- *Corresponding Author:
- H. Mourya
School of Studies in Pharmaceutical Sciences, Jiwaji University, Gwalior, Madhya Pradesh 474001,
India
E-mail: Hemant35143@gmail.com
| Date of Received | 01 July 2021 |
| Date of Revision | 25 September 2022 |
| Date of Acceptance | 16 February 2023 |
| Indian J Pharm Sci 2023;85(1):189-198 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
The purpose of this investigation is to formulate and evaluate the antihypertensive drug propranolol hydrochloride sustained-release bilayer tablets. In this formulation, one layer provides a loading dose through the immediate release of the drug and the other layer provides a maintenance dose for up to 12 h through controlled release. Different quantities of polymers such as Kyron T-314, Hydroxypropyl methylcellulose- K4M, and ethyl cellulose were used to make bi-layer tablets by direct compression. The compatibility study of pharmaceutical excipients was conducted through Fourier transform infrared spectroscopy studies and no interaction was found. The pre-compression parameter for the angle of repose, bulk density, tapped density and compressibility index was assessed on the produced granules and the findings were good. The tablets were evaluated for the post-compression parameters for thickness, hardness, friability and in vitro release studies. In vitro dissolution study was approved out for 12 h using United States Pharmacopeia dissolution apparatus I using phosphate buffer of pH 1.2 and 6.8 as dissolution medium. Hydroxypropyl methylcellulose- K4M and ethylcellulose were used in combination in all formulations but optimized formulation propranolol hydrochloride tablet 4 showed a higher rate of drug release up to 12 h as compared to the other formulation and optimized formulations propranolol hydrochloride tablet 4 follows the Higuchi model with non-fickian diffusion based on regression coefficient of the kinetics data of cumulative drug release from the dosage form.
Keywords
Propranolol hydrochloride, sustained release bilayer tablet, hydroxypropyl methylcellulose, ethyl cellulose
For decades, oral drug delivery has been recognized as the most widely used route of administration in the exploration of systemic drug delivery[1]. When administered in the traditional dosage form, many orally delivered medications have low bioavailability. i.e., the degree and level of absorption of the medications are less than desirable. Absorption of some medications can be as low as 30 % or less of the orally managed dosage. Furthermore, poorly absorbed drugs have a lot of variation in bioavailability within and also withinsubjects. This dilemma could be solved by using a revamped release drug delivery system with a longer stomach residence period[2]. Furthermore, conventional dosage forms cause a wide variety of drug concentration fluctuations in the bloodstream and tissues, resulting in decreased or lost drug potency or a rise in the rate of side effects, resulting in unwanted toxicity and inefficiency. Sustained or managed drug delivery systems, on the other hand, will reduce dosing frequency while still increasing drug efficacy by localizing the drug at the site of operation, lowering the dosage needed and ensuring uniform drug delivery[3]. An Immediate Release (IR) layer and a Sustained Release (SR) layer are used in a bilayer tablet, which is a recent idea for the effective production of a SR formulation with different features to provide a means of a successful drug delivery system. The IR layer maintains therapeutically efficient plasma drug concentrations for a limited period, while the SR layer maintains uniform drug levels for an extended period, reducing dosing cycles and side effects increasing the safety boundary for highly potent medications and thereby improving patient compliance. Hypertension is characterized as a systolic pressure greater than 140 mm Hg and a diastolic pressure greater than 90 mm Hg with a systolic pressure greater than 140 mm Hg. It can induce blood flow changes in the back of the eye (retina), irregular heart muscle thickening, kidney dysfunction, brain injury, myocardial infarction (heart attacks), heart failure, artery aneurysms (e.g. aortic aneurysm) and peripheral arterial disease[4]. Propranolol hydrochloride is a nonselective beta-adrenergic blocker that is commonly used to treat asthma, angina pectoris and a variety of other cardiovascular conditions[5]. Propranolol hydrochloride has poor oral bioavailability (15 %-23 %). It is a crystalline solid that is stable and soluble in water and ethanol. It has a molecular weight of 295.80[6]. It's an extremely water-soluble compound with a 3-5 h cellular half-life and the normal dosage is 40 mg thrice a day. This necessarily requires a high frequency of administration, resulting in oscillations in plasma medication concentrations. To lower the frequency of administration while increasing potency and oral bioavailability, a SR dosage type must be developed[7].
Materials and Methods
Propranolol Hydrochloride is taken as a gift sample from IPCA Lab. Ltd. Ratlam (m.p) India. Hydroxypropyl Methylcellulose-K4M (HPMCK4M) and Ethylcellulose from Loba Chemie Pvt. Ltd. (Mumbai), Starch from Hi-Media Laboratories Pvt. Ltd. (Mumbai), Microcrystalline cellulose (MCC) from Hi-Media Laboratories Pvt. Ltd. (Mumbai), Magnesium stearate from Central Drug House Pvt. Ltd. (New Delhi). All other chemicals used were of analytical grade.
