- *Corresponding Author:
- R. Shri
Department of Pharmaceutical Sciences and Drug Research, Punjabi University, Patiala-147 002, India
E-mail: rshri587@hotmail.com
| Date of Submission | 05 December 2016 |
| Date of Revision | 01 April 2017 |
| Date of Acceptance | 02 November 2017 |
| Indian J Pharm Sci 2018; 80(1):02-13 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
Myrica esculenta (Myricaceae) commonly known as box berry or kaphal is an important Indian medicinal plant. It is found in foothill tracks of Eastern Himalayas, Meghalaya, Nepal, China and Pakistan. Local tribes mainly use its fruits to prepare pickle and refreshing drinks. Traditionally, the bark has been used for the treatment of cough, asthma, fever, chronic bronchitis, diarrhoea, rheumatism and inflammation; roots have been used in bronchitis, asthma, cholera and flowers claimed to treat earache, diarrhoea, paralysis. Phytochemical studies of the different parts of plant revealed the presence of various bioactive phytoconstituents such as phenolic compounds, alkaloids, glycosides, triterpenoids and volatile oils. The plant is also reported to have innumerable significant pharmacological activities like analgesic, anxiolytic, antiallergic, antidiabetic, antimicrobial, antihypertensive, antiulcer, antioxidant and antiinflammatory evaluated by using various animal models. The objective of the present review article is to compile all the relevant published information regarding traditional uses, phytochemistry and therapeutic potential of M. esculenta. For this purpose various databases and books were examined. The review clearly demonstrates the importance of this plant in ethnomedicine and its immense potential in modern medicine.
Keywords
Myrica esculenta, kaphal, box berry, phytoconstituents, pharmacology
The genus Myrica consists about 97 species of small tree and aromatic shrubs belonging to family Myricaceae. These are reported to be globally distributed in both temperate and sub-tropical regions of the world [1]. There is only one species Myrica australiasica F. Muell, which has been reported in Australia [2] while M. cerifera L. (Wax myrtle/Southern wax myrtle) and M. persylvanica Mirb. (Northern bayberry) [3] are reported as official drugs of North America [4] and claimed to have same medicinal properties there, as M. esculenta in the Indian systems of medicine [5]. The another species of genus Myrica, such as M. rubra known as Chinese bayberry are commonly found in China and Japan only [6-8]. Some other species, which belong to genus Myrica are M. adenophora hance, M. caroliniesis (evergreen bayberry), M. cordifolia (waxberry/candle berry), M. californica (Californian bayberry), M. dentulata Baill., M. heterophylla Raf. (swamp bayberry), M. inodora W. Bartram (odourless bayberry), M. integra (A. chev.) Killick, M. nana A. Chev., M. quercifolia L. [6,9], M. faya Ait., M. gale L. (bog-myrtle/sweet gale) [6,8,10], M. hartwegi S. Watson (Sierra babyberry/mountain wax myrtle) [6,10].
M. esculenta commonly known as Boxberry, Kaiphal and Kathphala has been reported to be the only species found in India [11]. Its synonyms are M. nagi Hook. F. non Thunb [12], M. sapida Wall. [13] M. farqhariana Wall. and M. integrifolia Roxb. [10,14]. It is an economical medicinal plant with multipurpose uses [15]. Being actinorhizal, it is also useful in regeneration of nitrogen depleted soils [1]. The plant is primarily sourced for the fruits, which are one of the tastiest wild fruits of the sub-Himalayan region [16] and have recognized for its nutritional and therapeutic potential [17]. Fruits of the plant are also used to prepare jams, syrups, refreshing drinks and pickles [18]. It is the rich source of vitamin C and polyphenolic compounds such as tannins, phenols, flavonoids and flavonols [19]. Local tribes utilize tree as timber, fuel, fodder, wood [20] as well as used for tanning and obtaining yellow colored dye [21,22].
In spite of being a multipurpose tree, the cultivation of the plant is very limited and most of the traditional and commercial uses of M. esculenta plant depend exclusively on the collections from the wild sources by indigenous people [23]. Thus, the species is under imminent danger of extinction from wild sources due to increase in urbanization, over harvesting, negligence of sustainable utilization and over exploitation of forests and waste lands for commercial uses [18,24]. Poor regeneration in natural habitat due to high anthropogenic activity is another important factor, which affect the natural population of this plant species [25]. The present review article is an attempt to summarize the ethnomedical uses, phytochemistry and therapeutic potential of M. esculenta for its future prospects such as conservation, cultivation and sustainable utilization as well as to recognise the medicinal properties of this plant in the modern system of medicine.
Taxonomy
Botanical classification and vernacular names
Kingdom: Plantae, Division: Magnoliophyta, Class: Magnoliopsida, Order: Fagales, Family: Myricaceae, Genus: Myrica, Species: M. esculenta [12]. Assami: Ajooree, Nagatenga, Vdulbark, Bengali: Kayachhal, Kaiphal, Satsarila, English: Box myrtle, Bayberry, Gujrati: Kariphal, Hindi: Kapha, Kaiphal, Kannada: Kandujai kai, Kirishivani, Kirishivane, Malayalam: Maruta, Marathi: Kayaphala, Nepali: Kobuli, Katphala, Punjabi: Kaiphal, Kahela, Kahi, Sanskrit: Kathphala, Aranya, Krishnagarba, Mahavalkala, Tamil: Marudam, Marudampatai, Telugu: Kainaryamu [13,16,26].
Habitat
M. esculenta is a subtemperate evergreen dioecious small tree [26]. It is native to India and found to be widely distributed in the foothills track of mid Himachal Pradesh starting from Ravi eastwards to Assam including Arunachal Pradesh, Sikkim, Manipur, Uttranchal and Khasi, Jaintia, Naga and Lushai Hills of Meghalaya in between 900-2100 m above the sea level [27-29]. Apart from India, it is also found in Nepal [30], China, Japan [9], Pakistan, Singapore, and Malaya Islands [6].
Botanical Description
Morphological evaluation of the plant M. esculenta (Figure 1A) reveals that it is medium to large woody tree about 12-15 m in height with trunk diameter about 92.5 cm [28]. The outer bark is greyish dark in colour, rough, vertically wrinkled while inner bark is dark brown in colour with smooth surface (Figure 1B); fracture hard; bitter in taste and nauseating odour [5,26]. Leaves (Figure 1C) are lanceolate with entire or serrate margin, having pale green at lower surface and dark green at upper surface, about 9-12 cm in length and 3-3.5 cm in width and are mostly crowded toward the ends of branches [16,28]. Pistillate flowers are small, sessile, solitary and bracteates; sepals and petals are either absent or not visible; inflorescence (catkin), 4.2 cm long, axillary, bearing about 25 flowers in a thread-like style [28,31] while inflorescence of staminate flowers is compound raceme [25]. The flowering season starts from February and continues till second week of April but peak season for flowering was observed during first week of March while the fruiting season was observed during the first week of May, which continues till the end of May [22]. The tree yields a drupe fruit, red to dark brown in colour, ellipsoidal or oval in shape and about 2-7 mm in diameter (Figure 1D) having sweet and sour taste containing ovoid shaped, smooth surface light brown coloured seed of about 1-6 mm in diameter with oily taste [17,26].
