- *Corresponding Author:
- Liang Zhang
Department of Internal Medicine, Tongde Hospital of Zhejiang Province, Hangzhou, Zhejiang Province 310012, China
E-mail: zhangliang853@163.com
| Date of Received | 18 December 2021 |
| Date of Revision | 05 October 2022 |
| Date of Acceptance | 08 August 2023 |
| Indian J Pharm Sci 2023;85(4):1173-1177 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
To investigate the effect of simvastatin on ventricular remodeling in rats with acute myocardial infarction through the transforming growth factor beta/SMAD signaling pathway. A rat acute myocardial infarction model was established by ligating the left anterior descending coronary artery as the acute myocardial infarction group; treated with low, medium and high doses (10 mg/kg, 20 mg/kg and 40 mg/kg) of simvastatin as acute myocardial infarction+simvastatin-low, acute myocardial infarction+simvastatin-medium, acute myocardial infarction+simvastatin-high groups. Set up another sham operation group (sham group). 4 w later, the rats' body weight, hemodynamic acute myocardial infarction indicators, and blood lipid levels were detected; quantitative reverse transcription polymerase chain reaction and Western blot were used to detect the expression of transforming growth factor beta 1, SMAD2/3 messenger ribonucleic acid and protein. Compared with the sham group, the left ventricular weight and left ventricular weight index of the rats were significantly increased (p<0.05); compared with the acute myocardial infarction group, simvastatin significantly reduced the left ventricular weight and left ventricular weight index of the rats (p<0.05). Compared with the sham group, the left ventricular systolic pressure of the acute myocardial infarction group was significantly increased, and the left ventricular end-diastolic pressure, +dp/dtmax and -dp/dtmax were significantly decreased (p<0.05); compared with the acute myocardial infarction group, simvastatin significantly reduced the rat left ventricular end-diastolic pressure, significantly increased left ventricular end-diastolic pressure, +dp/dtmax and -dp/dtmax (p<0.05). Compared with the sham group, the expression of transforming growth factor beta 1, SMAD2, SMAD3 messenger ribonucleic acid and protein in the acute myocardial infarction group was significantly increased (p<0.05); compared with the acute myocardial infarction group, simvastatin significantly reduced the expression of transforming growth factor beta 1, SMAD2, SMAD3 messenger ribonucleic acid and protein in the rat (p>0.05). Simvastatin can alleviate ventricular remodeling in acute myocardial infarction rats by inhibiting the transforming growth factor beta/SMAD signaling pathway.
Keywords
Simvastatin, transforming growth factor beta, SMAD pathway, acute myocardial infarction, ventricular remodeling
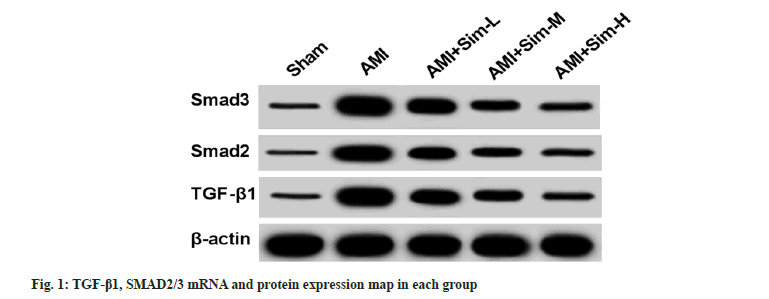
Acute Myocardial Infarction (AMI) is a disease that is highly harmful to humans. There is often severe and long-lasting retrosternal pain, which is not completely relieved by rest and nitrates, accompanied by increased serum cardiac enzyme activity and progressive electrocardiographic changes and can be complicated by arrhythmia, shock or heart failure[1,2]. Ventricular remodeling refers to the structural changes of the heart ventricle due to the coarsening of myocardial fibers in the heart, which leads to a series of changes in heart size, shape, wall thickness and tissue structure[3,4]. Simvastatin (SIM), which is refined from within Aspergillus terreus, began to medical use in 1992 and is one of the essential medicines in the basic medical system. This drug can reduce the chance of heart attack[5,6]. In this experiment, the effect of SIM on ventricular remodeling in AMI rats through Transforming Growth Factor-Beta (TGF-β)/SMAD signaling pathways is mainly discussed. Fifty healthy Specific-Pathogen Free (SPF) grade adult Sprague-Dawley (SD) rats with a body mass ranging from 200-220 g were purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd., qualification: