- *Corresponding Author:
- C. Y. Jagtap
Institute for Post Graduate Teaching and Research in Ayurveda, Gujarat Ayurved University, Jamnagar-361 008, India
E‑mail: drshikhar84@gmail.com
| Date of Submission | 05 July 2012 |
| Date of Revision | 11 March 2013 |
| Date of Acceptance | 08 April 2013 |
| Indian J Pharm Sci,2013,75(3):346-352 |
Abstract
The use of metals in traditional medicines is very often seen as matter of concern these days, especially the Bhasma preparations which are always under stringent observations for containing highly reactive inorganic elements such as lead, mercury, arsenic and others. One of the Bhasma extensively used in routine Ayurvedic practice is Tamra (copper) bhasma. If it is not prepared properly or Shodhana procedure is not done properly, it acts as a poison. To indicate its toxic potential, Ashtamahadoshas (eight major ill effects) have been quoted in classics and due emphasis have been given to its Shodhana procedure. In the present study, Tamra bhasma prepared from Shodhita and Ashodhita Tamra was subjected to oral toxicity study to ascertain the role of Shodhana process on safety profile of Tamra bhasma on subchronic administration to albino rats. Both the samples were administered to rats for 45 consecutive days at the doses of 5.5, 27.5, and 55 mg/kg. Animals were sacrificed on 46 th day and parameters like hematological, serum biochemical, and histopathology of various organs were studied. Results showed that Tamra bhasma prepared from Ashodhita Tamra has pathological implications on different hematological, serum biochemical and cytoarchitecture of different organs even at therapeutic dose level (5.5 mg/kg). Whereas, Tamra bhasma prepared from Shodhita Tamra is safe even at five-fold to therapeutic equivalent doses (27.5 mg/kg). These observations emphasize the role of Shodhana and importance of dose in expression of toxicity of the medicinal preparations.
Keywords
Amrutikarana, copper, herbomineral formulation, LD50, Marana, Shodhana
Introduction
In traditional medicines, use of metals is very often seen as matter of concern these days. Especially the Bhasma preparations; which are always under stringent observations for containing highly toxic inorganic elements such as lead, mercury, arsenic and others [1,2]. In this present era of scientific validation and good manufacturing practices, the metals and minerals that are transformed into drugs must have the excellent quality, safety and therapeutic efficacy. These metallic preparations have unique procedures of their preparation, involving Shodhana (purification and/or detoxification), Marana (incineration and/ or calcination), and are sustaining themselves since centuries in clinical practices. These methods were developed by practitioners of this science to detoxify the raw material by chemical transformations and modify the properties of therapeutic materials to enhance their potential [3,4]. Hence their use cannot be denied just for their heavy metal content. But unfortunately some metals and minerals have the potential to produce adverse effects [5]. Therefore, during their transmutation to drugs, it is essential to evaluate the margin of safety between the dose level that produces the therapeutic effects and that produces the adverse effects. Animal experimentation is the only way through which this evaluation can be made.
Tamra (copper) Bhasma is used in various Ayurvedic preparations. If it is not prepared properly or Shodhana procedure is not done; it acts as a poison [6]. To indicate its toxic potential, Ashtamahadoshas (eight major ill effects) have been quoted in classics and due emphasis has been given to its Shodhana procedure [6]. Previously Nayak et al. found that Tamra Bhasma has the tendency of producing toxicity [7] on oral administration. Pattanaik et al. observed that Tamra Bhasma given for longer period (90 days), in 5 mg/kg induced lipid peroxidation without any effect on the survival [8]. Vahalia et al. suggested that the safety of Tamra Bhasma may be attributed to Bhasmikarana (Incineration) procedure which converts the metal into its specially designed chemical compound which eliminates the toxicity of metal and has the necessary medicinal benefits [9]. Although several such studies were reported, till date no research work on role of Shodhana (Purification) on Tamra Bhasma has been reported to provide scientific basis to this. Hence in the present study, Tamra Bhasma prepared from Shodhita and Ashodhita Tamra was subjected to oral toxicity studies to ascertain the role of Shodhana on safety profile of Tamra Bhasma on subchronic administration to albino rats.
Materials and Methods
Copper wire containing 99.89% copper, which is used for the purpose of electrical earthing, was procured from local electrician. In one sample raw copper wire was directly subjected to Marana and Amritikarana procedures and coded as ATB (Tamra Bhasma prepared from Ashodhita Tamra). In another sample raw Tamra was subjected to Samanya (general) Shodhana, Vishesha (special) Shodhana, Marana [10] and Amritikarana [11] procedures as per the classical references and coded as STB (Tamra Bhasma prepared from Shodhita Tamra). These two coded samples were subjected to acute and sub‑chronic toxicity studies.
