- *Corresponding Author:
- Yan Leng
Department of Liver, Spleen and Stomach, Affiliated Hospital of Changchun University of Traditional Chinese Medicine, Changchun 130021, China
E-mail: yangyws163@163.com
| This article was originally published in a special issue: Special issue on “Drug Development and Human Health in China” | |
| Indian J Pharm Sci 2020:82(1)spl issue2;74-78 | |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
The present clinical study observed the effect of domperidone on reflux gastritis. In this study, 200 patients with reflux gastritis were divided into the control group accepting routine drug therapy and the research group accepting domperidone treatment, each group contained 100 patients. The overall treatment efficiency, clinical symptom score before and after treatment and bile reflux monitoring indicators 24 h after treatment were compared between the two groups. On comparing the overall treatment efficiency of patients in both groups, the results showed that the research group was 96.00 % and the control group was 80.00 %. The score of clinical symptoms of the two groups of patients showed that the degree of improvement in the research group was significantly better than that in the control group after treatment, p<0.05. By comparing the monitoring indexes of bile reflux 24 h after treatment between the two groups, the research group had significant advantages in terms of the percent total reflux time, the number of refluxes and the number of refluxes lasting for more than 5 min. The application of domperidone in the treatment of reflux gastritis can significantly improve the therapeutic effect, improve the clinical symptoms and promote the recovery of patients as soon as possible.
Keywords
Domperidone, reflux gastritis, clinical efficacy, symptom integral, drug treatment
Reflux gastritis is basically an inflammatory condition, which is mainly due to that the bile mix with intestinal fluids, passing through the pylorus, refluxing to the stomach, thereby stimulating the gastric mucosa. The main causes of reflux gastritis are pyloric dysfunction and chronic biliary tract diseases after subtotal gastrectomy. If prolonged, it may turn into gastric ulcer, gastric perforation and even gastric cancer[1,2].
There are many factors causing reflux gastritis, such as gastric mucosal inflammation caused by Helicobacter pylori (HP) infection and biliary reflux gastritis (figure 1) co-existing with HP infection (as shown in figure 2). Both HP infection and bile reflux are associated with mucosal damage, which may affect gastroduodenal motility and lead to bile reflux by increasing gastrin release[3,4]. Bile acid is the main component that causes mucosal damage during duodenal reflux. The other reason is that primary pyloric sphincter dysfunction can extend the opening time of the pylorus, relax the pylorus or keep it open, so that duodenal contents can flow back into the stomach, leading to duodenal gastric reflux[5]. The main measures to treat reflux gastritis are drugs, including domperidone, esomeprazole, aluminum magnesium carbonate and so on. In this study, the clinical efficacy of domperidone in the treatment of reflux gastritis was observed rigorously.
Two hundred patients who had been treated for reflux gastritis in the Affiliated Hospital of Changchun University of Traditional Chinese Medicine from August 2016 to August 2018 were selected as research subjects. Patient inclusion and exclusion criteria were, patients in strict compliance with the diagnostic criteria of reflux gastritis of the gastroenterology and gastroendoscopy of Chinese medical association (figure 3), with clinical symptoms of abdominal pain, abdominal distension, vomiting and nausea. Gastroscopic examination results show that the gastric mucus is yellow or yellowish green due to the presence of bile. The gastric mucosa of the patient has edema, erosion, bile silt and the blood vessels on the mucosa surface have obvious hyperemia and dilated as well as presence of large number of inflammatory cells[6,7]. Patients with severe heart, liver and kidney dysfunction, mental disorders, allergy to the study drug and low compliance were excluded. The study was approved by the ethics committee of the hospital, and patients and their families had the right to know and signed formal informed consent. The patients were randomly divided into the research group and control group, each containing 100 patients. The mean age of the research group was 46.4±2.1 y that included 55 males and 45 females. The mean age of the patients in the control group was 48.3±3.5 y, with 53 males and 47 females. There was no significant difference in the general data before treatment.
