- *Corresponding Author:
- T. Phaechamud
Department of Pharmaceutical Technology, Faculty of Pharmacy, Silpakorn University, Nakhon Pathom 73000, Thailand
E-mail: thawatchaienator@gmail.com
| Date of Submission | 14 October 2016 |
| Date of Revision | 15 February 2017 |
| Date of Acceptance | 23 July 2017 |
| Indian J Pharm Sci 2017;79(5):758-767 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
Cetirizine dihydrochloride has a very unpleasant bitter taste. To mask the bitter taste, complexation with β-cyclodextrin using ethanol as a kneading solvent was investigated and evaluated by Fourier transform infrared spectroscopy, differential scanning calorimetry, powder X-ray diffractometry, thermogravimetric analysis, simultaneous X-ray diffractometry-differential scanning calorimetry measurement and 1H-nuclear magnetic resonance spectroscopy. Taste was evaluated by human volunteers using an unstructured line scale. The basic characterization of the prepared tablets including in vivo disintegration time and drug dissolution was studied. Fourier transform infrared spectroscopy, differential scanning calorimetry and powder X-ray diffractometry data confirmed the inclusion complex formation. Thermogravimetric analysis, simultaneous X-ray diffractometry-differential scanning calorimetry measurements revealed the reduction of crystalline β-cyclodextrin due to the displacement of water molecules from complex formation. Proton nuclear magnetic resonance spectroscopy indicated an intermolecular interaction between protons of cetirizine dihydrochloride with the cavity of β-cyclodextrin. The prepared complex showed an unstructured line scale with apparent low average rating of 1.70. The inclusion complex-loaded fast disintegrating tablet prepared with direct compression has short disintegration time and good taste with an unstructured line scale with average rating of 0.28. Cetirizine dihydrochloride, β-cyclodextrin inclusion complex was successfully prepared by kneading method using ethanol as solvent. The inclusion complex-loaded fast disintegrating tablet had good taste and good perception in the mouth.
Keywords
Cetirizine, β-cyclodextrin, inclusion complex, fast disintegrating tablet, ethanol kneading
Fast disintegrating tablet (FDT) dissolves or disintegrates in saliva within seconds when placed on the tongue before it is swallowed [1-5]. Unfortunately, cetirizine dihydrochloride (CTZ) has a very unpleasant bitter taste. Many strategies have been reported to decrease the bitter taste of CTZ. Some strategy is to use lipophilic Gelucire® 33/01 in the presence of soybean lecithin to markedly reduce the bitterness without affecting CTZ release [6]. Besides, the efficient effect on masking CTZ bitterness, cyclodextrins (CD) as well could influence the drug release from the formulation. The presence of β-cyclodextrin (β-CD) resulted in an increase of cetirizine release [7]. The inclusion complex between CTZ and β-CD prepared using a supercritical antisolvent (SAS) process and freeze drying method presented the same efficacy in regards to the dissolution characteristics [8]. CD and its derivatives were used as taste masking agents for the preparation of rapidly dissolving films of CTZ by using a solvent casting method. The optimized ratio of 1:3 CTZ:hydroxypropyl β-CD was appropriate for taste masking [9,10]. CTZ was coated by a fluidized bed coating using Eudragit® RL30-D and taste-masking evaluation was performed by healthy human volunteers and their perceived levels were recorded on a numerical scale [11,12]. Moreover, rizatriptan benzoate and β-CD prepared by kneading with distilled water demonstrated effective taste masking [13]. Therefore one of the methods of CD complexation is kneading method. Ethanol is widely used for wet granulation process in industry thus the kneading method with this solvent is effective and simple for developing in large scale or industrial manufacturing.
The present work was conducted to investigate the application of β-CD for taste masking of CTZ, and the kneading method using ethanol as solvent, was employed to prepare the CTZ, β-CD inclusion complex. The formation of inclusion complexes with β-CTZ were characterized by Fourier transform infrared spectroscopy (FTIR), differential scanning calorimetry (DSC), powder X-ray diffractometry (PXRD), thermogravimetric analysis (TGA), simultaneous XRD-DSC measurement and 1H-nuclear magnetic resonance spectroscopy (1H-NMR). Finally, the taste perception study was carried out by human volunteers to evaluate the taste masking ability of inclusion complex and CTZ FDT. Therefore, the purpose of this research was to prepare and evaluate the inclusion complex between CTZ and β-CD using ethanol as solvent with kneading technique for taste masking. CTZ and β-CD FDTs were also prepared and evaluated.
Materials and Methods
CTZ, β-CD, sodium stearylfumarate (Alubra®) and aspartame were supplied by Pharmanueva Co., Ltd., Bangkok, Thailand. Ethanol (Absolute, AR grade) was purchased from VWR International SAS, France. Ludiflash® was supplied by BASF, Bangkok, Thailand. Aerosil 200 was purchased from Wacker-Chemie GmBH, Germany. Menthol and caffeine anhydrous was purchased from P. C. Drug Center Co., Ltd., Bangkok, Thailand. Lemon flavour (Reg. No. 199705186M) was supplied by Silesia flavour S.E.A. Pte, Ltd., Singapore. Potassium bromide (KBr, spectrograde) was purchased from Fisher Scientific UK limited, UK. Deuterium oxide (D2O 99.9%) was purchased from Sigma-Aldrich Co., Missouri, USA. Phosphoric acid 85% was purchased from Merck KGaA, Darmstadt, Germany and acetonitrile was purchased from V. S. ChemHouse, Bangkok, Thailand.
