- Corresponding Author:
- PAYAL JOSHI
C. B. Patel Research Center, 3rd floor, Opp. Bhaidas Hall, Vile Parle (West), Mumbai-400 056, India
E-mail: payalchem@gmail.com
| Date of Submission | 7 June 2010 |
| Date of Revision | 14 June 2011 |
| Date of Acceptance | 17 June 2011 |
| Indian J Pharm Sci, 2011, 73 (3): 287-291 |
Abstract
Degradation products of itopride formed under different forced conditions have been identified using LC-PDA and LC-MS techniques. Itopride was subjected to forced degradation under the conditions of hydrolysis, photolysis, oxidation, dry and wet heat, in accordance with the International Conference on Harmonization. The stress solutions were chromatographed on reversed phase C18 (250×4.6 mm, 5 μm) column with a mobile phase methanol:water (55:45, v/v) at a detection wavelength of 215 nm. Itopride degraded in acid, alkali and oxidative stress conditions. The stability indicating method was developed and validated. The degradation pathway of the drug to products II-VIII is proposed.
Keywords
Degradation products, forced degradation, itopride, stability indicating, validation
Itopride hydrochloride is a novel, synthesized, gastro prokinetic drug, which stimulates gastrointestinal motor activity through the synergistic effects of dopamine D2-receptor blockade and acetylcholine esterase inhibitors [1-3]. Chemically, it is N- [p- 2(2-(dimethylamino)ethoxy)-benzyl] veratramide hydrochloride. Benzamide structure, amide and ether linkages in the drug molecule make it susceptible to degradation. Due to lability of these linkages and functional groups to hydrolysis and oxidation, several degradation products were anticipated to be formed during stability testing of the drug [4].
Itopride is not official in any pharmacopoeia. Few analytical methods have been reported for the quantitative estimation of itopride. Mushiroda et al. [5] have reported HPLC method with fluorescence detection. The limitation of this method was the use of a high ratio of buffer solution in the mobile phase that can cause damage to the reverse phase column. Takahara et al. [6] have reported HPLC method for the simultaneous determination of itopride and its metabolites in human serum and urine using automated column switching. Two sensitive and reproducible HPLC and HPTLC methods have been described for the quantitative determination of itopride hydrochloride in the presence of its degradation products [7]. HPTLC method for the determination of itopride hydrochloride in pharmaceutical preparation and bulk drug has been reported [8]. However, no degradation products of itopride were identified in the reported methods.
Thus, the purpose of the present study was to develop a validated stability indicating HPLC method and identify the degradation products of itopride formed under ICH recommended stress conditions of hydrolysis, photolysis, oxidation, dry and wet heat [9-11] taking the aid of LC–PDA and LC–MS techniques.
Materials And Methods
Itopride was supplied by Hemco Pharma, (Mumbai, India). Methanol (HPLC grade), hydrochloric acid, sodium hydroxide pellets (AR Grade) were purchased from Merck (India), hydrogen peroxide solution (AR grade) was purchased from Qualigens (India). HPLC-grade water was obtained using a tripledistillation glass assembly (Qualigens, India). It was filtered through a 0.2 μm membrane (Millipore, Bangalore, India) before use. Precision water bath (Technosys Model TWC-3) with temperature control within ±1° was used for conducting stress studies. Thermal stability study was performed in a hot air oven (Thermo Lab, Mumbai, India). Photolytic degradation was carried out in a photostability chamber (Thermo Lab, Mumbai, India) equipped with fluorescent and UV light and capable of controlling temperature and humidity in the range of ±2° and ±5% RH, respectively. The light system complied with ‘Option 2’ of ICH guideline Q1B [12]. The HPLC system used for the method development, validation and degradation studies of itopride was Jasco 1500 Series, equipped with a pump (Jasco Model PU 1580 intelligent HPLC pump), auto-injecting facility (AS-1555 sampler) and ChromNAV 1.12.01 software with LC-Net II/ADC system. LC-MS study for identification of degradation products of itopride was performed on Applied Biosystems (Applied Biosystems/MDS Sciex 2000 Series) with LC part of PerkinElmer 200 Series (PerkinElmer Inc., Shelton, CT, USA) equipped with Analyst 1.3 Software.