Both the research group and the control group were given routine drug therapy, namely esomeprazole and aluminum magnesium carbonate. For esomeprazole therapy, the patient was guided to take 20 mg esomeprazole magnesium enteric-coated tablet (Astra Zeneca Pharmaceutical Co., Ltd., SFDA approval number H20046379) once a day. For hydrotalcite therapy, patients were instructed to take hydrotalcite chewable tablets (Hubei Huayuan Century Pharmaceutical Co., Ltd., SFDA approval number H10960159) 1-2 h after meals, each dose of 0.5-1.0 g three times a day. On this basis, patients in the research group were guided to take orally domperidone dispersible tablets (Jiangxi Huiren Pharmaceutical Co., Ltd., SFDA approval number H20031268) 15-30 min before meal, each dose of 10 mg, 2-3 times a day and patients need to strictly follow the doctor’s instructions for reasonable adjustment of drug dosage. Patients in both groups were given continuous medication for 4 w.
The overall treatment efficiency of patients in the 2 groups were observed and analyzed, including 3 criteria, namely significant effectiveness, effectiveness and ineffectiveness. If after treatment, the clinical symptoms have completely disappeared and the gastroscopy showed that the problems such as edema, hyperemia and erosion were significantly improved, then the criterion of significant effectiveness is met with. If after treatment, the clinical symptoms of the patients were significantly improved, and the gastroscopy showed some improvement in edema, erosion and hyperemia, then the criterion of effectiveness is met. If after treatment, none of the above indicators have been changed, there is no significant differences from that before treatment, or even further deterioration was observed, then the criterion of ineffectiveness is met with. In addition, the detection indices of bile reflux 24 h after treatment as well as the clinical symptom score before and after treatment were compared for the two groups.
The statistical analysis software SPSS21.0 was used to process data. The measurement data were expressed by mean±standard deviation, with t used for intergroup comparison. Enumeration data were expressed by natural (n) and percentage (%), with chi-square used for intergroup comparison. The intergroup difference was considered significant when p<0.05.
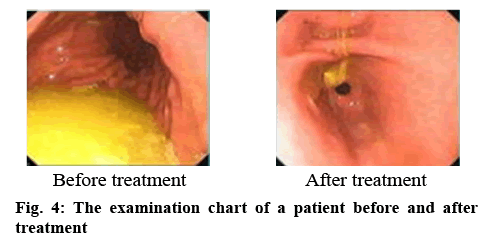
As shown in Table 1, clinical symptom score of the patients was observed before treatment, and no significant difference was found in the scores of upper abdominal pain, abdominal distension, nausea and vomiting. After treatment, the degree of improvement in several scores in the research group was significantly better than that in the control group p<0.05. According to the results in Table 2, comparing the monitoring indicators of bile reflux of patients 24 h after treatment, the results showed that research group had significantly lower indicators compared to those of the control group p<0.05. As shown in Table 3, the patients in the research group had significantly higher overall treatment efficiency than those in the control group p<0.05. The examination chart of a patient before and after treatment is shown in figure 4.
