- *Corresponding Author:
- V. M. Berlin Grace
Department of Biotechnology, Karunya Institute of Technology and Sciences, Coimbatore-641 114, India
E-mail: berlinbiochem@gmail.com
| Date of Submission | 27 March 2017 |
| Date of Revision | 23 March 2018 |
| Date of Acceptance | 24 September 2018 |
| Indian J Pharm Sci 2018;80(6):1039-1044 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
The main objective of the present work was to develop liposomal nano-formulation for 9-cis-retinoic acid using di-stearoylphosphocholin/cholesterol mixture, to characterise and to evaluate its anticancer effect on A549 human lung cancer cell lines. The liposomes were prepared using thin film formulation method and characterization of particle size and shape were carried out employing dynamic light scattering and scanning electron microscopy techniques, respectively. The level of drug entrapment into the liposomes and liposomal stability were analysed using spectrophotometry and expressed in terms of percent entrapment. The level of 9-cis-retinoic acid in treated cells also was assayed using spectrophotometry. In vitro drug release level evaluated using a dialysis bag. The anticancer effect was studied using MTT and trypsin blue assays in human lung cancer cell line. The drug entrapment level achieved was 83.33 %. Viability of cancer cells was significantly reduced after liposomal 9-cis-retinoic acid treatment. From these results it could be concluded that the liposomal 9-cis-retinoic acid was easily taken up by the A549 cells compared to free 9-cis-retinoic acid, which might have enhanced the anticancer activity observed in this study.
Keywords
9-cis-Retinoic acid, liposomes, lung cancer cell lines, SEM, DLS, anticancer activity
Lung cancer being one of the leading causes of cancer deaths in the US [1] and worldwide, need special attention pertaining to development of new therapeutic regimens. Targeted drug therapy using liposomes is an intense research area in recent years for cancers having complex molecular changes during carcinogenesis. A very common human lung cancer cell line, A549, established from lung adenocarcinoma of epithelial origin in 1972 is being used for most of the anticancer and genotoxic studies for lung cancer [2-4]. 9-cis-Retinoic acid (9-cis-RA) binds and activates both its receptors retinoid X receptor (RXR) and retinoic acid receptor (RAR) [5] for producing molecular actions, among the both it binds RXRs with greater affinity [6]. Many in vivo and in vitro assays on tumour cell proliferation, differentiation and apoptosis have proven the 9-cis-RA as a promising therapeutic agent [7]. It is synthesized and approved as a potential therapeutic agent by the USFDA to treat Kaposi sarcoma [8]. But still its clinical use is under trials due to limiting challenges such as instability, toxic side effects, chirality (isomerization), targeted reach, poor pharmacokinetics, and development of drug resistance on prolonged use [9]. It was reported that, among the isomers of retinoid, 9-cis-RA is least prevalent and does not accumulate over time in body as it is metabolized rapidly [10].
Liposomal drug delivery systems are found to be effective for a variety of drugs with such limitations through sustained drug release, prevention of exposure toxicity and enhanced drug stability [11]. This investigations aim was to formulate a liposomal 9-cis- RA delivery system using 1,2-distearoyl-sn-glycero- 3-phosphocholine (DSPC) and cholesterol as lipid core for the treatment of lung cancer. Characteristics of this liposomal 9-cis-RA preparation such as size, morphology, stability and in vitro drug release property evaluated. Percent encapsulation and retinoid level in cell lines also were measured. The anticancer effect of liposomal 9-cis-RA formulation on A549 cell line was evaluated using MTT and trypan blue assays and was compared with that of free 9-cis-RA.
Materials and Methods
Development of the liposomal 9-cis-RA formulation
The liposomal 9-cis-RA formulation was prepared according to a reported method with slight modification [12] using a mixture of DSPC, cholesterol and synthetic 9-cis-RA (50:40:10 molar ratio) in chloroform in a rotary evaporator, subjected to vacuum drying, desiccation and then hydration of dry film with PBS (pH=7.4). The developed lipo-9-cis-RA solution was then subjected to probe sonication at 70 W for 30 s. The unentrapped 9-cis-RA precipitated upon centrifugation was removed.