Preparation of inclusion complex
This study used CTZ to β-CD at 10:260 mg ratio as reported in a previous work [14]. Both compounds were mixed and ground in a mortar with a pestle for 1 min. Then, 1 ml of ethanol was added to the mixture and the obtained paste was further kneaded for 3 min. After mixing, the end product was dried in the hot air oven (UT6760, Heraeus, Kendro Laboratory Products, Germany) at 50° for 2 h and kept in desiccator before characterization. The physical mixture (PM) with the same ratio was also prepared by mixing without ethanol.
FTIR spectroscopy
Approximately 2 mg each sample was mixed and ground with 100 mg KBr before compressed into pellet using KBr die kit. Spectrums were scanned using FTIR spectrometer (FT/IR-4100 (JASCO International Co., Ltd., Tokyo, Japan), Spectra manager version 2.2.9.1 (JASCO International Co., Ltd., Tokyo, Japan) between the wave number ranges of 400-4000 cm-1 with 32 scans at 4 cm-1 resolution.
DSC
The thermal properties of samples were determined using DSC (DSC 8500, PerkinElmer, Inc., MA, USA). Small pieces of the samples were weighed accurately 5 mg into aluminium pan (PerkinElmer, Inc., MA, USA) and sealed. DSC was operated under dry nitrogen atmosphere. Measurement temperature was set at the ranges of 0-300° with heating rate of 10°/min.
PXRD
The samples were analysed under the X-ray powder diffractometer (Miniflex II, Rigaku Corp. Tokyo, Japan). The X-ray source was Cu-Kα, which employed the x-radiation of wavelength λ=1.5418 Å between a 2θ angle ranges of 4° to 60° with voltage of 30 kV and current of 15 mA. The samples were filled and compressed into a glass sample holder with 0.2 mm depth before test.
TGA
TGA was carried out by using a thermogravimetric analyser (TG8120, Rigaku, Tokyo, Japan). Samples of 10 mg were weighed into an open alumina crucible under the purging nitrogen gas. The dynamic experiment was carried out in the temperature ranges of 20° to 400° under the purging nitrogen gas at heating rate of 10°/min.
Simultaneous XRD-DSC measurement
The method for XRD-DSC measurement was studied using a Rigaku XRD-DSC II system (Riguku Corporation, Tokyo, Japan). Briefly, 5 mg of samples were weighed into opened aluminium pans. The heating rate of DSC was 10°/min and nitrogen gas was purged at flow rate of 100 ml/min. X-ray diffraction was then measured simultaneously at 40 kV and 40 mA.
1H NMR spectroscopy
One-dimensional NMR spectra (1H NMR, 500 MHz) were recorded on a Bruker Advance 300 (Karlsruhe, Germany) equipped with a 5 mm probe with actively shielded z-field gradient capability. All experiments were conducted using D2O (99.95% isotopic purity) as solvent. The probe temperature was regulated to 300 K.
Taste evaluation of inclusion complex
Gustatory sensation test was carried out by panel of human volunteers, which informed consent was approved by the Ethics Committee (ID 7/2557, 6/2557) of Faculty of Pharmacy, Silpakorn University. For this purpose, the 6 healthy human volunteers, of either sex, in the age group of 20-35 y were selected out from 20 volunteers based on taste sensitivity test from training. The 6 healthy human volunteers were trained to recognize a level of bitter taste of CTZ. Caffeine was used as the standard of bitter taste to compare and recognize a bitter taste level of CTZ [15-17].
Determination of threshold bitterness concentration of CTZ was determined using the series of caffeine standard solutions of different concentrations (0.05, 0.15 and 0.2 mg/ml). When the volunteers could pass the training, they would be accepted for testing the bitter taste of sample. CTZ powder was placed in mouth for 15 s to compare with inclusion complex equivalent to 10 mg CTZ and its PM. After expectoration, the bitterness level was recorded and thereafter the samples were spit out. The oral cavity was rinsed with distilled water twice (200 ml per each time). The time between the testing of each sample was 10 min. The scaling technique for this study was unstructured line scale for grading of the bitterness as following levels: 0=no bitterness and 12=strongly bitter as previously reported [15-17]. After evaluation by volunteer, the result was calculated for mean and standard deviation. Significant differences among the test scores were analysed using the R-stat test version 3.0.1; a value of P<0.05 was accepted as index of a significant difference.
Preparation of CTZ, β-CD FDT
The direct compression method was used to prepare CTZ, β-CD FDT complex equivalent to 10 mg of drug was taken along with diluent (Table 1). However, the β-CD amount was lowered to decrease the tablet weight and then other favouring agents were used to mask the tastes of drug (Table 1). Ludiflash®, Aerosil 200, Alubra®, aspartame, menthol and lemon flavour were used as fast disintegrating diluent, glidant, lubricant, sweetening, cooling agent and flavouring agent, respectively. All the ingredients were weighed and passed through sieve #30. Then, the powders except lubricant were mixed geometrically in plastic bottle. Finally, Alubra® was added and mixed in plastic bottle. Tablets were compressed on single punch tablet machine using round concave punch of 9 mm diameter. The hardness of the tablet was maintained at 3 kp.
| Ingredient | Content (mg/tab) |
|---|---|
| CTZ/β-CD complex (equivalent to 10 mg of CTZ) | 70.17 |
| Alubra® | 4 |
| Aerosil 200 | 0.2 |
| Aspartame | 4 |
| Menthol | 0.4 |
| Lemon flavor | 2.4 |
| Ludiflash® | 138.83 |
| Total | 220 |
β-CD: β-cyclodextrin, CTZ: cetirizine dihydrochloride
Table 1: Composition of CTZ fast-disintegrating tablet
Evaluation of CTZ FDTs
The prepared tablets were evaluated for the following different characteristics like weight variation, disintegration time, in vitro dissolution profile. The weight variation test was performed on 20 randomly collected tablets from a batch. The procedure described in USP 37 NF 32 [18] was followed for this test. Weight variation test was done by weighing 20 tablets individually. For content uniformity and assay were conducted with the HPLC method as described in USP 37 NF 32 [19].
