- *Corresponding Author:
- S. H. Gan
Human Genome Centre, School of Medical Sciences, Universiti Sains Malaysia 16150, Kubang Kerian, Kelantan, Malaysia
E-mail: shgan@usm.my
| Date of Submission | 13-Apr-2015 |
| Date of Revision | 05-Feb-2016 |
| Date of Acceptance | 15-Mar-2016 |
| Indian J Pharm Sci 2016;78(2):173-181 |
Abstract
Owing to the efficacy and popularity of anastrozole in the treatment of estrogen receptor-positive breast cancer, interest in analytical methods for its quantitative measurement both in pharmaceutical dosage formulations and Biological samples has risen sharply for a decade. This paper reviews the existing chromatographic methods, which are based on two popular detection methods: high performance liquid chromatography and gas chromatography mass spectrometry. The Current high performance liquid chromatography methods with UV detection so far reported; focus primarily on determining the concentration of anastrozole in pharmaceutical dosage formulations. However, such methods are insufficient in measuring the concentrations yielded in biological samples when the relatively low doses (1 mg daily) recommended for breast cancer treatment are used. Therefore, there is limited applicability of HPLC-UV detection methods for quantitative measurements of anastrozole in biological samples. A number of methods combining liquid chromatography with tandem mass spectrometry with limits of quantitation as low as 0.05 ng/ml have been developed for anastrozole measurement in plasma. A few methods on active pharmaceutical ingredients have also been reported. On the other hand, only a few gas chromatography methods have successfully been developed for the determination of anastrozole concentrations. In majority of the methods, where relevant, parameters such as selectivity, linearity, limit of detection, limit of quantitation, and accuracy have been considered, and recovery assessments during accuracy evaluation are generally satisfactory, with reported values ranging between 81 and 109%. However, a significant number of these studies did not use an internal standard to correct for the area of anastrozole, making their accuracies questionable. Overall, the current available detection methods for anastrozole are inadequate, and improved method validation techniques as well as simple and affordable anastrozole quantification methods in both pharmaceutical dosage formulations and especially biological samples are needed.
Keywords
Anastrozole, HPLC, LC-MS/MS, GC-MS, breast cancer
Globally, breast cancer is the most common malignancy encountered by women and is reported to be one of the leading causes of death especially in the less developed regions [1]. It has been estimated that approximately 80% of breast cancers are estrogenreceptor (ER) positive [2]. Both ERs and estrogen biosynthesis pathways are the main therapeutic targets for breast cancer. For about three and half decades, tamoxifen (TAM), which works by competitively blocking estrogen from binding the ERs, has been the major drug of choice for the treatment and prevention of ER-positive breast cancers in preand post-menopausal women [3]. On the other hand, anastrozole (ANA) and other aromatase inhibitors (AIs) target the estrogen biosynthesis pathway by inhibiting aromatase, a rate-limiting enzyme in the biosynthesis of estrogen [4].
Currently, TAM is still a valuable treatment option in breast cancer therapy [5]. However, its long term use is limited by life-threatening adverse effects [6-9], and some patients can become resistant [10,11]. As a result of these challenges, three AIs including ANA were introduced to either serve as TAM alternatives or to be used following several years of TAM therapy [4,10,12-14].
ANA is widely used in the adjuvant therapy of post-menopausal women with breast cancer. This drug significantly decreases plasma estradiol (E2) concentrations when administered at 1 or 10 mg/ day, with both doses being capable of suppressing E2 production [4,15,16]. ANA acts by inhibiting aromatase, an enzyme responsible for converting androgens into estrone and E2, subsequently suppressing these hormones in the peripheral tissues as well as at several locations in the central nervous system [4,17].
Chemistry and metabolism of ANA
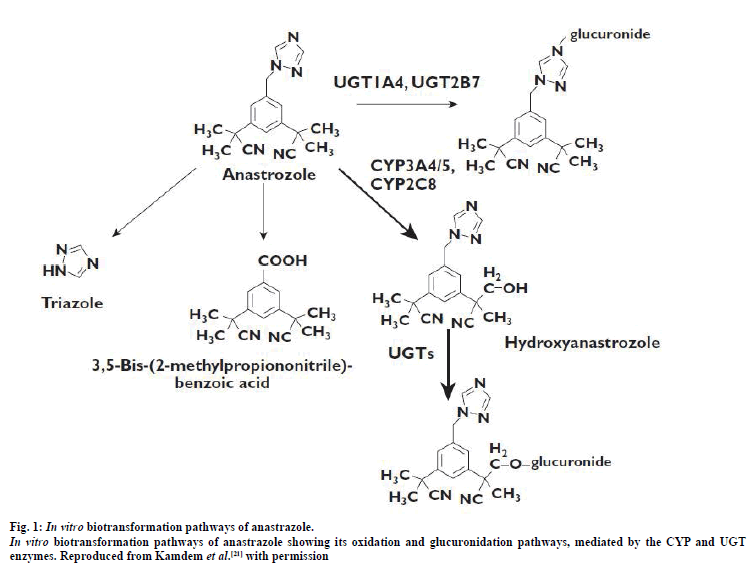
ANA (fig. 1) is a non-steroidal, third-generation achiral triazole derivative that is chemically known as 2,2- [5-(1H-1,2,4-triazol-1-ylmethyl)-1,3- phenylene]bis(2-methylpropiononitrile) [15]. The main site of ANA metabolism is in the liver [18]. Here, it undergoes oxidation (catalyzed by CYP3A4) to form hydroxy-anastrozole, which may further undergo glucuronidation (catalyzed by UGT1A4) to yield conjugated hydroxy-anastrozole. ANA can also be directly glucuronidated to anastrozole N-glucuronide [18-21]. A schematic representation of the biotransformation pathway is shown in fig. 1.