Estimation of the percent drug encapsulation
For estimating the percent entrapment of 9-cis-RA into the liposomes, liposomal solution was centrifuged in an ultracentrifuge and the unentrapped 9-cis-RA level in the supernatant was estimated on a spectrophotometer at 360 nm. The pellets were subjected to lysis in absolute alcohol in a bath sonicator since 9-cis-RA only dissolves in ethanol. The clear filtrate was then subjected to spectrophotometric analysis at 360 nm to measure the entrapped 9-cis-RA level. The percent entrapment of 9-cis-RA was calculated using the Eqn., percent entrapment (%) = (concentration of test 9-cis- RA/total concentration of 9-cis-RA)×100.
Particle size and morphology analysis
The Zetasizer Nano was used to measure the random size of the developed lipo-9-cis-RA in triplicate preparations. Briefly, 1 μm of lipo-9-cis-RA and bare liposomes were subjected to dynamic light scattering (DLS) analysis. The morphological and average particle size of the prepared encapsulated 9-cis-RA formulation was investigated in a scanning electron microscope (SEM) after gold coating the lyophilized samples of bare liposomes and 9-cis-RA-entrapped liposomes.
Stability analysis
The developed lipo-9-cis-RA formulation was divided into 3 portions and preserved in a sealed amber vial for 60 d in a refrigerator (4-8°), at room temperature (25±2°) and in an incubator (37±2°) to determine stability at different temperatures. The entrapped 9-cis-RA content was assayed at 15 d intervals in the pellets obtained after ultracentrifugation of the samples following the method described under estimation of percent drug encapsulation [13].
9-cis-RA drug release study in vitro
Membrane dialysis method [14] was employed for the evaluation of 9-cis-RA release from the liposomal formulation. Briefly, dialysis bags (12 000-14 000 MW cut off) were washed and processed in PBS, pH=7.4 and 75 % ethyl alcohol. The bags were then filled with free 9-cis-RA and liposomal 9-cis-RA formulation separately, tagged and then immersed in to buffer solution with constant stirring (100 rpm) using a magnetic stirrer. Portions of buffer solution (2 ml) was removed periodically and tested for the level of 9-cis- RA released using a spectrophotometer at 360 nm and the percent drug release was calculated using the Eqn., percent drug release = (concentration of released 9-cis- RA/total concentration of 9-cis-RA )×100.
Anticancer activity estimation with MTT assay
The A549 lung cancer cell line was procured from the NCCS, Pune and maintained in DMEM media with 10 % serum and antibiotic under a 5 % CO2 atmosphere at 37°. The cytotoxicity of liposomal 9-cis-RA formulation was analysed on A549 cells using the MTT assay [15]. As per the standard protocol, 96 well plates were seeded with 5000 cells/well and incubated for 24 h. The cell lines were then treated with liposomal 9-cis-RA and free 9-cis-RA in 2 different groups at 1 μM concentration of 9-cis-RA and incubated for different time interval such as 24, 48 and 72 h [16]. The untreated and blank liposome-treated cells served as control groups. Four hour before completion of the reaction, 20 μl of MTT and 100 μl of DMSO was added to each well. OD values were measured at 570 nm using an ELISA reader. The percent cell viability was then calculated using following Eqn., percent cell line viability (%) = (OD of treated cell lines/OD of control cells)×100.
Cell viability study by trypan blue assay
A549 cells were treated with 1 μM 9-cis-RA and liposomal 9-cis-RA at different time intervals of 24, 48 and 72 h [17]. The control groups were untreated cancer cell lines and the blank liposome-treated cancer cells. After the treatment, cells were washed with PBS and trypsinised followed by centrifugation at 100 g for 10 min. Then cells were stained with 0.4 % trypan blue solution and counted using a haemocytometer. The viable cells did not take up the stain and remained unstained. Percent viable cells (%) = no. of viable cell/ sum of viable and dead cells×100.
Analysis of retinoid content in the cell line
The A549 cell lines were treated with 1 μM 9-cis-RA and liposomal 9-cis-RA and incubated at 37° in a CO2 incubator for 24 h. The cells were then homogenized with 250 μl of 10 % ascorbic acid and 250 μl of ethanol to extract retinoid. The tubes were vortexed for 1 min followed by addition of 2 ml of n-hexane and further vortexed. The samples were centrifuged at 2000 rpm for 10 min. The hexane layer was removed, evaporated to dryness. The residue was then dissolved in 250 μl of methanol and the retinoid level was estimated at 360 nm on a spectrophotometer. From the OD values, the concentration of retinoid was calculated using standard 9-cis-RA.