Disintegration time was tested following method. Six tablets were placed individually in each tube of disintegration apparatus and the discs were placed. The distilled water was maintained at a temperature of 37±0.5° and the time taken for all tablets to disintegrate in this immersion fluid completely was recorded.
The dissolution of CTZ from FDT was determined by USP dissolution testing apparatus II (paddle method). The dissolution test was carried out using 900 ml water, at 37±0.5° and 50 rpm. A sample (10 ml) of the solution was withdrawn from the dissolution apparatus at different time interval. An aliquot of 10 ml was collected at predetermined time intervals and replaced with fresh dissolution medium. Samples were analysed by HPLC as assay method with some modification [18].
In vivo disintegration time and taste masking
Evaluation of taste masking was done using the intensity method on panel of 6 healthy human volunteers with informed consent approved by the ethics committee (ID 7/2557, 6/2557) of Faculty of Pharmacy, Silpakorn University. Prior to the test the volunteers were instructed to rinse their oral cavity with distilled water. Each volunteer was asked to place one FDT on the tongue. Volunteers were strictly informed not to chew or swallow the tablets, though licking was allowed. The end point for disintegration was recorded when there was no lump left in the oral cavity. After the test was finished, volunteers rinsed their mouth properly. Bitterness, mouth feeling and disintegration time were recorded immediately. The scaling technique for this study was unstructured line scale for grading of the bitterness as following levels: 0=no bitterness and 12=strongly bitter [15-17]. Significant differences among the test scores were analysed using the R-stat test version 3.0.1; a value of P<0.05 was accepted as index of a significant difference.
Statistical analysis
Statistical significance of the measurements was examined using one-way analysis of variance (ANOVA) followed by the least significant difference (LSD) post hoc test. The significance level was set at P<0.05. The analysis was performed by using SPSS for windows (version 11.5).
Results and Discussion
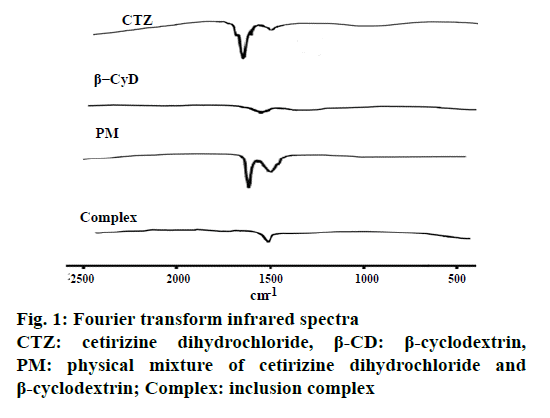
The prepared inclusion complex was white and fine powder without stickiness. Figure 1 shows the FTIR spectra for CTZ, β-CD, PM of CTZ and β-CD and also inclusion complex of CTZ and β-CD. The carbonyl group of CTZ exhibited an intense absorption peak between 1500 and 1700 cm−1 due to its stretching vibration. CTZ exhibited a strong absorption peak at 1700 cm-1 of the carbonyl stretching vibration. In the PM, as compared to pure CTZ, the spectrum showed a less intense absorption peak at 1700 cm−1. Moreover, the spectrum of the inclusion complex displayed almost similar to the pure β-CD.
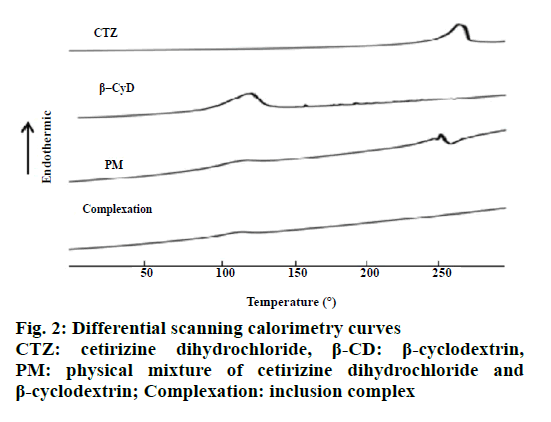
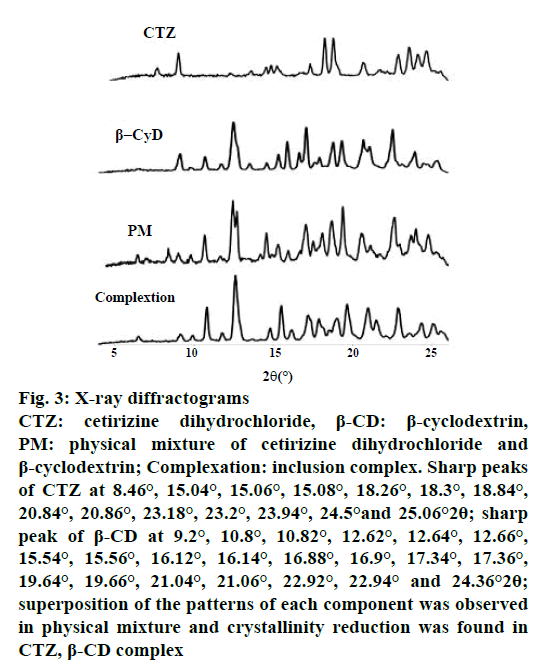
The DSC thermogram of CTZ (Figure 2) exhibited an endothermic peak at 220° corresponding to its thermal decomposition. β-CD showed a broad endothermic peak at about 90°. The PM showed two endothermic peaks, one for the β-CD at 90° and another for the CTZ at 220°. Moreover, the DSC curve displayed by the inclusion complex is similar to that of pure β-CD. The formation of inclusion complex was identified by PXRD as presented in Figure 3. CTZ showed the sharp peaks at 8.46°, 15.04°, 15.06°, 15.08°, 18.26°, 18.3°, 18.84°, 20.84°, 20.86°, 23.18°, 23.2°, 23.94°, 24.5° and 25.06° 2θ positions indicating the crystalline nature of this drug. The XRD of β-CD exhibited the sharp peak at 9.2°, 10.8°, 10.82°, 12.62°, 12.64°, 12.66°, 15.54°, 15.56°, 16.12°, 16.14°, 16.88°, 16.9°, 17.34°, 17.36°, 19.64°, 19.66°, 21.04°, 21.06°, 22.92°, 22.94° and 24.36° 2θ positions. The PXRD pattern of the PM was the superposition of the patterns of each component, but the intensity was decreased, while in the XRD of inclusion complex, it showed a significant reduction of crystallinity.
