- *Corresponding Author:
- Qiyin Dong
Department of Gynecology, Department of Reproductive Medicine, Huzhou Maternity and Child Care Hospital, Wuxing, Zhejiang 313000, China
E-mail: wangfang_hzyy@163.com
| This article was originally published in a special issue, “Current Trends in Pharmaceutical and Biomedical Sciences” |
| Indian J Pharm Sci 2022:84(5) Spl Issue “272-277” |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
The fundamental target of the present survey is to explore the clinical impact of resveratrol in treating polycystic ovary syndrome. A number of 104 cases suffering from polycystic ovary syndrome admitted to the hospital from January 2018 to December 2020 were chosen as investigation subjects. The cases were categorized into two groups, namely control and study groups through the random number table technique, with 52 patients in each group. Patients in the group of study were considered to receive 100 % pure resveratrol (once daily, 1000 mg) and cases in the group of control received matched placebo. 3 mo later, the treatment effects of the two groups were scrutinized. Following the treating process, the length of ovary, endometrial thickness and number of sinus follicles in the research group were improved and substantially superior to those in the group of control (all p<0.05). The improvement in menstrual cycle and hair loss in the group of observation was considerably greater than that in the group of control (both p<0.05). Following the treatment, the levels of luteinizing hormone, estradiol and testosterone in the group of observation were less than those in the group of control, with statistically meaningful discrepancies (all p<0.05), but the follicle-stimulating hormone levels had no significant difference. After 3 mo of treating, the expression level of mitofusin-2 in the study group was substantially enhanced in comparison to that in the control group (p<0.05). Resveratrol improved sex hormone levels, menstrual irregularities, hair loss and ovarian function in cases suffering from polycystic ovary syndrome.
Keywords
Resveratrol, polycystic ovary syndrome, hyperandrogenemia, hirsutism, insulin
Polycystic Ovarian Syndrome (PCOS) is a pervasive disease resulted from metabolic and endocrine disorders in females of reproductive years. The patients are mainly identified by hyperandrogenemia and chronic anovulation and the main clinical manifestations are irregular menstrual cycle, infertility and hirsutism, with the reproductive function of patients seriously affected[1,2]. At present, the pathogenesis of PCOS has not been meticulously defined clinically, but it is believed to be related to insulin resistance and hyperandrogenemia[3], which results in the decline of ovarian reserve function. At present, clomiphene is commonly used in Western medicine to promote ovulation, Diane 35 is used to reduce androgen level and metformin is used to improve insulin resistance; by these intervenes, pregnancy rate can be improved in a short term, but there are disadvantages such as severe adverse reactions and long treatment cycle[4,5]. Therefore, it is particularly important to find safe and effective therapies with less adverse reactions.
Resveratrol (RSV), a polyphenolic antioxidant discovered in many plants, has been the focus of recent investigations due to its life-extending effect in budding yeast. Multiple studies have shown that RSV helps prevent cardiovascular disorders[6], cancers[7] and age-related deteriorations[8]. Although, only a limited number of investigations have been earmarked to the advantageous influences of RSV on reproduction to date. A recent investigation on middle-aged mice showed that short-term injection of RSV can successfully improve the fragmentation and death of aging oocytes in oviduct induced through oxidative stress[9], suggesting that RSV has an advantageous task in the reproductive health of elderly women. In addition, results of a study evaluating the effects of the treatment of RSV on proinflammatory and endoplasmic reticulum stress markers in PCOS cases showed that RSV has anti-inflammatory effects by inhibiting Nuclear Factor Kappa B (NF-κB) and gene products regulated by NF-κB. In another aspect, RSV can regulate endoplasmic reticulum stress in Granulosa Cells (GCs) via modifying gene expressions participated in Unfolding Protein Response (UPR)[10].