Following administration of a single radiolabeled dose of ANA (1 mg) to human subjects, approximately 60% of the administered dose was excreted as metabolites while another 10% was excreted unchanged in the urine [22]. Triazole, hydroxyl-anastrozole, hydroxylanastrozoleglucuronide and anastrozoleglucuronide constitute major ANA metabolites, reported to be present in the plasma and the urine of patients taking the drug (fig. 1) [19,21]. However, to date, the exact pharmacological activity of these metabolites has yet to be fully established [18,20,23]. In a recent study, the triazole metabolite of ANA could not be identified [21,23], thus questioning the rationale for developing methods for their detection.
Chromatographic analyses of ANA
The chromatographic separation of ANA is valuable mainly because it can be used in the identification and quantification of the drug. Although ANA has only recently become available in the market (September 2002), a number of chromatographic methods have been developed which include UV- spectrophotometric method for estimation of ANA in bulk and in pharmaceutical dosage formulations [24], high performance liquid chromatography (HPLC) techniques for determination of ANA in bulk and pharmaceutical dosage forms [25-29], liquid chromatography-mass spectrometry/mass spectrometry (LC MS/MS) [25,30-33], ultra-performance liquid chromatography (UPLC)-tandem mass spectrometric method for ANA detection in human plasma [34], capillary gas chromatography [35,36] and high performance thin layer chromatography (HPTLC) [37]. However, owing to the growing interest in pharmacogenetic studies involving ANA and other active pharmaceutical ingredients (APIs), simple, sensitive, affordable and rapid chromatographic techniques for ANA quantification in biological samples are still needed. The type of chromatographic method selected is governed by a number of factors including the required sensitivity, the chromatographic resolution needed, the nature of the biological matrix. The different HPLC and LC-MS/MS methods previously reported for the determination of ANA are summarized in Tables 1 and 2.
| Mobile phase | Column/stationary phase | Detection wavelength (nm) |
Retention time (min) | LOD (µg/ml) | LOQ (µg/ml) | References |
|---|---|---|---|---|---|---|
| ACN:methanol (50:50) | Hichrom RPB 18 (250×4.6 mm, 5.0 µm) | 215 | 10.9 | - | - | [24] |
| ACN:ammonium acetate (50:50) | Inertstil ODS-3V (250×4.6 mm, 5.0 µm) | 210 | 6.5 | - | - | [25] |
| ACN:ammoniumformate (40:60) | Oyster ODS-3 (100×4.6 mm, 3.0 µm) | 215 | 10.0 | - | - | [26] |
| ACN:phosphate buffer (50:50) | Gracesmart RP18 (100×4.6 mm, 5.0 µm) | 215 | 5.3 | 0.351 | 1.053 | [27] |
| ACN:water (46.5:53.5) | Symmetry® C-8 (100×4.6 mm, 3.5 µm) | 270 | 4.8 | 2.200 | 6.700 | [28] |
| ACN:ammonium acetate (proportion unspecified) | Inertsil C-8 (250×4.6 mm, 5 µm) | 215 | 23.2 | 0.008 | 0.016 | [29] |
| ACN:water (45:55) | Inertsil ODS C18 (250×4.6 mm, | 215 | 4.4 | 0.615 | 1.863 | [30] |
ACN-acetonitrile, LOD-limit of detection, LOQ-limit of quantification
Table 1: Comparison of Existing HPLC Methods for Anastrazole Analyses Using UV Detection
| Mobile phase | Column/stationary phase | Extraction method | LOQ (ng/ml) | Analytical time (min) | References |
|---|---|---|---|---|---|
| ACN:AA (proportion unspecified) | Merck Purospher® Star RP-18 (55×4 mm, 3.0 µm) | Liquid-liquid | - | - | [31] |
| ACN:MeOH:AA (proportion unspecified) | Genesis C18 (100×2.1 mm, 4.0 µm) | Liquid-liquid | 0.05 | 2.5 | [32] |
| ACN/MeOH:AA (90:10) | YMC cyano (50×4.0 mm, particle size unspecified) | Liquid-liquid | 0.10 | 1.6 | [33] |
| ACN:water (50:50) | Inertsil ODS-3V (250×4.6 mm, 5.0 µm) | - | 0.06 | - | [34] |
| ACN:formic acid (88:12) | Hypurity C18 (50×4.6 mm, 5.0 µm) | Solid-phase | 0.30 | 2.5 | [35] |
| Methanol | Eurosphere-C18 (200×0.5 mm, 5.0 µm) | Solid phase | - | 16.0 | [36] |
| ACN:formic acid (proportion unspecified) | BEH C18 (100×2.1 mm, 1.7 µm) | Solid phase | 0.10 - 2.00 | - | [37] |
| ACN:formic acid (50:50) | Luna 3 µm C18(2) (100×2 mm) | Solid phase | 0.10 | - | [18] |
| ACN:formic acid (50:50) | Luna 3 µm C18(2) (100×2 mm) | Solid-phase | 0.10 | - | [21] |
ACN-acetonitrile, AA-ammonium acetate, LOQ-limit of quantification, MeOH-methanol
Table 2: Comparison of Existing Lc-Ms/Ms Methods for Analyses of Anastrazole
The use of an internal standard (IS) in quantitative determination of ANA based on chromatographic methods is indispensable because of the role it plays in correcting the unknown losses in the techniques used. The IS varies by method, depending on the chemical properties of the intended target drug planned to be used as an IS as well as its availability. As much as possible, drugs that are not naturally present in the biological samples to be studied are used as IS. In addition, the IS should ideally, yield a peak with good resolution, elute close to the analyte being measured, not chemically react with the sample and be present in approximately the same concentration as the analyte being quantified [38]. Table 3 summarizes the various ISs reported in the literature. It is important to note that due to the relatively low recommended dose of ANA for breast cancer therapy (1 mg daily), the quantification of ANA in biological samples such as plasma and urine has posed a lot of challenges. The low ANA concentration limits the number of analytical methods available for ANA detection in human plasma.