Figure 3: X-ray diffractograms
CTZ: cetirizine dihydrochloride, β-CD: β-cyclodextrin,
PM: physical mixture of cetirizine dihydrochloride and
β-cyclodextrin; Complexation: inclusion complex. Sharp peaks
of CTZ at 8.46°, 15.04°, 15.06°, 15.08°, 18.26°, 18.3°, 18.84°,
20.84°, 20.86°, 23.18°, 23.2°, 23.94°, 24.5°and 25.06°2θ; sharp
peak of β-CD at 9.2°, 10.8°, 10.82°, 12.62°, 12.64°, 12.66°,
15.54°, 15.56°, 16.12°, 16.14°, 16.88°, 16.9°, 17.34°, 17.36°,
19.64°, 19.66°, 21.04°, 21.06°, 22.92°, 22.94° and 24.36°2θ;
superposition of the patterns of each component was observed
in physical mixture and crystallinity reduction was found in
CTZ, β-CD complex
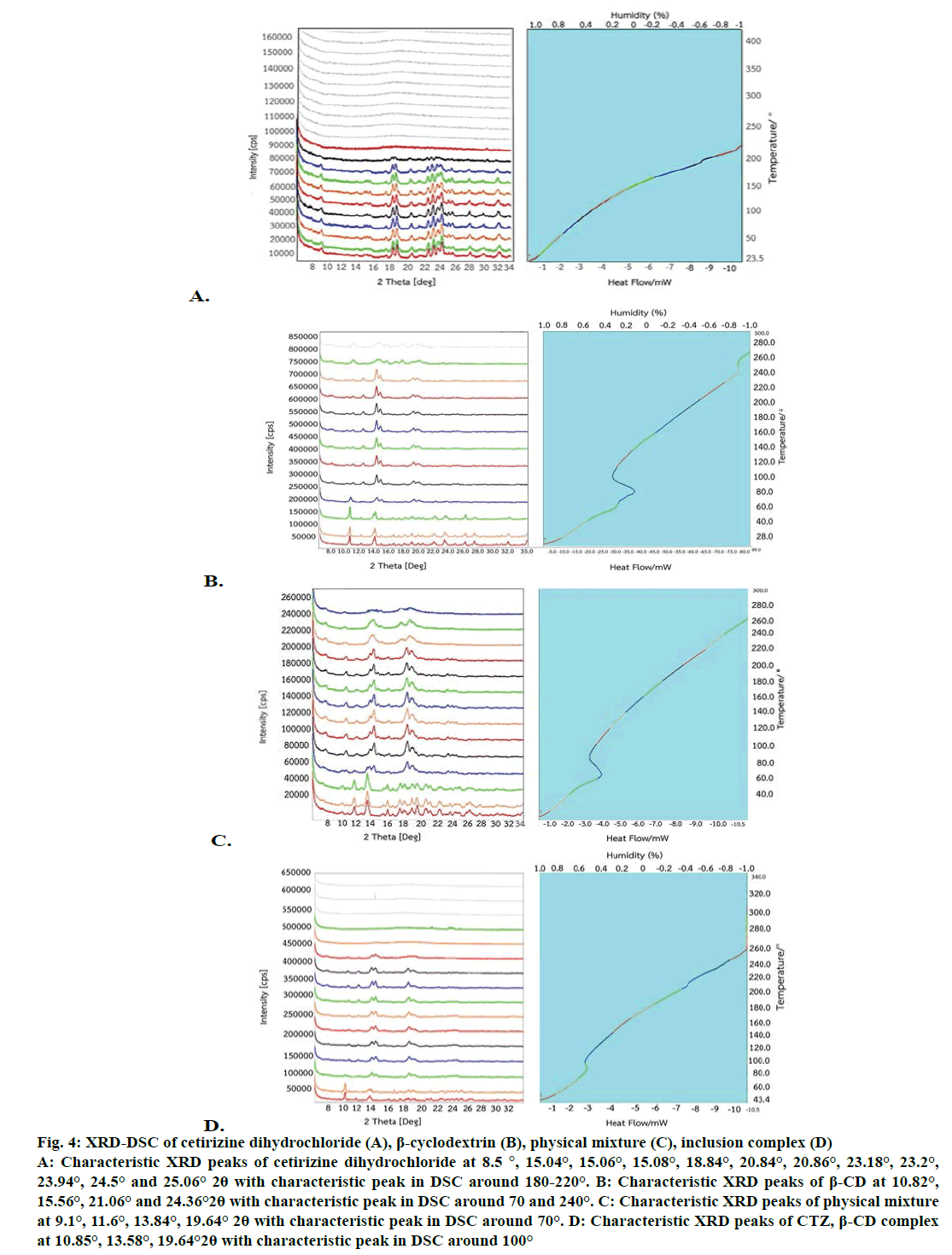
Parallel measurements of separate specimens using DSC and XRD and a combination of both results are one of the popular methods for characterization of solids. Simultaneous XRD and DSC measurement were used to validate the result of XRD and DSC of CTZ and inclusion complex (Figure 4). The characteristic peaks of CTZ were at 8.5°, 15.04°, 15.06°, 15.08°, 18.84°, 20.84°, 20.86°, 23.18°, 23.2°, 23.94°, 24.5°, 25.06° 2θ positions (Figure 4A) that exhibited at the same position as previous powder X-ray measurement. Moreover, DSC thermogram showed the characteristic peak of CTZ at around 180-220° in line with previous study, which reported at 220° [19]. The simultaneous XRD-DSC data of the β-CD, PM and inclusion complex, were also recorded (Figure 4B-D). There were three crystalline peaks of inclusion complex at around 10.85°, 13.58º, 19.64° 2θ positions. The first stage of inclusion complex showed characteristic peak of X-ray pattern at 10.85° 2θ position and showed the loss of water molecules of β-CD from DSC thermogram around 70°. Moreover, PM exhibited the characteristic peak of X-ray pattern at 9.1°, 11.6°, 13.84°, 19.64° 2θ positions whereas the inclusion complex showed at 10.85°, 13.58°, 19.64° 2θ positions.