Fourier Transform Infrared Spectroscopy (FTIR):
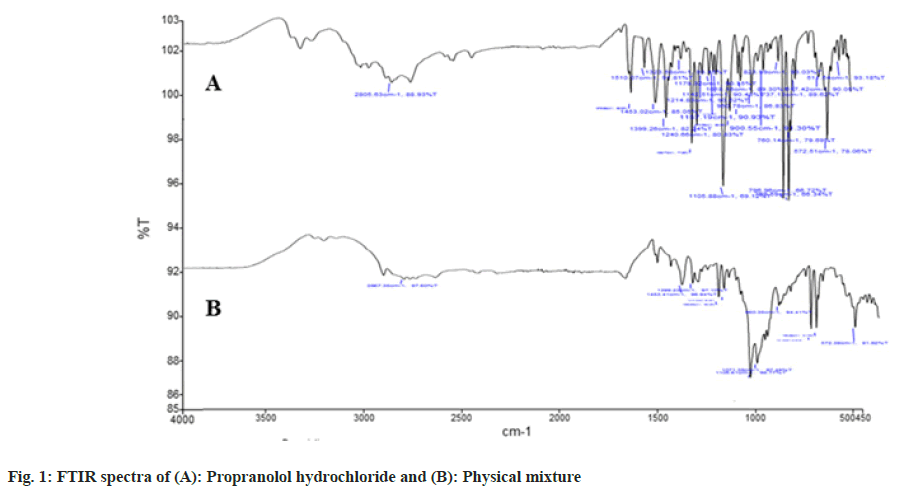
The functional groups in a molecule are identified using FTIR. The medication was scanned at 4 mm/s with a resolution of 2 cm on a wavenumber range of 400- 4000 cm-1 on a Potassium bromide (KBr) disc. Since the drug and polymer are nearby during the preparation of a bilayer pill, they can interfere, resulting in drug instability. Pre formulation experiments on drugpolymer interactions are also crucial in choosing the right polymers. The stability of propranolol hydrochloride and selected polymers was determined using FTIR spectroscopy (Perkin Elmer Model No. Spectrum Two Serial no. 105627 FT-IR). Separately, the actual medication and the drug with excipients were scanned (fig. 1)[8].
Differential Scanning Calorimetry (DSC):
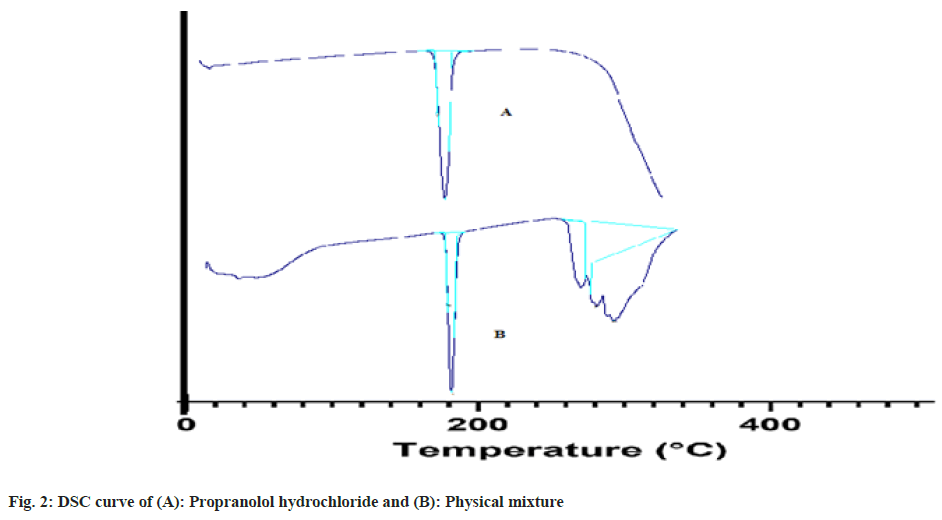
DSC measurements of propranolol hydrochloride were approved out using a thermo tropic transformation testing tool (DSC 60 plus Shimadzu Japan). Empty aluminum pans were used as controls and samples were carefully put in another aluminum pan. The test was carried out in an inert environment at a rate of 10° per minute in a temperature range of 30° to 305° (fig. 2)[9].
Pre-compression parameters:
The powder blend was evaluated for pre-compression properties like angle of repose, bulk density, tapped density, Carr’s index, Hausner’s ratio[10].
Preparation of bilayer tablet:
The tablets are manufactured by compressing powdered ingredients without changing their physical properties. Direct compression is usually used for crystalline materials with good physical features like flow, compressibility and so on. Direct compression has several advantages, including time savings, operational safety and cheap cost.