Preparation of stock and standard solutions
An accurately weighed sample (25 mg) of itopride was transferred in a 25 ml volumetric flask and dissolved in methanol to obtain solution strength of 1000 μg/ml. For preparing standard solution, 1 ml of this solution was transferred in 25 ml volumetric flask and made up to volume with mobile phase. Standard calibration solutions (5-500 μg/ml) for linearity assessment were prepared from 1000 μg/ml stock solution by diluting with mobile phase.
Forced degradation studies
All stress decomposition studies were performed at an initial drug concentration of 1 mg/ml. Acidic and alkaline hydrolysis were carried out in 1.0 N HCl and 1.0 N NaOH, respectively and heated at 80º for 4 h and neutral hydrolysis was performed in water at 80º for 12 h. The acid and alkaline stress solutions were neutralized and stored in cold cabinets. Oxidative stress was carried out in 15% H2O2 at 70º for 4 h in the dark to avoid photo-oxidation. Solid drug was exposed to thermal stress in dark at 100º for 24, 72 and 168 h for dry heat and wet heat treatment. Photolytic stress was determined by exposing the solid drug to 1.2 million lux h fluorescent light and 200 Wh/m2 UV. Later the intensity was increased by two to five times to establish the photostability of itopride. All the reaction solutions were suitably diluted with mobile phase before HPLC analysis. Peak purity analysis was performed to determine the percentage degradation and total loss of the API during the formation of forced degradants. A parallel set of three stress samples were generated for every stress condition viz, the blank subjected to stress in the same manner as the drug solution, zero time sample containing the drug stored under normal conditions and the drug solution subjected to stress treatment. The comparison of these stress solutions gave a real assessment of the changes in the intrinsic drug stability.
Separation studies
Itopride and its forced degradants were separated on a Kromasil C18 column (250×4.6 mm, 5μm) with 55:45 (v/v) methanol: water as the mobile phase. The flow rate was maintained at 1 ml/min and the eluent was monitored at 215 nm using diode-array detection controlled by ChromNAV software.
Method validation
The proposed method was validated as per ICH guidelines [13]. The linearity of the method was determined at a concentration range of 5-500 μg/ ml for itopride. 10 μl of each solution was injected in the system to obtain calibration graph of peak area against concentration. RSD (%) of the slope and intercept of the calibration plot was calculated. The limit of detection (LOD) and quantification (LOQ) was determined respectively at a signalto- noise ratio of 3 and 10 by injecting a series of dilute solutions of itopride of known concentration. Intra-day and inter-day precision was established by analyzing 100, 200 and 300 μg/ml drug solutions in triplicate on the same day and on consecutive days respectively. Accuracy was determined by spiking 0.1% of impurity in three known concentrations of the drug, viz., 100, 200 and 300 μg/ml in triplicate and the percent recovery of the analyte from the impurity was determined. Specificity of the method was established by the peak purity determination using a PDA detector. The resolution factor for the drug and nearest resolving peak was determined. Peak purity as well as the resolution was determined for all the degradation product peaks, in addition to the drug peak, to prove the selectivity of the developed method. Robustness of the method was performed by deliberate changes in chromatographic conditions such as change in flow rate (±10%), organic content in mobile phase (±2%), wavelength of detection (±5%) and pH of the mobile phase (±0.2%). The stability of itopride standard solutions and sample solutions was tested by analysis after 24, 48, and 72 h. The responses for the aged solution were evaluated by comparison with freshly prepared solutions.
Identification of degradation products
The stressed solutions, in which sufficient amounts of products were formed, was treated suitably and subjected to LC-PDA and LC-MS studies for the identification of forced degradation products. Two impurities are already known for itopride namely, dimethyl amino ethyl benzyl amine (DEBA) and veratric acid chloride. However, the drug was found to exhibit more than two impurities in the stress samples. LC–MS studies were carried out using positive electrospray ionization. The chromatographic conditions used for LC–MS analyses were the same as that for LC–PDA analyses, except that injection volume was 10 μl. A splitter was placed before the mass detector, allowing entry of only 30% of the eluent. LC–MS scans of the drug and all degradation products in ESI mode were recorded in the range of 50–3000 m/z and masses of the peaks were recorded for accurate mass measurements.
