- *Corresponding Author:
- Veeresham Ciddi
Faculty of Pharmaceutical Sciences, Kakatiya University, Warangal−506 009, India
E-mail: ciddiveeresham@yahoo.co.in
| Date of Submission | 15 June 2005 |
| Date of Revision | 18 November 2005 |
| Date of Acceptance | 25 July 2006 |
| Indian J Pharm Sci, 2006, 68 (4): 490-492 |
Abstract
Suspension cultures of Withania somnifera cells were established and shown to produce withaferin A. The identification of withaferin A was done by TLC, UV absorption, HPLC and electron spray mass spectroscopy. These cultures could be strongly elicited by exposure to salacin. Addition of salacin at the concentration of 750 µM to the cultures in production medium enhanced production levels of withaferin A to 25±2.9 mg/l compared to 0.47±0.03 mg/l in unelicited controls. This report is the first to demonstrate withaferin A production in plant suspension cultures and provides prerequisites for commercial scale, controlled production of withaferin A.
Withania somnifera Dunal, an erect evergreen shrub, commonly called Ashwagandha (Sanskrit), grows throughout India, Pakistan and Italy. The plant contains a group of compounds known as withanolides. There is a significant variation in the withanolide expressed from plants grown in different geographical areas. Withanolides are naturally occurring steroids built on ergostane skeleton, in which C-22 and C-26 are appropriately oxidized to form a d-lactone ring. Withaferin A has antibacterial, anti-tumor, anti-inflammatory and immunomodulatory properties [1].
The commercial cultivation of Withania somnifera has two major problems. The first problem is plant to plant variation in quality and quantity of active constituents and the second is the long gestation period (4-5 y) between planting and harvesting. To enhance the commercial prospects for the production of Withaferin A, an alternative choice could be the use of plant cell cultures [2]. Vitali et al. [3] described a method for initiation of callus from Withania somnifera (Israel) and withanolides production from them. Roja et al. [4] described a method for induction of callus of Withania somnifera but the callus failed to synthesize withanolides. However, multiple shoot cultures produced withanolides. The transformed root cultures of Withania somnifera produced withanolide-D [5]. Banerjee et al. [2] reported the hairy root cultures of Withania somnifera able to produce Withaferin A. However, commercial production would be much more practical with suspension cultures which have not yet been demonstrated to produce withanolides.
Laboratory scale studies for optimization of plant cell suspension culture conditions is usually performed in glass shake flasks. Six-well polystyrene plates have been used for mammalian and insect cell culture for several years; this technology has very limited application in the culturing of plant cells [6-7]. Srinivasan et al. [8] reported the combined use of six-well plates and TLC for rapid screening of Taxus sp cell suspension cultures for taxane production. Their studies revealed that six-well plates were superior to shake flasks for rapidly predicting which conditions may be most effective for growth and product formation. This communication reports the establishment of suspension cultures of Withania somnifera capable of production of Withaferin A and the use of six well plates to test various elicitors for enhancement of product formation.
Materials and Methods
Callus initiation and maintenance
The seeds of Withania somnifera were washed thoroughly with water and allowed to germinate aseptically on 3% sucrose-agar medium. The seeds germinated after 3 w. The hypocotyl portions of 3-w old seedlings were aseptically transferred onto MS medium9 containing 3% sucrose plus 2,4-D (1 mg/l) plus kinetin (0.2 mg/l). The callus induction was observed after 5 d and callus was sufficiently well formed by 3 w so that it could be transferred. The callus was maintained by subculturing onto the half strength B5 medium10 with 3% sucrose plus dicamba (2 mg/l) plus kinetin (1 mg/l) at 4 w intervals.
Initiation and maintenance of suspension cell cultures
The suspension cultures were initiated by transferring 10% w/v of callus into half strength B5 medium supplemented with dicamba (2 mg/l), kinetin (1 mg/l) and glutamine (292 mg/l) and incubated at 25±2° at 120 rpm. After one week the cultures were filtered aseptically through a 100 size sieve to remove large cells. The uniform suspension was filtered through Buchner funnel and transferred to fresh medium using an inoculum of 5% w/v and cultures were maintained by subculturing at 1 w intervals.
