- *Corresponding Author:
- Guosheng Xie
Department of Rehabilitation Medicine, West China Hospital, Sichuan University/West China School of Nursing, People's Republic of China
E-mail: 769602954@qq.com
| This article was originally published in a special issue,“Recent Progression in Pharmacological and Health Sciences” |
| Indian J Pharm Sci 2024:86(2) Spl Issue “183-188” |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
Investigating the efficacy of Yaobitong capsule in reducing pain and influencing interleukin-6 and interleukin-8 factors was the main goal of this research. The division of 80 patients diagnosed with lumbar disc herniation into a control group and an observation group, consisting of 40 patients each, was conducted during the period from June 2022 to June 2023. Throughout the 2 w treatment period, the control group underwent lumbar traction, massage therapy, and the oral administration of loxoprofen sodium capsules, taken at a frequency of one capsule, three times a day. Meanwhile, the same treatment as the control group was undergone by the observation group, with the addition of Yaobitong capsule, administered at a frequency of three capsules, three times a day. Subsequent to 2 w of treatment, a comparison was made between the two groups based on the clinical efficacy, visual analogue scale scores, Oswestry disability index scores, adverse reactions, and serum interleukin-6 and interleukin-8 levels. Additionally, a follow-up evaluation was conducted after 6 mo to appraise the long-term treatment outcomes. Following a treatment duration of 2 w, the observation group demonstrated a notably higher total effective rate (90.00 %) in comparison to the control group (67.50 %), revealing statistical significance (p<0.05). However, post 2 w of treatment, both groups demonstrated a decline in serum interleukin-6 and interleukin-8 levels compared to pre-treatment levels, with the observation group exhibiting lower levels than the control group. The occurrence rate of adverse reactions did not differ remarkably between the two groups (p>0.05). The outcomes of this study illustrate the notable clinical efficacy of Yaobitong capsule in managing pain linked to lumbar disc herniation. It has the potential to effectively alleviate pain, enhance function, and potentially impact inflammatory cytokines.
Keywords
Yaobitong capsule, lumbar disc herniation, interleukin-6, loxoprofen sodium
A factor contributing to lower back pain is Lumbar Disc Herniation (LDH)[1]. By utilizing Magnetic Resonance Imaging (MRI), diverse stages of degenerative transformations, particularly affecting the nucleus pulposus of the lumbar intervertebral disc, can be identified, potentially leading to the protrusion of herniated nucleus pulposus tissue to the rear or into the spinal canal due to a ruptured fibrous ring[2,3]. In recent years, conservative and surgical strategies have emerged as the principal methods of treatment[4,5]. The majority of patients find relief or recover through non-surgical interventions, with only 10 % to 15 % of cases requiring surgical intervention[6]. Conventional therapies encompass physical therapy[7,8], oral drugs (pain relievers or non-steroidal anti-inflammatory drugs)[9,10], and localized pharmaceutical injections[11]. Traction treatment, a key component of physical therapy, stimulates the relaxation of lower back muscles by applying sustained tension, expanding the space between vertebrae, promoting intervertebral disc reduction, and enhancing local tissue blood flow. As a result, this process reduces inflammatory responses, relieves tissue swelling, minimizes physical damage to affected nerve sites, and aids in the absorption of local inflammation[12,13]. However, the effectiveness of this therapy is restricted and typically requires supplementary pharmacological intervention. Of these, loxoprofen sodium capsules are frequently prescribed for this disorder, distinguished for their anti-inflammatory and pain-relieving effects, notwithstanding the numerous adverse reactions associated with their use[14]. Hence, the quest for a reliable and safe substitute medication is especially paramount. Yaobitong capsule, an herbal remedy from Traditional Chinese Medicine (TCM), is thought to possess attributes that enhance blood circulation to remove stasis and relieve pain, a conclusion drawn from years of clinical observation and investigation[15]. While the specific mechanism of action remains ambiguous[16], multiple studies have evidenced its effectiveness in mitigating pain associated with LDH. Consequently, this research seeks to assess the impact of Yaobitong capsule on the management of pain related to LDH and explore its influence on the inflammatory factors Interleukin (IL)-6 and IL-8. The gathering of data on clinical pain assessment and serum IL-6 and IL-8 levels will be the outcome of a comparative analysis between the group receiving Yaobitong capsule treatment and the commonly used loxoprofen sodium. The objective is to supply novel clinical evidence for the rehabilitation treatment of LDH and to serve as a point of reference for a more thorough examination of its mechanism of action.