Despite the fact that there exist some beneficial outcomes, information from randomized assessments of adequate duration and size to appraise the influences of RSV on clinical outcomes in patients with PCOS are insufficient and ineffective. Therefore, the main objective of the current research was to explore the clinical effect of RSV on patients with PCOS. To provide a new treatment plan and data support for the clinical treatment of PCOS patients.
Materials and Methods
Clinical data:
A number of 104 PCOS cases admitted to our hospital from January 2018 to December 2020 were chosen as the exploration objects.
Inclusion criteria: The patients should meet PCOS related criteria established at the 2003 Dutch PCOS Conference, comprising at least two of the following three clinical achievements; oligomenorrhea or amenorrhea, chemical or clinical testosterone >0.75 ng/ml, hyperandrogenemia and/or ultrasound Polycystic Ovarian Morphology (PCOM)[11]; aged between menarche and 40 y old; have a spouse with good reproductive function and normal semen quality in routine examination; have at least one unobstructed oviduct in hysterosalpingography/laparoscopic examination; received no other treatment 3 mo before enrollment; voluntary to receive drug treatment, convenient for follow-up and, be informed and consent to the research.
Exclusion criteria: Those who do not satisfy the diagnostic criteria; those with gonad hypoplasia, abnormal menstruation (caused by reproductive tract malformation or organic lesions of reproductive organs) and irregular menstrual cycle; those who have taken sex hormone drugs in the last 3 mo before the study; those with diabetes, liver and kidney diseases, thyroid dysfunction, hyperprolactinemia and other diseases; those have mental illness and cannot cooperate with the researcher normally. The present exploration was confirmed through the ethics Committee of Hospital, with informed consent of patients and their families. Using the random number table technique, the cases were categorized into two groups, namely study and control groups, with 52 patients in each group. There were no statistical significances in course of disease, age, body mass index and other general information between the two groups (all p>0.05), exhibiting comparability as shown in Table 1.
| Group | Age/year | Course/year | BMI/(kg/m2) |
|---|---|---|---|
| Study group | 28.98±4.25 | 6.73±4.43 | 24.92±1.56 |
| Control group | 27.12±5.32 | 5.82±4.28 | 24.86±1.62 |
| t | 1.97 | 1.065 | 0.192 |
| p | 0.052 | 0.289 | 0.848 |
Table 1: Comparison of Clinical Outcomes between the Two Groups (x̄±s, n=52)
Therapy:
Throughout the study period (3 mo), study group patients were considered to receive 100 % pure RSV (once daily, 1000 mg) and control group to receive matched placebo.
Observation indexes and detection methods:
Color Doppler ultrasound: Before treatment and on the 10th d after treatment, the length of ovary, endometrial thickness and number of sinus follicles were measured by transvaginal color Doppler ultrasonography.
Comparison of clinical symptoms: Measurements of acne, hirsutism, hair loss and menstrual frequencies were taken in the morning during the investigation visit. Acne and hirsutism and were assessed using the acne score and Ferriman Gallwey technique[12,13].
Comparison of sex hormone levels: Pre and post-treatment process, 5 ml of venous blood in fasting state was extracted from the two groups and centrifuged for 10 min at 3500 r/min. The upper serum was isolated. Enzyme linked immunosorbent assessment was employed to ascertain Luteinizing Hormone (LH), Testosterone (T), Estradiol (E2), Follicle-Stimulating Hormone (FSH) levels strictly according to kit instructions.
Statistical study:
GraphPad Prism 8.0 statistical analysis computer program was employed for statistical assessment. Quantitative outcomes were represented as mean±standard deviation (x̄±s) and compared by t-test. Qualitative outcomes were represented as the number of cases (%) and compared by Chi-square (χ2) test. The test level ɑ was set as 0.05 unless otherwise specified.
