- *Corresponding Author:
- Chaitanya Gadiko
Department of Pharmaceutical Sciences, Jawaharlal Nehru Technological University Hyderabad (JNTUH), Hyderabad, Telangana, 500085, 1Department of Pharmacology, Andhra University, Visakhapatnam, Andhra Pradesh 530003, India
E-mail: chaitanya.gadiko@gmail.com
| Date of Received | 30 April 2026 |
| Date of Revision | 02 May 2026 |
| Date of Accepted | 10 May 2026 |
| Indian J Pharm Sci 2026;88(1):61-65 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
The present study evaluated whether clarithromycin modifies the glucose-lowering response of gliclazide in experimental animal models. Studies were conducted in normal rats, alloxan-induced diabetic rats, and normal rabbits using a period-wise crossover design that included gliclazide alone, clarithromycin alone, and their combination following either single-dose or multiple-dose clarithromycin pretreatment, with a 10 d washout between periods. Blood glucose levels were measured at predefined time points using the glucose-oxidase-peroxidase method. Gliclazide produced a biphasic hypoglycaemic response in rats, with peak effects observed at 2 h and 8 h, whereas a single peak at approximately 3 h was observed in rabbits. Clarithromycin alone did not produce any consistent effect on blood glucose. In contrast, clarithromycin pretreatment significantly enhanced both the magnitude and duration of gliclazide-induced hypoglycaemia in rats and rabbits, with a more pronounced effect following multiple-dose pretreatment. These findings indicate a clinically relevant interaction, highlighting the importance of careful glucose monitoring when clarithromycin is prescribed in patients receiving gliclazide.
Keywords
Gliclazide, clarithromycin, pharmacodynamic interaction, hypoglycaemia, rats, rabbits
Type 2 diabetes mellitus is commonly managed with oral glucose-lowering medicines, and sulfonylureas continue to be widely used, particularly in resource-limited settings. Because infections occur frequently in patients with diabetes, antibiotics are often co-prescribed, increasing the likelihood of drug-drug interactions. In a large retrospective cohort of older adults receiving sulfonylureas, clarithromycin exposure was associated with a substantially increased risk of hypoglycaemia-related emergency visits or hospitalization compared with non-interacting antibiotics, underscoring its clinical relevance [1]. Similar population-based studies have also reported an increased hypoglycaemic risk when clarithromycin is combined with sulfonylureas or other glucose-lowering agents [2, 3].
Several case reports have described severe or refractory hypoglycaemia following co-administration of clarithromycin with sulfonylureas such as glipizide and glyburide, suggesting a class-related interaction [4, 5]. Clarithromycin is a time-dependent inhibitor of CYP3A, with inhibition evident in both intestinal and hepatic metabolism after repeated dosing [6]. Gliclazide undergoes oxidative metabolism primarily via CYP2C9, with a secondary contribution from CYP2C19 [7]. Although clarithromycin is not a potent CYP2C9 inhibitor, macrolide antibiotics may influence drug response through multiple mechanisms, including effects on drug-metabolizing enzymes, transporters and plasma protein binding [1-6].
Despite clinical signals suggesting an interaction, controlled experimental data evaluating the effect of clarithromycin and gliclazide-induced hypoglycaemia remain limited. Therefore, the present study was designed to assess the influence of single-dose and multiple-dose clarithromycin pretreatment on the pharmacodynamic response of gliclazide in rats and rabbits.
Gliclazide and clarithromycin were provided as gift samples from Dr. Reddy?s Laboratories (India). Alloxan monohydrate was procured from SD Fine-chemicals Ltd., (India). Commercial glucose estimation kits based on Glucose Oxidase-Peroxidase (GOD-POD) principle were used throughout the study.
Albino rats (160-250 g) and albino rabbits (1.45-1.65 kg) of either sex were used. Animals were housed under standard laboratory conditions with free access to pellet diet and water, except during fasting periods. Prior to each dosing period, animals were fasted for 18 h with free access to water. The study protocol was approved by the Institutional Animal Ethics Committee and conducted in accordance with CCSEA guidelines (approval no.: CCSEA/IAEC/JLS/24/11/25/021).