Statistical analysis
All data were expressed as mean±SD. The statistical analysis was done using one-way analysis of variance (ANOVA) followed by a Dunnett's test (using GraphPad InStat version 3.00; GraphPad Software, Inc., La Jolla, CA).
Results and Discussion
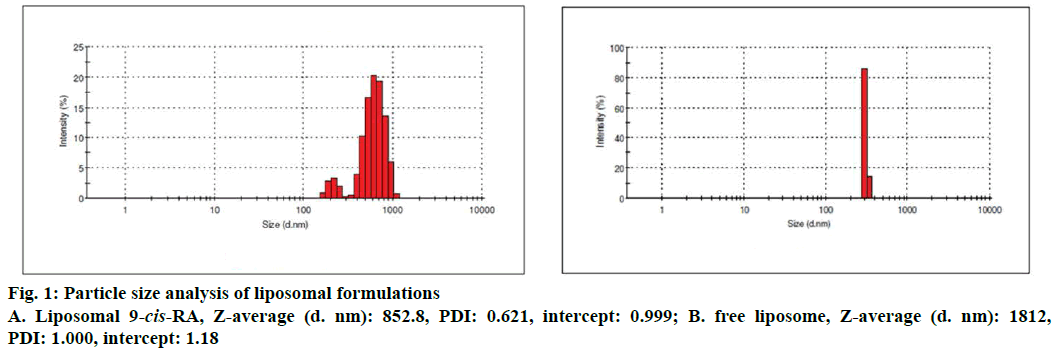
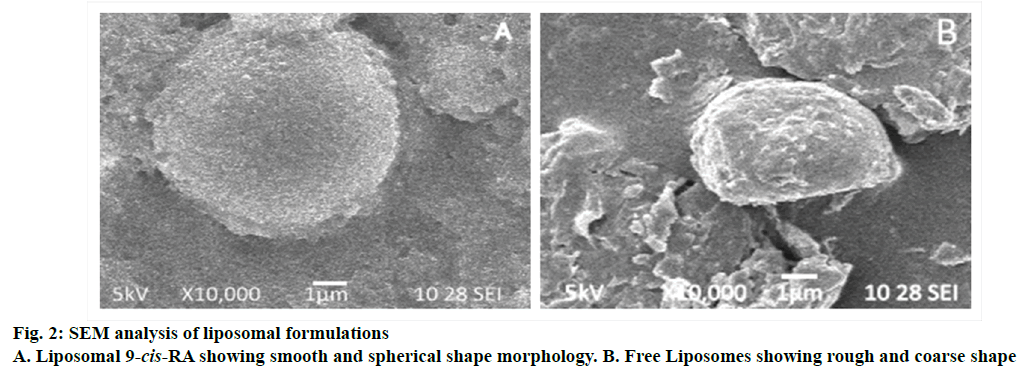
The entrapment efficiency of liposomes of 9-cis-RA was found to be 83.33 % at higher drug concentration. The particle size found in DLS analysis was 301 d nm for free liposome and 648.7 d nm for 9-cis-RAentrapped liposomes as shown in Figure 1. The average size and morphology of both bare and 9-cis-RA-entrapped liposomes are shown in SEM photomicrographs (Figure 2). The bare liposomes have shown a coarse sphere appearances whereas 9-cis-RA-entrapped liposomes appeared as smooth spheres.