| IS | Technique | References |
|---|---|---|
| Dexchlorpheniramine | LC-MS/MS | [32, 38] |
| Omeprozole | LC-MS/MS | [33] |
| Androsteroneglucuronide | LC-MS/MS | [31] |
| Letrozole | LC-MS/MS | [35] |
| Bunitrolol | LC-MS/MS | [36] |
| ANA-d12 | LC-MS/MS | [37] |
| Desmethyldiazepam | LC-MS/MS | [18, 21] |
| Verapamil | LC-MS/MS | [39] |
| Diazepam | GC-ECD | [40, 41] |
| Clomipramine | GC-FID | [42] |
LC-MS/MS= LC coupled with tandem mass spectrometry; GC-ECD= GC with electron capture detection; GC-FID= gas chromatography with flame ionization detection; ANA-d12= deuterium-labeled anastrozole.
Table 3: Internal Standards Used for Anastrazole Analyses
API is a term defined in ICH Q7 as “any substance or mixture of substances intended to be used in the manufacture of a drug product and that, when used in the production of drug, becomes an active ingredient in the drug product. Such substances are intended to furnish pharmacological activity or other direct effect in the diagnosis, cure, mitigation, treatment or prevention of disease or to affect the structure and function of the body” [39]. The process of API involves a number of steps that may include multi-step chemical synthesis and fermentation, purification, crystallization, drying, milling, packing, labeling and testing.
High-Performance Liquid Chro-Matography
Chromatographic system is a technique that encompasses separation of components or solutes of a mixture according to the relative concentration of each solute distributed between the mobile phase and the stationary phase. The mobile phase may be either a liquid or a gas, while the stationary phase can either be a solid or a liquid. Quantitation of ANA by HPLC has been conducted with UV detection; however, the weakness of most of the methods reported in the literature is that many of them were not validated in biological samples. In general, the mobile phases used in HPLC systems were either acetonitrile (ACN) and/or methanol in combination with water or buffer.
UV detection
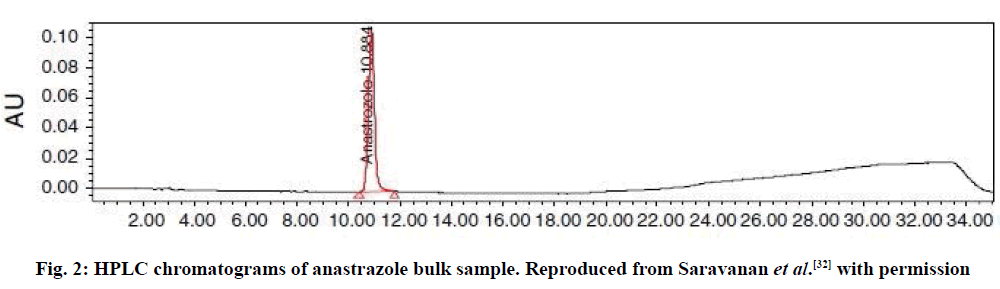
Using a mixture of ACN and methanol (50:50, v/v) as a mobile phase, a gradient reverse-phase liquid chromatography was developed for quantification of ANA in bulk which was achieved with Hichrom RPB 18 (250×4.6 mm, 5 μ) column [32]. The researchers used a flow rate of 1.0 ml/min and an injection volume of 20 μl, monitored at 215 nm. ANA was detected in the bulk drug sample at 10.88 min (fig. 2). The linearity test was performed using six different concentrations ranging between 25 and 150 μg/ml. The calibration graph was plotted using peak area versus concentration (expressed as a percentage). No IS was used to correct for ANA peak area. Interestingly, the intermediate precision was evaluated by an analyst using a different instrument in the same laboratory, and the method was reported to be robust.
Owing to the potential health hazards posed by the presence of impurities in active pharmaceutical ingredients, it is recommended that the levels of impurities should be maintained within an acceptable set limit [39]. Some researchers [40] developed a method of isolation and characterization of three process-related ANA impurities using an Inertsil ODS-3V (250×4.6 mm, 5μ) column. The mobile phase they employed consisted of ACN:ammonium acetate (50:50, v/v) at a flow rate of 1.0 ml/min. However, they used a long analytical time (40 min). In a later related study, Sitaram and co-workers characterized ANA degradation products, detecting two additional impurities [25]. Their method utilized ACN:ammonium formate (40:60, v/v), a flow rate of 0.5 ml/min, with a 20 μl injection volume, monitored at 215 nm. Based on the recommendations of the International Conference on Harmonisation (ICH) [48], these methods are stability-indicating and therefore provide a beneficial assessment of the final ANA purity in the presence of process-related impurities and degradation products.