Wistar strain albino rats of either sex, weighing 200±20 g were used as per the guidelines of the Institutional Animal Ethics Committee (IAEC). The animals were obtained from the animal house attached to the Pharmacology laboratory, Institute for Post Graduate Teaching and Research in Ayurveda, Gujarat Ayurved University, Jamnagar, and were exposed to natural day and night cycles, with ideal laboratory conditions in terms of ambient temperature and humidity. Temperature during the time of carrying out the experiment was between 24±2º and humidity around 50‑60%. Animals were fed ad libitum with Amrut brand rat pellet feed supplied by Pranav Agro Industries and tap water. The experiment was carried out after obtaining the permission from institutional animal ethics committee (Approval number: IAEC 07/2010/05/MD) and care of animals was taken as per the CPCSEA guidelines.
Dose fixation and schedule
Clinical dose of Tamra Bhasma is 30 mg twice a day (60 mg per day) [12]. The suitable dose for rats was calculated by referring to table of Paget and Barnes [13] and was found to be 5.5 mg/kg (considered as therapeutic equivalent dose, TED). The test drugs were administered in the form of suspension in distilled water orally with the help of rubber catheter attached to a disposable syringe. For the preparation of stock solution, both the test drugs samples were taken in requisite quantity in small porcelain mortar and 0.5 ml of 5% gum acacia suspension was added, the formed mixture was further grounded for 5 min and the volume was made up with distilled water, to obtain a concentration of 5.5 mg/ml test drug.
Acute toxicity study
Young, healthy, nulliparous, and nor pregnant Wistar albino female rats were selected and acclimatized for seven days before the experiment. The test drug was administered to overnight fasted animals at graded doses by following ‘Up and Down method’ with 2000 mg/kg (OECD TG 425). The animals were observed continuously for 6 h and then intermittently up to 48 h after the dosing to record changes in behavior pattern and mortality if any.
Study protocol for subchronic toxicity
Animals were divided in seven groups, each comprising three male and three females. First group was kept as control where as the second to fourth groups were administered with test drug ATB at the dose of 5.5 mg/kg (TED), 27.5 mg/kg (TED×5), and 55 mg/kg (TED×10). Fifth to seventh groups were administered with test drug STB at the dose of 5.5 mg/kg (TED), 27.5 mg/kg (TED×5) and 55 mg/kg (TED×10). Initial body weight of all the groups was recorded and test drugs were administered for 45 consecutive days to overnight fasted rats. The general behavioral pattern was observed once every week by exposing each animal to open arena. On 46th day, animals were weighed again and anaesthetized with diethyl ether. Blood was drawn from supraorbital plexus by puncturing and collected in two different types of tubes, one containing anticoagulant fluid for hematological parameters and another plain tube for serum biochemical investigations. Then the rats were sacrificed by overdose of diethyl ether anesthesia and important organs were dissected out and transferred immediately to a glass bottle containing 10% formalin for histopathological studies.
Hematological analysis was performed using an automatic hematological analyzer (Swelab AC 970EO+, AC 920EO+, AC 910EO+ , Boule Medical AB, Stockholm, Sweden). Hemoglobin (Hb),hematocrit, total red blood cell (RBC), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), white blood cell (WBC), neutrophils, lymphocyte percentage, eosinophils percentage, monocyte percentage, packed cell volume (PCV), and platelet count were measured from the blood samples.
For estimation of biochemical parameters serum was separated from collected blood. Requisite quantity of serum was fed to the auto analyzer (Fully automated Biochemical Random Access Analyzer, BS‑200; Lilac Medicare Pvt. Ltd., Mumbai) which was automatically drawn in to the instrument for estimating different parameters. The biochemical parameters were recored a per the commercial kit instructions for blood glucose [14], blood urea [15], serum creatinine [16], serum total cholesterol [17], serum high density lipoproteins (HDL) cholesterol [18], serum triglyceride [19], serum total protein [20], serum alkaline phosphatase [21] activity, serum glutamate oxaloacetate transaminase (SGOT) [22], serum glutamate pyruvate transaminase activity (SGPT) [23], uric acid [24], direct bilirubin [25], and total bilirubin [26].
The histopathological slides of different organs like liver, kidney, heart, lung, trachea, jejunum, spleen, thymus, lymph node, testis, seminal vesicle, prostate, uterus, ovary, pituitary, brain, and adrenal gland were prepared by referring to standard procedure of Raghuramulu et al. [27]. The slides were viewed under trinocular research Carl‑Zeiss’s microscope at various magnifications to note down the changes in the microscopic features of the tissues studied.