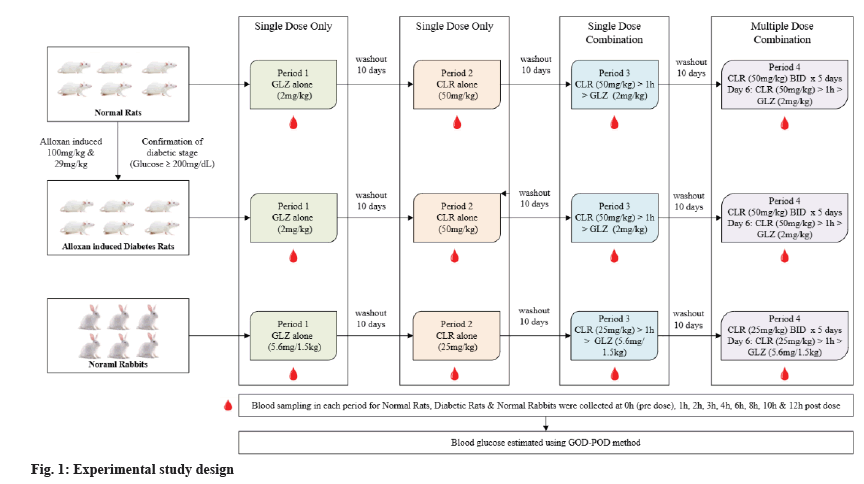
Gliclazide was administered orally at a dose of 2 mg/kg in rats and 5.6 mg/1.5 kg body weight in rabbits. These doses were selected based on previous studies demonstrating a consistent hypoglycaemic response without producing severe hypoglycaemia [8]. Clarithromycin was administered orally at 50 mg/kg in rats and 25 mg/kg in rabbits, corresponding to body-surface-area-scaled equivalents of clinically used therapeutic doses (500 mg twice daily in humans) [9]. Two clarithromycin pretreatment conditions were evaluated: Single-dose pretreatment (interaction day: clarithromycin given 60 min before Gliclazide) and multiple-dose pretreatment (clarithromycin twice daily (12 h apart) for 5 d; on d 6, clarithromycin given 60 min before Gliclazide).
A period-wise crossover design was employed using six animals per species (n=6). Each animal received the following treatments across four study periods: Period 1: Gliclazide alone; Period 2: Clarithromycin alone; Period 3: Single-dose clarithromycin pretreatment followed by gliclazide and Period 4: Multiple-dose clarithromycin pretreatment followed by gliclazide.
A 10 d washout interval was maintained between successive treatment periods. The experimental protocol is illustrated in fig. 1.
After an overnight fast (16 h), diabetes was induced in rats by intraperitoneal administration of alloxan monohydrate (100 mg/kg initially, followed by 29 mg/kg). Fasting blood glucose was measured 72 h after the final injection; and rats with glucose ?200 mg/dl were considered diabetic and included in the study.
In both normal and diabetic rats, pharmacodynamic evaluation was conducted during each treatment period following oral drug administration. During interaction periods (Periods 3 and 4), clarithromycin was administered 60 min prior to gliclazide. A similar period-wise pharmacodynamic evaluation was conducted in rabbits.
Blood samples were collected from the retro-orbital plexus in rats and from the marginal ear vein in rabbits at predefined time points. Blood glucose concentrations were estimated using a commercial GOD-POD enzymatic method, according to the manufacturer?s instructions.
Data were expressed as mean±standard error of the mean. Statistical comparisons were performed using a one-way Analysis of Variance (ANOVA). When significance was detected, Dunnett?s post-hoc method was performed to identify specific comparisons against the control group. A p-value<0.05 was considered statistically significant.
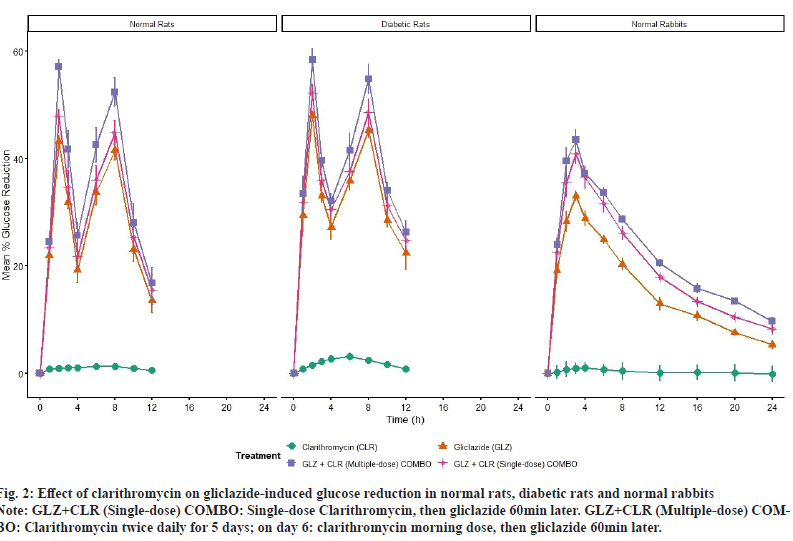
Gliclazide administered alone produced a reproducible biphasic hypoglycaemic response across species. In rats, the response was biphasic with peaks observed at 2 h and 8 h, whereas rabbits exhibited a single peak effect at approximately 3 h. Clarithromycin alone did not produce any meaningful or consistent reduction in blood glucose levels.