Dose calculation:
For sustained drug release up to 12 h, the immediate dose of drug was calculated from total dose of propranolol hydrochloride extended release tablet, which was 80 mg. According pharmacokinetic data[11], DT=AD(1+0.693×t/t1/2) Where, DT=Total dose, AD=Adult dose, t=Total time period for which SR is required, t1/2=Half-life of drug. Halflife of propranolol hydrochloride ranges from 3 h to 5 h. For example, propranolol hydrochloride: 80=Dose[1+(0.693×12)/3)], Dose=21.208 mg propranolol hydrochloride; Propranolol hydrochloride: 80=Dose [1+(0.693×12)/5)], Dose=30.039 mg Propranolol hydrochloride.
According to dose calculation, IR dose of the drug can be taken in an in-between range of 21.208 mg to 30.039 mg for the preparation of bilayer tablets; thus 22 mg of propranolol hydrochloride was taken in the IR layer and 58 mg of Propranolol hydrochloride was taken in SR layers.
Formulation of the IR layer:
Formulation of the IR layer has been done as follows. The powder combination was blended for 15 min to achieve a uniform distribution of the medicine in the formulation after the composition of the IR layer was weighed properly and added to the blender. The mixture was blended for 2 min with talc and magnesium stearate before being stored in a desiccator until needed (Table 1).
| S. no. | Ingredients (mg) | Formulation of IR layer (mg) |
|---|---|---|
| 1 | Propranolol hydrochloride | 22 |
| 2 | Kyron T-314 | 4 |
| 3 | Starch powder | 10 |
| 4 | Microcrystalline cellulose | 59.8 |
| 5 | Talc | 2 |
| 6 | Magnesium stearate | 2 |
| 7 | Color | 0.2 |
| Total weight | 100 | |
Table 1: Formulation of LR Layer
Formulation of the SR layer:
Formulation of the SR layer has been done as follows. The powder combination was blended for 20 min to achieve a uniform distribution of the medicine in the formulation after the composition of the SR layer was correctly weighed and added to the blender (Table 2).
| S. no. | Ingredients (mg) | Formulation of SR layer | |||||||
|---|---|---|---|---|---|---|---|---|---|
| PHT 1 (mg) | PHT 2 (mg) | PHT 3 (mg) | PHT 4 (mg) | PHT 5 (mg) | PHT 6 (mg) | PHT 7 (mg) | PHT 8 (mg) | ||
| 1 | Propranolol hydrochloride | 58 | 58 | 58 | 58 | 58 | 58 | 58 | 58 |
| 2 | Hpmc-K4M | 5 | 10 | 15 | 20 | 25 | 30 | 35 | 40 |
| 3 | Ethylcellulose | 40 | 35 | 30 | 25 | 20 | 15 | 10 | 5 |
| 4 | Starch powder | 20 | 20 | 20 | 20 | 20 | 20 | 20 | 20 |
| 5 | Microcrystalline cellulose | 69 | 69 | 69 | 69 | 69 | 69 | 69 | 69 |
| 6 | Talc | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 |
| 7 | Magnesium stearate | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 |
| Total weight | 200 | 200 | 200 | 200 | 200 | 200 | 200 | 200 | |
Table 2: Formulation of SR Layer
Compression of bilayer tablet:
The compression of the bilayer tablet was done manually using single rotatory compression machines (CLIT Single Rotatory 16). An accurately weighed amount of SR powder mixture was fed manually into the die cavity. SR was compressed at mild compression force (2-3 kg/cm2). After that, an accurately weighted IR powder mixture was manually fed into the die on a SR layer and compressed using 9 mm circular punches. Both the layers were identified based on color since the IR layer had pink color and the SR layer has a white color (Table 3).
| Formulation code | Angle of repose | Bulk density | Tapped density | Compressibility index (%) | Hausner’s ratio |
|---|---|---|---|---|---|
| PHT 1 | 32.68±0.36 | 0.47±0.04 | 0.554±0.01 | 13.24±0.65 | 1.10±0.06 |
| PHT 2 | 31.38±0.60 | 0.47±0.01 | 0.558±0.03 | 13.56±0.42 | 1.11±0.14 |
| PHT 3 | 33.51±0.54 | 0.48±0.06 | 0.551±0.05 | 11.43±0.64 | 1.11±0.21 |
| PHT 4 | 30.87±0.12 | 0.48±0.05 | 0.559±0.02 | 12.49±0.32 | 1.10±0.07 |
| PHT 5 | 34.71±0.71 | 0.49±0.03 | 0.553±0.01 | 12.23±0.49 | 1.12±0.14 |
| PHT 6 | 32.26±0.18 | 0.49±0.07 | 0.569±0.04 | 13.78± 0.42 | 1.13±0.18 |
| PHT 7 | 33.19±0.96 | 0.50±0.02 | 0.555±0.02 | 11.56±0.32 | 1.12±0.07 |
| PHT 8 | 34.56±0.73 | 0.47±0.03 | 0.563±0.04 | 12.98±0.14 | 1.11±0.21 |
Note: Values are expressed as mean±Standard Deviation (SD), n=3
Table 3: Precompression Parameters of Powder Blend
Post compression parameters:
Thickness: Using a digital Vernier scale, the thickness of 20 pre-weighed tablets from each batch was assessed and the normal thickness in mm was calculated. The thickness of the pill should be kept within a 3 % range of a standard[12].