Statistical analysis
The results were presented as Mean±SEM for six rats in each group. Statistical comparisons were performed by both unpaired Student’s t test and one way ANOVA with Dunnets’ multiple t test as post‑hoc test by using Sigmastat software (version 3.1) for all the treated groups with the level of significance set at P<0.05.
Results
No change in gross behavior was observed in any group during acute toxicity study. Both ATB and STB did not show any mortality up to 14 days when given in dose up to 2000 mg/kg.
Subchronic administration of ATB at TED dose level leads to significant increase in total RBC count, neutrophil percentage and PCV, while significantly decrease in lymphocyte percentage. However, it did not produce any significant impact at TED×5 and TED×10 dose levels. In contrast, administration of STB did not affect the majority of hematological parameters except for significant increase in platelet count at TED×5 and significant decrease in total RBC count at TED×10 dose level (Table 1).
| Parameters | NC | ATB TED (5.5 mg/kg) |
STB TED (5.5 mg/kg) |
ATB TED×5 (27.5 mg/kg) |
STB TED×5 (27.5 mg/kg) |
ATB TED×10 (55 mg/kg) |
STB TED×10 (55 mg/kg) |
|---|---|---|---|---|---|---|---|
| Hb (g %) | 14.8 ± 0.5 | 16.9 ± 0.8 | 15.5 ± 0.3 | 15.6 ± 0.5 | 14.8 ± 0.6 | 16.04 ± 0.6 | 14.2 ± 0.3 |
| Hematocrit (%) | 48.7 ± 1.2 | 54.7 ± 2.9 | 47.8 ± 2.0 | 50.3 ± 2.1 | 45.9 ± 2.5 | 50.8 ± 2.5 | 43.9 ± 2.2 |
| RBC(×106 cells/mm3) | 8.6 ± 0.1 | 9.7 ± 0.5a | 8.5 ± 0.3 | 8.9 ± 0.4 | 8.1 ± 0.4 | 8.8 ± 0.4 | 7.8* ± 0.2 |
| MCH (pg) | 17.3 ± 0.6 | 17.3 ± 0.2 | 18.2 ± 0.5 | 17.5 ± 0.4 | 18.4 ± 0.2 | 18.2 ± 0.2 | 18.2 ± 0.4 |
| MCHC (g/dl) | 30.5 ± 0.9 | 30.9 ± 0.3 | 32.4 ± 0.8 | 31.2 ± 0.3 | 32.4 ± 0.8 | 31.6 ± 0.4 | 32.2 ± 0.5 |
| MCV (fl) | 56.9 ± 1.1 | 56.1 ± 0.6 | 55.4 ± 0.7 | 56.2 ± 0.7 | 56.8 ± 0.4 | 57.4 ± 0.3 | 56.2 ± 0.6 |
| WBC (×103/mm3) | 7966.0 ± 656.59 | 8390.3 ± 847.00 | 8933.33 ± 868.58 | 8696.00 ± 996.2 | 8166.66 ± 841.29 | 7040.0 ± 518.3 | 8240.00 ± 700.40 |
| Neutrophils (%) | 27.12 ± 0.44 | 36.66 ± 3.09a | 25.40 ± 2.87 | 26.00 ± 2.62 | 27.66 ± 2.81 | 29.40 ± 6.76 | 26.16 ± 2.08 |
| Lymphocytes (%) | 68.83 ± 0.4 | 58.0 ± 3.3a | 68.40 ± 3.76 | 69.8 ± 2.7 | 67.8 ± 2.9 | 66.4 ± 7.6 | 69.2 ± 2.4 |
| Eosinophils (%) | 2.25 ± 0.16 | 3.0 ± 0.0 | 2.20 ± 0.2 | 2.20 ± 0.2 | 2.50 ± 0.50 | 2.40 ± 0.51 | 2.50 ± 0.22 |
| Monocytes (%) | 2.00 ± 0.18 | 2.33 ± 0.21 | 2.0 ± 0.31 | 2.0 ± 0.0 | 2.0 ± 0.0 | 1.8 ± 0.37 | 2.16 ± 0.16 |
| PCV | 47.38 ± 1.23 | 54.73 ± 2.89a | 47.83 ± 2.03 | 50.28 ± 2.10 | 45.93 ± 2.53 | 50.82 ± 2.47 | 43.95 ± 1.37 |
| Platelet count(103/µl) | 1047.7 ± 49.3 | 1202.5 ± 72.9 | 1215.8 ± 70.7 | 949.2 ± 125.3 | 1317.6 ± 58.4ab | 1208.0 ± 140.2 | 1005.7 ± 47.9 |
| Data presented as=Mean±SEM, SEM is standard error of means, aP<0.05 (Unpaired t test), bP<0.05 (Dunnett’s multiple t test), PCV=Packed cell volume, WBC=White blood cell, MCV=Mean corpuscular volume, MCHC=Mean corpuscular hemoglobin concentration, MCH=Mean corpuscular hemoglobin, RBC=Red blood cell, TED=therapeutic equivalent dose. | |||||||
Table 1: Effect on hematological parameters.