Materials and Methods
General information:
The study included 80 previously untreated patients diagnosed with LDH who received treatment at our hospital between June 2022 and June 2023. Categorized according to the treatment they received, the patients were split into a control group and an observation group, with 40 individuals in each. The control group encompassed 18 male and 22 female participants, ranging in age from (38-72) y and averaging (51.72±6.13) y. The condition persisted for an average of (31.25±4.64) d, with a range from (10-60) d. Comprising 21 males and 19 females, the observation group’s ages ranged from (40-73) y, with an average of (52.18±6.24) y. They experienced the condition for an average of (32.31±4.72) d, within a range of (11-58) d. No statistically noteworthy variances were detected in the clinical data of the two groups (p>0.05), indicating their similarity. Authorization for the analysis of clinical data was acquired from the Hospital Ethics Committee, with patient informed consent being waived.
Inclusion criteria: Individuals diagnosed with LDH according to lumbar spine X-rays, Computed Tomography (CT) scans, MRI, and clinical manifestations[17]; patients aged 18 y to 80 y, of either gender and patients with normal language, consciousness, and communication abilities were included in this study.
Exclusion criteria: Patients with spinal tumors, spinal stenosis, or spinal tuberculosis; patients with severe systemic diseases such as heart, liver, or kidney conditions and pregnant or lactating women.
Methods:
Control group treatment: Over a 2 w period, the patients were treated with lumbar traction and massage therapy, supplemented with loxoprofen sodium capsules (60 mg) from Shanxi Tianyuan Pharmaceuticals at a dose of one capsule three times daily.
Observation group treatment: In observation group, in addition to the lumbar traction and massage therapy enforced in the control group, was provided with Yaobitong capsules (0.42 g) manufactured by Jiangsu Kangyuan Pharmaceuticals, administered at a dosage of three capsules three times daily over a 2 w period.
Observation indicators:
Following the 2 w treatment period, an analysis was conducted to compare the clinical efficacy, Oswestry Disability Index (ODI) scores, Visual Analogue Scale (VAS) scores, adverse reactions, IL-8 and IL-6 levels between the two groups. A subsequent follow-up evaluation was undertaken after 6 mo to evaluate the long-term treatment outcomes. A 3 ml fasting sample of venous blood from the elbow was obtained, and the DG3022 enzyme-linked immune detector was utilized to conduct an Enzyme-Linked Immunosorbent Assay (ELISA) to monitor changes in IL-6 (normal range of 56.37–150.33 pg/ml) and IL-8 (normal range of <156 pg/ml) concentrations. The VAS, graded from 0 to 10, was employed to assess lumbar pain, with 0 indicating no pain and 10 indicating unbearable pain. The ODI serves as the benchmark for evaluating functional impairment associated with lumbar pain. The questionnaire includes 10 items concerning the severity of pain, daily activities, sleep, and walking. Each item offers six response options, rated from 0 to 5 points. An elevated score signifies a heightened constraint on daily activities and heightened lumbar and leg pain intensity[18].
Efficacy assessment: Raising the hips and legs by more than 70° without experiencing lumbar and leg pain indicated effectiveness; a prominent reduction in lumbar and leg pain, and an improvement in lumbar function, indicated improvement; persistent symptoms and signs were indicative of ineffectiveness. Adverse reactions includes urticaria, drowsiness, and nausea.
Overall efficacy rate=(Effectiveness+improvement)/ total cases×100 %
Statistical analysis:
Utilizing the Statistical Package for the Social Sciences (SPSS) 25.0, the analysis was performed, comparing the measurement data, presented as mean±standard deviation, between the two groups via an independent sample t-test. Utilizing the Chisquare (χ2) test, the count data were analyzed. The criteria for establishing statistical significance were defined at a level of p<0.05.