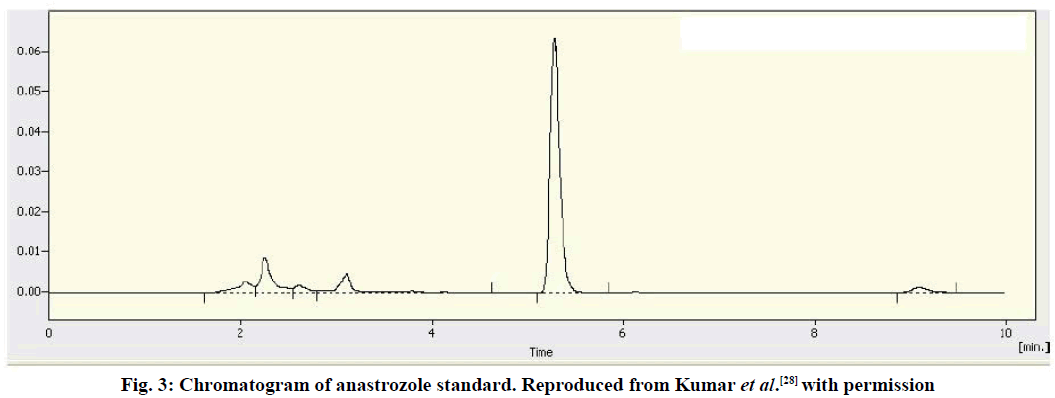
In another study, a reverse-phase isocratic HPLC method for the detection of ANA in tablet dosage forms was developed by Kumar and colleagues using Gracesmart RP18 (100×4.6 mm, 5 μ) [28]. In this approach, a mobile phase mixture of ACN:potassium dihydrogen orthophosphate(50:50, v/v) with a pH of 6 (adjusted with triethylamine) was monitored at 215 nm with a flow rate of 1 ml/min. The total analytical time was 15 min. Manual injections were conducted using a Rheodyne injectable valve (20 μl loop), and the method was validated using three different concentrations of ANA (18, 20 and 22 μg/ml). A linearity test was performed using concentrations ranging between 10 and 30 μg/ml against which the peak area of ANA was used to plot the calibration graph. Again, although the method was linear and comparatively fast (15 min), no IS was used to correct for the peak area (fig. 3).
In another study, a reverse-phase isocratic HPLC method was developed for simultaneous detection of ANA, bicalutamide, TAM and their synthetic impurities using Symmetry®C8 column (100×4.6 mm, 3.5 μm) [29]. The method employed a mobile phase consisting of ACN:water containing N,Ndimethyloctylamine (pH adjusted to 3.0 with orthophosphoric acid) at a flow rate of 1.0 ml/min with an injection volume of 20 μl, monitored at 270 nm. A wave length of 270 nm was selected because it yielded a good resolution of all the analytes without interference peaks from the excipients. The total analytical time was 20 min, and ANA was detected at approximately 4.8 min. The linearity test was conducted using seven different concentrations of standard solutions ranging between 10 and 150 μg/ml. As expected, this method is not without challenges, especially because of the similarities in the chemical properties of the tested drugs and their respective synthetic impurities. Selecting optimal analytical conditions may therefore yield good analyte separation with optimal column pressure; this can be achieved by using a mobile phase consisting of ACN and water at a pH of 3.0 to improve the resolution. Consequently, the method can provide a simple, rapid and cost effective means of simultaneous determination of ANA, bicalutamide, TAM and their synthetic impurities in tablets that can applied by pharmaceutical companies.
Another gradient reverse phase HPLC method was developed for ANA detection in pharmaceutical dosage forms using an Inertsil C8 column (250×4.6 mm, 5 μm) [26]. In this experiment, the mobile phase consisted of ammonium acetate buffer and ACN, at 1 ml/min flow rate with an injection volume of 10 μl. Specific monitoring was conducted at 215 nm. The linearity test was conducted using a concentration of 0.0165-15.000 μg/ml. The intermediate precision (which expresses within-laboratories variations in different days, different analysts and different equipments) was reported to be good (relative standard deviation, RSD=1.58), but the total analytical time was relatively long considering that the retention time for ANA was 23.16 min.
Recently, another group of researchers also developed a method based on an isocratic technique for the quantification of ANA in bulk and in pharmaceutical dosage forms, using water and ACN as the mobile phase which was achieved using an Inertsil ODS column (250×4.6 mm, 5 μ) [27]. In this method, both ANA standard and tablets were used in a solvent system consisting of water. ACN (55:45) and the signals were monitored at 215 nm for 8.00 min. However, in this approach, an IS was not used. Moreover, a higher range of ANA concentrations (2.5-15.0 μg/ml) was used in the linearity test, which may only be applicable in the quantitative analysis of ANA in bulk and pharmaceutical concentrations but not in biological samples such as plasma. In summary, to date, ANA detection based on HPLC methods in human plasma is still lacking.