Figure 4: XRD-DSC of cetirizine dihydrochloride (A), β-cyclodextrin (B), physical mixture (C), inclusion complex (D)
A: Characteristic XRD peaks of cetirizine dihydrochloride at 8.5 °, 15.04°, 15.06°, 15.08°, 18.84°, 20.84°, 20.86°, 23.18°, 23.2°,
23.94°, 24.5° and 25.06° 2θ with characteristic peak in DSC around 180-220°. B: Characteristic XRD peaks of β-CD at 10.82°,
15.56°, 21.06° and 24.36°2θ with characteristic peak in DSC around 70 and 240°. C: Characteristic XRD peaks of physical mixture
at 9.1°, 11.6°, 13.84°, 19.64° 2θ with characteristic peak in DSC around 70°. D: Characteristic XRD peaks of CTZ, β-CD complex
at 10.85°, 13.58°, 19.64°2θ with characteristic peak in DSC around 100°
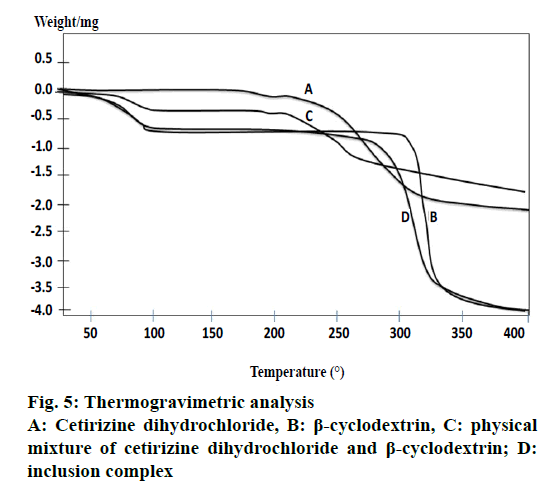
TGA was done on samples to identify the change in weight percentage with respect to temperature change. Figure 5 shows the TGA thermograms of pure CTZ, β-CD, PM, inclusion complex. β-CD exhibited two stages of weight loss. The first stage was due to the loss of water molecules at 50°, which was located in the cavity of β-CD and the weight loss at 300° was also found. The inclusion complex showed the weight losses in three stages.
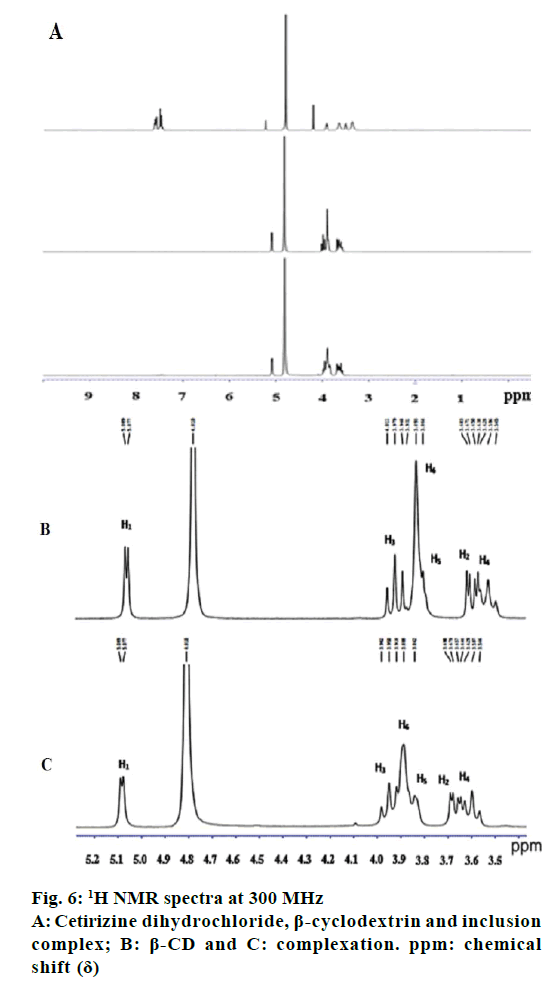
1H NMR spectroscopy of CTZ, β-CD, PM, CTZ, β-CD inclusion complex prepared by kneading method is showed in Figure 6 and the chemical shift together with assignments are presented in Table 2. In this study, only β-CD and inclusion complex protons were discussed because too many peaks were overlapped in 7.4-7.7 ppm region of CTZ, which related to the aromatic ring resonance [8]. From Table 2 and Figure 4C, some chemical shifts were observed for H1, H2, H3, H4, H5 and H6. The largest chemical shift changes of CTZ were investigated for H3 and H5 cavity protons, resulting from intermolecular interactions with guest protons residing in the CTZ cavity. Changes in chemical shift were determined to some extent for all CTZ protons, and the highest change was observed at the phenyl and chlorophenyl ring protons.