SCXK (J 2012-0001). AMI model was established by ligating the anterior descending branch of the left coronary artery[7]. After the rats fasted for 12 h, they were anesthetized, prepped, sterilized and the heart was exposed and the left anterior descending coronary artery was ligated. The color of left ventricular anterior wall turned white and the ST-T arc of electrocardiogram was raised upward, indicating that the model of myocardial infarction was successfully established. Fifty rats, 40 of which were established as AMI models in rats and 10 as AMI groups; another 30 were treated with low, medium, or high doses (10 mg/kg, 20 mg/kg and 40 mg/kg) of SIM as AMI+SIM-Low (SIM-L), AMI+SIM-Medium (SIM-M) and AMI+SIM-High (SIM-H) groups. 10 rats were born as a sham group, only threaded without ligation. After 4 w of administration, the rats were anesthetized using chloral hydrate, and the rat Body Weight (BW) was weighed. The common carotid arteries were isolated, and a multi-lead physiological recorder was connected to determine the Left Ventricular Systolic Pressure (LVSP), Left Ventricular Diastolic Pressure (LVEDP), and maximal rate of rise and fall (±dp/dtmax) in the left ventricle, and the rats were sacrificed. Blood 2 ml was collected for blood lipid testing. After the rats were sacrificed, the large blood vessels, atria and right ventricle tissues were removed, the Left Ventricular Weight (LVW) was weighed, and the LVW Index (LVWI)=LVW/BW was calculated. Total Cholesterol (TC), Triglyceride (TG), Low-Density Lipoprotein Cholesterol (LDL-C) and High-Density Lipoprotein Cholesterol (HDL-C) levels were measured using a blood lipid assay kit and AU640 automatic biochemical analyzer according to the instructions. Total Ribonucleic Acid (RNA) was extracted using Trizol reagent and messenger RNA (mRNA) was synthesized into complementary Deoxyribonucleic Acid (cDNA) according to the reverse transcription kit. And using cDNA as template, for amplification, β-actin was used as an internal reference, and TGF-β1 and SMAD2/3 mRNA relative expression levels were calculated with 2-△△Ct method as shown in Table 1. Protein concentration was determined by Bicinchoninic acid (BCA) protein kit and electrophoresed by 10 % Sodium Dodecyl Sulphate-Polyacrylamide Gel Electrophoresis (SDS-PAGE). Transferred to Polyvinylidene Difluoride (PVDF), blocked in 5 % non-fat dry milk respectively. Placed in primary antibody incubation solution and incubated overnight at 4°. Place in goat anti-rabbit secondary antibody incubation solution and incubate at 37° for 1 h. ImageJ software was used to analyze the relative expression levels of proteins of interest. Data were analyzed using Statistical Package for the Social Sciences (SPSS) 22.0. Metrology data are presented as (x̄±s). One way Analysis of Variance (ANOVA) was used for multi-group comparison, and Student–Newman–Keuls (SNK)-q test was used for further pairwise comparison. p<0.05 indicated that the difference was statistically significant. Compared with sham group, there were no significant changes in BW in AMI group rats (p>0.05), and LVW and LVWI increased significantly (p<0.05); no significant changes of BW were observed in AMI+SIM-L, AMI+SIM-M and AMI+SIM-H groups compared with AMI group (p>0.05), and LVW and LVWI reduced significantly (p<0.05) as shown in Table 2. Compared with the sham group, the LVSP of the AMI group was significantly increased, and the LVEDP, +dp/dtmax, and -dp/dtmax were significantly decreased (p<0.05); compared with the AMI group, rat LVSP in AMI+SIM-L, AMI+SIM-M and AMI+SIM-H groups reduced significantly, LVEDP, +dp/dtmax and -dp/dtmax significantly increased (p<0.05) as shown in Table 3. Compared with sham group, there were no significant changes in TC, TG, LDL-C, and HDL-C in AMI group rats (p>0.05); no significant changes were observed in AMI+SIM-L, AMI+SIM-M and AMI+SIM-H groups compared with AMI group (p>0.05) as shown in Table 4. Compared with the sham group, the expression