Gas chromatography
Few gas chromatography (GC) methods have been developed for the quantification of ANA in biological samples. This may be because such techniques are labor-intensive and require extensive sample cleanup to remove the co-extracted compounds from the biological matrix prior to use. A validated analysis for the quantification of ANA in human plasma by GC with electron capture detection (GC-ECD) has been developed [36]. A liquid-liquid extraction method was used to extract the samples with an LOQ of 3.0 ng/ml using Durabond 17 cross-linked fused-silica capillary, 50% phenyl-methyl silicone (15×0.32 mm, 0.25 μm) column. However, the use of a temperature program (an initial temperature of 180° with an initial time of 1 min, at a program rate of 50°/min with a temperature of 240°, followed by time 2 (5.8°/ min) at a rate of 50°/min, temperature of 280° and a final time of 1.2 min) is tedious. In addition, the high temperature may destroy some heat-sensitive compounds such as proteins. Therefore, its application in analyses of large amount of samples may be laborintensive. Subsequently, a similar GC-ECD method was developed [47] to compare the pharmacokinetics data between domestic and imported ANA tablets. The method can be used in bioequivalence studies to differentiate several ANA formulations from different sources.
In an attempt to develop a more comprehensive report of the ANA pharmacokinetic profile, in another study, Duan and co-workers [46] developed a GC-ECD method for the quantification of ANA in plasma samples of healthy subjects using a single step liquid-liquid extraction (LLE) procedure. The LLE involved the addition of 100 μl of diazepam (1 μg/ml) as an internal standard and a drop of ammonia water to 1 ml of plasma. The samples were then extracted by vortexing with 7 ml ethyl ether for 2 min. The organic layer was transferred to a tube and allowed to evaporate to dryness under a nitrogen stream in a water bath at 40°. The residues were redissolved in 150 μl ethyl acetate before injection. The separation was achieved using a HP-50 (50% phenyl-methyl silicone, 30 m×0.53 mm, 1 μm) at a temperature of 260, 230 and 260° for inlet, oven and detector, respectively. This method demonstrated that the two compounds tested were bioequivalent.
Another GC method coupled with flame ionization detection (FID) had a sensitivity of 32 ng/ml LOQ for ANA [35]. The method was applied in the simultaneous detection of TAM, letrozole and ANA in pharmaceutical preparations using an HP-5 column (5% phenyl-methylsilicone, 15 m×0.25 mm, 025 μm) and may serve as an alternative to the traditional analytical methods for quantification of these drugs. However, the application of this method may still be limited in many laboratories due to a lack of availability of FID-equipped instruments.
Liquid chromatography mass spectrometry
There has been significant progress in liquid chromatography mass spectrometry (LC-MS/MS) methods for ANA quantification based on both atmospheric pressure chemical ionization (APCI) and electrospray ionization (ESI) interfaces. Although these methods are very sensitive and specific, they are unpopular in most laboratories in developing countries, perhaps due to their high cost and tedious and complex, time-consuming sample preparations.
An LC-MS/MS method for the simultaneous detection of ANA and exemestane in human urine using liquid-liquid extraction with a low LOQ for ANA has been reported [41]. Samples were analyzed via LC coupled to triple quadrupole mass spectrometer using APCI on an RP 18 column (4×55 mm, 3 μm). This method was demonstrated to be capable of detecting exemestane and its main metabolites in urine samples of women with breast cancer receiving both ANA (1 mg) and exemestane (25 mg). Nevertheless, due to its high cost, this method would be more suitable for the determination of ANA and exemestane levels in situations where blood sample collection is not feasible. A common example is the screening of athletes for some selected anabolic androgenic steroids [41].
One of the earliest reports of ANA quantification in human plasma by LC-MS/MS was published [42]. In this method, a liquid-liquid extraction without cumbersome cleanup procedures was successfully performed using a C 18 column (100×2.1 mm, 4 μ). The 2.5 min analytical time was also the shortest reported at that time. The extracts were analyzed using LC coupled with photospray tandem mass spectrometry. The chromatographic conditions were a mobile phase, which consisted of ACN:methanol:water:acetone (60:20:15:5, v/v/ v/v) containing 0.1% acetic acid and 10 mM of ammonium acetate at a flow rate of 0.45 ml/min. Although as a dopant, toluene may offer better results than acetone, the later was used due to its friendly nature with the selected column (Genesis® C 18, 100×2.1 mm, 4 μm), yielding an LOQ of 0.05 ng/ml of ANA in human plasma, making it suitable for pharmacokinetic studies. However, the mobile phase mixture containing ACN, methanol, water, acetone and acetic acid was quite complex and the atmospheric photospray source may not be readily available in most analytical laboratories, potentially limiting its routine application.
The development and validation of analytical methods based on APCI LC-MS/MS and ESI LC-MS/MS for the determination of ANA levels in human plasma have also been published [30]. Using this method, APCI was significantly more sensitive and specific than ESI for ANA quantification in human plasma. The chromatographic conditions consisted of a mixture of ACN:methanol and ammonium acetate (90:10, v/v) at a pH 3.5 (adjusted by addition of acetic acid), running at 0.6 ml/min with an injection volume of 30 μl and an analytical time of only 1.6 min achieved on a YMC cyano column (50×4.0 mm). Using methyl t-butyl ether (MTBE) as the organic solvent and omeprazole as the IS, a liquid-liquid extraction method was applied to samples containing ANA with a LOQ of 0.10 ng/ml. Overall, the method indicated that, in developing an ANA quantification method, a preliminary comparison between the two ion sources is necessary, particularly when the ANA to be quantified is present in very low concentrations.