Results and Discussion
Before treatment, there were no substantial discrepancies in ovarian length diameter, endometrial thickness and number of sinus follicles between 2 groups (all p>0.05). Following the treatment, the length of ovary, endometrial thickness and number of sinus follicles in the group of study were improved and were remarkably better than those in the group of control (all p<0.05) as shown in Table 2.
| Group | Ovarian long diameter (mm) | Endometrial thickness (mm) | Number of sinus follicles | |||
|---|---|---|---|---|---|---|
| Before treatment | After treatment | Before treatment | After treatment | Before treatment | After treatment | |
| Study group | 17.05±2.41 | 21.65±2.50* | 3.70±0.22 | 5.25±0.36* | 3.01±0.48 | 5.56±1.15* |
| Control group | 17.21±2.43 | 18.01±2.46 | 3.72±0.32 | 3.75±0.32 | 3.05±0.47 | 3.01±0.49 |
| t | 0.337 | 7.484 | 0.371 | 22.46 | 0.429 | 14.71 |
| p | 0.737 | <0.0001 | 0.711 | <0.0001 | 0.669 | <0.0001 |
Table 2: Comparison Of Color Ultrasound Examination Achievements Between The Two Groups Pre And Post-Treatment (x̄±s, n=52)
After treatment, acne score (p=0.26) and hirsutism (p=0.44) did not change considerably. Following 3 mo of treatment, 39 cases in the RSV group (75 %) and 25 cases in the group of placebo (48.1 %) had normal menstrual cycle length, with significant differences (p=0.005). Hair loss was observed in 16 patients (30.1 %) in the RSV group and 35 (67.3 %) in the placebo group, with a statistically significant difference (p<0.001) as shown in Table 3.
| Group | Acne score | Ferriman Gallwey score | Number of cases with hair loss | Number of cases with normal menstrual cycle | ||||
|---|---|---|---|---|---|---|---|---|
| Before treatment | After treatment | Before treatment | After treatment | Before treatment | After treatment | Before treatment | After treatment | |
| Study group | 1.36±0.2 | 1.06±0.2 | 20.08±1.2 | 18.02±1.2 | 41 | 16 | 11 | 39 |
| Control group | 1.32±0.18 | 1.10±0.16 | 18.84±1.3 | 18.21±1.3 | 31 | 35 | 12 | 25 |
| t | 1.126 | 0.774 | ||||||
| p | 0.26 | 0.44 | <0.001 | 0.005 | ||||
| χ2 | 13.89 | 7.96 | ||||||
Table 3: Comparison of Clinical Symptoms between the Two Groups Pre and Post-Treatment (n=52)
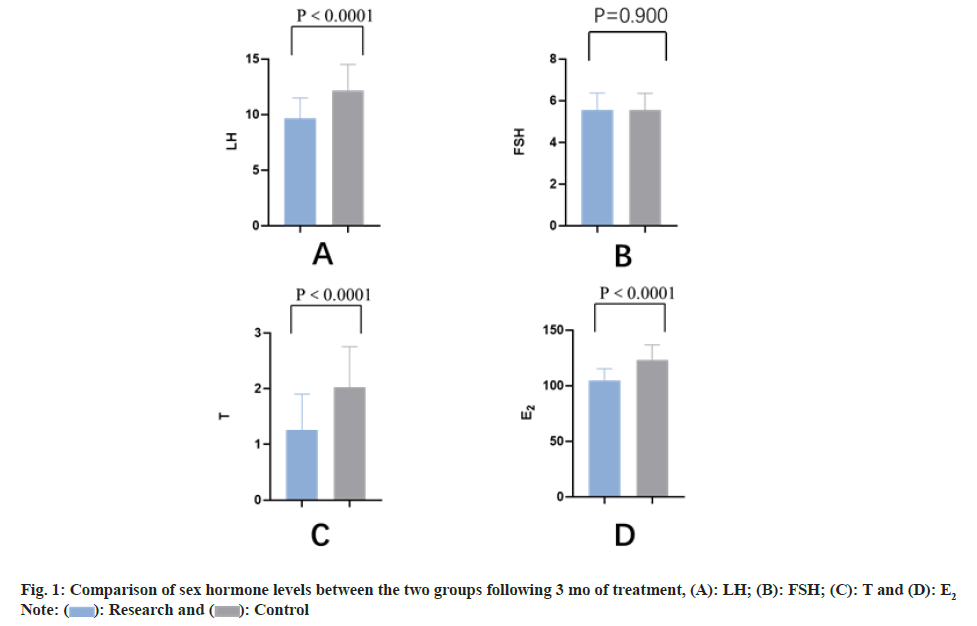
There existed no statistical discrepancy in blood glucose level between 2 groups prior to the treatment (p>0.05). Following 1 mo of treatment, fasting blood glucose, 2 h postprandial blood glucose and hemoglobin A1C levels in both groups, namely study and control were notably reduced than those prior to the treatment (all p<0.05) and the index levels in the study group were less than those in the control group (all p<0.05) as shown in Table 4 and fig. 1.