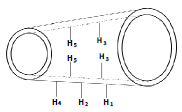 |
||||||
|---|---|---|---|---|---|---|
| Substance | H1 | H2 | H3 | H4 | H5 | H6 |
| β-CD | 5.089 | 3.683 | 3.979 | 3.638 | 3.864 | 3.893 |
| Complexation | 5.089 | 3.69 | 3.95 | 3.646 | 3.842 | 3.888 |
| Δδ | 0 | 0.007 | -0.029 | 0.006 | -0.022 | -0.005 |
Δδ is change of chemical shift. β-CD: β-cyclodextrin
Table 2: Change of chemical shift (δδ, ppm) for protons of β-cd and complexation
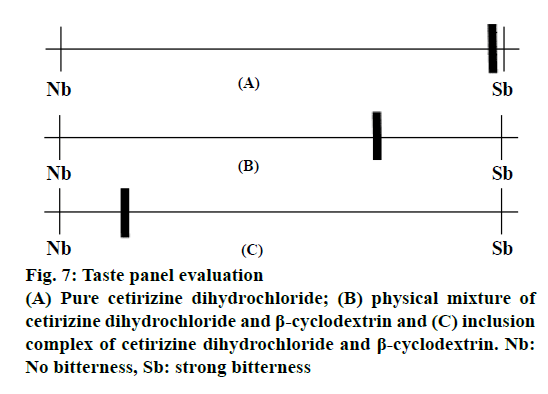
An unstructured line scale was used for the quantification of the results from taste sensory study as presented in Figure 7. Generally, all examiners demonstrated overall the same pattern of bitterness perception for the CTZ. The overall ratings for various samples were obtained and the mean±SD rating was used to qualify the taste masking efficiency. Inclusion complex showed an average rating of 1.75 (SD=0.22) as compared to 11.55 (SD=0.09), 8.4 (SD=0.28) in case of CTZ powder and CTZ, β-CD PM, respectively.
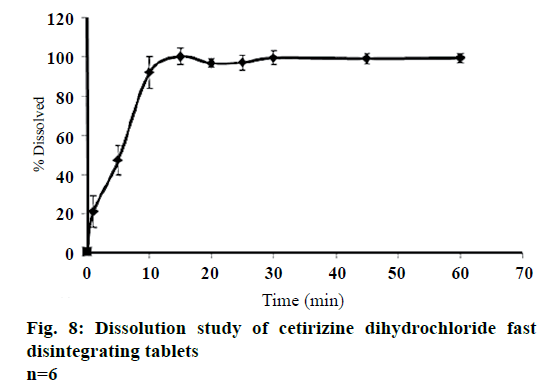
Percent weight variation was within the prescribed official limits, and in an assay was found to be in the range of 97-98% (Table 3) within the acceptable limit. Content uniformity for the tablets is shown in the Table 3. The low value of standard deviation indicated the uniformity of drug content. The disintegration time of tablets was less than 60 s. The in vitro fast drug dissolution of tablets was attained with 99.5% within 10 min (Figure 8). The disintegration time of the CTZ, β-CD FDTs in the mouth of the volunteers was 33 s (Table 3). Thus, a fast disintegration tablet was successfully achieved. CTZ, β-CD FDT showed an improved to no bitterness perception with which an average rating of was 0.28 (SD=0.26) and significantly different from that of CTZ (average rating of 11.55, SD=0.82, P<0.05). Moreover, all the volunteers experienced a pleasurable sensation in the mouth when FDT dissolved in the mouth with a minimum of grit with the fragrance of lemon and mint with sweet taste of aspartame.
| Properties | Value (mean±SD) |
|---|---|
| Assay (%, n=20) | 97.67±0.38 |
| Content uniformity (%, n=10) | 98.69±1.34 |
| Disintegration time (s, n=6) | 31.33±1.37 |
| Oral disintegration time (s, n=6) | 33.83±4.49 |
SD is standard deviation; s is second
Table 3: Physicochemical properties of CTZ FDT
The FTIR spectrum of the inclusion complex displayed almost similar to the pure β-CD indicating that during the formation of the inclusion complex, CTZ was introduced into the cavities of β-CD. Therefore FTIR analysis demonstrated the interaction and complex formation between CTZ and β-CD. Previous study reported that cetirizine showed strong absorption peak at 1741 cm−1 and the spectra of inclusion complexes were similar to that of β-CD [8]. From DSC, CTZ exhibited an endothermic peak at 220° corresponding to its thermal decomposition which was in agreement with previous investigation [19]. The endothermic peak of β-CD showed a broad peak about at 90°, which attributed to a dehydration process. β-CD exhibiting a broad peak about 91.82° has been reported previously [20]. Moreover, the DSC curves of the inclusion complex displayed almost similar to that of the pure β-CD indicating that during the formation of the inclusion complex, CTZ was introduced into the cavities of β-CD. Loss of crystallinity from XRD of the complexes may signify the presence of reciprocal interactions between drug and β-CD [21,22]. Moreover, the X-ray pattern of the inclusion complex displayed almost similar to the pure β-CD. Therefore, these results confirmed that CTZ was included in the β-CD cavity.