Appropriate stability-indicating analytical methods for ANA tablet formulations are also very important because they can be used for stability testing and routine quality control analyses and in other related procedures. Reddy and colleagues have developed a stability-indicating LC-MS/MS method for estimation of the impurity levels present in ANA tablets using an Inertsil ODS-3V (250×4.6 mm, 5 μm) [31]. This method may be useful for separation of ANA from its degradation products, impurities and other related compounds present in the tablet dosage formulation.
Another LC-MS/MS method developed yielded a sensitivity of 0.30 ng/ml LOQ for ANA [43]. The technique was used in conjunction with the solidphase extraction of plasma samples. The extracts were separated by an isocratic method (achieved on Hypurity C18, 50×4.6 mm, 5 μm column) using a mobile phase that consisted of ACN:formic acid (0.1%) (88:12, v/v) at a flow rate of 0.4 ml/min and an injection volume of 5 μl. Monitoring was conducted using tandem mass spectrometry in positive ion mode for a total analytical time of 2.5 min, which is short and time-saving. Nevertheless, the high (88%) percentage of organic solvent used makes the method unaffordable if used over a long time period.
A group of researchers developed an LC-MS/ MS method, using a triple quadrupole ion trap spectrometry, for the simultaneous detection of TAM, letrozole and ANA using Si C18 column (200×0.5 mm, 5 μm) [44]. A linear gradient separation technique was employed for an analytical time of 16 min. Plasma samples were analyzed following solid-phase extraction with LOQ of 26.1, 17.0 and 6.0 ng/ml for TAM, letrozole and ANA respectively. Although the sensitivity of this method in terms of LOQ for ANA (6.0 ng/ml) is relatively lower than previous published reports, it offers the advantage of monitoring patients’ compliance to treatment with TAM, letrozole and ANA, especially in clinical studies. Furthermore, the method can also guide clinicians for the objective evaluation of drug concentration-dependent side effects and treatment failure. Likewise, Liu et al [45] reported an LC-MS/ MS method for the simultaneous detection of ten different endocrine drugs (raloxifene, mifepristone, finastride, TAM, N-desmethyltamoxifen, clomiphene, toremifene, exemestene, letrozole and ANA) in hospitals and in municipal wastewater samples. The chromatographic conditions were a mobile phase consisting of formic acid and ACN with a flow rate of 0.3 ml/min in a gradient elution. The separation was achieved on a Waters ACQUITY™ UPLC™ BEH C18 column (100×2.1 mm, 1.7 μm). The target analytes were analyzed using triple quadrupole mass spectrometer. The LOQ of this method was reported to be between 0.1 and 2.0 ng/ml.
Two interesting LC-MS/MS methods have recently been developed to measure the levels of ANA and its metabolites in vitro and in vivo using a triple quadrupole system [18,21]. These methods can assess inter-individual variation in ANA metabolism and pharmacodynamics among breast cancer patients receiving the recommended daily dose (1 mg) and are therefore useful in pharmacogenetic studies of ANA. However, variation may still exist in the mass spectrometric properties of ANA and its metabolites due to the altered molecular weight, making the interpretation of formation rates (pmol per minute per mg protein) less accurate.
In terms of sensitivity and sample throughput for the analysis of drugs in biological samples, ultraperformance liquid chromatography (UPLC)- MS/MS has advantages over the conventional LCMS/ MS [49]. To test this hypothesis with respect to the quantification of ANA in human plasma, Yu and coworkers developed and validated an UPLC-MS/MS method involving a one-step liquid-liquid extraction with diethyl ether that required only 500 μl of plasma sample [34]. The chromatographic conditions included a mobile phase of methanol:ammonium acetate (75:25, v/v) set at a flow rate of 0.30 ml/min. the separation was performed on a Waters ACQUITY™ UPLC™ BEH C18 column (50×2.1 mm, 1.7 μm). Detection was conducted using triple quadrupole tandem mass spectrometry via multiple reactions monitoring (MRM) by ESI, with a total analytical time of only 1.5 min. The method achieved excellent sensitivity with an LOQ of 0.055 ng/ml.
Conclusions
Many different chromatographic methods have been reported for ANA quantification, both in pharmaceutical dosage formulations and in biological matrices. Some HPLC methods with sensitivity as high as 0.0165 μg/ml LOQ have been developed for ANA quantification. However, due to the low recommended dosage (1 mg daily) of ANA in breast cancer treatment, application of these methods for the quantification of ANA in biological matrices is rather limited. In addition, some methods require improvements in their approaches to method validation and linearity testing. A number of LC-MS/ MS methods with higher sensitivity (as high as 0.05 ng/ml) and shorter analytical time have also been developed and validated for ANA quantification in human plasma. On the contrary, very few GC-MS methods have been developed for ANA measurement in plasma. In general, potential areas in which chromatographic methods need to be improved include developing appropriate method validations and linearity testing based on internationally recognized guidelines. Therefore, simpler and more affordable, potential UPLC-based methods coupled with mass spectrometry are required, especially in developing countries where access to instruments is limited by insufficient research funding.
Financial Assistance
None.
Conflict of Interests
None declared.
References
- GLOBOCAN. Estimated Cancer Incidence, Mortality and Prevalence Worldwide in 2012. Available from: http://globocan.iarc.fr/Default.aspx. [last accessed on June 10, 2014].