Hardness: It was determined by using Monsanto hardness tester and the average pressure of (kg/cm2) applied for crushing the tablet was determined[13].
Friability: It was determined by first weighing 10 tablets and placing them in Roche Friabilator, which was rotated for 100 revolutions at 25 rpm. After dusting, the total remaining mass of the tablets was recorded and the percent friability was calculated[14].
Weight variation: According to United States Pharmacopeia (USP) 20 tablets were weighed individually which were randomly selected for the determination of weight variation. The mean and standard deviation were determined[15].
Drug content assay: Ten pills were coarsely pulverized and 100 mg of propranolol hydrochloride was properly weighed and placed in a 100 ml volumetric flask, followed by 70 ml of buffer pH 1.2 (0.01 N Hydrochloric acid (HCl)). For 10 min, the flask was shaken. Finally, using the same buffer solution, the volume was brought up to par. The resulting solution was filtered completely using Whatman filter paper (No.41), and 1 ml of the filtrate was diluted to 100 ml with the same buffer solution and analyzed for propranolol hydrochloride content at 290 nm using a double beam Ultra Violate (UV)/ Visible spectrophotometer (Shimadzu 1800, Japan) and 0.01 N HCl as a blank (Table 4)[16].
| Formulation code | Thickness (mm) | Hardness (Kg/cm2) | Friability (%) | Weight variation (mg) | % Drug content |
|---|---|---|---|---|---|
| PHT 1 | 4.06±0.32 | 5.21±0.12 | 0.12±0.09 | 299.89±0.29 | 93.25±0.54 |
| PHT 2 | 4.18±0.49 | 4.87±0.35 | 0.19±0.14 | 299.92±0.21 | 94.29±0.36 |
| PHT 3 | 4.59±0.13 | 4.23±0.89 | 0.25±0.12 | 299.90±0.12 | 96.13±0.12 |
| PHT 4 | 4.02±0.46 | 4.12±0.18 | 0.31±0.13 | 300.01±0.26 | 98.06±0.48 |
| PHT 5 | 4.49±0.56 | 4.01±0.65 | 0.26±0.06 | 299.96±0.21 | 97.14±0.32 |
| PHT 6 | 4.32±0.03 | 4.32±0.56 | 0.19±0.12 | 299.94±0.30 | 95.32±0.64 |
| PHT 7 | 4.21±0.09 | 4.17±0.23 | 0.21±0.09 | 299.96±0.91 | 96.14±0.47 |
| PHT 8 | 4.11±0.29 | 4.19±0.12 | 0.07±0.04 | 299.90±0.29 | 95.12±0.17 |
Note: Values are expressed as mean±SD, n=3
Table 4: Post Compression Parameter
In vitro drug release study and release kinetics:
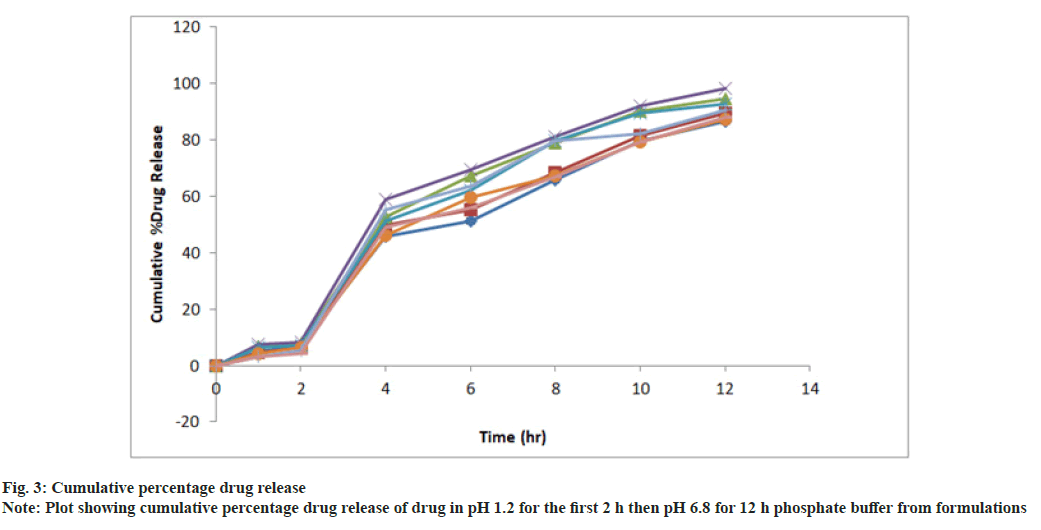
In vitro drug release study of all the developed formulations were tested utilizing a paddle-type tablet dissolving apparatus (USP I type) spinning at 50 rpm. The study was performed in phosphate buffer pH 1.2 for the first 2 h and then it was replaced by phosphate buffer 6.8 pH and the paddle was rotated continuously for up to 12 h dissolving medium was kept at 37° (fig. 3 and Table 5). Samples were withdrawn at definite intervals, diluted appropriately and evaluated for cumulative drug release using a UV-Visible spectrophotometer at 275 nm. The proportion of propranolol hydrochloride dissolved in buffer was determined and the graph was plotted as percent cumulative drug release vs. time.