of TGF-β1, SMAD2, SMAD3 mRNA and protein in the AMI group was significantly increased (p<0.05); compared with the AMI group, the expression of TGF-β1, SMAD2, SMAD3 mRNA and protein in the rat of AMI+SIM-L, AMI+SIM-M and AMI+SIM-H groups significantly reduced (p>0.05) as shown in fig. 1 and Table 5. AMI is a very harmful disease to humans, which refers to the acute necrosis of part of the myocardium caused by long-lasting and severe myocardial ischemia, and in a short time, due to a certain reason, the human coronary artery has undergone occlusion, so that the human heart will be ischemic, causing a certain area of myocardial necrosis[8,9]. In this study, compared with the sham operation group, the LVW and LVWI of rats were significantly increased; SIM significantly reduced LVW and LVWI in rats compared with AMI group, compared with sham operation group, there were no significant changes in BW and lipid levels in rats of AMI group compared with those of sham operation group; compared with AMI group, SIM did not significantly affect the BW and lipid levels of rats. Xiao et al. found that consistent with this experiment, there was no statistical difference in the BW of rats in each group[10]. Compared with sham group, LVW and LVWI significantly increased by SIM and there were no statistical differences in all lipid levels among groups. This study shows that compared with the sham operation group, the LVSP of the AMI group was significantly increased and the LVEDP, +dp/dtmax, and -dp/dtmax were significantly decreased; compared with the AMI group, SIM significantly reduced the rat LVSP, significantly increased LVEDP, +dp/dtmax and -dp/dtmax. Studies have reported that similar to this experiment, treatment with SIM suppresses ventricular remodeling in patients with AMI and has positive effects on controlling the condition[11]. After using SIM, cardiac function indexes were improved in patients with AMI[12]. TGF-β signaling pathways are involved in many other cellular functions such as cell growth, differentiation, apoptosis and homeostasis. TGF-β class ligands bind to type II receptors, which phosphorylate type I receptors, and type I receptors rephosphorylate receptor regulated SMAD proteins, which in turn bind to coSMAD. R-SMAD/coSMAD complex assembles in the nucleus as a transcription factor and is involved in the regulation of target gene expression[13,14]. In this study, we found that compared with the sham operation group, the expressions of genes and proteins related to TGF-β/SMAD signaling pathway were significantly elevated; SIM significantly reduced TGF-β/SMAD signaling pathway related genes and protein expression in rats compared with AMI group. It has been shown that TGF-β/SMAD signal transduction pathway is involved in the process of ventricular remodeling after myocardial infarction[15]. TGF-β/SMAD signaling pathways are closely related to myocardial fibrosis after myocardial infarction[16]. In conclusion, SIM can alleviate ventricular remodeling in AMI rats by inhibiting the TGF-β/SMAD signaling pathway.
| Group | Upstream primer sequences (5'→3') | Downstream primer sequences (5'→ 3') |
|---|---|---|
| TGF-β1 | CTCCCGTGGCTTCTAGTGC | GCCTTAGTTTGGACAGGATCTG |
| SMAD2 | ATGTCGTCCATCTTGCCATTC | AACCGTCCTGTTTTCTTTAGCTT |
| SMAD3 | CACGCAGAACGTGAACACC | GGCAGTAGATAACGTGAGGGA |
| β-actin | TTGGCATAGAGGTCTTTA | AGATGACCCAGATCATGTTTGA |
Table 1: Reverse Transcription-Polymerase chain reaction primer sequences.
| Group | BW (g) | LVW (mg) | LVWI (mg/g) |
|---|---|---|---|
| Sham | 219.19±16.33 | 423.68±68.10 | 1.93±0.38 |
| AMI | 246.38±56.28 | 847.36±96.12* | 3.43±0.92* |
| AMI+SIM-L | 240.92±25.82 | 724.83±88.16# | 3.00±0.71# |
| AMI+SIM-M | 234.44±37.90 | 658.34±62.30## | 2.8±0.55# |
| AMI+SIM-H | 222.30±41.09 | 546.35±55.28### | 2.45±0.46## |
| F | 382.617 | 483.129 | 128.62 |
| p | 0.000 | 0.000 | 0.000 |
Note: Compared with sham group, *p<0.05 and compared with the AMI group, #p<0.05
Table 2: Effect of SIM on BW, LVW and LVWI in AMI rats (x̄±s, n=9).