Among the 13 serum biochemical parameters, ATB at all the three levels significantly decreased HDL cholesterol. Other changes observed were decrease in serum triglyceride level and serum alkaline phosphatase activity in TED dose levels of ATB, decrease in serum alkaline phosphatase activity in TED×5 dose level of ATB, decrease in total protein in TED×10 dose level of ATB in comparison to control group. Changes that occurred in STB‑administered groups were; significant decrease in serum alkaline phosphatase activity in TED and TED×5 doses, significant decrease in total protein and HDL cholesterol at TED×10 dose (Table 2).
| Parameters | NC | ATB TED (5.5 mg/kg) |
STB TED (5.5 mg/kg) |
ATB TED×5 (27.5 mg/kg) |
STB TED×5 (27.5 mg/kg) |
ATB TED×10 (55 mg/kg) |
STB TED×10 (55 mg/kg) |
|---|---|---|---|---|---|---|---|
| Blood glucose (mg/dl) | 117.5 ± 8.9 | 108.7 ± 2.1 | 108.2 ± 9.7 | 116.8 ± 5.5 | 116.0 ± 12.4 | 110.9 ± 4.2 | 98.5 ± 15.5 |
| Total cholesterol (mg/dl) | 77.5 ± 9.9 | 63.3 ± 6.02 | 91.2 ± 8.2 | 51.5 ± 4.4a | 78.5 ± 16.5 | 56.0 ± 3.7 | 53.8 ± 5.6 |
| Triglyceride (mg/dl) | 97.5 ± 11.9 | 61.7 ± 6.9a | 121.2 ± 22.6 | 93.0 ± 11.07 | 87.0 ± 14.6 | 94.2 ± 10.4 | 124.3 ± 18.9 |
| HDL cholesterol (mg/dl) | 39.2 ± 5.4 | 26.3 ± 2.2ab | 37.3 ± 3.7 | 24.8 ± 2.2ab | 32.7 ± 3.7 | 25.0 ± 1.2a | 24.0 ± 3.3a |
| Blood urea (mg/dl) | 100.3 ± 11.4 | 112.0 ± 10.5 | 96.0 ± 5.7 | 114.8 ± 11.8 | 97.3 ± 6.9 | 115.8 ± 12.6 | 83.0 ± 3.4 |
| Serum creatinine (mg/dl) | 0.6 ± 0.2 | 0.6 ± 0.07 | 0.6 ± 0.05 | 0.6 ± 0.03 | 0.6 ± 0.02 | 0.6 ± 0.06 | 0.6 ± 0.03 |
| S.GPT (IU) | 77.3 ± 6.9 | 89.0 ± 9.8 | 62.7 ± 3.4 | 93.5 ± 4.08 | 70.8 ± 8.3 | 83.7 ± 6.2 | 86.2 ± 15.3 |
| S.GOT (IU) | 332.0 ± 42.2 | 279.2 ± 9.9 | 246.5 ± 9.9 | 321.7 ± 25.6 | 302.0 ± 19.9 | 335.3 ± 54.9 | 309.0 ± 36.6 |
| Total protein (g/dl) | 7.6 ± 0.3 | 7.9 ± 0.26 | 7.7 ± 0.2 | 7.15 ± 0.1 | 7.5 ± 0.1 | 6.9 ± 0.1a | 6.7 ± 0.2ab |
| Alkaline phosphatase (IU/l) | 236.2 ± 20.7 | 176.7 ± 16.4a | 146.7 ± 11.4a | 300.3 ± 60.3 | 146.8 ± 30.3a | 250.0 ± 29.8 | 170.3 ± 34.72 |
| Total bilirubin (mg/dl) | 0.7 ± 0.2 | 0.9 ± 0.2 | 0.5 ± 0.04 | 0.6 ± 0.1 | 0.5 ± 0.04 | 0.9 ± 1.1 | 0.5 ± 0.04 |
| Direct bilirubin (mg/dl) | 0.2 ± 0.04 | 0.3 ± 0.6 | 0.15 ± 0.02 | 0.2 ± 0.03 | 0.15 ± 0.02 | 0.7 ± 0.5 | 0.2 ± 0.2 |
| Uric acid (mg/dl) | 2.08 ± 0.4 | 2.6 ± 0.5 | 1.6 ± 0.1 | 1.7 ± 0.3 | 1.6 ± 0.2 | 2.7 ± 0.4 | 1.9 ± 0.4 |
| Data presented as=Mean±SEM, SEM is standard error of means, aP<0.05 (Unpaired t test), bP<0.05 (Dunnett’s multiple t test), TED=therapeutic equivalent dose, ATB=Ashodhita Tamra Bhasma, STB=Shodhita Tamra Bhasma, GPT=Glutamate pyruvate transaminase, GOT=Glutamate oxaloacetate transaminase. | |||||||
Table 2: Effect on serum biochemical parameters.
