- *Corresponding Author:
- K. U. Kothule
Department of Pharmaceutical Science, Jawarlal Nehru Technological University, Kukatpally, Hyderabad-500 085, India
E-mail: kothule@rediffmail.com
| Date of Submission | 27 April 2012 |
| Date of Revision | 25 January 2013 |
| Date of Acceptance | 08 February 2013 |
| Indian J Pharm Sci, 2013;75(2):138-142 |
Abstract
The objective of this work was to increase the amount of acyclovir in the basal epidermis, site of herpes virus simplex infection, using the solid lipid nanoparticles loaded gel cream as carriers. Solid lipid nanoparticles were prepared by high pressure homogenisation method and incorporated in a semisolid submicron gel cream. Acyclovir distribution into rat skin after topical application of solid lipid nanoparticles loaded gel cream was determined by fabricated Franz diffusion cell. The results showed that, the quantity of the acyclovir in the basal epidermis with the solid lipid nanoparticles loaded submicron gel cream was two folds times more than marketed acyclovir gel cream. This type of carrier can improve acyclovir loaded therapy since it increases drug retention in the basal epidermis.
Keywords
Acyclovir, homogenisation, solid lipid nanoparticles, medium chain triglyceride
Acyclovir (ACV), a synthetic analogue of 2′‑deoxiguanosine, is a potent and highly selective inhibitor of viruses of herpes group, such as herpes simplex virus type 1 (HSV 1) and type 2 (HSV 2), varicella‑zoster virus, and to a lesser extent against Epstein Barr virus and cytomegalovirus [1]. Although, ACV is commercially available for intravenous, oral and topical administration, but its oral bioavailability is low (~20%) and systemically administered ACV may cause mild to severe side effects like nephrotoxicity. Therefore, topical route is considered more desirable [2].
Topical delivery of drugs to the skin, one of the most readily accessible organs of the body, has been studied for a long time to understand the pathophysiology of skin diseases or for their effective treatment [3]. Skin separates the vital organs from the outside environment and serves as a protective barrier against physical, chemical and microbial attacks. Although topical application of drug offers many advantages, including avoidance of first‑pass metabolism, lower fluctuations in plasma drug levels, targeting of the active ingredient for a local effect and good patient compliance [4], but the barrier nature of skin makes it difficult for most drugs to penetrate into and permeate through it [5]. There has been wide interest in exploring new techniques to enhance drug absorption through the skin during the past decades [4,6,7].
ACV is effective against cutaneous infections due to HSV‑1, whose target site is the basal epidermis. However, it has been suggested that ACV topical therapy has a low efficacy, due to the lack of penetration of a sufficient amount of the drug to the target site [8]. In this way, Zanowiak and Jacobs [2] found a good relationship between the free drug concentration at the basal epidermis and the in vivo antiviral efficacy for a variety of ACV topical formulations. The development of an effective ACV topical preparation prompted researchers to try several approaches. In some studies superior antiviral activity was demonstrated using different vehicles [10], percutaneous absorption enhancers [11] and iontophoresis [9]. Another possible strategy to achieve site specific drug delivery [12] would be the use of particulate drug carriers (microparticles and nanoparticles). These carriers possess certain advantages for topical application, since sustained release is important to supply the skin with the drug over a prolonged period of time. The objective of this work was to obtain a high concentration of ACV in the basal epidermis, where viral lesions are usually located, with the use of ACV-loaded solid lipid nanoparticles (ACV-SLNs).
SLNs have been utilized to enhance the skin/dermal uptake of several pharmacological agents such as triptolide, isotretinoin, podophyllotoxin, and prednicarbate [13‑17], demonstrating the potential of SLNs to be employed as the carrier for the topical delivery of penciclovir. The dermal location of SLNs is desirable to interfere with the establishment of the latent HSV infection. This would prevent the diseases associated with HSV and the transmission of the virus among men. The present work is focused on the preparation, characterisation, in vitro percutaneous permeation and skin targeting behaviors of ACV‑loaded SLNs. In addition, the interaction between SLNs and the skin surface was also investigated.
Materials and Methods
ACV was procured from Indoco Remedies Ltd. Rabale, Mumbai. The medium chain triglyceride used in this experiment was Miglyol 810, purchased from Sasol, Germany. Lipoid S 100 was used as the lecithin and was obtained as a gift sample from Lipoid GmbH, Germany. Pluronic‑68, carbopol, methyl paraben and propyl paraben was purchased from Gujarat Organics Ltd., Anakleshwar, Gujarat, India.