| Group | LH (IU/L) | FSH (IU/L) | T( nmol/l) | E2 (pg/ml) | ||||
|---|---|---|---|---|---|---|---|---|
| Before treatment | After treatment | Before treatment | After treatment | Before treatment | After treatment | Before treatment | After treatment | |
| Study group | 17.08±2.53 | 9.64±1.85* | 5.64±0.78 | 5.56±0.81 | 2.88±0.76 | 1.26±0.64* | 130.05±18.99 | 104.80±10.46* |
| Control group | 17.02±2.44 | 12.16±2.35* | 5.61±0.80 | 5.54±0.81 | 2.83±0.73 | 2.03±0.72 | 131.89±18.91 | 123.04±13.60 |
| t | 0.123 | 6.076 | 0.194 | 0.126 | 1.101 | 5.764 | 0.495 | 7.666 |
| P | 0.902 | <0.0001 | 0.847 | 0.9 | 0.274 | <0.0001 | 0.622 | <0.0001 |
Note: Compared with before treatment, *p<0.05
Table 4: Comparison of Sex Hormone Levels between the Two Groups Pre and Post-Treatment (x̄±s, n=52)
PCOS is the most pervasive endocrine disorder influencing females of child-bearing years, characterized by hyperandrogenemia and ovulation dysfunction. PCOS can be caused by heredity, hypothalamic-pituitary-ovarian axis dysfunction and other factors and its induction mechanism is mainly as follows; the binding of gonadotropin-releasing hormone and the receptor on the pituitary membrane will activate enzymes related to LH synthesis and secretion and increase LH synthesis; when LH level is too high, it will promote the hyperplastic changes of ovarian interstitial cells and follicular membrane cells[14], resulting in the increase of androgen secretion and gonadotropin sensitivity. The estrogen secreted by a large number of ovarian follicles can inhibit the apoptosis of follicles, affect the normal advancement of sinus follicles and promote the formation of polycystic ovary. PCOS is an important factor leading to menstrual disorder and infertility in women of childbearing age. Effective treatment of PCOS is of substantial significance to ameliorate the prognosis of studied cases.
The achievements of the current research illuminated that the levels of LH, E2 and T in the grop of study were lower than those in the group of control after treating and the differences were statistically meaningful (all p<0.05). There existed no substantial discrepancy in FSH level. This indicated that RSV treatment is able to ameliorate the level of sex hormones in patients. Ovarian androgens (mainly androstenedione and some testosterone) react to LH in follicular membrane cells and diffuse to GCs through the basal membrane. Under the control of FSH, GCs induce the expression of aromatase, which converts androgens to estrogen[15]. According to reports, RSV has been illustrated to hinder the proliferation of follicular membrane interstitial cells, boost the growth of ovarian GCs, and reduce the messenger RNA expression of CYP17A1 in rat follicular membrane interstitial cells via inhibiting the signaling pathway of Protein Kinase B (AKT)/ Phosphatidylinositol 3-Kinase (PI3K)[16]. Therefore, RSV may prerelease excessive intraovarian androgens by reducing the volume of follicular membrane cells and the sensitivity of follicular membrane cells to LH stimulation and causing the proliferation of GCs, leading to normal ovulation of PCOS patients[17].