Induction of product synthesis
The cell cultures were filtered aseptically through a Buchner funnel with vacuum and washed with medium. Then the cells were transferred to production medium, ¼ B5 medium supplemented with 2,4-D (2 mg/l), zeatin (0.5 mg/l) and sucrose (5% w/v) in six-well plates or shake flask in 20% w/w inoculum and incubated for 2 w.
Elicitor treatments
The solutions of elicitors were prepared by dissolving in a suitable solvent and added to the cultures before incubation. The elicitors used were: salacin dissolved in water; methyl jasmonate and arachidonic acid dissolved in ethanol.
Extraction of withaferin A
Cell culture samples were first filtered to obtain a cell fraction and cell-free filtrate. The cell mass was weighed to determine fresh weight. Cells were then frozen and thawed and then extracted three times with three volumes of dichloromethane. The combined dichloromethane extract was evaporated and the residue was dissolved in methanol and saved for analysis.
TLC analysis
The methanolic extract was analyzed by using pre-coated silica gel plates of 250 μm layer, UV 254 (Whatman Ltd, Maidstone, Kent, England) by co-chromatography with authentic sample of withaferin A. The plates were developed in a solvent system of chloroform: methanol (95:5) and detected using a vanillin-sulfuric acid reagent [11].
HPLC analysis
The HPLC analysis of extracts was carried out by Phyton Inc, USA. The conditions of the analysis includes, gradient method with water with 0.1% TFA (tri-fluoro acetic acid) in Pump A, recovered acetonitrile (86%, 14% water) with a 0.1% TFA in pump B, with a column of Waters delta pack C-18, 150x4.6 mm, 4 μm with a guard column. The flow rate of mobile phase was 0.85 ml/min.
Results and Discussion
The growth index (GI) after 7 d was 1.8 (five independent wells or n=5) for six well plates and 2.2 (n=3) for flask cultures. The growth index is the final dry weight divided by the initial dry weight. The value of GI changed little after day 7 (measured at day 10, day 15 and day 20 in replicate experiments) and was always in the range of 1.8 to 2.2. Thus, on growth medium the doubling time was approximately 7 d or less.
The production of withaferin A for cultures transferred to production medium was confirmed by TLC analysis, HPLC and ES mass spectra. With TLC analysis the Rf values of presumptative withaferin A had the same value as the authentic withaferin A; when derivatized with vanillin sulfuric acid reagents both spots were blue. The HPLC analysis gave the same retention time as the standard (9.9 min) for a presumptive withaferin A peak. HPLC was used for quantification. The extracts also had an ES mass spectra signal the same as the standard with a peak at 473.5. These measurements taken together provide strong evidence for production of withaferin A from these suspension cultures.
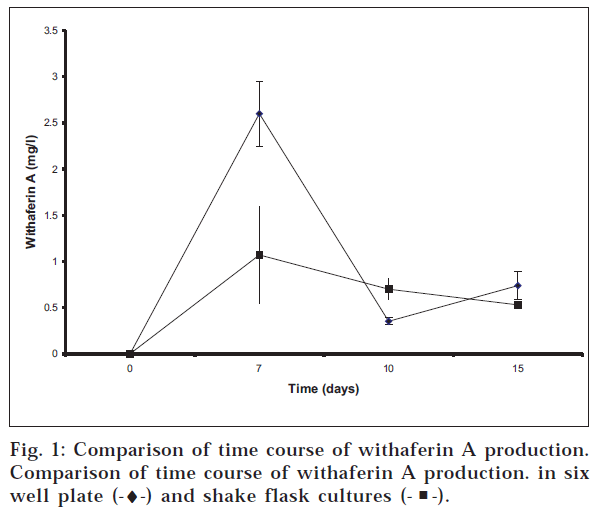
The kinetic pattern for production of withaferin A is shown in fig. 1. Measurements from four independent cultures were made at each time point. Total withaferin A is reported. The cellular fraction was about 60% of the total withaferin A with the remainder being in the medium. The maximum value observed was 2.6±0.35 mg/l on day 7 in the 6 well plate system. With the flasks the maximum value 1.07±0.52 mg/l was also observed on day
7. In both cases the values of withaferin A decreased at day 10 and 15. These data demonstrate that the same production trend is observed in both methods of culture confirming the suitability of 6-well plate cultures for use in evaluating secondary metabolite production in plant cell suspension cultures.
