- *Corresponding Author:
- V Kavitha
Department of Pharmacognosy, School of Pharmacy, Dhanalakshmi Srinivasan University, Trichy, Tamil Nadu 621112, India
E-mail: vkavitha.pharmacognosy@gmail.com
| Date of Received | 08 February 2026 |
| Date of Revision | 23 February 2026 |
| Date of Accepted | 28 March 2026 |
| Indian J Pharm Sci 2026;88(1):51-60 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
Breast cancer is still the commonest cancer among females across the globe; BRCA1 gene mutations play a great role in inducing breast cancer. In the exploration for non-traditional therapeutic players, this study assesses the anti-cancer effectiveness of phytoconstituents isolated from two red seaweed species namely; Kappaphycus alvarezii and Gracilaria debilis using in silico molecular docking. The 3D structure of BRCA1 (PDB ID: IGK (4IGK) was used as docking template in Molegro virtual Docker. Ligands obtained through gas chromatography-mass spectrometry analysis were filtered for drug-likeness properties by Lipinski’s rule and docking simulations were done to determine the binding affinities. According to the results, it was found out that the highest score of MolDock by Bromoindole-3-acetic acid obtained from Kappaphycus alvarezii and Aplysiatoxin derived from Gracilaria debilis was very favourable and good Hydrogen bonding interaction between the aforementioned compounds and BRCA1 was very strong compared to These results show the prospects of these marine-derived compounds as lead candidates for breast cancer treatment and are worth pharmacological confirmation.
Keywords
BRCA1, breast cancer, Kappaphycus alvarezii and Gracilaria debilis, molecular docking
A breast cancer is the number one cancer afflicting women everywhere in the world. It manifests in the form of uncontrolled growth and division of cells in the breast tissues. In the last three decades, the incidence of breast cancer has been almost doubled. It is estimated that between 50–75% of the breast cancers begin in the duct, while 10 %-15 % come from the lobules and a little proportion of the same arises from other breast tissues[1]. Approximately, 1 376 111 new cases of breast cancer were reported among women in the world in 2008, according to the Global cancer Facts and Figures by the American Cancer Society. As a rule, the breast cancer rates are higher in developed countries in contrast with developing countries. Regardless of the reason that causes this disparity, lifestyle and reproductive factors are said to be contributing significantly. There are women who have got mutations in the BRCA1 and BRCA2 genes and they are at high risk of having breast and ovarian cancers. Patients with mutations in these genes have a 60-80 % life time risk of breast cancer and 15 %-60 % risk of ovarian cancer by the age 70[2]. BRCA1 gene (breast cancer 1, early onset) is a tumor suppressor gene, which encodes breast cancer type 1 susceptibility protein. Because of its two main domains, i.e., Zinc finger C3HC4 type (RING finger) and BRCA1 C Terminus (BRCT) the protein BRCA1 is also called RING finger protein 53[3].
BRCA1 gene is located in chromosome 17q21 and has 24 exons whose total length is estimated to be around 100 kilobases. It’s coding sequence starts somewhere in the middle of the fourth exon. Upon the end, the protein obtained is phosphorylated and contains 1863 amino acids, molecular weight of 220 kDa[5]. RING finger domain, having Rad51 binding site, is present at the N-terminus; while, the two BRCT domains involved in cell cycle checkpoint control and Deoxyribonucleic Acid (DNA) repair are located at the C-terminus. These BRCT domains promote chromatin remodeling and transcription processes activation[6].
The location of the BRCA1 protein is in the nucleus of the cell[7]. According to recent studies, small deletion, insertion, nonsense mutation and splicing errors cover 87 % of all pathogenic mutation of BRCA1 gene, resulting in the production of abnormal BRCA1 protein[8]. This protein is very important in sustaining genomic stability since it assists in DNA repair or induces cell death in situations where repair of DNA is impossible. A BRCA1 gene that is faulty interferes with the repair of DNA leaving one most susceptible to cancer[9].