Preparation of ACV‑loaded SLNs
SLNs were prepared by high pressure homogenization method. An accurately weighed quantity of ACV (0.5 g) was mixed with varying quantities (Table 1) of medium chain triglyceride (MCT), i.e., Miglyol 810 (optimized 9 g) and lecithin (Lipoid S 100, optimised 1 g) until a homogeneous oily phase is achieved. The oil phase was then dispersed into 90 ml of aqueous phase comprising of 2 g of Pluronic‑F68 and 0.1 g of a mixture of methyl and propyl parabens by initial mixing with a magnetic stirrer followed by mixing on a high shear mixer (Polytron K3000) for 5 min at 20,000 rpm to form an emulsion. Further, the emulsion was subjected to a high pressure homogenizer (Aluminium Plant and Vessles co. ltd (APV)‑Gaulin) at 800 bar for 6 min (about 10 cycles) at 45‑55º. Thereafter, the emulsion was cooled to room temperature (RT) resulting in formation of SLNs. The mean droplet size was measured and found to be of 120 nm size having a very narrow distribution with practically no droplet above one micron (less than 0.5%) being detected.
| Batch | Acyclovir(g) | Miglyol 810(g) | Lipoid S100 (g) |
|---|---|---|---|
| FM‑1 | 0.5 | 5 | 0.25 |
| FM‑2 | 0.5 | 7 | 0.50 |
| FM‑3 | 0.5 | 9 | 1.00 |
| FM‑4 | 0.5 | 11 | 1.50 |
| FM‑5 | 0.5 | 13 | 2.00 |
SLN=solid lipid nanoparticle, FM=formulation material
Table 1: Formulation Plan For Acv-Slns
Preparation of ACV‑SLNs gel cream
SLN incorporated gel cream was prepared by adding 0.3% of carbopol to the above emulsion. Finally, the pH was adjusted to 7.0 with sodium hydroxide and a semisolid submicron droplet preparation (gel cream) was obtained [18].
Particle characterisation
The size distribution of SLNs was carried out in one set of triplicates, in a multimodal mode, by dynamic light scattering technique using a computerized Cilas 1900 Particle Size Analyzer Instrument (Cilas, France) (Table 2). Scanning electron microscopy (SEM) (Philips CM12 Electron Microscope, Eindhoven, Netherlands) was used as a visualizing aid for SLNs (fig. 1).
| Formulation | Particle size (nm) | Zeta potential (mV) | Polydispersity index | Entrapment efficiency (%) |
|---|---|---|---|---|
| FM‑1 | 81.67±1.53 | −20.21±3.12 | 0.4752±0.026 | 54.63±2.07 |
| FM‑2 | 85.33±2.08 | −22.39±2.19 | 0.4445±0.019 | 51.10±2.13 |
| FM‑3 | 98.33±2.52 | −27.31±1.38 | 0.5537±0.038 | 63.65±2.52 |
| FM‑4 | 121.00±2.65 | −24.15±5.57 | 0.4592±0.040 | 52.79±3.55 |
| FM‑5 | 142.67±3.06 | −20.22±4.48 | 0.5739±0.032 | 55.97±2.83 |
Data were expressed as mean±SD, n=3, FM=formulation material, SD=standard deviation
Table 2: Particle Size, Polydispersity Index And Entrapment Efficiency Of Optimised Formulations
Entrapment efficiency was determined by dialysis method. The prepared drug loaded gel cream (0.2 g) was diluted to 10 ml with chloroform/methanol (1:1) was placed inside the dialysis bag and it was suspended suitably in a beaker containing 100 ml diffusion medium (distilled water), which was constantly stirred at 37±0.5° using a magnetic stirrer for 4 h. Samples were withdrawn at various time intervals and assayed spectrophotometrically at 253 nm using ultraviolet (UV) spectrophotometer (Shimadzu‑UV Pharmaspec 1700, Japan). The time required to release the unentrapped drug was noted [20]. Entrapment efficiency was calculated by subtracting the amount of the drug dialyzed from the total drug used in the formulation (Table 2).