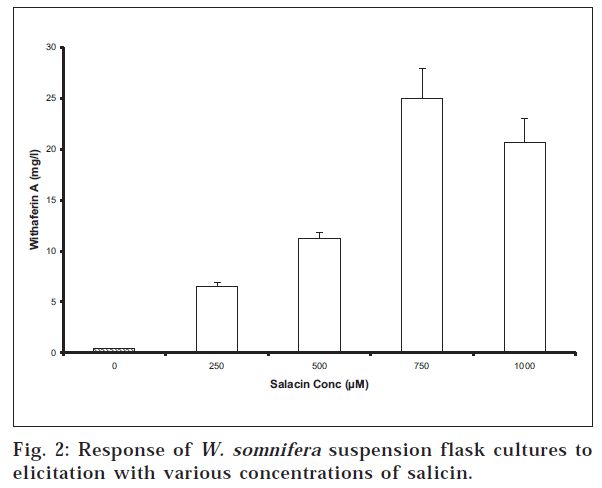
A strategy that has often proved successful in enhancing secondary metabolite production from plant cell cultures has been the use of elicitors [7,12]. Elicitors typically cause the cells to activate their defense system through an incompletely understood signal transduction system. The use of methyl jasmonate (at 100 μM) and arachidonic acid (1 mg/l) did not improve withaferin A production over non-elicited controls. However, salacin was an effective elicitor of withaferin A production (fig. 2). With the introduction of salacin on day 0 the amount of withaferin A on day 15 was 25±2.9 mg/l with 750 μM salacin in flask cultures (n=4) versus 0.47±0.03 mg/l for control flasks that received no elicitor. Using a dose of 500 μM salacin introduction on day 7 rather than day 0 improved day 15 withaferin A production to 21±2.6 mg/l versus 11±0.6 mg/l for salacin addition on day 0. While withaferin A production could be further optimized, these results demonstrate that 50 fold enhancement using salacin as an elicitor is possible.
Addition of the sterol inhibitor, chlorocholinechloride (at 1 mM) inhibited the production of withaferin A. This result suggests that precursors enter through the acetate-mevalonate pathway rather than through the non-mevalonate pathway.
In summary, we have shown for the first time production of withaferin A from suspension culture (using Withania somnifera) and a strong positive response to elicitation by salacin. These results provide a basis for controlled production of withaferin A as a pharmaceutical.
Acknowledgements
The author is thankful to Dr. M. D. Kharya, Dr. H.S. Gaur University, Sagar, M.P., India for providing the seeds of Withania somnifera, Prof. M. L. Shuler, Paula Miller and Dr. S. Y. Yoon for their help during the work, Dr. V. Bringi, Phyton Inc. for analysis of the extracts, Prof. Atta-ur-Rahman of HEJ Research institute, Karachi, Pakistan for supplying authentic sample of Withaferin A and to UNESCO for financial assistance.
References
- Archana, R. and Namasivayam, A., J. Ethnopharmacol., 1999, 64, 91.
- Banerjee, S., Naqvi, A.A., Mandal, S. and Ahuja, P.S., Phytotherapy Res., 1994, 8, 452.
- Vitali, G., Conte, L. and Nicoletti, M., Planta Med., 1996, 62, 287.
- Roja, G., Heble, M.R. and Sipahimalani, A.T., Phytotherapy Res., 1991, 5, 185.
- Ray, S., Ghosh, B., Sen, S. and Jha S., Planta Med., 1996, 62, 571.
- Schumacher, H.M., Gundlach, H., Fieldler, F. and Zenk, M.H., Plant Cell Rep., 1987, 6, 410.
- Gundlach, H., Mueller, M.J., Kutchan, T.M. and Zenk, M.H., Proc. Natl. Acad. Sci. USA, 1992, 89, 2389.
- Srinivasan, V., Roberts, S.C. and Shuler, M.L., Plant Cell Rep., 1997, 16, 600.
- Murashige, T. and Skoog, F., Physiol Plant., 1962,15, 473.
- Gamborg, O.L., Miller, R.A. and Ojima, K., Exp. Cell Res., 1968,50,151.
- Wagner, H. and Bladt, S., Plant Drug Analysis − A Thin-Layer Chromatography Atlas, Springer-Verlag, 1996.
- Ciddi, V., Srinivasan, V. and Shuler, M.L., Biotechnol. Lett.,1995, 17,1343.