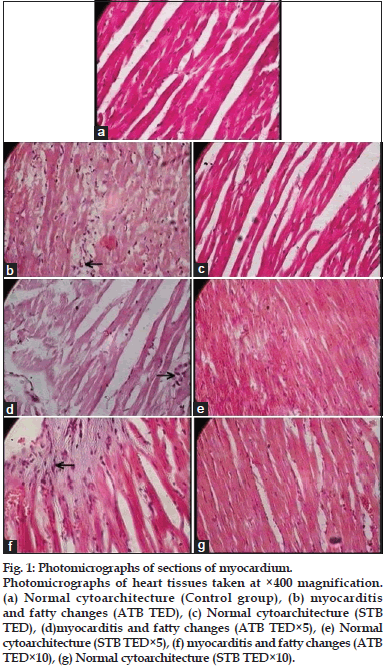
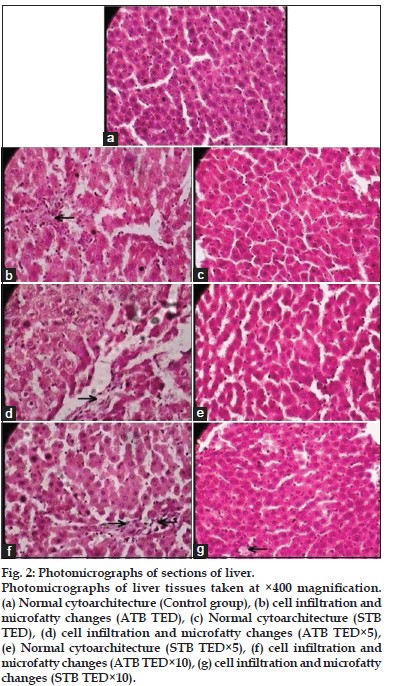
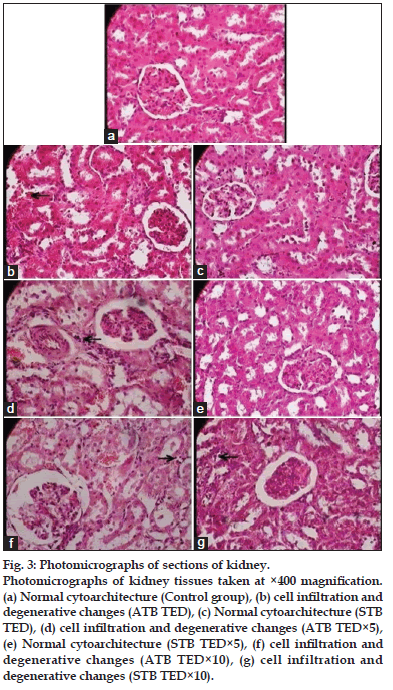
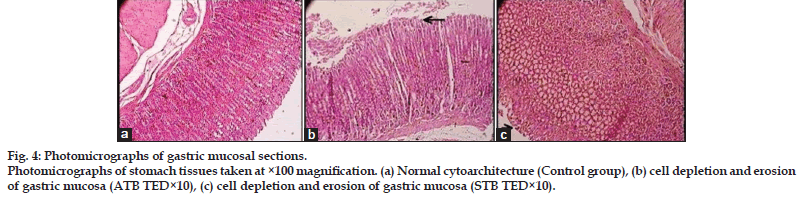
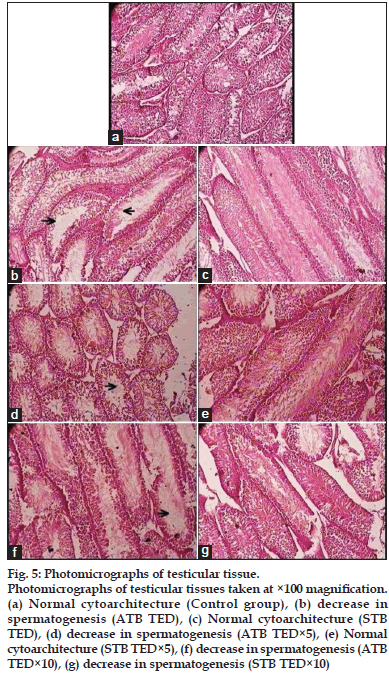
Microscopic examination of all the organs obtained from control group exhibited normal cytoarchitecture (figs 1a, 2a, 3a, 4a and 5a). All the doses of ATB and STB did not affect the cytoarchitecture of brain, pituitary, spleen, lung, stomach, lymph node, ovary, thymus, and uterus. Administration of ATB at all the three doses lead to myocarditis and fatty changes in heart (figs 1b, 1d, and 1f), cell infiltration and microfatty changes in liver (figs 2b, 2d, and 2f), cell infiltration and degenerative changes in kidney (figs 3b, 3d, and 3f), and decrease in spermatogenesis (figs 5b, 5d, and 5f). It also caused cell depletion and erosion of gastric mucosa at TED×10 dose (fig. 4b). In contrast, STB at TED and TED×5 dose showed normal cytoarchitecture in all of these organs; however, at TED×10 dose it caused pathological changes in myocardium (figs 1c, 1e and 1g), liver (figs 2c, 2e, and 2g), kidney (figs 3c, 3e, and 3g) stomach (fig. 4c) and testicular tissue (figs 5c and 5e) similar to that of ATB.
Fig. 1: Photomicrographs of sections of myocardium. Photomicrographs of heart tissues taken at ×400 magnification. (a) Normal cytoarchitecture (Control group), (b) myocarditis and fatty changes (ATB TED), (c) Normal cytoarchitecture (STB TED), (d)myocarditis and fatty changes (ATB TED×5), (e) Normal cytoarchitecture (STB TED×5), (f) myocarditis and fatty changes (ATB TED×10), (g) Normal cytoarchitecture (STB TED×10).
Fig. 2: Photomicrographs of sections of liver. Photomicrographs of liver tissues taken at ×400 magnification. (a) Normal cytoarchitecture (Control group), (b) cell infiltration and microfatty changes (ATB TED), (c) Normal cytoarchitecture (STB TED), (d) cell infiltration and microfatty changes (ATB TED×5), (e) Normal cytoarchitecture (STB TED×5), (f) cell infiltration and microfatty changes (ATB TED×10), (g) cell infiltration and microfatty changes (STB TED×10).
Fig. 3: Photomicrographs of sections of kidney. Photomicrographs of kidney tissues taken at ×400 magnification. (a) Normal cytoarchitecture (Control group), (b) cell infiltration and degenerative changes (ATB TED), (c) Normal cytoarchitecture (STB TED), (d) cell infiltration and degenerative changes (ATB TED×5), (e) Normal cytoarchitecture (STB TED×5), (f) cell infiltration and degenerative changes (ATB TED×10), (g) cell infiltration and degenerative changes (STB TED×10).