| Time | Cumulative % drug Release | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| pH | F1 | F2 | F3 | F4 | F5 | F6 | F7 | F8 | |
| 1 | 1.2 | 04.89±0.51 | 04.46±0.14 | 06.90±0.35 | 07.47±0.49 | 06.47±0.36 | 04.23±0.29 | 03.29±0.32 | 03.05±0.71 |
| 2 | 1.2 | 07.14±0.12 | 06.39±0.38 | 07.82±0.14 | 08.31±0.31 | 07.28±0.17 | 06.49±0.87 | 05.31±0.13 | 04.23±0.29 |
| 4 | 6.8 | 45.62±0.97 | 49.63±1.56 | 52.79±1.02 | 58.98±0.12 | 51.14±1.13 | 46.28±0.21 | 55.38±0.46 | 49.12±0.74 |
| 6 | 6.8 | 51.31±1.05 | 55.19±0.53 | 67.28±0.34 | 69.41±1.09 | 62.18±0.93 | 59.67±1.41 | 63.76±0.18 | 56.17±1.32 |
| 8 | 6.8 | 65.78±0.41 | 68.47±0.28 | 79.13±0.03 | 81.29±0.97 | 79.64±0.49 | 67.49±.0.38 | 79.68±0.97 | 66.87±0.43 |
| 10 | 6.8 | 79.54±0.03 | 81.55±0.78 | 90.28±0.42 | 92.14±0.42 | 89.49±0.31 | 79.45±0.17 | 82.16±0.41 | 79.38±0.19 |
| 12 | 6.8 | 86.71±0.87 | 89.64±0.17 | 94.54±0.69 | 98.36±1.67 | 92.71±0.21 | 87.21±0.29 | 90.56±0.38 | 88.15±0.48 |
Note: Values are expressed as mean±SD, n=3
Table 5: Cumulative % Drug Release
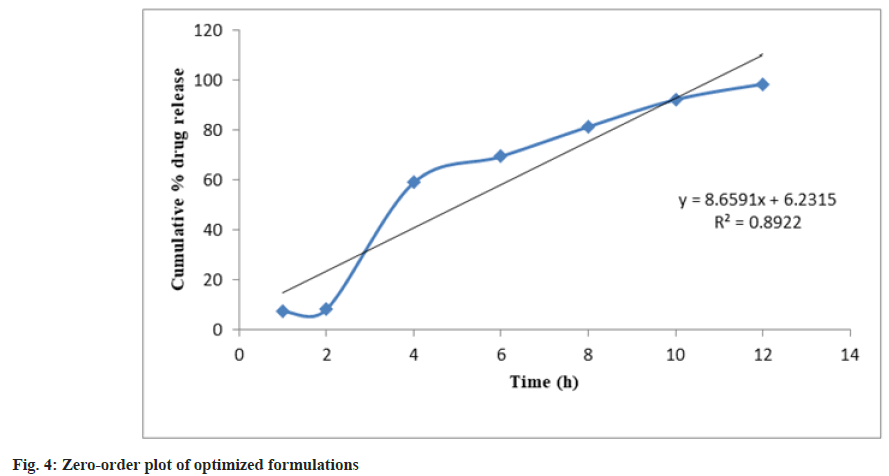
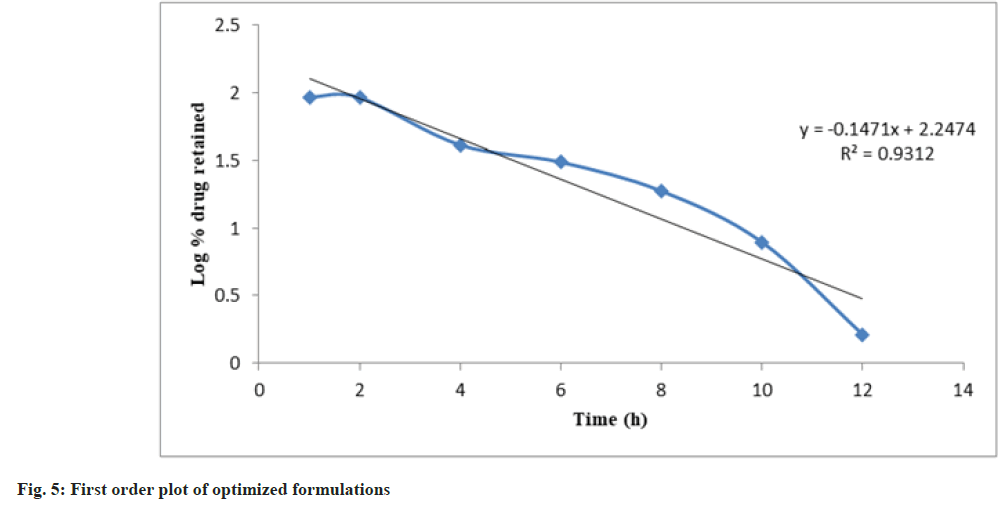
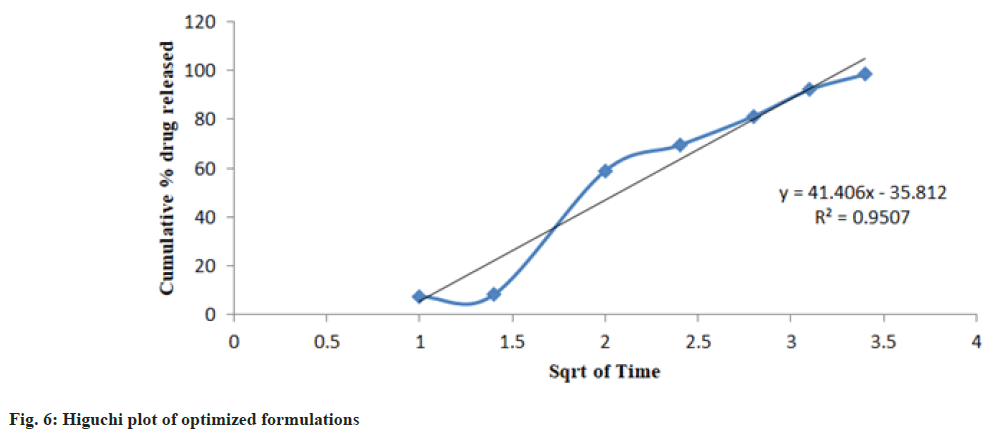