Co-administration of clarithromycin with gliclazide significantly enhanced and prolonged the hypoglycaemic effect of gliclazide in normal rats, alloxan-induced diabetic rats and normal rabbits. The magnitude of potentiation was greater following multiple-dose clarithromycin pretreatment compared with single-dose pretreatment (fig. 2).
Fig. 2: Effect of clarithromycin on gliclazide-induced glucose reduction in normal rats, diabetic rats and normal rabbits
Note: GLZ+CLR (Single-dose) COMBO: Single-dose Clarithromycin, then gliclazide 60min later. GLZ+CLR (Multiple-dose) COMBO:
Clarithromycin twice daily for 5 days; on day 6: clarithromycin morning dose, then gliclazide 60min later.
The present study demonstrates that clarithromycin pretreatment enhances both the magnitude and duration of gliclazide-induced hypoglycaemia in rats and rabbits. The effect was consistently greater after multiple-dose pretreatment, supporting a time-dependent interaction. Since clarithromycin alone did not meaningfully affect blood glucose levels, the enhanced response observed during combination treatment is unlikely to reflect additive glucose-lowering effects.
Clinical evidence indicates that clarithromycin can precipitate clinically significant hypoglycaemia when used with sulfonylureas and other glucose-lowering agents [1-5]. Clarithromycin is known to produce time-dependent inhibition of CYP3A with repeated dosing [6], while gliclazide is primarily metabolized by CYP2C9, with additional involvement of CYP2C19 [7]. Although mechanistic evaluation was beyond the scope of the present study, the findings are consistent with altered gliclazide disposition and/or response following clarithromycin pretreatment.
A limitation of this study is the absence of gliclazide concentration and insulin measurements, which would have helped distinguish pharmacokinetic and pharmacodynamic contributions to the interaction. However, the primary objective was to establish a reproducible pharmacodynamic interaction signal under controlled experimental conditions. Caution is warranted when extrapolating these findings to clinical settings, though the consistency of the results across multiple animal models provides a strong preclinical signal.
In conclusion, clarithromycin pretreatment potentiated and prolonged the hypoglycaemic response to gliclazide in rats and rabbits, with a greater effect observed following multiple dose pretreatment. These findings supports the need for closer glucose monitoring when clarithromycin is prescribed to patients receiving gliclazide.
Acknowledgements:
The authors thank R. RajBharath for assistance with animal experimentation.
Conflict of interests:
The authors declared no conflict of interests.
References
- Parekh TM, Raji M, Lin YL, Tan A, Kuo YF, Goodwin JS. Hypoglycemia after antimicrobial drug prescription for older patients using sulfonylureas. JAMA Inter Med 2014;174(10):1605-12.
[Crossref] [Google Scholar] [PubMed]
- Kennedy KE, Teng C, Patek TM, Frei CR. Hypoglycemia associated with antibiotics alone and in combination with sulfonylureas and meglitinides: an epidemiologic surveillance study of the FDA adverse event reporting system (FAERS). Drug Saf 2020;43(4):363-9.
[Crossref] [Google Scholar] [PubMed]
- Wang YC, Chen YT, Kuo SC, Chen TL, Chang FY. Rapid hypoglycemia onset associated with antimicrobial use in patients with diabetes: A nationwide population-based case-crossover study. Eur J Inter Med. 2016;34:e14-5.
[Crossref] [Google Scholar] [PubMed]
- Ruan XC, Tan PY, Tan Y. Clarithromycin and glipizide drug-drug interaction leading to refractory hypoglycemia. Cureus 2019;11(6):e4800.
- Leiba A, Leibowitz A, Grossman E. An unusual case of hypoglycemia in a diabetic patient. Ann Emer Med. 2004;44(4):427-8.
[Crossref] [Google Scholar] [PubMed]
- Quinney SK, Malireddy SR, Vuppalanchi R, Hamman MA, Chalasani N, Gorski JC, et al. Rate of onset of inhibition of gut-wall and hepatic CYP3A by clarithromycin. Eur J Clin Pharmacol 2013;69(3):439-48.
[Crossref] [Google Scholar] [PubMed]
- Elliot DJ, Suharjono N, Lewis BC, Gillam EM, Birkett DJ, Gross AS, et al. Identification of the human cytochromes P450 catalysing the rate?limiting pathways of gliclazide elimination. Br J Clin Pharmacol 2007;64(4):450-7.
[Crossref] [Google Scholar] [PubMed]
- Raju MG, Satyanarayana S, Kumar E. Improved efficacy of gliclazide with polyherbal formulation on pharmacodynamic and pharmacokinetic activity in normal rabbits. Mortality 2015;1(2):123-9.
- BIAXIN® (clarithromycin) prescribing information. United States (US) Food and Drug Administration; 2017.