Microscopical evaluation
Transverse section through midrib shows upper and lower epidermis, few epidermal cells on upper surface elongated to form non glandular/covering unicellular trichomes. Below the epidermis, single layer of palisade cells present followed by spongy parenchyma and vascular strands. Midrib region of leaf shows vascular bundle containing xylem and phloem surrounded by collenchyma cells. Prism shaped calcium oxalate crystals, unicellular thin walled covering trichomes, starch grains, anisocytic stomata and non-lignified fibres were observed as microscopical diagnostic features of powder microscopy of leaves [32].
The transverse section of matured M. esculenta bark shows multi-layered rectangular shaped thick lignified cork cells followed by 5-8 layers of phelloderm cells consisting of narrow and tangentially flattened parenchyma cells with single or small groups of round to slightly oval shaped thick walled lignified stone cells, spheroidal shaped starch grains and prismatic calcium oxalate crystals. The phloem tissue was composed of thick walled sieve tubes, companion cells, thick lignified phloem parenchyma phloem fibres, stonecells and some of the crystal fibres and was traversed by medullary rays. Prisms of calcium oxalate were present in crystal fibres and in phloem parenchyma. The reported diagnostic microscopical characters of M. esculenta powder are numerous calcium oxalate crystals up to 24.8 μm in size, spheroidal shaped starch grains up to 15.5 μm in diameter, rounded or rectangular shaped thick lignified stone cells up to 370 μm in length, thick walled sclerenchyma fibres usually accompanied by crystal fibres and fragments of the parenchyma, some of them rich in starch grains [5,26].
Epicarp shows single layer, unlignified, thin walled, reddish brown parenchymatous cells with numerous unicellular trichomes, a few elongated tubercled cell with smooth walls; endocarp hard and stony consisting of sclerenchymatous cells [6,26,31]. Seed coat shows single layered, thick, brown coloured cell; cotyledons composed of single layered, thin walled epidermal cells containing oil globules and aleurone grains; mesophyll cell thin-walled, isodiametric densely packed with oil globules and aleurone grains [6,26,31].
Ethnobotany
A large part of population living in rural areas of Uttrakhand use stem bark to cure chronic cough, asthma and ulcers [33] and bark powder is inhaled to cure headache [34]. Local people of Sub-Himalayan region use decoction of bark as mouth freshener and to cure toothache [35] while paste of bark is applied on wounds, joints pains and paralysis as well as used to cure cold and headache [36]. Bark is also used in the treatment of mental illness by different ethnic groups of the rural region of Orissa [37]. Fruit are eaten raw or used to prepare refreshing drinks while its juice is used against bacterial dysentery by tribal communities of Meghalaya [38-40] and bark is chewed to relieve toothache and for washing putrid sores [22,41]. The paste of leaves is applied externally by local tribe of Uttranchal to cure headache [42].
Traditional uses
M. esculenta is well recognized medicinal plant since from ancient Ayurveda and Unani system of medicine [43]. The detailed report on traditional uses of M. esculenta plant are summarised in Table 1 [44-48].
| Part | Traditional uses | References |
|---|---|---|
| Fruits | Claimed to act as sedative, stomachic, carminative, antiulcer | [28,44] |
| Used in abdominal tumours, asthma, fever, piles, irregular bowel function, anaemia, nausea, oral disorders, cough, dyspnoea | [26] | |
| Useful to retain placenta and bone fracture | [13] | |
| Juice of the unripe fruit is used as an antihelmintic. | [45] | |
| Fruit wax or oil is used to cure bleeding piles, toothache, menorrhagia and other menstrual disorders | [6,13] | |
| Bark | Reported to be used as astringent, stimulant, antiseptic, carminative, antirheumatic | [13,46,47] |
| claimed to be useful in the treatment of abdominal tumours, asthma, chronic bronchitis, fever, piles, ulcer, anaemia, diarrhoea, dysentery, nausea, oral disorders, cough, dyspnoea, indigestion, anorexia, ear, nose and throat disorders | [13,26,28,46] | |
| Bark oil is used in earache | [28] | |
| Act as fish poison | [12,48] | |
| Decoction of bark in combination with Quercuslanatabark used in the treatment of dysentery and in form a gelatinous mass it is applied as a poultice on sprains | [45] | |
| Bark powder mixed with ginger is used as a rubefacient in the treatment of cholera. Externally bark juice is applied to heal cuts and wounds while internally it is used to cure catarrh and headache | [45] | |
| Flowers | Oil from flower has been found to be useful in earache, diarrhoea, paralysis and inflammation | [16,28,34] |
| Roots | Used in bronchitis, asthma, cholera and cough | [12] |
Table 1: Traditional uses of M. Esculenta plant
Ayurvedic formulations
Fruits and roots are used to prepare Ayurvedic formulations such as Chwayanprash and Brahmarasayan to enhance digestion, memory, intelligence, concentration and physical strength [49]. The bark of M. esculenta is used as one of the common ingredient of Ayurvedic formulation Visweshwara rasa to treat fever of kapha and pitta origin [50]. Other Ayurvedic formulations, which contain fruits or bark of M. esculenta as an important ingredient include Katphaladi churna, Kaas-har churna, Katphala taila, Katphala kvatha, Khadiradi gutika, Maha vatagajankush rasa, Brihatphala ghrita, Pusyanuga churna, Arimedadi taila, Bala taila, Mahavisagarbha taila used for the treatment of various ailments such as rheumatoid arthritis, diarrhoea, dysentery, headache, menorrhagia and other menstrual disorders [26,51].
Nutritional Value
The proximate analysis of nutrients such as crude fibre, crude protein, crude fat, crude carbohydrates, ash value, moisture content and mineral contents such as Na, K, Ca, Mg, Fe, Zn, Mn, Cu of M. esculenta fruits were evaluated [52,53]. The results reported in Table 2 supported the use of fruit for nutritional purpose and adequate protection may be obtained against diseases arising from malnutrition if consumed in sufficient amount.