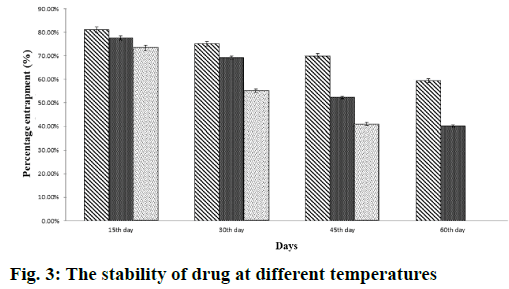
The stability of liposomal 9-cis-RA in terms of percent entrapped 9-cis-RA under different storage temperature at different time interval is shown in Figure 3. On observation, the percent entrapment of liposomal drug on 30 d of storage at 4° was found to be more stable, in comparison to the stability of the drug at different temperatures 25° and 37°, which degraded significantly. Complete degradation of liposomal 9-cis- RA occurred on 60th d at 37°. For the measurement of release rate of drug 9-cis-RA from liposome was done at 37° in PBS, and compared with free 9-cis-RA release. Over a period of nine hours the OD measurement was taken after every hour at 360 nm. The drug release rate is then expressed in terms of percent of 9-cis- RA released into the buffer through the dialysis bags as shown in Figure 4. The results shown a delayed and constant release of 9-cis-RA from liposome-9-cis-RA, where as a significantly rapid release were observed for free 9-cis-RA.
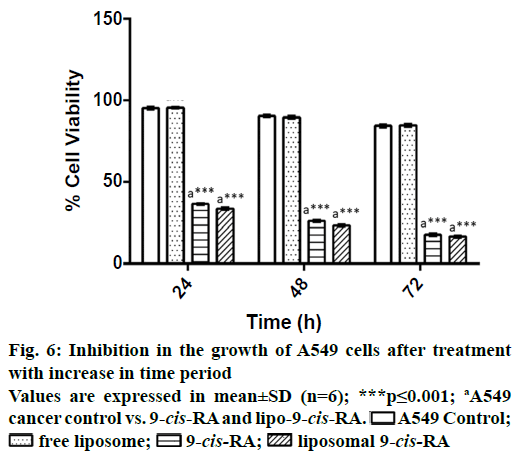
The toxicity of liposomal 9-cis-RA was quantitatively estimated by MTT assay (Figure 5), in which the formation of formazan dye depended on mitochondrial integrity. As a whole, the viability loss was found to be timedependent. At 72 h, 1 μM liposomal 9-cis-RA was found to be effective in inhibiting the cancer cells as indicated by the significantly less viability compared to the inhibition shown by free 9-cis-RA.
Viable cells excluded the trypan blue dye, whereas dead cells taken up the dye as an indication of cytotoxicity of 9-cis-RA. The percent viability was then calculated from the dead cell count and compared with the untreated control cell lines and between the treatments. Viability was found to be less in liposomal 9-cis-RA that is 18±2 % when compared with free 9-cis-RA (Figure 6).
The uptake of 9-cis-RA by the liposomal 9-cis-RAand free 9-cis-RA-treated A549 cell lines was assayed by measuring the absorbance of extracted retinoid at 360 nm. From the standard value for 9-cis-RA, the levels of 9-cis-RA present in the retinoid extracts of treated cell lines was calculated and were found to be 6.43±0.5033 μg/ml and 10.9±0.5291 μg/ml of 9-cis-RA, respectively. From this it was inferred that the uptake level of 9-cis-RA level was significantly more in liposomal 9-cis-RA-treated cancer cells when compared with that of in free 9-cis-RA-treated cells.
Lung cancer is known to be one of the most deathcausing disease in the world. There are different treatments available to treat lung cancer such as radiation therapy, chemotherapy, and surgery [18], in spite of which the lung cancer death rate is very high. This demand for newer strategies of treatments such as moleculartargeted therapy as the gene level control may respond better than the other methods of treatment. 9-cis-RA is a metabolite of vitamin A, grouped as retinoid said to have antimigration, antiinvasion properties and to inhibit the growth of tumor cell lines [19]. The therapeutic effect of another retinoid all-trans retinoic acid has already been studied extensively in different in vivo lung cancer mice models as well as on HeLa cell line and reported to have an efficient therapeutic effect [20-23]. 9-cis-RA regulates cell differentiation, and proliferation by binding to the RXR and also to the RAR, which forms the heterodimer complex and then binds with retinoic acid receptor responsive element [24].