| Group | LVSP (mmHg) | LVEDP (mmHg) | +dp/dtmax (mmHg/s) | -dp/dtmax (mmHg/s) |
|---|---|---|---|---|
| Sham | 4.62±0.92 | 163.82±14.82 | 3587.31±182.49 | 3118.75±258.96 |
| AMI | 14.62±2.82* | 118.25±18.92* | 1354.92±136.67* | 115.86±127.82* |
| AMI+SIM-L | 8.63±1.21# | 128.92±15.64# | 2186.25±283.15# | 1987.36±225.03# |
| AMI+SIM-M | 7.41±0.88# | 135.60±16.66# | 2563.72±246.31## | 2363.85±246.69## |
| AMI+SIM-H | 6.34±0.15## | 148.21±18.20## | 2876.34±287.36### | 2512.64±173.24### |
| F | 198.637 | 188.637 | 286.341 | 487.325 |
| p | 0.000 | 0.000 | 0.000 | 0.000 |
Note: Compared with sham group, *p<0.05 and compared with the AMI group, #p<0.05
Table 3: Effects of SIM on LVSP, LVEDP and ±dp/dtmax in AMI rats (x̄±s, n=9).
| Group | TC (mmol/l) | TG (mmol/l) | LDL-C (mmol/l) | HDL-C (mmol/l) |
|---|---|---|---|---|
| Sham | 1.46±0.27 | 0.55±0.05 | 0.42±0.04 | 0.66±0.06 |
| AMI | 1.22±0.82 | 0.50±0.05 | 0.46±0.05 | 0.62±0.06 |
| AMI+SIM-L | 1.16±0.18 | 0.48±0.06 | 0.40±0.04 | 0.69±0.07 |
| AMI+SIM-M | 1.27±0.27 | 0.52±0.05 | 0.41±0.04 | 0.64±0.05 |
| AMI+SIM-H | 1.18±0.96 | 0.49±0.05 | 0.47±0.06 | 0.62±0.06 |
| F | 98.634 | 128.932 | 82.376 | 176.354 |
| p | 0.000 | 0.000 | 0.000 | 0.000 |
Note: Compared with sham group, *p<0.05 and compared with the AMI group, #p<0.05.
Table 4: Effect of SIM on Lipid levels in AMI rats (x̄±s, n=9).
| Group | TGF-β1 mRNA | TGF-β1 protein | SMAD2 mRNA | SMAD2 protein | SMAD3 mRNA | SMAD3 protein |
|---|---|---|---|---|---|---|
| Sham | 1.00±0.10 | 0.25±0.02 | 1.02±0.11 | 0.25±0.02 | 1.00±0.10 | 0.27±0.02 |
| AMI | 2.38±0.16* | 0.88±0.08* | 2.89±0.47* | 0.89±0.08* | 2.55±0.33* | 0.87±0.08* |
| AMI+SIM-L | 1.88±0.65# | 0.65±0.06# | 2.00±0.88# | 0.62±0.06# | 1.82±0.37# | 0.63±0.06# |
| AMI+SIM-M | 1.52±0.72## | 0.56±0.05## | 1.78±0.63## | 0.50±0.05## | 1.42±0.33## | 0.52±0.05## |
| AMI+SIM-H | 1.33±0.18### | 0.39±0.03### | 1.50±0.53### | 0.40±0.04### | 1.18±0.26### | 0.40±0.03### |
| F | 128.960 | 152.371 | 154.632 | 168.963 | 114.257 | 137.268 |
| p | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
Note: Compared with Sham group, *p<0.05 and compared with the AMI group, #p<0.05.
Table 5: Effect of SIM on TGF-β1, SMAD2/3 mRNA and protein expression in AMI rats (x̄±s, n=9).
Funding:
The work was supported by clinical study of new thrombolytic drugs combined with ultra-long cardiopulmonary resuscitation in the treatment of suspected sudden myocardial infarction (020KY087).
Author’s contributions:
Yunfang Wang and Bo Wu have contributed equally to this work.
Conflict of interests:
The authors declared no conflict of interests.
References
- Li L, Ma Y, Geng XB, Tan Z, Wang JH, Cui C, et al. Platelet-to-lymphocyte ratio relates to poor prognosis in elderly patients with acute myocardial infarction. Aging Clin Exp Res 2021;33:619-24.