Simultaneous XRD-DSC data indicated that the inclusion complex started to lose water from β-CD cavity at 71° as reported previously [21,23]. Consequently, the second stage of inclusion complex showed X-ray pattern at 13.58° 2θ and the exothermic peak of inclusion complex from a decomposed β-CD was found around 230°. Some research reported the second decomposition of inclusion complex at 225° [24]. The third stage at 19.64° 2θ was probably owing to the decomposition of CTZ which showed the exothermic peak around 220° [19]. Additionally, XRD pattern of the inclusion complex was similar to that of β-CD indicated that CTZ was included into cavity. The changes in diffractogram clearly indicated the transformation of the crystalline form to another by removal of water molecules. When the water molecules were released from the cavity of β-CD, the crystallinity of complexes deteriorated [25]. The main driving force of complex formation was the release of enthalpy-rich water molecules form the cavity. Water molecules were displaced by more hydrophobic guest or partial guest. Furthermore, the bindings of CTZ within β-CD cavity decreased the ring strain as well as increasing the number of hydrogen bonds formed as the displaced water returned to the larger pool resulting in the changing 2θ positions of inclusion complex compared with PM. Therefore, water molecules played an important role for formation of inclusion complex. Ethanol has been used as solvent for kneading method because it could dissolve CTZ and β-CD [26,27].
Weight loss from TGA found at 220° related to the endothermic peak of DSC corresponding to its thermal decomposition while that at 300° indicated its decomposition temperature as mentioned previously [28]. The inclusion complex showed the weight losses in three stages. The first stage was the dehydration of water molecules, the second stage was due to the decomposition of CTZ and the third stage was the decomposition of β-CD. In comparison, to confirm the formation of the inclusion complex, the first weight loss for the inclusion complex started around 30° whereas the first weight loss for the PM started around 50° which was almost similar to the water loss of β-CD. In addition, the second weight loss for the inclusion complex started at 250° whereas it started at 200° for the PM but for the β-CD it occurred at 310°. Previous study mentioned that the formation of inclusion complex decreased the thermal stability of β-CD [29].
Insertion of a guest molecule into the hydrophobic cavity of the β-CD resulted in the chemical shift (Δδ) of guest and host molecules in the NMR spectra. The largest chemical shift changes of CTZ were investigated for H3 and H5 cavity protons, resulting from intermolecular interactions with guest protons residing in the CTZ cavity. Chemical shift changes were determined to some extent for all CTZ protons, and the highest change was observed at the phenyl and chlorophenyl ring protons. Therefore, this shift provided the indication for the formation of the inclusion complex between β-CD and CTZ [30-34].
From taste sensory study, the bitterness of complex is significantly different from CTZ powder and PM (P<0.05). CTZ was notably bitter and slightly sour due to its dihydrochloride salt form [7]. Based on the average ratings it could be concluded that CTZ was successfully complexed in the cavity of β-CD using kneading method, which prevented a contact of CTZ with taste bud receptors. The prepared CTZ, β-CD FDTs exhibited proper physical properties with fast in vitro and in vivo disintegration and also fast drug dissolution. CTZ, β-CD FDT showed good taste and low bitterness, which all the volunteers experienced a pleasurable sensation in the mouth. In conclusion, FDT of CTZ, β-CD exhibited good palatability.
CTZ, β-CD inclusion complex was prepared successfully with the kneading method using ethanol as a solvent. All the data obtained from the FTIR, DSC, X-ray diffraction, TGA, simultaneous XRD-DSC measurement, 1H NMR spectroscopy and SEM studies confirmed the formation of an inclusion complex. In addition, inclusion complex reduced the bitter taste of CTZ after testing with an unstructured line scale. The taste evaluation by human volunteers demonstrated that the CTZ, β-CD FDT had good taste, good perception in the mouth and reduced bitterness.
Acknowledgements
This work was supported through The Royal Golden Jubilee funding (no. PhD/0052/2552). Materials and instruments were kindly made available for use by the Faculty of Pharmacy, Silpakorn University and Pharmanueva Co., Ltd. The authors would like to thank Prof. Katsuhide Terada, Mr. Peeracha Thanawattanawanich and Dr. Napasinee Aksornkoae for their kindness, valuable support and great enthusiasm in my research work. The authors are grateful to volunteers for their cooperation. We would like to thank Henry James Erskine-Hill for valuable proof reading.
Conflicts of interest
There are no conflicts of interest.
Financial support and sponsorship
Nil.
References
- Fu Y, Yang S, Jeong SH, Kimura S, Park K. Orally fast disintegrating tablets: developments, technologies, taste-masking and clinical studies. Crit Rev Ther Drug2004;21:433-76.
- Fukami J, Yonemochi E, Yoshihashi Y, Terada K. Evaluation of rapidly disintegrating tablets containing glycine and carboxymethylcellulose. Int J Pharm2006;310:101-9.
- Sunada H, Bi Y. Preparation, evaluation and optimization of rapidly disintegrating tablets. Powder Technol2002;122:188-98.
- Wilson CG, Washington N, Peach J, Murray GR, Kennerley J. The behavior of a fast-dissolving dosage form (expidet) followed by g-scintigraphy. Int J Pharm1987;40:119-23.
- Ishikawa T, Watanabe Y, Utoguchi N, Matsumoto M. Preparation and evaluation of tablets rapidly disintegrating in saliva containing bitter-taste-masked granules by the compression method. Chem Pharm Bull (Tokyo)1999;47:1451-4.