Cancer treatment with suitable vehicle molecule is now gaining clinical acceptance and is under clinical trials for many cancer drugs, which are found to be ineffective when used in free form. Liposomes are gaining popular areas of research interest owing to the inherit, nature which includes high degree of biocompatibility, flexibility in their fabrication techniques and encapsulation of both the hydrophilic and hydrophobic drugs at large amount inside the vesicles [25]. Liposomes has drug retention control and increase cell uptake of drug by target cell fusion technique and liposome encapsulated drug has been found to be effective in inhibiting cancer with minimized side effects [26,27]. However liposomes prepared using different constituents have shown defects too in terms of charge, stability and targeted delivery. Several comparative studies have shown that the neutral liposome such as DSPC is a better choice for drug delivery as it induce very less immune response with reduced drug clearance, resulting in long term blood circulation [28]. Based on the literature, DSPC liposome was used in this study. In the present study, a DSPC/cholesterol liposomal 9-cis- RA nano-formulation was developed for better drug delivery to the lung cancer cells, thereby enhancing the therapeutic effect. Liposome encapsulated 9-cis- RA was prepared by incorporating 9-cis-RA into lipid emulsion. This liposomal formulation has the ability to alter the distribution characteristics and also enhances the inhibition of lung cancer. The liposomal 9-cis-RA developed in our study has shown good entrapment efficiency and was found to be in nano size with great stability and controlled drug release capacity compared with free form of 9-cis-RA. The previous reports suggested that the 9-cis-RA actively inhibits the cell proliferation, regulates cell differentiation and induce apoptosis in various types of cancers [29-31]. Especially in lung cancer, 9-cis- RA showed an efficient therapeutic effect as a combinatorial drug on in vitro and in vivo lung cancer models [32,33]. The in vitro anticancer study has shown a significant cytotoxicity as indicated by the percent inhibition on cancer cell growth. Choi and his research group used different concentration of 9-cis-RA to treat A549 cells and showed that 1 μM concentration of 9-cis-RA resulted in efficient growth inhibition [16]. Thus, we used 1 μM concentration of 9-cis-RA for treatment of lung cancer cell line in this study. Our results revealed that the lipo-9-cis-RA at this concentration has shown a potent growth inhibition than the free 9-cis-RA. This may be due to the reason that the uptake level of 9-cis-RA in cancer cells is more for lipo-9-cis-RA treatment when compared with free form treatment, which might have initiated the molecular induction within cells leading to cell death. There was no significant cytotoxicity observed in liposome alone-treated group compared with untreated cancer cells. However pre-clinical in vivo trials with such lipo-9-cis-RA are required to further validate its efficiency.
In this study A549 cell lines derived from a lung carcinoma is used to study cytotoxic effect. The liposomal 9-cis-RA controls the cell line growth more than the free 9-cis-RA does, by enhanced delivery of the 9-cis-RA in to the cells which is indicated by the uptake level of 9-cis-RA by the A549 cell line. The developed formulation of liposomal 9-cis-RA showed high stability, gradual drug release and efficient delivery to the target site. This showed that this formulation would be the effective drug to treat lung cancer, and further studies are needed to analyse the targeted molecular genes, which helps to suppress the cancer.
Acknowledgements
The authors would like to acknowledge the financial support of Karunya Institute of Technology and Sciences through the Karunya Short Term Research Grant and the Department of Science and Technology - Science and Engineering Research Board (DST-SERB) for their partial financial support. Authors would also like to acknowledge the valuable technical help of Dr. C. Vani, Assistant Professor, Karunya Institute of Technology and Sciences, Coimbatore and Department of Biotechnology, Karunya Institute of Technology and Sciences for providing laboratory and instrument facilities.
Conflict of interest
The authors declare that there was no conflict of interest in this research.
References
- Cardenal F, López-Cabrerizo MP, Antón A, Alberola V, Massuti B, Carrato A, et al. Randomized phase III study of gemcitabine-cisplatin versus etoposide-cisplatin in the treatment of locally advanced or metastatic non-small-cell lung cancer. J Clin Oncol 1999;17(1):12-8.
- Giard DJ, Aaronson SA, Todaro GJ, Arnstein P, Kersey JH, Dosik H, et al. In vitro cultivation of human tumors: establishment of cell lines derived from a series of solid tumors. J Natl Cancer Inst 1973;51:1417-23.
- Lee PJ, Alam J, Wiegand GW, Choi AM. Overexpression of heme oxygenase-1 in human pulmonary epithelial cells results in cell growth arrest and increased resistance to hyperoxia. Proc Natl Acad Sci USA 1996;93(19):10393-8.