In vitro drug release studies
The in vitro skin permeation of ACV‑SLN formulation was studied using locally fabricated Franz diffusion cell with an effective permeation area and receptor cell volume of 1.0 cm2 and 10 ml, respectively. The temperature was maintained at 37±0.5°. The receptor compartment contained 10 ml PBS (Phosphate Buffer Solution) (pH 6.5) and was constantly stirred by a magnetic stirrer (Expo India Ltd., Mumbai, India) at 100 rpm. The hairless (80‑100 μm) rat skin from abdominal areas were obtained from MET college, Adgaon and stored at −20°. The skin was then carefully checked through a magnifying glass to ensure that samples were free from any surface irregularity such as tiny holes or crevices in the portion that was used for transdermal permeation studies. The skin was mounted on a receptor compartment with the stratum corneum side facing upward into the donor compartment. The gel cream formulation (250 μl) was applied on the skin in donor compartment, which was then covered with parafilm to avoid any evaporation process. Samples were withdrawn through the sampling port of the diffusion cell at predetermined time intervals over 24 h and analyzed for drug content by UV/Vis spectrophotometry at 254 nm, after that the receptor phase was immediately replenished with equal volume of fresh diffusion buffer. Experiment was conducted in triplicate for each study. Similar experiments were performed with plain ACV solution (1% aqueous dispersion) as well. Sink conditions were maintained throughout the experiment. The amount of ACV retained in the skin was determined at the end of the in vitro permeation experiment (24 h). The skin was washed with methanol and homogenized for 5 min with an electric stirrer. The resulting solution was centrifuged for 10 min at 7000 rpm. The supernatant was analyzed for drug content by UV/Vis spectrophotometry. The cumulative amount of drug permeated per unit area was plotted as a function of time; the steady‑state permeation rate (Jss) and lag time (LT, h) were calculated from the slope and x‑intercept of the linear portion, respectively.
Determination of drug content in the epidermis and dermis
The drug amount taken up in the epidermis and dermis was determined at 2, 4, 6, 8, 10 and 12 h after treating with the same method described in the previous section. The excessive SLNs loaded gel cream dispersion and marketed cream was removed and the skin samples with a penetration area of 3.8 cm2 were rinsed with alcohol and water to eliminate the ACV remained on the surface and gently dried with a cotton wool [19]. Heat‑separated membrane separation was carried out based on the reported method [20]. The skin samples were immersed in distills water at 60±1° for 2 min, and then the epidermis was removed from the dermis using a dull scalpel blade. The epidermis sample or dermis cut into small pieces was soaked in 5 ml of 0.4% perchloric acid solution for 24 h and subjected to homogenization with a Diax 900 homogenizer (Heidolph Electro, 2.8 Kelhaim, Germany) for 1 min and ultrasonication for 60 min in an ultrasound bath (CX‑250, Peking Medical Equipment’s Ltd., Peking, China), followed by centrifugal separation (0412‑1 Shanghai Surgical Instruments Co. Ltd., Shanghai, China). The drug extracted from epidermis or dermis was analyzed by UV/Vis spectrophotometry, respectively.
Storage/physical stability of SLNs
The ability of SLNs to retain the drug (drug retentive behavior) was assessed by keeping the SLN formulation at different temperatures, i.e., 5±3°, 25±2° (RT) and 40±2° for different periods of time (1, 30, 60, 90, and 180 days). The SLNs formulation was kept in sealed vials (10 ml capacity) after flushing with nitrogen. The stability of SLNs was also assessed quantitatively by monitoring size, zeta potential, and entrapment efficiency of the vesicles (Table 3).
| Particle size | Zeta potential | Drug content | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Initial | 5±3° | 25±2° | 40±2°/75% RH | Initial | 5±3° | 25±2° | 40±2°/75% RH | Initial | 5±3° | 25±2° | 40±2°/75% RH |
| 98.33± | 139.11± | 125.45± | 132.79± | −27.31± | −22.31± | −25.31± | −17.39± | 63.65± | 60.74± | 61.35± | 58.91± |
| 2.52 | 6.10 | 4.23 | 5.30 | 1.38 | 1.59 | 2.01 | 3.46 | 2.52 | 2.95 | 1.58 | 3.15 |
Data were expressed as mean±SD, n=3. FM=formulation material, RH=relative humidity, SD=standard deviation
Table 3: Stability Studies Of Formulation Fm‑3
Results and Discussion
ACV‑SLNs were prepared by high pressure homogenization method as reported by Friedman et al. The SLNs prepared using varying concentration of MCT and lecithin when examined by SEM appeared as spheroids with a spherical shape (fig. 1). Particle size, polydispersity index (PDI), and entrapment efficiency (Table 2) are the basic parameters on the basis of which the formulations were optimized.
The stability of the formulation was tested for the entrapment efficiency and the particle size by incubating the formulation at different temperatures viz. 5±3°, 25±2°/60% RH (RT) and 40±2°/75% RH for a period of 6 months (30, 60, 90, and 180 days). It was found that ACV‑SLNs exhibited a good stability during the period of 6 months. No significant changes in formulation clarity and degradation were found. The critical parameters monitored during accelerated testing are physical appearance, particle size, entrapment efficiency, and degradation of the drug. The mean diameter of ACV-SLNs was 132.79±5.3 nm and the entrapment efficiency was decreased by 4.74% when examined in accelerated conditions, i.e., 180 days at 40±2°/75% RH, whereas, the particle size increased to 139.11±6.1 nm and entrapment efficiency was reduced by 2.91% when store at 5±3° for 180 days. This indicates a marginal particle size enhancement and permissible drug leaching on storage.