| Parameters | Value |
|---|---|
| Ash (%) | 2.18±0.02 |
| Moisture content (%) | 72.33±0.23 |
| Crude fat (%) | 4.93±0.06 |
| Crude fibre (%) | 5.22±0.08 |
| Crude protein (%) | 9.62±0.03 |
| Carbohydrates (%) | 78.03±0.14 |
| Energy (Kcal/g) | 395.04±0.54 |
| Minerals (mg/g): | |
| Calcium | 4.63±0.06 |
| Magnesium | 8.4±0.20 |
| Potassium | 7.75±0.11 |
| Phosphorus | 0.24±0.25 |
| Sodium | 0.81+0.013 |
| Manganese | 0.032+0.0001 |
| Zinc | 0.216+0.0016 |
| Iron | 0.404+0.0021 |
| Copper | 0.004+0.0002 |
Table 2: Nutitional value of M. Esculenta fruit
Phytochemistry
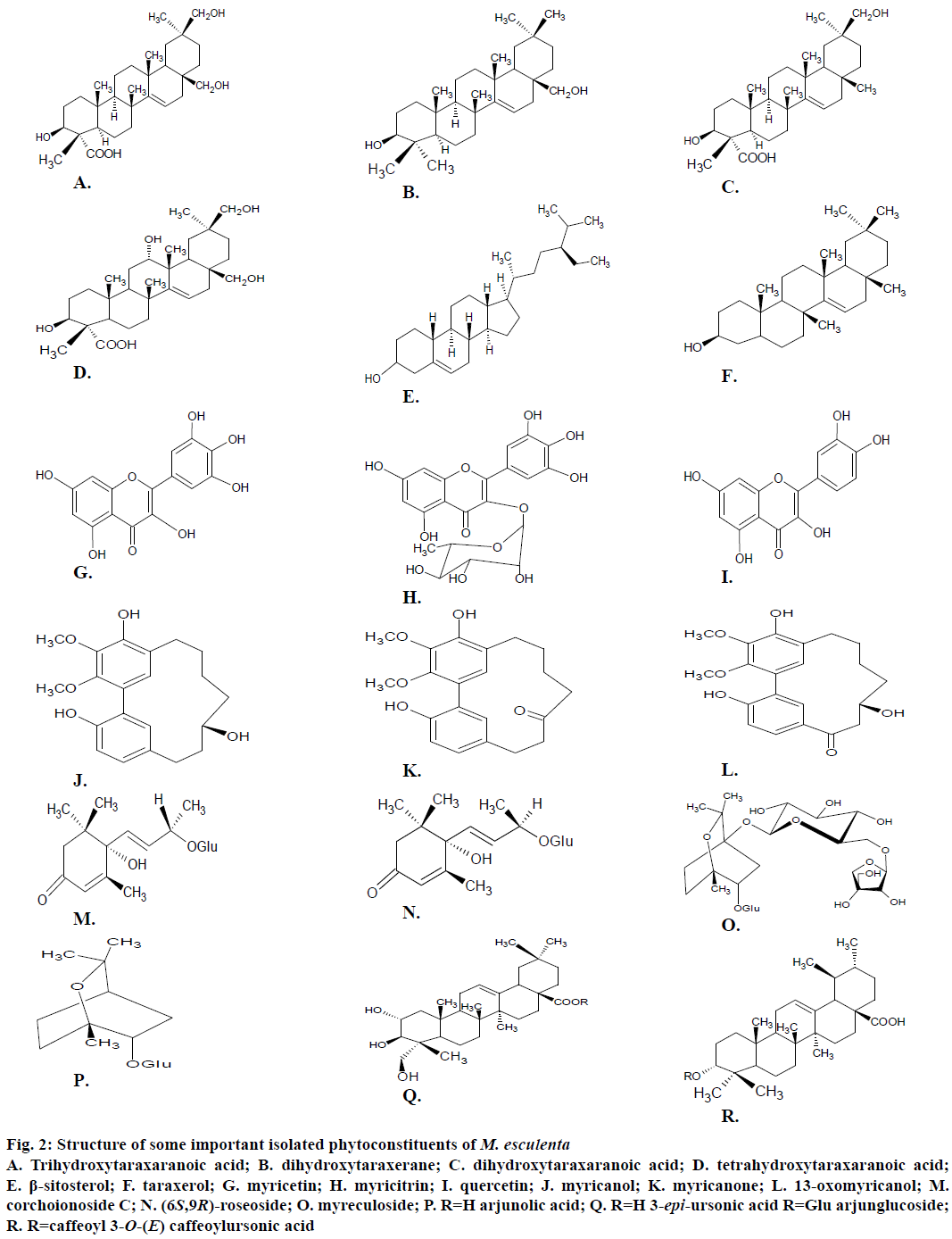
Various preliminary phytochemical studies carried out on the fruits [53], leaves [54] and bark [5,24,53] of M. esculenta showed the presence of various active phyto-constituents that exhibit a variety of biological effects. This plant is found to be a rich source of phenolic compounds, flavonoids and flavonols [25]. Other bioactive compounds reported in the plant belong to the class of alkaloids, glycosides, diarylheptanoids, ionones, steroids, saponins, triterpenoids, volatile compounds, which are listed in the Table 3 [55-73]. The structure of some important bioactive phytoconstituents reported in M. esculenta plant is presented in Figure 2.
| Phyto-constituent | Plant source and reference |
|---|---|
| Tannins and phenolic acids: | |
| Tannin, ascorbic acid | Fruits [13,21,55] |
| Gallic acid | Bark [56,57], fruits [58], leaves [59] |
| Catechin, cholorogenic acid, p-coumaric acid, caffeic acid, trans-cinnamic acid, ellagic acid | Fruit [58] |
| Ethyl-β-D-glucopyranoside; 3-hydroxybenzaldehyde, isovanillin, 4-(hydroxymethyl) phenol, 4-methoxybenzoic acid | Leaves [59] |
| castalagin, epigallocatechin-3-O-gallate; epigallocatechin-(4β→8)-epigallocatechin-3-O-gallate; 3-O-galloylepigallocatechin-(4β→8)-epigallocatechin-3-O-gallate | Bark [56] |
| Flavonoids: | |
| Myricetin | Bark [60,61], fruits [19], leaves [62] |
| Quercetin | Leaves [63] |
| Flavonoid Glycosides: | |
| Myricitrin (myricetin 3-O-rhamnoside) | Bark [61], leaves [59,62] |
| Flavone 4'-hydroxy-3',5,5'-trimethoxy-7-O-β-D-glucopyranosy) (1→4)-α-L-rhamnopyranoside; flavone 3',4'-dihydroxy-6-methoxy-7-O-α-L-rhamnopyranoside | Leaves [63] |
| myricetin-3-O-(3''-Ogalloyl)-α-L-rhamnoside; myricetin-3-O-(2''-Ogalloyl)-α-L galactopyranosideside; myrecetin 3-O-(2''-O-galloyl)-α-L-rhamnopyranoside | Bark [61] |
| Steroids: | |
| β–sitosterol | Bark [12,64], leaves [59,63] |
| Taraxerol | Bark [12,64] |
| Stigmasterol | Bark [57] |
| β-rosasterol, daucosterol | Leaves [59] |
| β-sitosterol-β-D-glucopyranoside | Leaves [63] |
| Ionones: | |
| Corchoionoside C; (6S,9R)-roseoside | Leaves [62] |
| Diarylhetanoids: | |
| Myricanol | Bark [65,56,66], leaves [59,62] |
| Myricanone | Bark [56,66], leaves [59,62] |
| 16 bromomyricanol | Bark [66] |
| 5-O-β-D-glucopyranosylmyricanol | Leaves [62] |
| 13-Oxomyricanol | Root [67] |
| Terpenes: | |
| 3-epi-ursolic acid; 3-O-(E)-caffeoylursonic acid | Leaves [62] |
| Lupeol; oleanolic acid; | Bark [57] |
| Triterpenediol (3β, 28-dihydroxytaraxerane) | Bark [12,68] |
| 3β,30-dihydroxy-taraxerane-23-oic acid; 3β, 28, 30-trihydroxy- taraxara-23-oic acid; 3β, 12α, 28, 30-tetrahydroxytaraxeran-23-oic acid | Bark [68] |
| Monoterpinoidal glycosides: myresculoside (4-hydroxy-1,8-cineole 4-O-β-dapiofuranosyl (1→6)-β-D-glucopyranosie); (1S,2S,4R)-2-hydroxy-1,8-cineole β-D-glucopyranoside | Leaves [62] |
| Triterpinoidal glycosides: arjunglucoside | Leaves [62] |
| Proanthocyanidin: | |
| Proanthocyanidin acetate, proanthocyanidin methyl-ether | Bark [69] |
| prodelhinidin | Bark [70] |
| Volatile compounds: | |
| Nerolidol, α-pinene, α-selinene, β-caryophyllene, β-selinen, α-caryophyllene, α-cadinol, linalool | Leaves [71] |
| n-Hexadecanol; eudesmol acetate; palmitic acid; cis-β-caryophyllene; n-pentadecanol; n-octadecanol | Bark [72] |
| Saponin: | |
| Arjunolic Acid | Leaves [62] |
| Others: | |
| Amino acids: L-Hydroxyproline, iso-leucine, valine, 2-aminobutyric acid, L-cystein hydroxyl, L-cysteinhydroxychloride, alanine, leucine, tryptophan, glutamic acid, tyrosine, threonine, lysine monochloride | Fruit [53] |
| 2-Furancarboxyaldehyde, 2,5-furandionedihydro-3-methylene, furfural, oxirane, myo-inositol, 1-ethyl-4-methylcyclohexane, methyl-d-lyxofuranoside | Fruits [73] |
Table 3: Various Phytoconstituents isolated from different parts of M. Esculenta
Figure 2: Structure of some important isolated phytoconstituents of M. esculenta
A. Trihydroxytaraxaranoic acid; B. dihydroxytaraxerane; C. dihydroxytaraxaranoic acid; D. tetrahydroxytaraxaranoic acid; E. β-sitosterol; F. taraxerol; G. myricetin; H. myricitrin; I. quercetin; J. myricanol; K. myricanone; L. 13-oxomyricanol; M. corchoionoside C; N. (6S,9R)-roseoside; O. myreculoside; P. R=H arjunolic acid; Q. R=H 3-epi-ursonic acid R=Glu arjunglucoside; R. R=caffeoyl 3-O-(E) caffeoylursonic acid
Pharmacology