Results and Discussion
After 2 w of treatment, the observation group exhibited an overall effective rate of 90.00 %, in contrast to the control group’s rate of 67.50 %. The distinction was notably significant (p<0.05) as shown in Table 1.
| Group (n=40) | Cured | Improvement | Ineffectiveness | Overall effective rate |
|---|---|---|---|---|
| Observation | 17 (42.50) | 19 (47.50) | 4 (10.00) | 36 (90.00) |
| Control | 14 (35.00) | 13 (32.50) | 13 (32.50) | 27 (67.50) |
| χ2 | 6.05 | |||
| p | 0.014 |
Table 1: Curative Effect
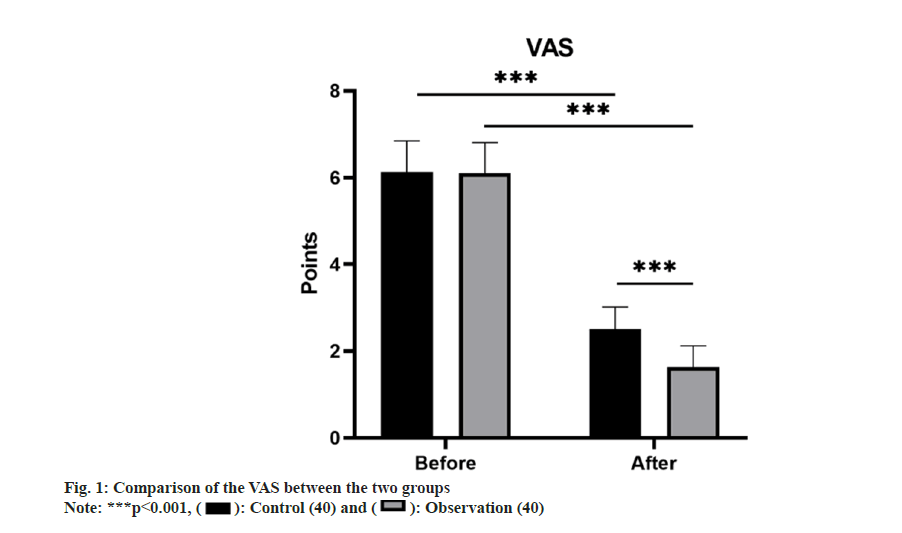
Comparing the VAS scores before treatment revealed no substantial distinction between the control group (6.13±0.72) and the observation group (6.10±0.71) (p>0.05). However, following treatment, both groups experienced a substantial reduction in VAS scores compared to pre-treatment (p<0.01). Notably, the observation group exhibited a more notable reduction in VAS scores (1.63±0.49) as opposed to the control group (2.50±0.51) with a remarkable difference (p<0.05) as shown in fig. 1.
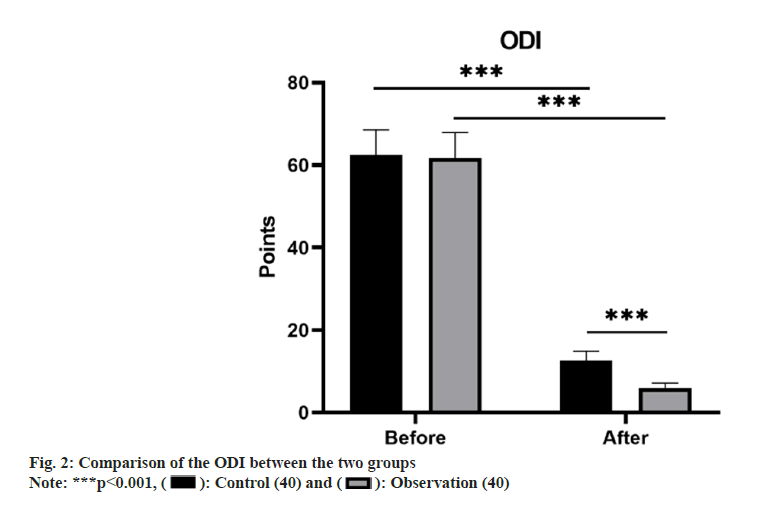
There was no substantial variance in the ODI scores of the control group (62.50±6.04) and the observation group (61.73±6.19) before treatment (p>0.05). Posttreatment, both groups saw a marked reduction in ODI scores, with the observation group achieving a remarkably lower ODI score (5.88±1.26) than the control group (12.60±2.28), demonstrating a notable difference (p<0.05) as shown in fig. 2.