Fig. 5: Photomicrographs of testicular tissue. Photomicrographs of testicular tissues taken at ×100 magnification. (a) Normal cytoarchitecture (Control group), (b) decrease in spermatogenesis (ATB TED), (c) Normal cytoarchitecture (STB TED), (d) decrease in spermatogenesis (ATB TED×5), (e) Normal cytoarchitecture (STB TED×5), (f) decrease in spermatogenesis (ATB TED×10), (g) decrease in spermatogenesis (STB TED×10)
Discussion
The right dose distinguishes the drug from a poison. So the safety and toxicity profile of the drug is important which is achieved through animal experimentation. Acute toxicity study was done to record immediate adverse signs and symptoms after the administration of single dose of drug at dose levels that are several folds higher than the therapeutic equivalent dose and 2000 mg/kg was set as limit test. In this test, mortality was not observed in any of the groups which suggest that the LD50 is much higher than this dose for both ATB and STB.
Globally Harmonized System (GHS) is an internationally agreed system of classification and labeling of chemicals, which was developed under the auspices of the United Nations (UN) [28]. The GHS document, which is known as the “purple book”, describes the harmonized classification criteria and the hazard communication elements by the type of hazard. It provides decision logics for each hazard, examples of classification of chemicals and mixtures and illustrates how to apply the criteria. The GHS includes harmonized criteria for the classification of physical hazards (e.g., flammable liquids); health hazards (e.g., carcinogens); and environmental hazards (e.g., aquatic toxicity). Since the test formulation is used for health care it was analyzed for probable health hazards. According to this classification if the LD50 in acute oral toxicity test is more than 2000 mg/kg it can be considered as hazardous and put in category 4. Material containing heavy metals are categorized as hazardous [29]. As per UN Classification any substance which has oral LD50 of more than 200 mg/kg is considered as low hazard potential and categorized as UN 6.1 PG III [30]. Thus as per the above criterion both ATB and STB can be categorized as substances with low health hazard potential (Class 4 of GHS and UN 6.1PGIII).