The traditional uses of M. esculenta have inspired researchers to verify its utility through scientific pharmacological screening. Several crude extracts from various parts of the plant and isolated bioactive compounds have been evaluated for different biological activities such as analgesic, antiasthmatic, anticancer, antioxidant, antiinflammatory, antidiabetic, antiulcer, anxiolytic, hepatoprotective, chemopreventive, hypotensive and wound healing activity by using a number of in vitro and in vivo animal models, which prove the traditional utilization of this plant scientifically.
Analgesic activity
Oral administration of methanol extract of the fruits of M. esculenta manifested a significant (p<0.05) analgesic activity in dose-dependent manner by increasing the paw licking and jumping time on hot plate until 50 min as compare to control by using Eddy’s plate method whereas hot plate reaction time in indomethacin treated mice was maximum at 60 min [74]. The ethyl acetate fraction of methanol extract (ME-EtAC) of leaves was found to possess significant analgesic effect in acetic acid-induced writhing assay and tail immersion assay [32] while methanol extract of leaves showed 54.56 % inhibition of writhing in acetic acid-induced writhing model at the dose of 200 mg/kg [75].
Antiasthmatic activity
Following ethnomedicinal approach, screening of M. esculenta bark was done for the treatment of asthma. The ethanol extract of bark at dose of 75 mg/kg given by oral route was found to exhibit remarkable antiasthmatic activity through several mechanisms which include: antianaphylactic activity in guinea pigs induced by egg albumin, spasmolytic activity by relaxation of guinea pig smooth muscle (tracheal muscle) in histamine and acetylcholine (Ach)- induced contraction [76], bronchodilatory activity by protecting against Ach- and histamine aerosolinduced bronchospasm in guinea pigs [76,77], inhibition of total and differential leucocytes in bronchoalveolar lavage fluid as well as inhibition of histamine release from chopped lung tissues of egg albumin sensitized guinea pigs [77]. The antiasthmatic potential of ethanol extract of the bark (75 mg/kg and 150 mg/kg, p.o.) was further supported by exhibiting antiallergic activity due to marked inhibition of eosinophil accumulation (p<0.05) in allergic pleurisy test as well as significant inhibition in the rise in plasma exudation (p<0.05) in acetic acid-induced vascular permeability [78]. However, water extract of the bark even at lower doses (27 and 54 mg/kg, p.o) was found to possess more potent antiasthmatic activity than ethanol extract by showing significant protection against histamine aerosol-induced bronchospasm in guinea pigs and by relaxing histamine-induced guinea pig tracheal chain contraction [79]. Additionally, polar extracts (200 mg/kg) [80] as well as ethyl acetate and water extracts (100 and 200 mg/kg) [81] of M. nagi bark showed significant dose-dependent better mast cell protection in treated animals as compared to control group in compound 48/80 and egg albumin-induced allergy test [80,81]. Thus, all these results scientifically support the use of M. esculenta bark in traditional medicine for the treatment of allergic reactions including asthma and bronchitis.
Anticancer activity
Preclinical studies have shown that acetone and acidmethanol extracts of M. esculenta fruits showed potent anticancer proliferative activities resulted in 70-92 % reduction in the viability of C33A, SiHa, and HeLa cancer cells while exhibiting no cytotoxicity towards normal transformed cell lines [82]. The methanol extract of fruit showed moderate anticancer activity leading to 50, 48.29 and 46.19 % inhibition of Hep G2, Hela and MDA-MB-231 cancer cell lines at concentration 5 mg/ml in methylthiazolyltetrazolium (MTT) assay. It was observed that effect of extract on percent inhibition of cancer cells was found to be increased with increase in dose which may be attributed due to presence of bioactive compounds such as ferulic acid and gallic acid determined by LC/MS analysis [73].
Antidepressant activity
Screening of ethanol extract of M. nagi bark at doses 300 and 500 mg/kg for neuro-pharmacological activities showed that bark extract exhibited significant dose-dependent antidepressant activity with respect to control group in open-field test, cage-crossing test, head-dip test, rearing test, traction test and forced swimming test [83], which contradicts the previous report that ethanol extract of M. nagi bark exhibited significantly and dose-dependent CNS-depressant effect as evident by increased duration of immobility in animals of extract treated groups as compare to control evaluated by tail immersion test and forced swimming test [37].
Antidiabetic activity
A significant hypoglycaemic effect was observed in dose-dependent manner by methanol extracts of M. esculenta leaves in streptozotocin-induced diabetic rats as the results showed that oral administration of extract produced significant reduction (p<0.05) in the blood glucose, blood cholesterol and body weight as well as showed beneficial effect (p<0.05) on lipid profile of extract treated group as compared to positive vehicle treated group [84].