After treatment, the length of ovary, endometrial thickness and number of sinus follicles were improved in the group of study and the indexes were remarkably better than those in the group of control (all p<0.05). The degree of improvement of menstrual cycle and hair loss in the group of observation was substantially greater than that in the group of control (both p<0.05). These results illuminated that RSV treatment can improve hyperandrogenemia in PCOS patients. The effects of RSV on menstrual regularity have not been fully studied. But the achieved findings were in agreement with those of Benrick et al.[18]. They appraised the role of RSV in a rat model of androgen-induced PCOS, showing that substantial enhancement in menstrual cycles was detected within 5-6 w of treating. RSV intake reducing hair loss makes biological sense. RSV intake is biologically plausible in reducing hair loss and this effect may be mediated by several biological mechanisms, including inhibition of 5 alpha (α)-reductase activity[19] and reduction of serum deoxytestosterone levels[20].
Mitochondrial fusion proteins are a class of guanosine triphosphate enzymes placed in the outer membrane of mitochondria and they directly affect mitochondrial function by promoting mitochondrial fusion. There are two subtypes of Mitofusin (Mfn)-1 and Mfn2. Mfn2 is involved in maintaining insulin sensitivity in multiple tissues and organs and may be involved in regulating insulin resistance in PCOS patients. In addition, Mfn2 and PI3K-Aktmammalian target of rapamycin pathway showed co-low expression in the rat PCOS model[21], suggesting that follicular dysplasia in PCOS may be associated with Mfn low expression. The findings of this exploration suggested that as a mitochondrial nutritional drug, RSV may improve ovarian function in patients with PCOS.
Taken together, this study provided some preliminary evidence that RSV can improve sex hormone levels, menstrual irregularities, hair loss, as well as ovarian function in patients with PCOS.
Author’s contributions:
Fang Wang and Yinyuan Zheng contributed equally to this work.
Conflict of interests:
The authors declared no conflict of interests.
References
- Abiad F, Khalife D, Safadi B, Alami R, Awwad J, Khalifeh F, et al. The effect of bariatric surgery on inflammatory markers in women with polycystic ovarian syndrome. Diabetes Metab Syndr 2018;12(6):999-1005.
[Crossref] [Google Scholar] [PubMed]
- Maas K, Mirabal S, Penzias A, Sweetnam PM, Eggan KC, Sakkas D. Hippo signaling in the ovary and polycystic ovarian syndrome. J Assist Reprod Genet 2018;35(10):1763-71.
[Crossref] [Google Scholar] [PubMed]
- Wang XL.Effect of Daying-35 Combined with ovulation-promoting therapy on infertility of polycystic ovary syndrome on its ovarian function. World J Complex Med 2020;6(6):175-7.
- Zhang ML, Yu CB, Luo XM. Effect of self-made ovulation induction formula and Diane-35 on polycystic ovary syndrome. Pract Clin J Integr Tradit Chin Western Med 2019;19(9):83-5.
- Kumar Y, Kotwal N, Singh Y, Upreti V, Somani S, Kumar KH. A randomized, controlled trial comparing the metformin, oral contraceptive pills and their combination in patients with polycystic ovarian syndrome. J Family Med Prim Care 2018;7(3):551-6.
[Crossref] [Google Scholar] [PubMed]
- Johnson WD, Morrissey RL, Usborne AL, Kapetanovic I, Crowell JA, Muzzio M, et al. Subchronic oral toxicity and cardiovascular safety pharmacology studies of resveratrol, a naturally occurring polyphenol with cancer preventive activity. Food Chem Toxicol 2011;49(12):3319-27.
[Crossref] [Google Scholar] [PubMed]
- Jang M, Cai L, Udeani GO, Slowing KV, Thomas CF, Beecher CW, et al. Cancer chemopreventive activity of resveratrol, a natural product derived from grapes. Science 1997;275(5297):218-20.