Prior to treatment, no marked difference was identified in IL-6 and IL-8 levels between the two groups (p>0.05). However, following the 2 w treatment, both groups showed a decrease in serum IL-8 and IL-6 levels compared to before treatment, with the observation group exhibiting lower levels than the control group, with considerable differences (p<0.05) as shown in Table 2.
| Group (n=40) | IL-6 (pg/ml) | IL-8 (pg/ml) | ||
|---|---|---|---|---|
| Before | After | Before | After | |
| Observation | 322.35±15.88 | 282.36±15.55* | 449.05±27.74 | 345.28±25.60* |
| Control | 325.99±15.94 | 304.97±15.02* | 444.79±27.72 | 400.23±23.28* |
| t | 1.022 | 6.615 | -0.686 | 10.041 |
| p | 0.310 | 0.000 | 0.494 | 0.000 |
Note: (*) indicates noteworthy difference following treatment compared with prior to treatment
Table 2: Inflammatory Cytokine Levels
The observation group reported 2 cases of nausea, 0 cases of urticaria, and 1 case of drowsiness, while the control group had 3 cases of nausea, 1 case of urticaria, and 1 case of drowsiness. No notable variance was found in the occurrence rate of adverse reactions between the two groups (p>0.05) as shown in Table 3.
| Group (n=40) | Nausea | Urticaria | Drowsiness | Overall incidence |
|---|---|---|---|---|
| Observation | 2 (5.00) | 0 (0.00) | 1 (2.50) | 3 (7.50) |
| Control | 3 (7.50) | 1 (2.50) | 2 (5.00) | 6 (15.00) |
| χ 2 | 1.127 | |||
| p | 0.288 |
Table 3: Complications n (%)
At the 6 mo mark post-treatment, the observation group displayed a total effective rate of 77.50 %, contrasting to 52.50 % in the control group, with the dissimilarity being remarkably significant (p<0.05) as shown in Table 4.
| Group (n=40) | Cured | Improvement | Ineffectiveness | Overall effective rate |
|---|---|---|---|---|
| Observation | 10 (25.00) | 21 (52.50) | 9 (22.50) | 31 (77.50) |
| Control | 6 (15.00) | 15 (37.50) | 19 (47.50) | 21 (52.50) |
| χ2 | 5.495 | |||
| p | 0.019 |
Table 4: Curative Effect
LDH frequently presents as a recurring and prevalent condition in clinical orthopedics, commonly affecting individuals in the (30-50) y age bracket. Degenerative changes, particularly in the intervertebral discs, transpire in various body tissues with advancing age, contributing to reduced elasticity and tension maintenance chiefly due to dehydration of the nucleus pulposus, thereby diminishing its capacity to endure pressure. Prolonged exposure to significant and repetitive injuries or physical trauma can weaken the fibrous ring, potentially leading to protrusion of the nuclear material. This, in turn, may cause clinical symptoms such as lower back and/ or lower limb pain and numbness due to nerve and vascular compression. Patients in severe cases may encounter disruptions in bowel and bladder control, or even paralysis, leading to a bedridden lifestyle, which significantly impacts their quality of life and ability to engage in daily activities and work[19].
Presently, the primary clinical treatment consists of a combination of traction, massage, and medication. Traction and massage therapies promote muscle relaxation in the lower back, expand intervertebral spaces, support disc reduction, and alleviate compression symptoms, all while enhancing blood circulation. Pain relievers and non-steroidal antiinflammatory drugs are administered concurrently to alleviate patient pain and accelerate the absorption of the inflammatory response. While loxoprofen sodium capsules show favorable analgesic effects, the post-discontinuation pain relief is not ideal, and adverse reactions are prevalent[14]. In TCM, LDH falls within the categories of Bi syndrome, lumbago, lumbosacral pain, etc. According to TCM, the causes of this condition are attributed to the imbalance of qi and blood circulation, organ dysfunction, and lumbar pain. The treatment focuses on promoting blood circulation, relaxing tendons, and relieving pain. Yaobitong capsule consists of a combination of herbs and is believed to aid in promoting blood circulation, eliminating stasis, consolidating vital energy, dispelling stasis, promoting blood flow, and relieving pain[20,21].