On subchronic administration ATB nonsignificantly increased WBC count in all the three doses. Increased WBC count is indicative of inflammatory conditions of certain organs especially of liver [31]. Histopathological studies showed severe fatty changes and degenerative changes not only in liver but in other organs like kidney and heart also. This may be the reason for the elevation of WBC count. Although significant changes occurred in other hematological parameters like total RBC count, neutrophil percentage, and PCV, they cannot be taken as pathological as they were not occurred in dose‑dependant manner.
ATB administration at all the three levels significantly decreased HDL cholesterol. Smoking, anabolic steroids, beta‑blockers, malnutrition and obesity lead to low HDL [32]. However these factors in the experimental conditions studied are unlikely to be the cause of the observed decrease in serum HDL‑cholesterol level in treated groups. Two other possibilities that can be proposed to explain the decrease are the test drugs may impair the transfer of cholesterol from both very low density lipoproteins and tissue to HDL fraction or they may be promoting the metabolism of this fraction by enhancing the activity of the key enzymes involved in HDL cholesterol metabolism. In contrast to this observation, administration of STB at TED and TED×5 did not alter the HDL cholesterol to significant extent, however only at TED×10 dose levels it decreased the HDL cholesterol to significant extent. Significant decrease in serum total protein only at higher dose level (TED×10) of both ATB and STB indicates possibility of hypoproteinemia which is most commonly associated with the decreased production of albumin in liver due to significant destruction of hepatocytes [33]. It can also be seen in nephritic syndrome due to excessive loss in urine [34]. Observed decrease in serum protein level may be attributed to impairment of liver function because histopathological study of liver sections from this dose level shows cell infiltration and microfatty changes. It is interesting to note that at TED and even TED×5 dose levels both the test samples did not produce any significant changes in total protein level. This clearly indicates importance of drug dosage in production of toxicity.
Tamra Bhasma prepared from Ashodhita Tamra is toxic even in the dose of therapeutic equivalent dose and highly toxic at TED×5 and TED×10 doses on subchronic administration for 45 days as revealed by hematological, biochemical, and histopathological parameters. Tamra Bhasma prepared from Shodhita Tamra is safe even five‑fold to therapeutically equivalent doses. However at ten fold dose it is not only prone to cause haepatotoxicity and nephrotoxicity, but also cause gastric mucosal damage. These observations reveal the role of Shodhana and importance of dose in expression of toxicity of the medicinal preparations.
References
- Kumar A, Nair AG, Reddy AV, Garg AN. Availability of essential elements in Bhasmas: Analysis of Ayurvedic metallic preparations by INAA. J Radioanalytical Nuclear Chem 2006;270:173‑80.
- Chan K. Some aspects of toxic contaminants in herbal medicines. Chemosphere 2003;52:1361‑73.
- Krishnamachary B, Rajendran N, Pemiah B, Krishnaswamy S, Krishnan UM, Sethuraman S, et al. Scientific validation of different purification steps involved in the preparation of an Indian Ayurvedic medicine, LauhaBhasma. J Ethnopharmacol 2012;142:98‑104.
- Kohli KR. Ayurvedic medicines and heavy metals issue. Ayurveda Herit 2005;1:5‑6.
- Wikipedia, The free encyclopedia. Wikimedia Foundation, Inc. [updated 2012 Feb 22]. Available from: http://en.wikipedia.org/wiki/Toxic_ metal [Last accessed on 2012 Jun 10].
- Upadhyay M. AyurvedPrakasha. In: Mishra GS, editor. 3rd chapter. Verse 115‑116. Varanasi: ChaukhambaBharatiya Academy; 1999. p. 368.
- Prajapati PK, Sarkar P, Nayak SV, Joshi RD, Ravishankar B. Safety and toxicity profile of some metallic preparations of Ayurveda. AncSci Li 2006;25:57‑63.
- Pattanaik N, Singh AV, Kumar M, Dixit SK, Tripathi YB. Toxicology and free radical scavenging property of TamraBhasma. Indian J ClinBiochem 2003;18:181‑9.
- Vahalia MK, Thakur KS, Nadkarni S, Sangle VD. Chronic toxicity study for TamraBhasma (A generic Ayurvedic mineral formulation) in laboratory animals. Rec Res SciTechnol 2011;3:76‑9.