- Foster KA, Oster CG, Mayer MM, Avery ML, Audus KL. Characterization of the A549 cell line as a type II pulmonary epithelial cell model for drug metabolism. Exp Cell Res 1998;243:359-66.
- Kane MA. Analysis occurrence and function of 9-cis retinoic acid. Biochim Biophys Acta 2012;1821(1):10-20.
- Heyman RA, Mangelsdorf DJ, Dyck JA, Stein RB, Eichele G, Evans RM, et al. 9-cis retinoic acid is a high affinity ligand for the retinoid X receptor. Cell 1992;68:397-406.
- Szabó DR, Baghy K, Szabó PM, Zsippai A, Marczell I, Nagy Z, et al. Antitumoral effects of 9‑cis retinoic acid in adrenocortical cancer. Cell Mol Life Sci 2015;71:917-32.
- Walmsley S, Northfelt DW, Melosky B, Conant M, Friedman-Kien AE, Wagner B. Treatment of AIDS-related cutaneous Kaposi’s sarcoma with topical alitretinoin (9-cis-retinoic acid) gel. Panretin Gel North American Study Group. J Acquir Immune Defic Syndr 1999;22:235-46.
- Retinoids. IARC Hand Books of Cancer Prevention. Vol. 4. Lyon, France: International Agency for Research On Cancer, WHO 1999. p. 187-207.
- Nagao A, Olson JA. Enzymatic formation of 9-cis, 13-cis and all-trans retinals from isomers of β-carotene. FASEB J 1994;8:968-73.
- Kawakami S, Wong J, Sato A, Hattori Y, Yamashita F, Hashida M. Biodistribution characteristics of mannosylated, fucosylated, and galactosylated liposomes in mice. Biochim Biophys Acta 2000;1524:258-65.
- Chansri N, Kawakami S, Yamashita F Hashida M. Inhibition of liver metastasis by all-trans retinoic acid incorporated into O/W emulsions in mice. Int J Pharm 2006;321:42-9.
- Begum MY, Shaik MR, Abbulu K, Sudhakar M. Ketorolac Tromethamine Loaded Liposomes of Long Alkyl Chain Lipids: Development, Characterization and in vitro Performance. Int J Pharm Tech Res 2012;4:218-25.
- Patil JS, Yadav AK, Mokale VJ, Naik JB. Development of surfactant free nanoparticles by a single emulsion high pressure homogenization technique and effect of formulation parameters on the drug entrapment and release. Int J Pharm 2013;3:843-52.
- Siddikuzzaman, Grace VM. Inhibition of metastatic lung cancer in C57BL/6 mice by liposome encapsulated all-trans retinoic acid (ATRA). Int Immunopharmacol 2012;14:570-9.
- Choi EJ, Whang YM, Kim SJ, Kim HJ, Kim YH. Combinational treatment with retinoic acid derivatives in non-small cell lung carcinoma in vitro. J Korean Med Sci 2007;22(Suppl):S52-60.
- Tatebe H, Shimizu M, Shirakami Y, Tsurumi H, Moriwaki H. Synergistic growth inhibition by 9-cis-retinoic acid plus trastuzumab in human hepatocellular carcinoma cells. Clin Cancer Res 2008;14(9):2806-12.
- Stewart BW, Wild CP. World Cancer Report. Lyon, France: IARC Press; 2014. p. 350-2.
- Hong WK, Endicott J, Itri LM, Doos W, Batsakis JG, Bell R, et al. l3-cis-Retinoic acid in the treatment of oral leukoplakia. N Engl J Med 1986;315:1501-5.
- Ramya D, Siddikuzzaman, Manjamalai A, Berlin Grace VM. Chemoprotective effect of all-trans retinoic acid (ATRA) on oxidative stress and lung metastasis induced by benzo(a)pyrene. Immunopharmacol Immunotoxicol 2012;34:317-25.
- Siddikuzzaman, Grace VM. Antioxidant potential of all-trans retinoic acid (ATRA) and enhanced activity of liposome encapsulated ATRA against inflammation and tumor-directed angiogenesis. Immunopharmacol Immunotoxicol 2013;35:164-73.
- Siddikuzzaman, Grace VM. Anti-Metastatic Study of Liposome-Encapsulated All trans Retinoic Acid (ATRA) in B16F10 Melanoma Cells-Implanted C57BL/6 Mice. Cancer Invest 2014;32:507-17.