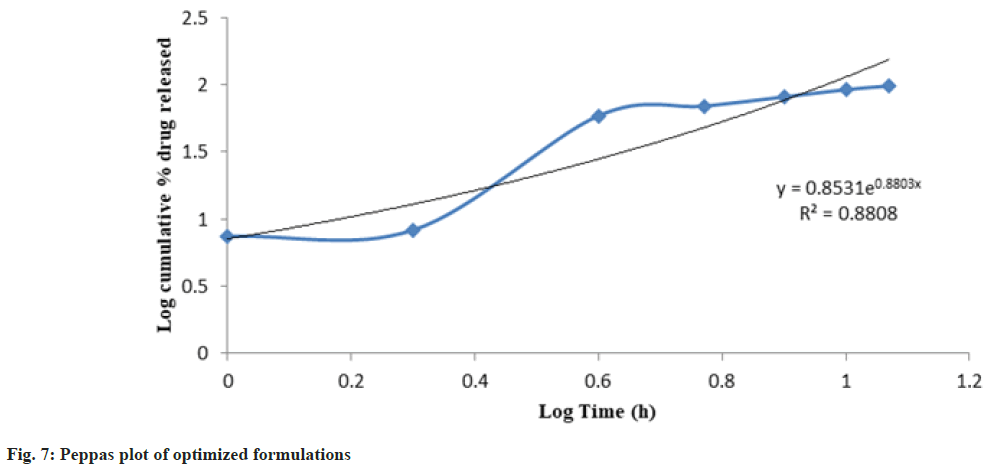
Release kinetics studies: In vitro release data was applied to kinetic models such as zero-order (Cumulative percent drug release vs. time), firstorder (Log Mean percent drug unreleased vs. time), Higuchi (Mean percent cumulative drug release vs. square root of time), and Korsmeyer-Peppas (Log mean percent cumulative drug release vs. Log time) to study the release kinetics and mechanism as shown in fig. 4-fig. 7 and Table 6[17].
| Formulation code | Zero-order | First-order | Higuchi | Korse Meyer-Peppas | |
|---|---|---|---|---|---|
| r2 | r2 | r2 | r2 | N | |
| PHT 4 | 0.8922 | 0.9312 | 0.9507 | 0.8808 | 0.8803 |
Table 6: Drug Release Kinetics
Stability studies:
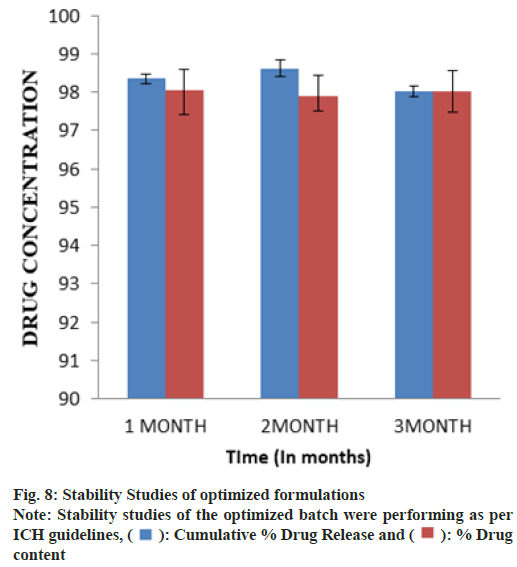
The investigation was carried out on a Stability Control Chamber (Remi Instruments Ltd.) at 40°/75 % Relative Humidity (RH) for 3 mo of the optimized formulation. The sample was put in an accelerated stability chamber after being wrapped in coated aluminum foil. Inspections took place at predetermined intervals of 1, 2 and 3 mo. The tablets were tested for a variety of physicochemical properties[18,19].