Our study revealed that the combination of Yaobitong capsules with lumbar traction and massage therapy significantly outperformed the sole use of lumbar traction and massage therapy in treating pain associated with LDH. The observation group’s remarkably higher overall effective rate than the control group’s displays the clinical efficacy of Yaobitong capsule in alleviating the pain symptoms associated with LDH. In terms of pain assessment, the VAS scores showed no significant difference between groups prior to treatment. However, following treatment, both groups exhibited a reduction in VAS scores compared to before treatment, with a more significant reduction observed in the observation group, indicating the effective relief of pain with Yaobitong capsule. In terms of functional disabilities, no substantial difference was observed in the ODI scores prior to treatment. However, following treatment, both groups showed a notable reduction in ODI scores, and the observation group displayed a significantly greater improvement in functional recovery compared to the control group. Furthermore, the observation group displayed notably lower levels of IL-6 and IL-8 following treatment compared to the control group, hinting at the potential role of Yaobitong capsule in suppressing the release of inflammatory factors. In terms of adverse reactions, no noteworthy difference in the occurrence rate was found between the two groups, indicating the relative safety of Yaobitong capsule at the utilized dosage. Long-term follow-up results indicated that the overall effective rate in the observation group remained higher than that in the control group, underscoring the sustained effectiveness of Yaobitong capsule in addressing the pain and functional impairment associated with LDH.
To summarize, the outcomes of this study suggest that Yaobitong capsule exhibits significant clinical efficacy in addressing pain associated with LDH. It effectively diminishes the perception of pain, enhances functional limitations, and may potentially achieve its effects by suppressing the inflammatory response. However, the study is subject to certain limitations such as a small sample size and a retrospective clinical design, highlighting the need for additional large-scale, multicenter, randomized controlled trials to further establish the effectiveness and safety of Yaobitong capsule in managing LDH.
Conflict of interests:
The authors declared no conflict of interest.
References
- Zhang AS, Xu A, Ansari K, Hardacker K, Anderson G, Alsoof D, et al. Lumbar disc herniation: Diagnosis and management. Am J Med 2023;136(7):645-51.
[Crossref] [Google Scholar] [PubMed]
- Hwang D, Kim S, Abeydeera NA, Statum S, Masuda K, Chung CB, et al. Quantitative magnetic resonance imaging of the lumbar intervertebral discs. Quant Imaging Med Surg 2016;6(6):744.
[Crossref] [Google Scholar] [PubMed]
- Bradley WG. Use of contrast in MR imaging of the lumbar spine. Magn Reson Imaging Clin N Am 1999;7(3):439-57.
[Google Scholar] [PubMed]
- Zhang L, Zhang C, Song D, Chen G, Liu L. Combination of percutaneous endoscopic lumbar discectomy and platelet-rich plasma hydrogel injection for the treatment of lumbar disc herniation. J Orthop Surg Res 2023;18(1):609.
- O’Connor SB, Holmberg KJ, Hammarstedt JE, Acosta JR, Monahan K, Sauber RD, et al. Return-to-play outcomes of athletes after operative and nonoperative treatment of lumbar disc herniation. Curr Rev Musculoskelet Med 2023;16(5):192-200.
[Crossref] [Google Scholar] [PubMed]
- Dydyk AM, Ngnitewe MR, Mesfin FB, Disc Herniation, StatPearls, Treasure Island (FL) ineligible companies. Fassil Mesfin declares no relevant financial relationships with ineligible companies: StatPearls Publishing Copyright© 2023, StatPearls Publishing LLC; 2023.
- Li X, Han Y, Cui J, Yuan P, Di Z, Li L. Efficacy of warm needle moxibustion on lumbar disc herniation: A meta-analysis. J Evid Based Complementary Altern Med 2016;21(4):311-9.
[Crossref] [Google Scholar] [PubMed]
- Xu J, Ding X, Wu J, Zhou X, Jin K, Yan M, et al. A randomized controlled study for the treatment of middle-aged and old-aged lumbar disc herniation by Shis spine balance manipulation combined with bone and muscle guidance. Medicine 2020;99(51):e23812.