[Crossref] [Google Scholar] [PubMed]
- Baxter RA. Anti-aging properties of resveratrol: Review and report of a potent new antioxidant skin care formulation. J Cosmetic Dermatol 2008;7(1):2-7.
[Crossref] [Google Scholar] [PubMed]
- Liang QX, Lin YH, Zhang CH, Sun HM, Zhou L, Schatten H, et al. Resveratrol increases resistance of mouse oocytes to postovulatory aging in vivo. Aging (Albany NY) 2018;10(7):1586-96.
[Crossref] [Google Scholar] [PubMed]
- Brenjian S, Moini A, Yamini N, Kashani L, Faridmojtahedi M, Bahramrezaie M, et al. Resveratrol treatment in patients with polycystic ovary syndrome decreased pro-inflammatory and endoplasmic reticulum stress markers. Am J Reprod Immunol 2020;83(1):e13186.
[Crossref] [Google Scholar] [PubMed]
- Rotterdam ESHRE/ASRM-Sponsored PCOS consensus workshop group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum Reprod 2004;19(1):41-7.
[Crossref] [Google Scholar] [PubMed]
- Al Kindi MK, Al Essry FS, Al Essry FS, Mula-Abed WA. Validity of serum testosterone, free androgen index and calculated free testosterone in women with suspected hyperandrogenism. Oman Med J 2012;27(6):471.
[Crossref] [Google Scholar] [PubMed]
- Kolodziejczyk B, Duleba AJ, Spaczynski RZ, Pawelczyk L. Metformin therapy decreases hyperandrogenism and hyperinsulinemia in women with polycystic ovary syndrome. Fertil Steril 2000;73(6):1149-54.
[Crossref] [Google Scholar] [PubMed]
- Shi YQ. Effect of compound Xuanju capsule combined with Diane-35 on sex hormone, follicle number, ovulation and pregnancy in patients with polycystic ovary syndrome. Chin J Mod Drug Appl 2020;14(19):213-5.
- Franks S, Hardy K. Androgen action in the ovary. Front Endocrinol 2018;9:452.
[Crossref] [Google Scholar] [PubMed]
- Ortega I, Villanueva JA, Wong DH, Cress AB, Sokalska A, Stanley SD, et al. Resveratrol reduces steroidogenesis in rat ovarian theca-interstitial cells: The role of inhibition of Akt/PKB signaling pathway. Endocrinology 2012;153(8):4019-29.
[Crossref] [Google Scholar] [PubMed]
- Alpañés M, Fernández-Durán E, Escobar-Morreale HF. Androgens and polycystic ovary syndrome. Exp Rev Endocrinol Metab 2012;7(1):91-102.
- Benrick A, Maliqueo M, Miao S, Villanueva JA, Feng Y, Ohlsson C, et al. Resveratrol is not as effective as physical exercise for improving reproductive and metabolic functions in rats with dihydrotestosterone-induced polycystic ovary syndrome. Evid Based Complement Alternat Med 2013;2013.
[Crossref] [Google Scholar] [PubMed]
- Hirano Y, Kondo R, Sakai K. 5α-Reductase inhibitory compounds produced by polymerization of resveratrol with horseradish peroxidase. J Wood Sci 2002;48(1):64-8.
- Rahman Siddique H, Nanda S, Parray A, Saleem M. Androgen receptor in human health: A potential therapeutic target. Curr Drug Targets 2012;13(14):1907-16.
[Crossref] [Google Scholar] [PubMed]
- Luo Q, Huo P, Wang L, Wu X. The influencing mechanism of mTOR signal pathway mediated by mitofusin-2 in development of follicle. Eur Rev Med Pharmacol Sci 2018;22(8):2212-7.
[Crossref] [Google Scholar] [PubMed]
 : Research and
: Research and  : Control
: Control