Results And Discussion
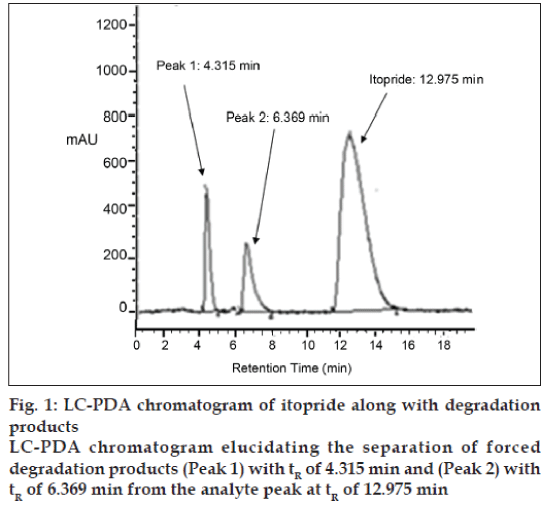
Itopride was susceptible to acid, alkali and oxidative stress conditions. The drug was stable to photolytic and thermal stress as no decrease in area of drug peak was observed. The detailed results of forced degradation studies of itopride are presented in Table 1. The stressors, choice of their concentration and preparation of stress samples were established to achieve a target degradation of 10-20% with respect to the active ingredient for development of stability indicating method. Different logical modifications like the change in pH, different mobile phase compositions and column temperature adjustment were tried to get good separation between the drug and the degradation products, as well as between the degradation products. Use of phosphate buffer was avoided so as to extend the separation method to LC-MS analysis. The best separation was achieved on a C18 column with methanol: water in the ratio of 55:45 at a flow rate of 1 ml/min as the mobile phase and detection wavelength of 215 nm. The method was found to unequivocally separate the forced degradation products from the analyte as shown in fig. 1.
| Stress condition | Exposure condition | Percentage degradation | Peak purityb |
|---|---|---|---|
| Hydrolysis | |||
| Acid | 1.0 N HCl, 80º for 4 h | 14.022 | 999.917 |
| Alkaline | 1.0 N NaOH, 80º for 4 h | 19.846 | 999.838 |
| Neutral | Drug solution heated in water at 80º for 12 h | No degradation | 999.996 |
| Oxidation | 15% H2O2, 70º for 4 h | 13.610 | 999.536 |
| 30% H2O2, 70º for 4 h | 28.633 | 999.748 | |
| Photolysisa | 1.2×106 and 2.4×106 lux h of fluorescent | Negligible | 999.516 |
| light and 200 Wh/m2 UV light | |||
| Thermal | Dry and wet heat at 100º for 24, 72 and 168 h | No degradation | 999.471 |
aSolid itopride was exposed to photolysis for 15 days to assess photo lability. bPeak purity values in the range of 990–1000 indicate a homogeneous peak
Table 1: Summary Of Forced Degradation Studies For Itopride
The summarized results of method validation are presented in Table 2. Response of the drug was a linear function of concentration over the range 60-300 μg/ml. The mean values of the slope, intercept, and correlation coefficient (±RSD) were 297.585, 9134.009 and 0.9983, respectively. The method was precise as the RSD values from studies of intra-day and inter-day precision were <1% and <2%, respectively, which is in accordance with the ICH guidelines. Accuracy of the method was determined and found to be 98.8- 100.1%. The signal/noise ratios 3:1 and 10:1 were considered as LOD and LOQ, respectively. LOD and LOQ were found to be 0.019 μg/ml and 0.353 μg/ml respectively. Specificity of the method was established by determining the peak purity using a PDA detector. The resolution factor for the drug and nearest resolving peak was determined. Peak purity as well as the resolution was determined for all the degradation product peaks, in addition to the drug peak, to prove the selectivity of the developed method. Influence of deliberate changes in chromatographic conditions such as change in flow rate (±10%), organic content in mobile phase (±2%), wavelength of detection (±5%) and pH of buffer in mobile phase (±0.2%) studied to determine the robustness of the method were also in favor of the developed LC method for the analysis of itopride. The solution stability experiment data confirmed that sample solutions used during assay were stable up to 48 h.