[Crossref] [Google Scholar] [PubMed]
- Rouzbahani M, Azimivghar J, Moghadam RH, Montazeri N, Janjani P, Rai A, et al. Acute myocardial infarction: Circadian, daily, monthly and seasonal patterns of occurrence in diabetics. J Diabetes Metab Disord 2021;20(1):765-70.
[Crossref] [Google Scholar] [PubMed]
- Espinola-Zavaleta N, Antonio-Villa NE, Briseño-Diaz N, Oregel-Camacho D, Camacho-Camacho G, Keirns C, et al. Left ventricular and atrial global strain evaluation within subtypes of ventricular remodeling. Echocardiography 2021;38(2):280-8.
[Crossref] [Google Scholar] [PubMed]
- Sinclair N, Ordenana C, Lee J, Onwuzulike K, Recinos V, Papay F, et al. Cranial vault remodeling in children with ventricular shunts. J Craniofac Surg 2020;31(4):1101-6.
[Crossref] [Google Scholar] [PubMed]
- Kotlovskiy MY, Udut EV, Kairov GT, Fisenko VP, Udut VV. Effects of simvastatin on the metabolism of fatty acids in combined secondary prevention of coronary heart disease: Dosage and gender differences between the effects. Cardiovasc Haematol Disord Drug Targets 2020;20(2):93-107.
[Crossref] [Google Scholar] [PubMed]
- Hsieh CC, Li CY, Hsu CH, Chen HL, Chen YH, Liu YP, et al. Mitochondrial protection by simvastatin against angiotensin II-mediated heart failure. Br J Pharmacol 2019;176(19):3791-804.
[Crossref] [Google Scholar] [PubMed]
- Tang YL, Zhao Q, Qin X, Shen L, Cheng L, Ge J, et al. Paracrine action enhances the effects of autologous mesenchymal stem cell transplantation on vascular regeneration in rat model of myocardial infarction. Ann Thorac Surg 2005;80(1):229-37.
[Crossref] [Google Scholar] [PubMed]
- Looi JL, Verryt T, McLeod P, Chan C, Pemberton J, Webster M, et al. Incidence of Takotsubo syndrome vs. acute myocardial infarction in New Zealand (ANZACS-QI 45). N Z Med J 2020;133(1511):90-4.
[Google Scholar] [PubMed]
- Zhao C, Liu J, Ge W, Li Z, Lv M, Feng Y, et al. Identification of regulatory circRNAs involved in the pathogenesis of acute myocardial infarction. Front Genet 2021;11:626492.
[Crossref] [Google Scholar] [PubMed]
- Xiao Xi, Qin S, Zhang D. Effect of simvastatin on ventricular remodeling after myocardial infarction in rats and its relation to TGF-β1/TAK1 signaling. Med J Chin People's Liberation Army 2009;34(9):1085-8.
- Gao C. Therapeutic effect of post interventional simvastatin on left ventricular remodeling in the early stage of acute myocardial infarction. Chin J Med 2017;9(19):147-8.
- Xie X, Zou J, Zhang L. Effect of simvastatin on early left ventricular remodeling after emergency interventional therapy for acute myocardial infarction. Chin J Geriatr Heart Brain Vessel Dis 2013;15(4):349-52.
- Zhang Y, Zhao G. Research progress of TCM prevention and treatment of myocardial fibrosis based on TGF-β/SMAD signaling pathway. Chin J Integr Med Cardiovasc Dis 2020;18(9):55-9.
- Liang Y, He Y. Research progress on the related mechanisms of transforming growth factor β/SMAD signaling pathway activation induced atrial fibrillation. Pract J Cardiac Cerebral Pneumal Vasc Dis 2019;27(5):117-20.
- Gao Y, Xie S, Wang Y. Mechanism of Jiashen prescription inhibiting TGF-β/SMAD signaling pathway to improve ventricular remodeling after myocardial infarction in rats. China J Tradit Chin Med Pharm 2017;32(3):1328-32.
- Huang M, Wen Z, Pang Y. Research progress on the relationship between TGF-β/SMAD signaling pathway and myocardial fibrosis after myocardial infarction. Chongqing Med 2018;47(24):3213-5.