Results and Discussion
The obtained infrared spectra of propranolol hydrochloride show peaks at 2805 cm-1 which correspond to a secondary amine group at 3300 cm-1 due to the hydroxyl group(secondary) at 1105 cm-1 due to the aryl alkyl ether stretching band and a peak at 796 cm-1. However, additional peaks were absorbed in physical mixtures which could be due to the presence of polymers and indicated that there was no chemical interaction between propranolol hydrochloride and other excipients.
The DSC thermo gram of the pure drug (propranolol hydrochloride) displayed an endothermic peak of 169° corresponding to its melting point and DSC thermo grams of the physical mixture of drug and excipients showed the melting peak of the drug at 167.85°.
Before compression powder blend was evaluated for their characteristic parameters such as angle of repose, bulk density, Tapped density, Compressibility index and Hausner’s ratio. Pre-compression Parameters like the angle of repose was found to be within the range of 30.87±0.12 to 34.56±0.73 which indicate the good flow ability. The bulk density of the powder is an important parameter in the compressibility of the powder. The bulk density was found between 0.47±0.01 to 0.50±0.02 gm/cm3. The tapped density was found between 0.551±0.05 to 0.569±0.04 gm/cm3. Carr's index is an indicator of compressibility. A value below 21 % shows fair to passable compressibility. It was found to be 11.43±0.64 to 13.78±0.42. Hausner's ratio is another parameter indicating the flow properties. The value of a ratio below 1.25 indicates good flow while above 1.25 indicates poor flow. It was found to be 1.10±0.06 to 1.13±0.18 (Table 3).
The prepared SR formulations were estimated for drug-excipients interaction hardness, friability, weight variation, drug content uniformity, in vitro drug release. The results are quoted in a different section of the result and discussion chapter. Various evaluation parameters, we summarize as from IR and physical observation it was observed that there was no significant drugexcipient interaction. The melting point of propranolol hydrochloride was found to be in a range between 161°-164°. Tablet thickness (n=3) was almost uniform in all formulations and values of tablets ranged from 4.02±0.46 mm to 4.59±0.13 mm. Weight variation of tablets ranged from 299.89±0.29 to 300.01±0.26 mg. The hardness of all formulations was in ranged from 4.01±0.65 to 5.21±0.12 kg/cm2. The values of friability of all formulations ranged from 0.07±0.04 to 0.31±0.13 %. The percentage drug content of all the formulated tablets was found within 93.25±0.54 to 98.06±0.48. On the basis of result of drug content studies, the formulation Propranolol Hydrochloride Tablet (PHT) 4 was selected as an optimized formulation for further examination.
In vitro drug release study was approved out using a tablet dissolution test apparatus (single). The release of propranolol hydrochloride from prepared formulation followed the order PHT4>PHT3>PHT5>PHT7>PHT2>PHT8>PHT6> PHT1 respectively. HPMC-K4M and ethyl cellulose was used in combination to get the enhanced effect for SR. PHT1, PHT2 exhibited 86.71 %, 89.64 % release due to an increase in the concentration of ethyl cellulose. The formulation PHT3, PHT4, PHT5 shows drug release 94.54 %, 98.36 %, 92.71 %. Because of the increase in the concentration of HPMC-K4M and ethyl cellulose in the formulation it will give an increased combined effect. PHT6, PHT7, PHT8 showed less drug released 87.21 %, 90.56 %, 88.15 % over 12 h, due to an increase in the concentration of HPMC-K4M in the formulation and decreased concentration of ethyl cellulose.
The optimized formulation PHT4 showed a higher rate of drug release up to 12 h as compared to the other formulation. Due to the combined effect of HPMCK4M with hydrophilic effect and ethyl cellulose and with hydrophobic effect to get an improved rate of drug release. The optimized formulations PHT4 follows the Higuchi model with non-fickian diffusion based on the regression coefficient of the kinetics data of cumulative drug release from the dosage form.
The stability study of the optimized batch was performing as per International Conference on Hormonisation (ICH) guidelines and when put in a stability chamber at temperature and humidity conditions of 40°/75 % RH reveals no significant changes in physical parameters, drug content and dissolution over a time of 3 mo as shown in fig. 8.
The present study has been a satisfactory attempt to formulate a SR drug delivery system of propranolol hydrochloride, an orally administrated anti-hypertensive drug with improves its oral bioavailability and giving SR of the drug for a prolonged period.