| Parameters | Results |
|---|---|
| System suitability | |
| Retention time (tR) | 12.9 min |
| Tailing factor | 1.08 |
| Theoretical Plates (N) | 16590 |
| Symmetry factor | 1.257 |
| Linear range | 60-300 µg/ml |
| Correlation coefficient (r2) | 0.9983 |
| Regression equation | y = 297.585x + 9134.009 |
| Precision (cRSD%) | |
| Intra-day (n = 6) | 0.74 |
| Inter-day (n = 6) | 1.85 |
| Accuracy | 98.8-100.1% |
| LOD and LOQ | 0.019 and 0.353 µg/ml |
RSDc indicates relative standard deviation
Table 2: Summary Of Method Validation Results
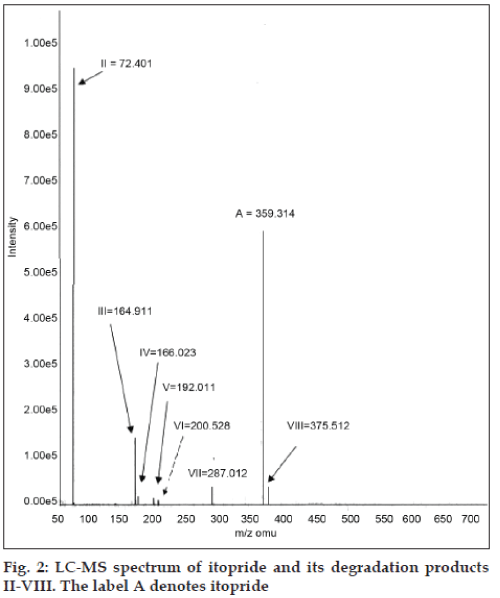
LC-PDA revealed the presence of two major impurities in itopride as shown in fig. 1. The forced degradation products were found to exhibit very poor response to ultraviolet absorption detection hence, LC-MS was employed for further detection of impurities [14]. The purity angle of each degradation product peak was less than its purity threshold, which indicated that all of them were pure. Degradation products (II-VIII) were generated on subjecting the drug to acid, alkaline hydrolysis and under oxidative stress conditions. The molecular masses of the degradation products were established by recording the LC-MS scans in ESI mode as shown in fig. 2. Two impurities each were formed in acid and alkaline conditions, whereas one impurity was formed in oxidative stress conditions. DP- V and DP-VI were identified as known impurities, while the products II, III, IV, VII and VIII were identified as major degradation products, as shown in Table 3. The m/z values and fragmentation patterns obtained for the degradation products through LC– MS studies helped to confirm the presence of known products and to propose the structures of unknown compounds.
| Drug/ | Experimental | Probable structure |
|---|---|---|
| DPsa | mass | |
| A | 359.314 | Itopride |
| II | 72.401 | N,N-Dimethylethanamine |
| III | 164.911 | Veratric aldehyde |
| IV | 166.023 | Veratric acid |
| V | 192.011 | Dimethyl amino ethyl benzyl amine |
| VI | 200.528 | Veratric acid chloride |
| VII | 287.012 | Hydrolysed form of Itopride |
| VIII | 375.512 | Oxidation product of Itopride |
aDegradation Products
Table 3: Interpretation Of The Lc-Ms Data And Fragments Of The Drug
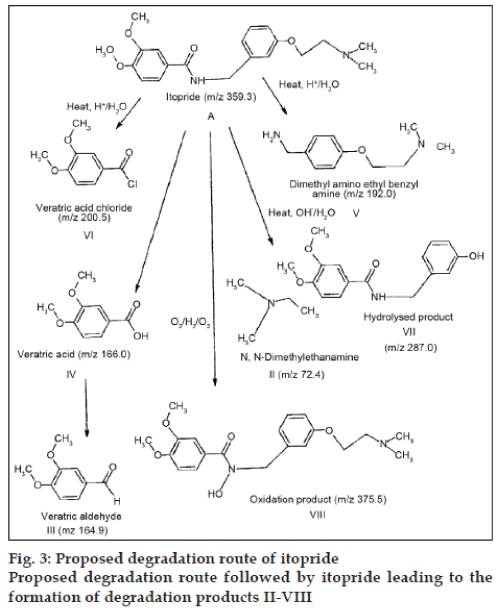
The postulation of degradation route followed by itopride is presented in fig. 3. The drug readily underwent amide hydrolysis under acidic and alkaline conditions to form DP-IV, DP-V and DP-VI. Formation of DP-IV is attributed to alkaline hydrolysis with poor amine as the leaving group. DP-VIII was formed under oxidative stress conditions. The fragmentation of ether linkage resulted in formation of low UV absorbing species, DP-II and DP-VII. The drug showed lability towards photolysis due to the presence of benzamide moiety, but the impurity was not quantified due to low ionization.