Antihelmintic activity
In a study, 50 % ethanol extract of M. esculenta bark was evaluated for antihelmintic activity against Indian earthworm Pheretima posthuma at different doses (50, 25 and 12.5 mg/ml) compared with piperazine citrate as standard. The result of such activity revealed that the crude extract at low dose (12.5 mg/ml) showed both paralysis and death in 20.11 and 41.25 min, respectively. The effect of extract was increased in a dose-dependent manner [85].
Antihypertensive activity
The effect of active phyto-constituents isolated from M. esculenta leaves for the management of hypertension was investigated by angiotensin I-converting enzyme inhibition. The results revealed that the compounds corchoionoside C and (6S,9R)-roseoside were found to be most potent ACE inhibitors with rates 29.97 and 25.63 % at the concentration of 100 μM, while myricanol, 5-O-β-D-glucopyranosyl myricanol and myricetin showed weak hypotensive activity with inhibition rate 0.07-1.41 % at same concentration [62].
Antiinflammatory activity
The methanol extract of M. esculenta leaves (200 mg/kg) has proven its potential for amelioration of acute inflammation as it showed a significant reduction (21.71 %) in inflammation in treated animals after 4th h of treatment, which was comparable to diclofenac (10 mg/kg; 32.75 %) treated group [75]. The essential oil isolated from M. esculenta bark exhibited significant topical antiinflammatory activity compared to standard drug in Swiss albino mice ear [72]. ME-EtAC of leaves was found to have significant (p<0.05) antiinflammatory effect against carrageenaninduced paw edema and the results were compared with diclofenac 10 mg/kg [32]. The ethyl acetate extract of M. nagi bark (200 mg/kg/p.o) showed significantly greater antiinflammatory activity than the aqueous extract with 27.51 and 26.82 % inhibition of edema in the carrageenan- and the histamine-induced rat paw edema models, respectively, which was almost as effective as the standard aspirin (100 mg/kg/p.o; 28 %) [86].
Antimicrobial activity
Volatile oil isolated from M. esculenta bark was reported to exhibit potent antibacterial activity against Gram-positive and Gram-negative bacteria [72]. The comparative in vitro antibacterial screening using disc diffusion assay on M. nagi bark and fruit extracts showed that methanol and chloroform extracts of the bark possessed greater antibacterial activity than fruit extracts when compared to the standard drug [87]. Gram-positive bacteria (S. aureus) was found to be most sensitive to methanol extract of bark while Gram-negative bacteria (E. coli) was found to be less sensitive [88]. Methanol extract of M. esculenta fruits showed antibacterial activity against S. epidermis and S. aureus comparable to that of the standard tetracycline with maximum zone of inhibition 18±0.5 and 16±0.5 mm, respectively. The antimicrobial activity of the extract could be attributed to the presence of dodecanol, phytol, furfurals and 4-H-pyran-4-one, which were reported to possess antimicrobial activity [73]. However, the ethanol extract of M. nagi fruit pulp (10 and 50 mg/ml), in disc diffusion assay inhibited food poisoning bacteria such as E. coli (MTCC 729), Streptococcus pyogenes (MTCC 1925) and E. coli (MTCC 443) in a dose-dependent manner [53]. It was also reported that methanol extract [74], ethanol and aqueous extract [53] of fruits showed significant antifungal activity.
Antioxidant activity
Three different in vitro radical scavenging assay viz. 1,1-diphenyl-2-picrylhydrazyl (DPPH) assay, 2,2-azinobis(3-ethylbenzothiazoline-6-sulphonic acid (ABTS) assay and ferric reducing antioxidant power (FRAP) assay were used to access the antioxidant potential of methanol extract of M. esculenta fruit [73] and fruit pulp [58]. The results had showed significant antioxidant activity by both extracts in all tested assays which is probably due to the presence of phenols, flavonoids and flavonols [58,73]. Seal compared free radical scavenging activity of acetone and 20 % v/v methanol extracts of four wild edible fruits including M. esculenta by DPPH assay. The acetone extract of M. esculenta fruit, which contained highest amount of phenolic compounds showed highest DPPH radical scavenging activity [89]. On the other side, the acid acetone extract of fruit had proved much higher DPPH radical scavenging activity and ferric reducing ability than acetone extract of fruit [82]. In another comparative study on methanol extracts of wild edible fruits of same species collected from three different regions of Uttrakhand (India) showed variation in DPPH antioxidant activity and Fe2+ chelating activity maximum up to 96.98±0.1847 and 72.17±0.2367 %, repectively due to variation in phenolic and flavonoids content affected by various biological factors of natural climates [74]. Additionally, fresh fruit juice of M. esculenta showed much higher DPPH scavenging activity (89.62 %) and nitric oxide scavenging activity (75.11 %) at the concentration of 2 mg/ml as compared to standard drug ascorbic acid [90]. The ME-EtAC fraction of leaves exhibited weaker activity than ascorbic acid used as standard [32] and methanol extract [75] in DPPH assay. Preliminary study on antioxidant and radical scavenging activity of the aqueous extract of M. esculenta bark showed marked inhibition in lipid peroxidation, complex metal ions (Fe2+) and significant DPPH scavenging activity [91]. The polar extract of M. nagi bark also showed remarkable DPPH free radical scavenging activity than non-polar and methanol extracts which might be owing to the presence of higher phenolic and flavonoid compounds like myricetin, myricanol, myricanone [92]. These results strongly supported the use of M. esculenta plant as a source of natural antioxidants.
Antipyretic effect
Screening of methanol extract of M. esculenta fruits for antipyretic activity in the Brewer’s yeast-induced pyrexia model in the mouse demonstrated that oral administration of the extract produced significant antipyretic activity comparable to that produced by paracetamol [74].
Antiulcer effect
Oral administration of ethanol extract of M. esculenta bark at the dose of 100 and 200 mg/kg showed protection against pylorus ligated ulcer in rats by significant reduction in gastric secretions, acidity, lipid peroxidation and myeloperoxidase enzyme as compared to control. The antiulcerogenic potential of the bark could be related to antioxidant mechanism as evident from significant increase in the catalase activity, nitrite and glutathione level [93]. Thus, the study provided scientific evidence for the traditional use of M. esculenta in ulcer treatment.
Anxiolytic activity
The ethanol extract of M. nagi bark tested at three different doses (100, 200 and 400 mg/kg, p.o.) has been reported to exhibit a dose-dependent significant anxiolytic activity as compare to standard drug diazepam evaluated by using two in vitro animal models such as elevated plus-maze and light/dark exploration test [37].
Chemopreventive activity
Pretreatment of mice with ethanol extract of M. nagi (2.0 and 4.0 mg/kg) showed significantly ameliorating the cumene hydroperoxide-mediated inhibition of cutaneous glutathione and the activities of antioxidant enzymes such as catalase, glucose-6-phosphate dehydrogenase, glutathione peroxidase, glutathione reductase and phase II metabolizing enzymes in a dose-dependent manner due to the presence of flavonoids, terpenoids and alkaloids in crude extract. Thus, suggested that it might be used as an effective chemopreventive agent [94].