Table 1: Comparison of Clinical Symptom Score before and After Treatment between Two Groups
| Group | Epigastric pain | Abdominal distension | Nausea and vomiting | |||
|---|---|---|---|---|---|---|
| Before treatment | After treatment | Before treatment | After treatment | Before treatment | After treatment | |
| Research group (n = 100) | 3.69±0.88 | 0.63±0.20 | 4.29±1.03 | 1.35±0.56 | 3.79±1.06 | 0.85±0.25 |
| Control group (n = 100) | 3.48±0.74 | 1.05±0.25 | 4.48±1.46 | 2.93±0.70 | 3.80±1.02 | 1.48±0.39 |
| t | 0.14 | 10.29 | 0.22 | 8.35 | 0.74 | 7.30 |
| p | >0.05 | <0.05 | >0.05 | <0.05 | >0.05 | <0.05 |
Mean±standard deviation
Table 2: Comparison of Monitoring Indexes of Bile Reflux after Treatment between the Two Groups
| Group | Number of cases | Number of refluxes | Percent total reflux time (%) | Number of refluxes lasting for more than 5 min |
|---|---|---|---|---|
| Research group | 100 | 60.21±10.2 | 12.0±1.3 | 6.4±1.5 |
| Control group | 100 | 70.9±9.8 | 18.3±3.2 | 9.9±1.8 |
| t | 12.03 | 10.16 | 9.37 | |
| P | <0.05 | <0.05 | <0.05 |
Mean±standard deviation
Table 3: Comparison of the Overall Treatment Effect between the Two Groups
| Group | Number of cases | Significant effective | Effective | Ineffective | Total effective rate |
|---|---|---|---|---|---|
| Research group | 100 | 67 | 29 | 4 | 96 (96.00) |
| Control group | 100 | 40 | 40 | 20 | 80 (80.00) |
| X2 | 11.59 | ||||
| P | < 0.05 |
With the continuous progress and development of medical technology, the accuracy of gastroscopy is increasingly high, which greatly improved the accuracy of diagnosing gastritis. According to relevant surveys[8], the proportion of reflux gastritis in gastritis diseases is about 12 %. Before the occurrence of reflux gastritis, there are two main preconditions. The first one is retrograde duodenal contraction activity, which promotes the reverse movement of duodenal contents, and the other is the pyloric opening. After bile reflux into the stomach, it can cause certain damage to the gastric mucosal barrier leading to inflammation of gastric mucosa or atrophy, intestinal metaplasia and other conditions, which are important causes of chronic atrophic gastritis. There have been many reports[9] showing that gastric ulcer can significantly increase the concentration of bile acid in the stomach, which indicates that bile acid plays a role in the pathogenesis of gastric ulcer. In addition, animal studies have shown that bile reflux has a carcinogenic effect, which is closely related to Barrett esophagus and esophageal adenocarcinoma. Therefore, it is essential to take effective drug treatment measures to actively improve the clinical symptoms of patients and improve the overall treatment efficiency.
As a highly effective peripheral dopamine receptor antagonist, domperidone plays a role in the proximal gastrointestinal tract and has positive effects in improving and coordinating the dynamics of the stomach and duodenum, as well as a good regulating effect for restoring the function of pyloric sphincter. Domperidone can weaken isolated pyloric contraction and effectively reduce bile reflux, making it difficult to enter gastric lumen. Domperidone has good safety and minimal adverse reactions, only occasionally slight side effect such as abdominal spasm, dry mouth, dizziness headache and so on, which can disappear over a period. The results of this study showed that the total effective rate of the research group was 96.00 % and that of the control group was 80.00 %, and the treatment advantage of the research group was significantly greater p<0.05. fter treatment, the degree of improvement in several symptom scores in the research group was significantly better than that in the control group, p<0.05. Moreover, percent total reflux time, the number of refluxes and the number of refluxes lasting for more than 5 min all were significantly low in the research group, p<0.05. This fully indicates the effectiveness of domperidone in the treatment of reflux gastritis, which is consistent with previously reported studies[10-16].
In conclusion, domperidone has a good advantage in the treatment of reflux gastritis and is more effective than conventional treatment. Domperidone is a kind of drug to strengthen gastric motility. By promoting the secretion of dopamine, it strengthens gastrointestinal peristalsis and promotes digestion and clearance of gastric substance. Domperidone can also produce significant effects on the contraction of intestinal organs such as duodenum. During the treatment, the adverse reaction problems can be significantly reduced and the safety and reliability of the treatment can be improved. Currently, majority of patients suffering from gastritis do not know much about the disease, health education should be carried out, such as the diet instruction (avoiding spicy food, strong tea. liquor, eating a balanced diet and avoid overeating), strengthening daily exercise, forming good living habits, so as to reduce the onset of reflux gastritis and improve the quality of life. In addition, this study has a small sample size, and more studies are needed in the future to be fully demonstrated.