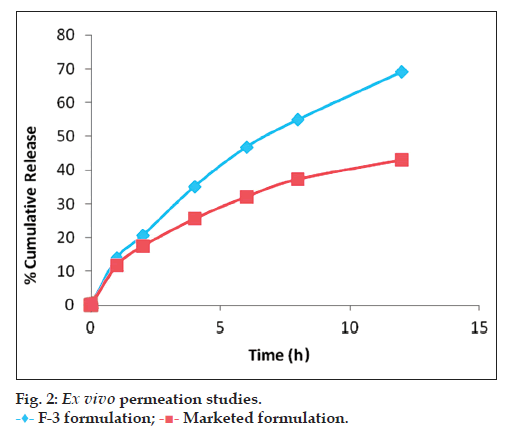
Ex vivo permeation studies were performed to compare the cumulative drug permeation from different formulations (Marketed cream and SLNs loaded gel cream) all having the same quantity 0.5% of ACV as shown in fig. 2. Permeability coefficient (Kp) values of ACL loaded SLNs gel cream and ACV marketed cream were found to be 7.57×10−3 and 8.03×10−3, respectively.
It indicated that the steady‑state flux (Jss) of marketed cream and SLNs loaded gel cream were 11.75±1.21 and 14.06±2.25 g/cm2/h, respectively. The cumulative amounts of ACV from cream and SLNs at 12 h after dosing were 43.12±3.13 and 69.13±3.37 g/cm2/h, respectively. In other words, the cumulative amount of ACV from SLNs incorporated gel cream penetrating through the rat skin was more than 2‑fold that of the marketed cream at 12 h, which was coincident with previous reports [21]. The cumulative amounts of ACV in the epidermis from the marketed cream and the SLNs incorporated gel cream up to 12 h after administration are shown in fig. 2. There was no significant difference in cumulative amounts of ACV in the epidermis between the gel cream and SLNs (P>0.05) while the SLNs significantly increased the cumulative uptake of ACV in dermis (P>0.05). The amount of ACV penetrated into dermis from SLNs at 12 h were approximately two times that of commercial cream. The small size and close interaction between SLNs and the stratum corneum are the possible reasons for the increased drug amount penetrating into the viable skin via SLNs. The similar findings were reported by Maia et al. and Sivaramakrishnan et al. [16,22]. The dermal location of SLNs is desirable to interfere with the establishment of the latent HSV infection and the effect of SLNs would prevent the diseases associated with HSV and inhibit human‑to‑human transmission of the virus [17].
On increasing the MCT concentration, an increase in particle size with less homogeneity was obtained. Though with an increase in lipid emulsifier lecithin concentration the particle size decreased, but beyond a certain limit the PDI of the particles was found to increase that compelled us to optimize 1:9 ratio of lecithin:MCT, respectively. At this particular concentration of MCT and lecithin entrapment efficiency was also found to be highest, i.e., 63.65±2.52%, justifying the optimized formulation. As with lowest particle size there is more opportunity for the carrier system to penetrate deeper into the skin passing through the tough barrier of the stratum corneum, with high payload.
The increase amount of drug in the dermis is also related to the occlusion properties. An adhesive layer occluding the skin surface is formed after the evaporation of water from the SLNs dispersion applied to the skin surface. Then the hydration of the stratum corneum can increase, which can facilitate drug penetration into deeper skin strata by reducing corneocyte packing and widening the intercorneocytes gaps [23]. Occlusive effects appear strongly related to particle size. Nanoparticles turned out 15‑fold more occlusive than microparticles, and particles smaller than 400 nm were found to elicit more intense effect [24]. So, ACV-SLNs with the mean diameter of 98.33 nm should have the ability to form an occlusive film on the surface of skin, increase the hydration of the stratum corneum, and improve the permeation of penciclovir into skin.
Acknowledgements
The authors thank Indoco Remedies Limited, Mumbai, for providing Acyclovir and Lipoid, Germany as gift samples from Lipoid S 100 for this work. They also thank MET Institute of Pharmacy, Nashik for providing required facilities to carry out this research work.
References
- Parry GE, Dunn P, Shah VP, Pershing LK. Acyclovir bioavailability in human skin. J Invest Dermatol 1992;98:856‑63.