Hepatoprotective activity
A polyherbal Ayurvedic formulation, Herbitars (50 and 100 mg/kg) containing 5 mg/g of M. esculenta as one of the ingredient exhibited hepatoprotective effect against carbon tetrachloride (CCl4)-induced hepatotoxicity in Wistar rats by significantly decreasing the levels of thiobarbituric acid reactive substance and hydroperoxides along with significantly increasing the antioxidant enzyme activities of superoxide dismutase, catalase, glutathione peroxidase and the levels of reduced glutathione in tissues (liver and kidney) of CCl4-induced rats [95].
Wound healing activity
Ethnotherapeutic claim of M. esculenta bark for wound healing was scientifically proven by using wound excision and incision model. Application of ointment prepared from the aqueous extract of bark facilitated wound healing process in the treated animals as evident by significant increase in the tensile strength, hydroxyproline content, faster wound contraction and decrease in the epithelization period found comparable with standard drug 0.2 % w/w nitrofurazon [45]. Thus, the ethanol extract of bark might be used as wound healing agent.
Toxicological Studies
Generally, M. esculenta is considered safe and only a few toxicity studies have been carried out. Rawat et al. studied toxic effects of the methanol extract of M. esculenta leaf. They did not find any sign of toxicity up to the dose of 300 mg/kg on oral administration of extract for two weeks. But, at 2000 mg/kg dose of the methanol extract toxic effects were observed in Wistar rats [84]. In addition, acute toxicity studies performed with ethyl-acetate and water extracts of M. nagi bark at three different i.v. doses, 100, 200 and 1000 mg/kg, showed that the LD50 of the ethyl-acetate and water extracts in mice was 1000 mg/kg [81].
M. esculenta has been used for its therapeutic and nutritional potential since from the ancient Ayurveda and Unani system of medicine. It is evident from this review that M. esculenta contains a number of phytoconstituents, which are responsible for medicinal value of this plant. Now, the road ahead is to establish the active therapeutic compound with specific mode of actions, which might be responsible for the medicinal properties of the plant. M. esculenta has been reported to have numerous pharmacological effects to treat various disorders including asthma, diabetes, cancer, ulcer, anxiety but being a rich source of vitamin C and polyphenolic compounds there is the need to explore the potential of this plant for immunomodulatory, cardioprotective, nephroprotective, and neuroprotective activity. As the population of this medicinal and economical plant species is in the verse to extinction due to overexploitation of forests and wastelands, negligence of sustainable resources, poor cultivation as well as poor regeneration of the species in natural habitat. Hence, it is high time to take preliminary necessary action to increase its population size, productivity, conservation and utilization.
Conflict of interest
The authors report no declarations of interest.
Financial support and sponsorship
Nil.
References
- Yanthan M, Misra AK. Molecular approach to the classification of medicinally important actinorhizal genus Myrica. Indian J Biotechnol 2013;12:133-6.
- Lutzow-Felling C, Gardner DE, Markin GP, Smith CW. Myricafaya: review of the biology, ecology, distribution, and control, including an annotated bibliography. Honolulu, Hawaii: Cooperative National Park Resources Studies Unit, University of Hawaii; 1995.
- http://edis.ifas.ufl.edu/pdffiles/ST/ST41100.pdf.
- Gathercoal EN, Wirth EH. Pharmacognosy. 2nd ed. Philadelphia: Lea &Febiger; 1943.
- Singh J, Lan VK, Trivedi VP. Pharmacognostic evaluation of Katphala (The bark of MyricaesculentaBuch–Ham). AncSci Life 1986;6:85-7.
- Kumar A, Rana AC. Pharmacognostic and pharmacological profile of traditional medicinal plant: Myricanagi. Int Res J Pharm 2013;3:32-7.
- Sun C, Huang H, Xu C, Li X, Chen K. Biological activities of extracts from Chinese bayberry (MyricarubraSieb. et Zucc.): a review. Plant Foods Hum Nutr 2013;68:97-106.
- Silva BJC, Seca AML, Barreto CMD, Pinto DCGA. Recent breakthroughs in the antioxidant and anti-Inflammatory effects of Morella and Myrica species. Int J MolSci 2015;16:17160-80.
- Kuang KZ, Lu AM. Myricaceae. Beijing: Flora ReipublicaePopularisSinicae; 1979. p. 1-6.
- Huguet V, Gouy M, Normand P, Zimpfer JM, Fernandez MP. Molecular phylogeny of Myricaceae: A reexamination of host-symbiont specificity. MolPhylogenetEvol 2005;34:557-68.
- Haridasan K, Rao RR. Forest flora of Meghalaya. Caprifoliaceae to Salicaceae. Dehradun (India): Bishen Singh Mahendra Pal Singh; 1987.
- http://www.niscair.res.in/activitiesandservices/products/wealth-of-indiaFolder2010.pdf.
- Nadkarni KM. Indian MateriaMedica. 3rd ed. Mumbai: Popular Book Depot; 2002. p. 871.
- Patel RK, De LC. Soh-phie (Myrica species)-An unexploited fruit of the future for Meghalaya. ENVIS Bulletin Himalayan Ecology 2006;14:34-7.
- Maikhuri RK, Semwal RL, Singh A, Nautiyal MC. Wild fruits as a contribution to sustainable rural development: A case study from the Garhwal Himalaya. Int J Sustain Dev World Ecol 1994;1:56-68.
- Parmar C, Kaushal MK. Myricanagi. In: Parmar C, Kaushal MK, editors. Wild Fruits of the Sub-Himalayan Region. New Delhi: Kalyani Publishers; 1982. p. 49-53.
- Dhani A. Major wild edible fruits used by locals of Garhwal Himalaya. Int J AdvLifSci 2013;6:145-9.
- Ksanbok M, Lynser MB, Pala KHM. Marketing of indigenous fruits: a source of income among Khasi women of Meghalaya, North East India. J AgriSci 2014;5:1-9.
- Panthari P, Kharkwal H, Kharkwal H, Joshi DD. Myricanagi: A review on active constituents, Biological and therapeutic effects. Int J Pharm PharmSci 2012;4:38-42.
- Dollo M, Samal PK, Sundriyal RC, Kumar K. Environmentally sustainable traditional natural resource management and conservation in Ziro valley, Arunachal Himalaya, India. J Am Sci 2009;5:41-52.
- Kumar JK, Sinha AK. Resurgence of natural colourants: a holistic view. Nat Prod Res 2004;18:59-84.
- Jeeva S, Lyndem FB, Sawian JT, Laloo RC, Mishra BP. MyricaesculentaBuch.-Ham. ex D. Don.- a potential ethnomedicinal species in a subtropical forest of Meghalaya, northeast India. Asian Pac J Trop Biomed 2011;1:S174-7.
- Kala CP. Prioritization of cultivated and wild edibles by local people in the Uttaranchal hills of Indian Himalaya. Indian J Tradit Know 2007;6:239-43.
- Srivastava B, Sharma VC, Pant P, Pandey NK, Jadhav AD. Evaluation for substitution of stem bark with small branches of Myricaesculentafor medicinal use-A comparative phytochemical study. J Ayurveda Integr Med 2016;7:218-23.