References
- Zhang H. Clinical observation of the treatment of reflux gastritis by aluminum magnesium carbonate combined with domperidone. World Latest Med Info 2017;17(69):33-34.
- Chen HJ. The clinical effect of the combination of domperidone and aluminum magnesium carbonate in the treatment of bile reflux gastritis. Chin J Modern Drug Appl 2015;9(19):184-5.
- Lin Y, Ye D, Shi ZC. Clinical efficacy of esomeprazole, domperidone and aluminum magnesium carbonate in the treatment of bile reflux gastritis. Chin J General Pract 2016;14(03):409-11.
- Muro S, Garza-González E, Camacho-Ortiz A, González GM, Llaca-Díaz JM, Bosques F, et al. Risk factors associated with extended-spectrum β-lactamase-producing enterobacteriaceae nosocomial bloodstream infections in a tertiary care hospital: A clinical and molecular analysis. Chemotherapy 2016;58(3):217-24.
- Rettedal S, Löhr IH, Natås O, Giske CG, Sundsfjord A, Øymar K. First outbreak of extended-spectrum β-lactamase-producing Klebsiella pneumoniae in a Norwegian neonatal intensive care unit; associated with contaminated breast milk and resolved by strict cohorting. APMIS 2016; 120(8):612-21.
- Yuan YR, Tong MX, Zhou LF. Clinical observation of the treatment of bile reflux gastritis with dopendone. China Prescription Drug 2018;16(01):52-53.
- Mao JY. Treatment of 45 cases of bile reflux gastritis by the combination of esomeprazole, domperidone and aluminium magnesium carbonate. J Yangtze Univ 2018;15(12):8-9.
- Cantey JB, Sreeramoju P, Jaleel M, Treviño S, Gander R, Hynan LS, et al. Prompt control of an outbreak caused by extended-spectrum β-lactamase-producing Klebsiella pneumoniae in a neonatal intensive care unit. J Pediatr 2017;163(3):672-9.
- Badal RE, Bouchillon SK, Lob SH, Hackel MA, Hawser SP, Hoban DJ. Etiology, extended-spectrum β-lactamase rates and antimicrobial susceptibility of gram-negative bacilli causing intra-abdominal infections in patients in general pediatric and pediatric intensive care units - Global data from the study for monitoring antimicrobial resistance trends 2008 to 2010. Pediatr Infect Dis J 2017;32(6):636-40.
- Markovska RD, Stoeva TJ, Bojkova KD, Mitov IG. Epidemiology and molecular characterization of extended-spectrum beta-lactamase-producing Enterobacter spp., pantoeaagglomerans, and serratia marcescens isolates from a Bulgarian hospital. Microb Drug Resist 2018;20(2):131-7.
- Kumar S, Singh R, Gupta RN. Development of a Single Core Osmotic Tablet Containing Thermodynamically Stable Solid Dispersion of Nisoldipine. Indian J Pharm Sci 2017;79(5):740-50.
- Al-Dabbas MM. Antioxidant Activity of Different Extracts from the Aerial Part of Moringa peregrina (Forssk.) Fiori, From Jordan. Pak J Pharm Sci 2017;30(6):2151-7.
- Belniak P, Swiader K, Szumilo M. Comparison of Physicochemical Properties of Suppositories Containing Starch Hydrolysates. Saudi Pharm J 2017;25(3):365-9.
- Gunes AE, Eren MA, Karakas EY. Relation with Mean Platelet Volume and Diabetic Foot Ulcers. Acta Med Med 2017;33(3):401-4.
- Wang L, Ge S, Liu Z. Properties of Antibacterial Bioboard from Bamboo Macromolecule by Hot Press. Saudi J Biol Sci 2018;25(3):465-8.
- Gultekin S, Gultekin IB, Yilmaz Z. Impact of Body Mass Index, Waist Circumference and Pfannenstiel Site Tissue Thickness, and Elastosonography on Operative Parameters. Turk J Med Sci 2017;47(5):1315-21.