- Massik ME. Development of cetirizine chewable tablets with inhibited bitterness using liquisolid compact technology. Alex J Pharm Sci2006;20:11-6.
- Stojanov M, Larsen KL. Cetirizine release from cyclodextrin formulated compressed chewing gum. Drug Dev Ind Pharm2012;38:1061-7.
- Lee CW, Kim SJ, Youn YS, Widjojokusumo E, Lee YH, Kim J, et al. Preparation of bitter taste masked cetirizine dihydrochloride/β-cyclodextrin inclusion complex by supercritical antisolvent (SAS) process. J Supercrit Fluids 2010;55:348-57.
- Mishra R, Amin A. Optimization and characterization of rapidly dissolving films of cetirizine hydrochloride using cyclodextrins for taste masking. Int J PharmTech Res2013;5:536-52.
- Patel AR, Vavia PR. Preparation and evaluation of taste masked famotidine formulation using drug/β-cyclodextrin/polymer ternary complexation approach. AAPS J2008;9:544-50.
- Douroumis DD, Gryczke A, Schminke S. Development and evaluation of cetirizine HCl taste-masked oral disintegrating tablets. AAPS J2011;12:141-51.
- Khan S, Kataria P, Nakhat P, Yeole P. Taste masking of ondansetron hydrochloride by polymer carrier system and formulation of rapid-disintegrating tablets. AAPS PharmSciTech 2007;8:E127-33.
- Birhade SVH, Gaikwad PD, Pawar SP. Preparation and evaluation of cyclodextrin based binary systems for taste masking. Inter J Pharm Sci Drug Res2010;2:199-203.
- Szejtli J, Szente L. Elimination of bitter, disgusting tastes of drugs and foods by cyclodextrins. Eur J Pharm Biopharm 2005;61:115-25.
- Lawless HT, Heymann H. Sensory Evaluation of Food: Principles and practices. 2nd ed. New York: Springer-Verlag; 1998.
- Morten CM, Carr BT, Gail VC. Sensory Evaluation Techniques. 4th ed. Boca Raton, Florida: CRC Press; 2007.
- Herbert S, Joel S. Sensory Evaluation Practices. 3rd ed. San Diego: Academic Press; 2004.
- http://app.uspnf.com/uspnf/display?cmd=jsp&page=chooser.
- Sovizi M, Hosseini S. Studies on the thermal behavior and decomposition kinetic of drugs cetirizine and simvastatin. J Therm Anal Calorim2013;111:2143-8.
- Chandrakant DS, Lingaraj SD, Abdual S, Mallikarjun BK. Preparation and evaluation of inclusion complexes of water insoluble drug. Int J Res Pharm Biomed Sci2011;2:1599-616.
- Ribeiro L, Loftsson T, Ferreira D, Veiga F. Investigation and physicochemical characterization of vinpocetine-sulfobutyl ether beta-cyclodextrin binary and ternary complexes. Chem Pharm Bull (Tokyo) 2003;51:914-22.
- Zarif MS, Afidah AR, Abdullah JM, Shariza AR. Physicochemical characterization of vancomycin and its complexes with β-cyclodextrin. Biomed Res India2012;23:513-20.
- Arii T, Kishi A, Kobayashi Y. A new simultaneous apparatus for X-ray diffractometry and differential scanning calorimetry (XRD-DSC). Thermochim Acta1999;325:151-6.
- Sambasevam K, Mohamad S, Sarih N, Ismail N. Synthesis and characterization of the inclusion complex of β-cyclodextrin and azomethine. Int J Mol Sci2013;14:3671-82.
- Del Valle EMM. Cyclodextrins and their uses: a review. Process Biochem2004;39:1033-46.
- Kawasaki J, Satou D, Takagaki T, Nemoto T, Kawaguchi A. Structural features of inclusion complexes of γ-cyclodextrin with various polymers. Polymer2007;48:1127-38.
- Yoshii H, Kometani T, Furuta T, Watanabe Y, Linko YY, Linko P. Formation of inclusion complexes of cycldextrin with ethanol under anhydrous conditions. Biosci Biotechnol Biochem 1998;62:2166-70.
- Giordano F, Novak C, Moyano JR. Thermal analysis of cyclodextrins and their inclusion compounds. Thermochim Acta2001;380:123-51.
- Chen M, Diao G, Zhang E. Study of inclusion complex of β-cyclodextrin and nitrobenzene. Chemosphere2006;63:522-9.
- Djedaini F, Lin SZ, Perly B, Wouessidjewe D. High-field nuclear magnetic resonance techniques for the investigation of a beta-cyclodextrin:indomethacin inclusion complex. J Pharm Sci1990;79:643-6.
- Fernandes CM, Carvalho RA, Costa S, Veiga FJB. Multimodal molecular encapsulation of nicardipine hydrochloride by β-cyclodextrin, hydroxypropyl-β-cyclodextrin and triacetyl-β-cyclodextrin in solution. Structural studies by 1H NMR and ROESY experiments. Eur J Pharm Sci2003;18:285-96.
- Harata K. The structure of the cyclodextrin complex. IV. The crystal structure of α-cyclodextrin-sodium 1-propanesulfonate nonahydrate. Bull Chem Soc Jpn1977;50:1259-66.
- Saenger W. Crystal packing patterns of cyclodextrin inclusion complexes. J Incl Phenom Macrocycl Chem 1984;2:445-44.
- Youming Z, Xinrong D, Liangcheng W, Taibao W. Synthesis and characterization of inclusion complexes of aliphatic-aromatic poly (Schiff base)s with β-cyclodextrin. J Incl Phenom Macrocycl Chem 2008;60:313-9.