- Gusain YS, Khanduri VP. Myricaesculentawild edible fruit of Indian Himalaya: need a sustainable approach for indigenous utilization. Eco Env Cons 2016;22:S267-70.
- Anonymous. Ayurvedic Pharmacopoeia of India. Part 1, Vol III. New Delhi: Ministry of Health and Family Welfare, Department of Indian System of Medicine and Homeopathy; 2007. p. 90-6.
- Rymbai H, Roy AR, Deshmukh NA, Jha AK, Shimray W, War GF et al. Analysis study on potential underutilized edible fruit genetic resources of the foothills track of Eastern Himalayas, India. Genetic Resour Crop Evol 2016;63:125-39.
- Kirtikar KR, Basu BD. Indian Medicinal Plants. 2nd ed. Vol. III. New Delhi: International book distributors; 1999. p. 1699.
- Osmaston AE. A Forest Flora for Kumaun. Dehradun: Bishen Singh Mahindra Pal Singh; 1987.
- Mahat RB, Chaudhary RP. Ethnomedical study and antibacterial activities of selected plants of Palpa district Nepal. Sci World J 2005;3:26-31.
- Sahu S, Sahu CR, Yadav A, Rathod P, Chaturvedi S, Tripathi R. Review on MyricaesculentaA popular plant of Himalayan region. J Chem Pharm Sci 2013;6:93-7.
- Pundir S, Tomar S, Upadhyay N, Sharma V. Antioxidant, anti-inflammatory and analgesic activity of bioactive fraction of leaves of MyricaesculentaBuch.-Ham along with its pharmacognostic and chromatographic evaluation. Int J Biol Pharm Allied Sci 2015;4:6509-24.
- Gangwar KK, Deepali, Gangwar RS. Ethnomedicinal plant diversity in KumaunHimalaya of Uttarakhand, India. Nat Sci 2010;8:66-78.
- Kumari P, Joshi GC, Tewari LM. Diversity and status of ethnomedicinal trees of Almora district in Uttarakhand, India. Int J BiodiversConserv 2011;3:298-326.
- Pandey NC, Joshi GC, Tiwari LM. Ethnobotanical plant diversity of Betalghat region, Kumaun Himalaya. Biolife 2016;4:629-49.
- Arya D, Khan AH, Adhikari M. Plant species used by locals as ethno-medicine in Kumaun region of Western Himalaya (India). Int J Pharm Sci Res 2014;5:3128-32.
- Khan MY, Sagrawat H, Upmanyu N, Siddique S. Anxiolytic properties of Myricanagibark extract. Pharm Biol 2008;46:757-61.
- Maikhuri RK, Gangwar AK. Ethnobiological notes on the Khasi and Garo tribes of Meghalaya, Northeast India. Econ Bot 1993;47:345-57.
- Kayang H. Tribal Knowledge on wild edible plants of Meghalaya, Northeast India. Indian J Tradit Know 2007;6:177-81.
- Laloo D, Hemalatha S. Ethnomedicinal plants used for diarrhea by tribals of Meghalaya, Northeast India. Pharmacogn Rev 2011;5:147-54.
- Laloo RC, Kharlukhi L, Jeeva S, Mishra BP. Status of medicinal plants in the disturbed and the undisturbed sacred forests of Meghalaya, Northeast India: Population structure and regeneration efficacy of some important species. CurrSci 2006;90:225-32.
- Bhatt UVP, Negi GCS. Ethnomedicinal plant resources of Jaunsari tribe of Garhwal Himalaya. Indian J Tradit Knowledge 2006;5;331-5.
- Chatterjee A, Pakrashi SC. The Treatise on Indian Medicinal Plants. Vol I. New Delhi: Publications and Information Directorate; 1994. p. 32-3.
- Chauhan, NS. Medicinal and Aromatic Plants of Himachal Pradesh. New Delhi: Indus Publishing Company; 1999. p. 226.
- Nainwal P, Kalra K. Study on the wound activity potential on the aqueous extract of the bark of MyricaesculentaBuch. & Ham. Int J Pharm Clin Res 2009;1:85-7.
- Rastogi RP, Mehrotra BN. Compendium of Indian Medicinal Plants. 4th ed. New Delhi: Council of Scientific and Industrial Research Publication; 1995. p. 490-2.
- Chopra RN, Nayar SL, Chopra IC. Glossary of Indian Medicinal Plants (Including the Supplement). 3rd ed. New Delhi: Council of Scientific and Industrial Research Publication; 1986. p. 203.
- Pala NA, Negi AK, Todaria NP. Traditional uses of medicinal plants of PauriGarhwal, Uttrakhand. Nat Sci 2010;8:57-61.
- Mishra RN. Rasayan–The Ayurvedic Perspective. Res J Pharm BiolChemSci 2011;2:269-82.
- Suresh P, Dhannapunei VK. Rasendra Sara Sangrah of Sri Gopal Krishna Bhatt. 1st ed. Varanasi: Chaukhambha Sanskrit Sansthan; 2007.
- Pandey MM, Rastogi S, Khatoon S, Mehrotra S, Rawat AKS. Evaluation of Ayurvedic compound formulation 5-Katphaladi Churna. Indian J Tradit Know 2013;12:295-99.
- Seal T. Nutritional composition of wild edible fruits in Meghalaya state of India and their ethnobotanical Importance. Res J Bot 2011;6:58-67.
- Chandra S, Saklani S, Mishra AP, Badoni PP. Nutritional evaluation, antimicrobial activity and phytochemical screening of wild edible fruit of Myricanagipulp. Int J Pharm PharmSci 2012;4:407-11.
- Kharwal H, Panthari P, Kharkwal H, Joshi DD. Investigations on Myricanagileaves: Phytochemical screening and physicochemical evaluation. World J Pharm PharmSci 2013;2:2867-73.
- Barnes J. Herbal medicines. 2nd ed. London: Pharmaceutical press; 2002. p. 71.
- Sun D, Zhao Z, Wong H, Foo LY. Tannins and other phenolics from Myricaesculentabark. Phytochemistry 1988;27:579-83.
- Singh N, Khatoon S, Srivastava N, Rawat A, Mehrotra S. Qualitative and quantitative standardization of MyricaesculentaBuch.-Ham. Stem bark by use of HPTLC. J Planar Chromat 2009;22:287-91.
- Rawat S, Jugran A, Giri L, Bhatt ID, Rawal RS. Assessment of antioxidant properties in fruits of Myricaesculenta: A popular wild edible species in Indian Himalayan Region. Evid Based Complement Alternat Med 2011:2011;512787.
- Wei Y, Chang-ming T, Xian L, Ya Z, Li W, Liang L. Study on the chemical constituents of Myricaesculenta. J Yunnan University (Nat Sci) 2011;33:453-7.
- Patel K, Patel V, Patel K, Gandhi T. Validated HPTLC method for quantification of myricetin in the stem bark of MyricaesculentaBuch. Ham. Ex D. Don, myricaceae. J Planar Chromat 2010;23:326-31.
- Dawang S, Zuchun Z, Foo LY, Wong H. Flavonols from Myricaesculentaa bark. Chem Indus Forest Prod 1991;04:251-57.