- Vihari M, Siddikuzzaman, Berlin Grace VM. Growth inhibitory and apoptosis induction effects of All-Trans Retinoic Acid (ATRA) on cervical cancer cell line (HeLa). Int J Adv Pharm Med Bioallied Sci 2014;2:2-6.
- Lotan R. Effects of vitamin A and its analogs (retinoids) on normal and neoplastic cells. Biochim Biophys Acta 1980;605:33-91.
- Sharma A, Sharma US. Liposomes in drug delivery: Progress and limitations. Int J Pharm 1997;154:123-40.
- Ozpolat B, Lopez-Berestein G, Adamson P, Fu CJ, Williams AH. Pharmacokinetics of intravenously administered liposomal all-trans-retinoic acid (ATRA) and orally administered ATRA in healthy volunteers. J Pharm Pharma Sci 2003;6:292-301.
- Bangham AD, Standish M, Klatins JC. Diffusion of univalent ions across the lamella of swollen phospholipids. J Mol Biol 1995;13:238-52.
- Boman NL, Masin D, Mayer LD, Cullis PR, Bally MB. Liposomal vincristine which exhibits increased drug retention and increased circulation longevity cures mice bearing P388 tumors. Cancer Res 1994;54(11):2830-3.
- Silveira ER, Naves MM, Vannucchi H, Jordão Júnior AA, Dagli ML, Moreno FS. Vitamin A and all-trans and 9-cis retinoic acids inhibit cell proliferation during the progression phase of hepato carcinogenesis in Wistar rats. Nutr Cancer 2001;39(2):244-51.
- Chen Q, Ross AC. Retinoic acid regulates cell cycle progression and cell differentiation in human monocytic THP-1 cells. Exp Cell Res 2004;297(1):68-81.
- Güzey M, Demirpençe E, Criss W, DeLuca HF. Effects of Retinoic Acid (All-trans and 9-cis) on Tumor Progression in Small-Cell Lung Carcinoma. Biochem Biophys Res Commun 1998;242(2):369-75.
- Han S, Fukazawa T, Yamatsuji T, Matsuoka J, Miyachi H, Maeda Y, et al. Anti-tumor effect in human lung cancer by a combination treatment of novel histone deacetylase inhibitors: SL142 or SL325 and retinoic acids. PloS one 2010;5(11):e13834.
- Nagy Z, Baghy K, Hunyadi-Gulyás É, Micsik T, Nyírő G, Rácz G, et al. Evaluation of 9-cis retinoic acid and mitotane as antitumoral agents in an adrenocortical xenograft model. Am J Cancer Res 2015;5(12):3645.
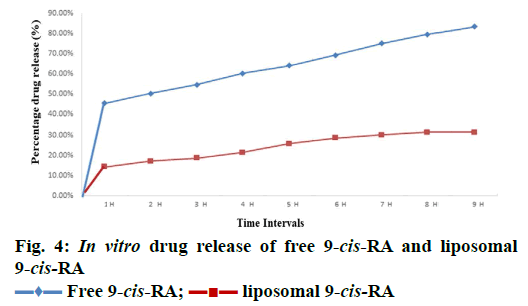
 Free 9-cis-RA;
Free 9-cis-RA;  liposomal 9-cis-RA
liposomal 9-cis-RA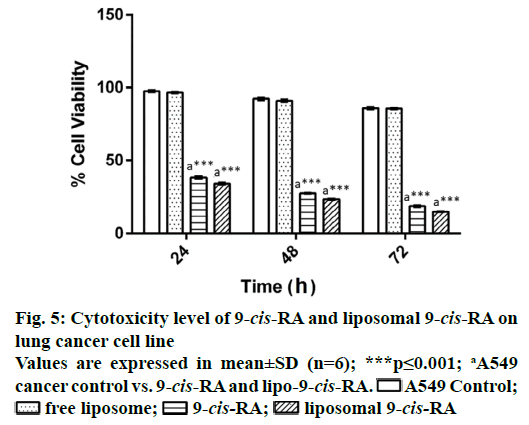
 A549 Control;
A549 Control;  free liposome;
free liposome;  9-cis-RA;
9-cis-RA;  liposomal 9-cis-RA
liposomal 9-cis-RA