorothiazide were 99.48%±0.52 and 99.13%±0.72, respectively. The values of percent recovery and low values of standard deviation indicate the accuracy, precision and reproducibility of the proposed method. The robustness of the method was studied by carrying out analyst to analyst and intra day variations The low values of standard deviation for analyst to analyst variation (±0.44 for telmisartan and ±0 .37 for hydrochlorothiazide) and intra-day variation (1.18 for telmisartan and ±0.24 for hydrochlorothiazide) indicate the robustness of the method. Based on the above results it can be concluded that the proposed method is accurate, precise, reproducible and specific and can be used for routine analysis of these drugs in combination tablets.
Acknowledgements
The authors are thankful to Head, Department of Pharmaceutical Sciences, Nagpur University, Nagpur for providing facilities for the research work and Aristo Pharmaceuticals Ltd., Raisen for providing the gift samples of the drugs.
References
- Maotian, Xu, Junfeng, S. and Yaodong, L., J. Pharm. Biomed. Anal. , 2004, 34, 681.
- Maotian, Xu, Junfeng S. and Ning, L., J. Pharm. Sci. , 2000, 89, 1465.
- Palled, M.S., Rajesh, P.M.N., Chatter, M. and Bhat, A.R., Indian J. Pharm. Sci ., 2005, 67, 108.
- Veerasekaran, V., Katakdhond, S.J., Kadam, S.S. and Janhavi, R., Indian Drugs , 2001, 38, 187.
- Shankar, M.B., Mehta, F.A., Bhatt, K.K., Mehta, R.S. and Geetha, M., Indian J. Pharm. Sci ., 2003, 65, 167.
- Kanumulu, G.V. and Bhanu, R., Indian Drugs , 2000, 37, 38.
- Belal, F., J. Pharm. Biomed. Anal ., 2001, 24, 335, through Int. Pharm. Abstr ., 2001, 38, 1676, 3808913