- Nhiem NX, Kiem PV, Minh CV, Tai BH, Cuong NX, Thu VK, et al. A new monoterpenoid glycoside from Myricaesculenta and the inhibition of Angiotensin I-Converting Enzyme. Chem Pharm Bull 2010;58:1408-10.
- Bamola A, Semwal DK, Semwal S, Rawat U. Flavonoid glycosides from Myricaesculentaleaves. J Indian ChemSoc 2009;86:535-6.
- Agarwal KP, Roy AC, Dhar ML. Triterpenes from the Bark of MyricaesculentaBuch.-Ham. Indian J Chem 1963;1:28-30.
- Krishnamoorthy V, Krishnaswamy NR, Seshadri TR. Myriconol from the stem bark of Myricanagi. CurrSci 1963;32:16-7.
- Begley MJ, Campbell RVM, Crombie L, Tuck B, Whiting DA. Constitution and absolute configuration of meta, meta-bridged, stained biphenyls from Myricanagi; X-ray analysis of 16-bromomyricanol. J ChemSoc C 1971;3634-42.
- Malterud KE, Anthonsen T. 13-oxomyricanol, a new [7.0]-metacyclophane from Myricanagi. Phytochemistry 1980;19:705-7.
- Agnihotri S, Wakode S, Ali M. Triterpenoids from the stem bark of MyricaesculentaBuch Ham. World J Pharm PharmSci 2016;5:1319-27.
- Krishnamoorthy V, Seshadri TR. A new Proanthocyanidin from the stem bark of Myricanagithumb. Tetrahedron 2001;22:2367-1.
- Mei WD, Hong CJ, Mei WY, Man X, Song WZ. Study on ultrasound-assisted extraction of proanthocyanidins from MyricaesculentaBark. Conference paper. Chem Indus Forest Prod 2009;29:105-9.
- Hui-fen M.A, Zheng-liang Y, Sang-zi ZE, Yong-jie L, De-lu N, Zhen YU. GC/MS analysis of volatile components from leaf of MyricaesculentaBuch.-Ham. Guangdong AgricSci 2011;16:18.
- Agnihotri S, Wakode S, Ali M. Essential oil of MyricaesculentaBuch. Ham: composition, antimicrobial and topical antiinflammatory activities. Nat Prod Res 2012;26:2266-9.
- Mann S, Satpathy G, Gupta RK. In-vitroevaluation of bioprotective properties of underutilized MyricaesculentaBuch.-Ham. ex D. Don fruit of Meghalaya. Indian J Nat Prod Resour 2015;6:183-8.
- Pant G, Prakash O, Chandra M, Sethi S, Punetha H, Dixit S, et al. Biochemical analysis, pharmacological activity, antifungal activity and mineral analysis in methanol extracts of Myricaesculentaand Syzygiumcumini: the Indian traditional fruits growing in Uttarakhand Himalaya. Indian J Pharm Biol Res 2014;2:26-34.
- Middha SK, Kumar GA, Talambedu U, Babu D, Misra AK, Prakash L. Evaluation of antioxidative, analgesic and anti-inflammatory activities of methanolic extract of Myricanagileaves - an animal model approach. Symbiosis 2016;1-3:179-84.
- Patel KG, Bhalodia PN, Patel AD, Patel KV, Gandhi TR. Evaluation of bronchodilator and antianphylactic activity of Myricasapida. Iran Biomed J 2008;12:191-6.
- Patel KG, Patel KV, Shah JH, Monpara KB, Gandhi TR. Evaluation of the effect of Myricasapidaon bronchoconstriction and bronchial hyperreactivity. Pharmazie 2008;63:312-6.
- Patel KG, Rao NJ, Gajera VG, Bhatt PA, Patel KV, Gandhi TR. Antiallergic activity of stem bark of MyricaesculentaBuch.-Ham.(Myricaceae). J Young Pharm 2010;2:74-8.
- Patel T, Ladani K, Shah S. Antiasthmatic activity of aqueous extract of Myricanagibark. Int J Phytopharm Res 2013;4:40-5.
- Rana RK, Patel RK. Pharmacological Evaluation of Antiasthmatic Activity of MyricanagiBark Extracts. AntiinflammAntiallergy Agents Med Chem 2016;15:145-52.
- Patel T, Rajshekar C, Parmar R. Mast cell stabilizing activity of Myricanagibark. J Pharmacognosy Phytother 2011;3:114-7.
- Saini R, Garg V, Dangwal K. Effect of extraction solvents on polyphenolic composition and antioxidant, antiproliferative activities of Himalayan bayberry (Myricaesculenta). Food SciBiotechnol 2013;22:887-94.
- Syed S, Ahmad M, Fatima N, Mahjabeen, Jahan N. Neuropharmacological studies of Myricanagibark. Int J Biol Biotech 2013;10:553-8.
- Rawat S, Kumar N, Kothiyal P. Evaluate the antidiabetic activity of Myricaesculentaleaves in streptozotocin induced diabetes in rat. Int J Univ Pharm Bio Sci 2013;2:510-25.
- Jain VK, Jain B. Anthihelmintic Activity of ethanolic extract of bark of Myricaesculenta. Int J Pharm Sci Res 2010;1:129-31.
- Patel T, Dudhpejiya A, Sheath N. Antiinflammatory activity of MyricanagiLinn. Bark. AncSci Life 2011;30:100-3.
- Suryawanshi JS, Karande KM, Udugade BV. Antibacterial activity of bark and fruits of M. nagi. Indian J Nat Prod 2009;25:21-3.
- Shan B, Cai YZ, Brooks JD, Corke H. The in vitroantibacterial activity of dietary spice and medicinal herb extracts. Int J Food Microbiol 2007;117:112-9.
- Seal T. Antioxidant Activity of Some Wild Edible Fruits of Meghalaya State in India. AdvBiol Res 2011;5:155-60.
- Goyal AK, Mishra T, Bhattacharya M, Kar P, Sen A. Evaluation of phytochemical constituents and antioxidant activity of selected actinorhizal fruits growing in the forests of Northeast India. J Biosci 2013;38:797-803.
- Chen J, Wang Y, Wu D, Wu Z. Preliminary study on antioxidative and radical scavenging activities of extracts from MyricaesculentaBuch.-Ham. Bark. Chem Industry Forest Prod 2007;S1:1-7.
- Rana RK, Patel RK. Antioxidant Activity of Bark of Myricanagi. Int J Pharm Sci Rev Res 2014;28:99-101.
- Swathi D, Prasad KVSRG. Antioxidant and antiulcer potential of ethanolic extract of bark of Myricaesculentain pyloric ligation ulcer model. Int J Pharm PharmSci 2015;7:195-8.
- Alam A, Iabal M, Saleem M, Ahmed SU, Sultana S. Myricanagiattenuates cumenehydroperoxide-induced cutaneous oxidative stress and toxicity in swiss albino mice. Pharmacol Toxicol 2000;86:209-14.
- Samundeeswari C, Rajadurai M, Periasami R, Kanchana G. Hepatoprotective effect of Herbitars, A polyherbal against CCl4 induced hepatotoxicity in rats. J Pharm Res 2011;4:676-9.