A stability indicating method for itopride was developed which separated all the degradation products formed under various stress conditions. The method proved to be simple, accurate, precise, specific and selective. Hence, it is recommended for analysis of the drug and degradation products in stability samples by the industry. Degradation products II-VIII formed during the forced degradation study on itopride were identified using LC-PDA and LC-MS techniques. The results in totality helped to draw out a more extensive degradation route of the drug.
Acknowledgements
The authors thank Hemco Pharma, Vile Parle, Mumbai, for providing Itopride and impurities. The authors also thank Dr. Sasikumar Menon, Assistant Director, TDM Laboratories, Sion, Mumbai, for the LC-MS facility necessary to carry out the research work.
References
- Iwanaga Y, Miyashita N, Mizutani F, Morikawa K, Kato H, Ito Y, et al. Stimulatory effect of N- [4- [2-(dimethylamino)ethoxy]benzyl]-3,4-dimethoxybenzamide hydrochloride (HSR-803) on normal and delayed gastrointestinal propulsion. Jpn J Pharamacol 1991;56:261-9.
- Kakiuchi M, Saito T, Ohara N, Hosotani T, Morikawa K. Pharmacological evaluation of itopride hydrochloride with drug-induced arrhythmia. JpnPharmacolTher 1997;25:811-7.
- Iwanga Y, Kimura T, Miyashita N, Morikawa K, Nagata O, Itoh Z, et al. Characterisation of acetylcholinesterase inhibition by itopride. JpnJ Pharmacol 1994;66:317-22.
- Baertschi SW, Alsante KM. Stress Testing: The Chemistry of Drug Degradation. In: Baertschi SW, editor. Pharmaceutical Stress Testing: Predicting Drug Degradation. 1st ed. New York: Taylor and Francis; 2005. p. 51-140.
- Mushiroda T, Douya R, Takahara E, Nagata O. The involvement of flavin containing monooxygenase but not CYP3A4 in metabolism of itopride hydrochloride, a gastrokinetic agent: Comparison with cisapride and mosapride citrate. Drug MetabDispos 2000;28:1231-7.
- Takahara E, Fukuoka. H, Takagi T, Nagata O, Kato H. Simultaneous determination of a new gastrointestinal prokinetic agent (HSR-803) and its metabolites in human serum and urine by high-performance liquid chromatography using automated column-switching. J Chromatogr B Biomed SciAppl 1992;576:174-8.
- Kaul N, Agrawal H, Maske P, Rao J, Mahadik KR, Kadam S, et al. Chromatographic determination of itopride hydrochloride inthe presence of its degradation products. J Sep Sci 2005;28:1566-76.
- Dighe V, Sane R, Menon S, Tambe H, Pillai S. High-performance thin-layer chromatographic determination of itopride hydrochloride in its pharmaceutical preparation and in the bulk drug, J Planar Chromatogr 2006;19:319-23.
- ICH, Q3A(R2) Impurities in New Drug Substances, International Conference on Harmonization, IFPMA, Geneva, 2006.
- ICH, Q3B(R2), Impurities in New Drug Products, International Conference on Harmonization, IFPMA, Geneva, 2006.
- ICH, Q1A(R2), Stability Testing of New Drug Substances and Products, International Conference on Harmonization, IFPMA, Geneva, 2003.
- ICH, Q1B, Stability Testing: Photostability Testing of New Drug Substances and Products, International Conference on Harmonization, IFPMA, Geneva, 1996.
- ICH, Q2(R1), Validation of Analytical Procedures: Text and Methodology, International Conference on Harmonization, IFPMA, Geneva, 2005.
- McCrossen SD, Bryant DK, Cook BR, Richards JJ. Comparison of LC detection methods in the investigation of non-UV detectable organic impurities in a drug substance. J Pharm Biomed Anal 1998;17:455-71.