- Vagbhattacharya. RasaratnaSamuchchaya, In: Kulkarni DA, editor. 5th chapter. Verse 13. Reprint. New Delhi: MeharchandLaxmandas publication; 1998. p. 93‑4.
- Sharma SN. Rasa Tarangini, In: Shastri KN, editor. 17th chapter. Verse 40‑42. Reprint. Delhi: MotilalBanarasi Das; 2004. p. 418.
- Sharma SN, Rasa Tarangini. In: Shastri KN, editor. 17th chapter. Verse 52. Reprint. Delhi: MotilalBanarasi Das; 2004. p. 422.
- Paget GE, Barnes JM. Evaluation of drug activities. In: Laurence DR, Bacharach AL, editors. Pharmacometrics. Vol. 1. London: Academic Press; 1964. p. 50.
- Pennock CA, Murphy D, Sellers J, Longdon KJ. A comparison auto analyzer method for the estimation of glucose in blood. ClinChimActa 1973;48:193‑201.
- Talke H, Schubert GE. Enzymatic urea determination in the blood and serum in Warburg optical test. KlinWochenschr 1965;42:174‑5.
- Slot C. Plasma creatinine determination. A new and specific Jaffe reaction method. Scand J Clin Lab Invest 1965;17:381‑7.
- Roeschlau P, Bernt E, Gruber WA. Enzymatic determination of total cholesterol in serum. J ClinChemClinBiochem 1974;12:226.
- Dominiczak M, McNamara J. The system of cardiovascular prevention. Nauk M, Wiebe D, Warnick G. Measurement of High‑Density‑Lipoprotein Cholesterol. In: Rifai, Warnick, Dominiczak, editors. Handbook of Lipoprotein testing. 2nd ed. Washington, DC: AACC Press; 2000. p. 819.
- Fossati P, Prencipe L. Serum triglycerides determined colorimetrically with an enzyme that produces hydrogen peroxide. ClinChem 1982;28:2077‑80.
- Tietz NW, editor. Text book of Clinical Chemistry. Philadelphia, PA: WB Saunders; 1986. p. 579.
- Wilkinson JH, Boutwell JH, Winsten S. Evaluation of a new system for kinetic measurement of serum alkaline phosphatase. ClinChem 1969;15:487‑95.
- Tietz NW, editor. Clinical Guide to Laboratory Tests. 3rd ed. Philadelphia, PA: WB Saunders; 1995. p. 76.
- Burtis CA, Ashwood ER, editors. Tietz textbook of Clinical Chemistry. 3rd ed. Philadelphia, PA: WB Saunders; 1999. p. 652.
- Kabasakalian P, Kalliney S, Wescott A. Determination of uric acid in serum, with use of uricase and tribromophenol‑aminoantipyrinechromogen. ClinChem 1973;19:522.
- Pearlman PC, Lee RT. Detection and measurement of total bilirubin in serum with use of surfactants as solubilising agents. ClinChem 1974;20:447.
- Burtis CA, Ashwood ER, editors. Tietz Textbook of Clinical Chemistry, 3rd ed. Philadelphia, PA: WB Saunders; 1999. p. 1136.
- Raghuramulu N, Nair KM, Kalyanasundaram S. A manual of laboratory techniques. Hyderabad: National Institute of Nutrition; 1983. p. 246‑53.
- Anonymous. Globally harmonized system of classification and labelling
- of chemicals, Part 3. Health Hazards. 3rd Revised ed. New York and Geneva: United Nations; 2009.Jarup L. Hazards of heavy metal contamination. Br Med Bull 2003;68:167‑82.
- Anonymous. UN recommendations on the transport of dangerous goods‑model regulations, Part – 2. In: Chapter 2.6, Class 6‑toxic and infectious substances. 12th revised ed. Geneva: United Nations Publications; 2001.
- Davidson S. Davidson’s principles and practice of medicine. 20th ed. Philadeilphia, PA: WB Saunders; 2006. p. 1006.
- Rader DJ, Hobbs HH. Disorders of lipoprotein metabolism. In: Longo DL, Kasper DL, Jameson JL, Fauci AS, Hauser SL, Loscalzo J, editors. Harrison’s principles of internal medicine. Vol. 2. New York: The McGraw‑Hill Companies; 2008. p. 3152.
- Mohan H. Textbook of Pathology. 4th ed. New Delhi: Jaypee Brothers Medical Publishers; 2002. p. 573.
- Longmore M, Wilkinson IB, Davidson EH, Foulkes A, Mafi AR. Oxford Handbook of Clinical Medicine. 8th ed. New York: Oxford University Press; 2010. p. 286.