[Crossref] [Google Scholar] [PubMed]
- Kataria D, Jumani L, Ahmed MU, Kumar V, Makda A. Comparison of pregabalin vs. placebo in reduction of pain due to lumber disc herniation. Cureus 2020;12(8):e9985.
[Crossref] [Google Scholar] [PubMed]
- Huang R, Meng Z, Cao Y, Yu J, Wang S, Luo C, et al. Nonsurgical medical treatment in the management of pain due to lumbar disc prolapse: A network meta-analysis. Semin Arthritis Rheum 2019;49(2):303-13.
[Crossref] [Google Scholar] [PubMed]
- Wongjarupong A, Pairuchvej S, Laohapornsvan P, Kotheeranurak V, Jitpakdee K, Yeekian C, et al. “Platelet-Rich Plasma” epidural injection an emerging strategy in lumbar disc herniation: A randomized controlled trial. BMC Musculoskelet Disord 2023;24(1):1-7.
[Crossref] [Google Scholar] [PubMed]
- Wang W, Long F, Wu X, Li S, Lin J. Clinical efficacy of mechanical traction as physical therapy for lumbar disc herniation: A meta-analysis. Comput Math Methods Med 2022;2022:5670303.
[Crossref] [Google Scholar] [PubMed]
- Cheng YH, Hsu CY, Lin YN. The effect of mechanical traction on low back pain in patients with herniated intervertebral disks: A systemic review and meta-analysis. Clin Rehabil 2020;34(1):13-22.
[Crossref] [Google Scholar] [PubMed]
- Xu WB, Wu DJ, Chen C, Zhao X, Hu ZJ, Fan SW, et al. Symptomatic postoperative discal pseudocyst after percutaneous endoscopic interlaminar discectomy: Case report and literature review. Orthop Surg 2021;13(1):347-52.
[Crossref] [Google Scholar] [PubMed]
- Deng Y, Gao X, Feng T, Wang Z, Xiao W, Xiong Z, et al. Systematically characterized mechanism of treatment for lumbar disc herniation based on Yaobitong capsule ingredient analysis in rat plasma and its network pharmacology strategy by UPLC-MS/MS. J Ethnopharmacol 2020;260:113097.
[Crossref] [Google Scholar] [PubMed]
- Zhang X, Yang K, Wang S, Tang B, Yin H, Lei Q, et al. Efficacy and safety of Yaobitong capsule for acute lumbar disc herniation: A protocol for a multi-center randomized controlled trial. Medicine 2022;101(47):e31533.
[Crossref] [Google Scholar] [PubMed]
- Kaito T, Yamato Y. The essence of clinical practice guidelines for lumbar disc herniation, 2021: 3. Diagnosis. Spine Surg Relat Res 2022;6(4):325-8.
[Crossref] [Google Scholar] [PubMed]
- Chen Z, Wang X, Cui X, Zhang G, Xu J, Lian X. Transforaminal vs. interlaminar approach of full-endoscopic lumbar discectomy under local anesthesia for L5/S1 disc herniation: A randomized controlled trial. Pain Phys 2022;25(8):E1191.
[Google Scholar] [PubMed]
- Taşpınar G, Angın E, Oksüz S. The effects of Pilates on pain, functionality, quality of life, flexibility and endurance in lumbar disc herniation. J Com Eff Res 2023;12(1):e220144.
[Crossref] [Google Scholar] [PubMed]
- Shi W, Deng Y, Zhao C, Xiao W, Wang Z, Xiong Z, et al. Integrative serum metabolomic analysis for preventive effects of Yaobitong capsule in adjuvant-induced rheumatoid arthritis rat based on RP/HILIC-UHPLC-Q-TOF MS. Anal Biochem 2022;637:114474.
[Crossref] [Google Scholar] [PubMed]
- Shi W, Ye H, Deng Y, Chen S, Xiao W, Wang Z, et al. Yaobitong capsules reshape and rebalance the gut microbiota and metabolites of arthritic rats: An integrated study of microbiome and fecal metabolomics analysis. J Chromatogr B 2022;1190:123096.
[Crossref] [Google Scholar] [PubMed]