- Zanowiak P, Jacobs MR. In: Laitin SC, editor. Handbook of Nonprescription Drugs. Washington, DC: American Pharmaceutical Association; 1982. p. 523‑9.
- Williams A. Transdermal and Topical Drug Delivery. 1st ed. London: Pharmaceutical Press; 2003.
- El Maghraby GM, Williams AC, Barry BW. Skin hydration and possible shunt route penetration in controlled estradiol delivery from ultradeformable and standard liposomes. J Pharm Pharmacol 2001;53:1311‑22.
- Barry BW. Novel mechanisms and devices to enable successful transdermal drug delivery. Eur J Pharm Sci 2001;14:101‑14.
- Bouwstra JA, Honeywell‑Nguyen PL. Skin structure and mode of action of vesicles. Adv Drug Deliv Rev 2002;54:S41‑55.
- Volpato NM, Santi P, Colombo P. Iontophoresis enhances the transport of acyclovir through nude mouse skin by electrorepulsion and electroosmosis. Pharm Res 1995;12:1623‑7.
- Volpato NM, Nicoli S, Laureri C, Colombo P, Santi P. In vitro acyclovir distribution in human skin layers after transdermal iontophoresis. J Control Release 1998;50:291‑6.
- Spruance SL, Crumpacker CS. Topical 5 percent acyclovir in polyethylene glycol for herpes simplex labialis. Antiviral effect without clinical benefit. Am J Med 1982;73:315‑9.
- Cooper ER, Merritt EW, Smith RL. Effect of fatty acids and alcohols on the penetration of acyclovir across human skin in vitro. J Pharm Sci 1985;74:688‑9.
- Berthold A, Cremer K, Kreuter J. Collagen microparticles: Carriers for glucocorticosteroids. Eur J Pharm Biopharm 1998;45:23‑9.
- Chen H, Chang X, Du D, Liu W, Liu J, Weng T, et al. Podophyllotoxin‑loaded solid lipid nanoparticles for epidermal targeting. J Control Release 2006;110:296‑306.
- Santos Maia C, Mehnert W, Schaller M, Korting HC, Gysler A, Haberland A, et al. Drug targeting by solid lipid nanoparticles for dermal use. J Drug Target 2002;10:489‑95.
- Liu J, Hu W, Chen H, Ni Q, Xu H, Yang X. Isotretinoin‑loaded solid lipid nanoparticles with skin targeting for topical delivery. Int J Pharm 2007;328:191‑5.
- Mei Z, Chen H, Weng T, Yang Y, Yang X. Solid lipid nanoparticle and microemulsion for topical delivery of triptolide. Eur J Pharm Biopharm 2003;56:189‑96.
- Sivaramakrishnan R, Nakamura C, Mehnert W, Korting HC, Kramer KD, Schäfer‑Korting M. Glucocorticoid entrapment into lipid carriers: Characterisation by parelectric spectroscopy and influence on dermal uptake. J Control Release 2004;97:493‑502.
- Smith RL, Morroni J, Wilcox CL. Lack of effect of treatment with penciclovir or acyclovir on the establishment of latent HSV‑1 in primary sensory neurons in culture. Antiviral Res 2001;52:19‑24.
- Friedman D, Carmei Y, Schwartz J, Haim A. Topical and transdermal delivery system utilizing submicron oil spheres. United States Patent (US 6,113,921).
- Dubey V, Mishra D, Asthana A, Jain NK. Transdermal delivery of a pineal hormone: Melatonin via elastic liposomes. Biomaterials 2006;27:3491‑6
- Bhaskar K, Anbu J, Ravichandiran V, Venkateswarlu V, Rao YM. Lipid nanoparticles for transdermal delivery of flurbiprofen: Formulation, in vitro, ex vivo and in vivo studies. Lipids Health Dis 2009;8:6.
- Mei Z, Chen H, Weng T, Yang Y, Yang X. Solid lipid nanoparticle and microemulsion for topical delivery of triptolide. Eur J Pharm Biopharm 2003;56:189‑96
- Maia CS, Mehnert W, Schäfer‑Korting M. Solid lipid nanoparticles as drug carriers for topical glucocorticoids. Int J Pharm 2000;196:165‑7.
- Cevc G. Lipid vesicles and other colloids as drug carriers on the skin. Adv Drug Deliv Rev 2004;56:675‑711.
- Wissing SA, Müller RH. Solid lipid nanoparticles (SLN) – A novel carrier for UV blockers. Pharmazie 2001;56:783‑6.

 F‑3 formulation;
F‑3 formulation; Marketed formulation.
Marketed formulation.