Bilayer tablets have been successfully developed and optimized using a direct compression method. SR propranolol hydrochloride layer was formulated using a combination of hydrophilic and hydrophobic polymers and IR was developed using Kyron T-314. Bilayer tablets showed good physicochemical attributes and were found to be stable under accelerated stability conditions. The combination of HPMC-K4M and ethyl cellulose successfully controlled the release of propranolol hydrochloride from the bilayer tablet. When concentration of ethylcellulose increases then its shows less drug release similarly on increases HPMC-K4M concentration drug release decreases but when we took both HPMC-K4M and ethylcellulose in equal quantity then it shows adequate drug release. A suggesting higuchi model for drug dissolution profile was approved via combining both HPMC-K4M and ethylcellulose and on increasing the concentration of both the components the release rate also increases. The formulation can be subjected to pharmacokinetic studies and clinical trials in the future for better management of hypertension medication and enhancing patient compliance.
In conclusion, the study was a successful attempt to develop a SR drug delivery system for propranolol hydrochloride, an orally taken hypertension medication, with the goal of enhancing oral bioavailability and ensuring long-term drug release. Several polymers, including HPMC-K4M and ethylcellulose, could be employed to construct a SR drug delivery system for propranolol hydrochloride using the direct compression method. The tablets were able to deliver the medication throughout a 12 h period.
Conflict of interests:
The authors declared no conflict of interests.
References
- Dandare MS, Sarage RD, Bhaskaran S. Bilayer tablet: A Novel approach for immediate release of telmisartan and hydrochlorthaizide combination. Int J Pharm Technol 2012;4(1):3970-83.
- Mukhopadhyay S, Goswami L, Satheesh Madhav NV, Upadhyaya K. Formulation and evaluation of floating bioadhesive tablets of ciprofloxacin hydrochloride by direct compression technique. Int J Pharm Pharm Sci 2010;2(3):113-5.
- Kumar KK, Mahesh M, Sasikanth K. Design, Development and characterization of sustained release of metformin and gliclazide bi-layered tablets. Int J Biopharm 2010;1(2):67-71.
- Medicine Net.in. 2013.
- Derle D, Joshi O, Pawar A, Patel J, Jagadale A. Formulation and evaluation of buccoadhesive bi-layer tablet of propranolol hydrochloride. Int J Pharm Pharm Sci 2009;1(1):206-12.
- Cid E, Mella F, Lucchini L, Cárcamo M, Monasterio J. Plasma concentrations and bioavailability of propranolol by oral, rectal and intravenous administration in man. Biopharm Drug Dispos 1986;7(6):559-66.
[Crossref] [Google Scholar] [PubMed]
- Ray D, Prusty AK. Designing and in vitro studies of gastric floating tablets of tramadol hydrochloride. Int J Appl Pharm 2010;2(4):12-6.
- Joshi R, Garud N. Development, optimization and characterization of flurbiprofen matrix transdermal drug delivery system using Box–Behnken statistical design. Future J Pharm Sci 2021;7(1):1-8.
- Akram W, Garud N. Design expert as a statistical tool for optimization of 5-ASA-loaded biopolymer-based nanoparticles using Box Behnken factorial design. Future J Pharm Sci 2021;7(1):1-7.
- Patric Sinko J, Martin’s. Physical pharmacy and pharmaceutical sciences. 6th ed. India: Wolters Kluwer; 2006. p. 442-67.
- Goodman and Gilman's. The pharmacology basis of therapeutics. London: McGraw-Hill medical publishing division; 2006. p. 1884.
- Remington JP. Remington: The science and practice of pharmacy. Lippincott Williams & Wilkins; 2006.
- Carter SJ. Cooper and Gunn’s. Tutorial Pharmacy. New Delhi: CBS Publishers and Distributors; 1986. p. 220-5.
- Cooper J, Gunn C. Powder flow and compaction. Tutorial Pharmacy. New Delhi, India: CBS Publishers and Distributors; 1986;6:211-33.
- Indian Pharmacopeia. 2007 ed. Delhi: The Controller of Publications, Government of India, Ministry of Health and Family Welfare; 2007.
- Senthilnathan B, Rupenagunta A. Formulation development and evaluation of venlafaxine hydrochloride or dispersible tablets. Int J Pharm Sci Res 2011;2(4):913.
- Mohd AH, Rao NG, Avanapu SR. Matrix-mini-tablets of lornoxicam for targeting early morning peak symptoms of rheumatoid arthritis. Iran J Basic Med Sci 2014;17(5):357-69.
[Google Scholar] [PubMed]
- Kanvinde SA, Kulkarni MS. Stability of oral solid dosage forms–A global perspective. Pharm Times 2005;37(5):9-16.
- Cartensen JT. Drug Stability Principles and Practices. 2nd ed. Marcel Dekker Inc; 1995.
 : Cumulative % Drug Release and
: Cumulative % Drug Release and  : % Drug
content
: % Drug
content