- Elledge RM, Osborne CK. Oestrogen receptors and breast cancer. BMJ 1997;314:1843-4.
- Osborne CK. Tamoxifen in the treatment of breast cancer. N Engl J Med 1998;339:1609-18.
- Smith IE, Dowsett M. Aromatase inhibitors in breast cancer. N Engl J Med 2003;348:2431-42.
- Pan K, Chlebowski RT. Adjuvant endocrine therapy of perimenopausal and recently postmenopausal women with hormone receptor positive breast cancer. Clin Breast Cancer 2014;14:147-53.
- Braithwaite RS, Chlebowski RT, Lau J, George S, Hess R, Col NF. Meta?analysis of Vascular and Neoplastic Events Associated with Tamoxifen. J Gen Intern Med 2003;18:937-47.
- Lewis S. Do endocrine treatments for breast cancer have a negative impact on lipid profiles and cardiovascular risk in postmenopausal women? Am Heart J 2007;153:182-8.
- Fisher B, Costantino JP, Wickerham DL, Cecchini RS, Cronin WM, Robidoux A, et al. Tamoxifen for the prevention of breast cancer: current status of the National Surgical Adjuvant Breast and Bowel Project P-1 study. J Nat Cancer Inst 2005;97:1652-62.
- Perez E. Safety profiles of tamoxifen and the aromatase inhibitors in adjuvant therapy of hormone-responsive early breast cancer. Annals Oncol 2007;18(suppl 8):26-35.
- Normanno N, Di Maio M, De Maio E, De Luca A, de Matteis A, Giordano A, et al. Mechanisms of endocrine resistance and novel therapeutic strategies in breast cancer. Endocrine-related Cancer 2005;12:721-47.
- Zilli M, Grassadonia A, Tinari N, Di Giacobbe A, Gildetti S, Giampietro J, et al. Molecular mechanisms of endocrine resistance and their implication in the therapy of breast cancer. BiochimBiophysActa (BBA)-Rev Cancer 2009;1795:62-81.
- Fabian C. The what, why and how of aromatase inhibitors: hormonal agents for treatment and prevention of breast cancer. Int J ClinPrac 2007;61:2051-63.
- Goldhirsch A, Ingle JN, Gelber RD, Coates AS, Thurlimann B, Senn H-J. Thresholds for therapies: highlights of the St Gallen International Expert Consensus on the primary therapy of early breast cancer 2009. Annals Oncol 2009;20:1319-29.
- National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology. Breast Cancer 2012, Available from: http://infoonco.es/wp-content/uploads/2011/08/Breast-Cancer-V1.2012.pdf.
- Plourde PV, Dyroff M, Dukes M. Arimidex®: a potent and selective fourth-generation aromatase inhibitor. Breast Cancer Res Treat 1994;30:103-11.
- Geisler J, King N, Dowsett M, Ottestad L, Lundgren S, Walton P, et al. Influence of anastrozole (Arimidex), a selective, non-steroidal aromatase inhibitor, on in vivo aromatisation and plasma oestrogen levels in postmenopausal women with breast cancer. Br J Cancer 1996;74:1286-91.
- Simpson E. Sources of estrogen and their importance. J steroid BiochemMolBiol 2003;86:225-30.
- Ingle JN, Buzdar AU, Schaid DJ, Goetz MP, Batzler A, Robson ME, et al. Variation in anastrozole metabolism and pharmacodynamics in women with early breast cancer. Cancer Res 2010;70:3278-86.
- AstraZeneca. Arimidexanastrozole tablets 2009. Available from: http://www1.astrazeneca-us.com/pi/arimidex.pdf.
- Lazarus P, Sun D. Potential role of UGT pharmacogenetics in cancer treatment and prevention: focus on tamoxifen and aromatase inhibitors. Drug Metab Rev 2010;42:182-94.
- Kamdem LK, Liu Y, Stearns V, Kadlubar SA, Ramirez J, Jeter S, et al. In vitro and in vivo oxidative metabolism and glucuronidation of anastrozole. Br J ClinPharmacol 2010;70:854-69.
- Sanford M, Plosker GL. Anastrozole: a review of its use in postmenopausal women with early-stage breast cancer. Drugs 2008;68:1319-40.
- Ingel JN, Kalari KR, Buzdar AU, Robson ME,Goetz MP, Desta Z, et al.Estrogens and their precursors in postmenopausal women with early breast cancer receiving anastrozole. Steroids 2015;99:32-8.
- Kumar S, Harani A, Reddy TR, Sucharitha G, Krishna P, Priyanka SJ. Development and validation of spectrophotometric method for estimation of anastrozole bulk and pharmaceutical dosage formulation.Int J Adv Pharm Sci 2011;1:D. .
- Sitaram C, Rupakula R, Reddy BN. Determination and characterization of degradation products of anastrozole by LC-MS/MS and NMR spectroscopy. J Pharm Biomed Anal 2011;56:962-8.
- Daphal V, Holkar G, Yadav R, Rokade M. Development and Validation of Simultaneous Detrmination of Anastrozole and Temozolomide in Pharmaceutical Dosage Forms. Int J TheoretApplSci 2012;4:48-55.
- Krishnaveni K, Nalini Y, Srinivas P. Development and Validation of stability indicating RP-HPLC method for estimation of Anastrozole in Bulk and Pharmaceutical Dosage Form. Int J Pharm Sci 2013;3:375-80.
- Kumar DS, Harani A, Sridhar D, Banji D, Rao K, Aran Y. Development and Validation of a HPLC Method for Determination of Anastrozole in Tablet Dosage Form. J Chem 2011;8:794-7.
- Gomes FP, Garcia PL. Development and validation of a simple and sensitive high performance liquid chromatographic method for the simultaneous determination of anastrozole, bicalutamide, tamoxifen, and their synthetic impurities. Talanta 2012;101:495-503.
- Apostolou C, Dotsikas Y, Kousoulos C, Loukas YL. Development and validation of an improved high-throughput method for the determination of anastrozole in human plasma by LC-MS/MS and atmospheric pressure chemical ionization. J Pharm Biomed Anal 2008;48:853-9.
- Reddy YR, Nandan SR. LC and LC-MS/MS study of forced decomposition behavior of anastrozole and establishment of validated stability-indicating analytical method for impurities estimation in low dose anastrozole tablets. J Pharm Biomed Anal 2009;50:397-404.
- Saravanan G, Suryanarayana MV, Jadhav MJ, Ravikumar M, Someswararao N, Acharyulu PVR. A stress stability behavior and development of an LC assay method for anastrozole. Chromatographia 2007;66:435-8.
- Daphal V, Holkar Ganesh, Yadav R, Rokade MD. Method Validation and Quantitative Determination of Anastrozole in Human Plasma by an CMS/MS. Bio Forum-Int J 2012;4:55-60.
- Yu J, He J, Zhang Y, Qin F, Xiong Z, Li F. An ultraperformance liquid chromatography-tandem mass spectrometry method for determination of anastrozole in human plasma and its application to a pharmacokinetic study. Biomed Chromatogr 2011;25:511-6.
- Berzas JJ, Rodriguez J, Contento AM, Cabello MP. Determination of drugs used in advanced breast cancer by capillary gas chromatography of pharmaceutical formulations. J Sep Sci 2003;26:908-14.
- Bock MJ, Bara I, LeDonne N, Martz A, Dyroff M. Validated assay for the quantification of anastrozole in human plasma by capillary gas chromatography-63Ni electron capture detection. J Chromatogr B Biomed SciAppl 1997;700:131-8.
- Bharati P, Vinodin A, Reddy A, Devi P. Development and validation of a planar chromatographic method with reflectance scanning densitometry for quantitative analysis of anastrozole in the bulk material and in tablet formulations. JPC-J Planar Chromatogr 2010;23:79-83.
- Willis DE. Internal standard method calculations. Chromatographia 1972;5:42-43.
- Guideline IHT. Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients. Q7A, Current Step 2005;4
- Hiriyanna S and Basavaiah K. Isolation and characterization of process related impurities in anastrozole active pharmaceutical ingredient. J BrazChemSociet 2008;19:397-404
- Mareck U, Geyer H, Guddat S, Haeelt N, Koch A, Kohler M, et al. Identification of the aromatase inhibitors anastrozole and exemestane in human urine using liquid chromatography/tandem mass spectrometry. Rapid Commun Mass Spectrom 2006;20:1954-62.
- Mendes GD, Hamamoto D, Ilha J, Pereira Ados, S, De Nucci. Anastrozole quantification in human plasma by high-performance liquid chromatography coupled to photospray tandem mass spectrometry applied to pharmacokinetic studies. J Chromatogr B AnalytTechnol Biomed Life Sci 2007;850:553-9.
- Jangid AG, Pudage AM, Joshi SS, Pabrekar PN, Tale RH, Vaidya VV. A simple, selective and rapid validated method for estimation of anastrazole in human plasma by liquid chromatography-tandem mass spectrometry and its application to bioequivalence study. Biomed Chromatogr 2010;24:727-31.
- Beer B, Schubert B, Oberguggenberger A, Meraner V, Hubalek M, Oberacher H. Development and validation of a liquid chromatography-tandem mass spectrometry method for the simultaneous quantification of tamoxifen, anastrozole, and letrozole in human plasma and its application to a clinical study. Anal BioanalChem 2010;398:1791-800.
- Liu X, Zhang J, Yin J, Duan H, Wu Y, Shao B. Analysis of hormone antagonists in clinical and municipal wastewater by isotopic dilution liquid chromatography tandem mass spectrometry. Anal BioanalChem 2010;396:2977-85.
- Duan G, Liang J,Zuo M. Rapid determination of anastrozole in plasma by gas chromatography with electron capture detection and its application to an oral pharmacokinetic study in healthy volunteers. Biomed Chromatogr 2002;16:400-3.
- Yuan J, Wang PQ, Ge SR, An FR, Shi AG, Chen J, et al. Pharmacokinetics of anastrozole in Chinese male volunteers. ActaPharmacol Sin 2001;22:573-6.
- ICH Q2B, Harmonized Tripartite Guideline: Validation of Analytical Procedures: Methodology, IFPMA in: Proceedings of the International Conference on Harmonization, Ontario, Canada.
- Churchwell MI, Twaddle NC, Meeker LR, Doerge DR. Improving LC-MS sensitivity through increases in chromatographic performance: comparisons of UPLC-ES/MS/MS to HPLC-ES/MS/MS. J Chromatogr B AnalytTechnol Biomed Life Sci 2005;825:134-43.