The BRCA1 protein associates with other tumor suppressors and DNA damage sensor proteins and signalling molecules to give form to the BRCA1- Associated Genome Surveillance Complex (BASC) [10]. It also attaches to Ribonuclic Acid (RNA) polymerase II and connects to histone deacetylase complexes via its C-terminal domain, being a part of DNA repair process, transcription regulation, and protein ubiquitination[9,11]. People with genetic abnormalities in either BRCA1 or BRCA2 gene have over 80 % lifetime risk of having breast cancer and most of the cases are connected with mutations in these genes[12]. Since it is more likely that BRCA1 is involved in the pathogenesis of breast cancer rather than BRCA2[13], the following study is devoted to the BRCA1 protein.
Red seaweeds (Rhodophyta) are yet another large group of algae that occur in various shores and are highly diverse with approximately 6000 known species which is more diversified than the other three major categories of seaweeds. Earlier, it has been reported that annually about 2.8 million tons of red seaweeds are harvested in the world. Species of nori gathered for edible purposes largely belong to the genus Porphyra, while Eucheuma and Kappaphycus are commonly employed in the production of carrageenan [14].
Among the most occurring red seaweeds in East Malaysia include Kappaphycus alvarezii (K. alvarezii). This species is cultivated in many countries around the world, namely Malaysia, Philippines, Indonesia, and Tanzania[15]. K. alvarezii is a well-known tropical red alga of great importance as producer of high contents of cell wall polysaccharides being the main source of kappacarrageenan- desirable hydrocolloid[16]. Kappacarrageenan is widely applied in food industry as gelling agent, emulsifying agent, thickening agent, and stabilizing agent[17]. Kappa-carrageenan is also used in pharmaceutical and nutraceutical products. Seaweeds, including the K. alvarezii have been known to be low in calorific values and have high contents of dietary fiber contents, essential minerals, proteins, vitamins, trace elements and secondary metabolites thus are also lacking in other organisms. In addition K. alvarezii has been suggested for the dietary fibre, cholesterol reduction agents, antioxidants, antiviral and anticancer and hemagglutination activities[18,19].
In terms of nutrition, K. alvarezii have 16.2 % protein (w/w), 29.4 % fiber (w/w) and 27.4 % carbohydrates (w/w). It also contains a high percentage of unsaturated fatty acids (44.5 % of the total fatty acids), such as oleic acid (11.0 %) and cis-heptadecenoic acid (13.5 %) as well as small amounts of linoleic acid (2.3 %). By contrast, saturated fatty acids such as the heptadecanoic acid account for 37.0 % of the total[20].
Carrageenan present in K. alvarezii are useful dietary fiber, which promotes digestion, safeguards gastric mucosa, and protects the intestine from exposure to potential carcinogens. Furthermore, this seaweed has exhibited potent antiviral activity against some of the enveloped viruses such as Human Immunodeficiency Virus (HIV), Herpes Simplex Virus (HSV) and Human Cytomegalovirus (HCMV) among others. It also has quite significant antitumor properties [21,22].
The bioactive compounds present in K. alvarezii were identified, the cytotoxic potential of the extracts against the Michigan Cancer Foundation-7 (MCF-7) breast cancer cell line was evaluated, and the toxicity of high-dose extracts in rats, along with their effect on 7, 12-dimethylbenz[a]anthracene-induced breast cancer, was assessed.
The Gracilaria genus of seaweed has a wide range of distribution in tropical and temperate places and is one of the largest within Gracilariaceae family[23]. We have in excess of 150 000 edible seaweeds in the intertidal zones and tropical marine setting where they are rich sources of natural products of medicinal and commercial value. About 300 species of Gracilaria have been identified in the world, and about 160 species of the same being taxonomically described. Most of these species are basic sources of agar production.
Furthermore, compounds isolated from red, brown and green algae compounds including Gracilaria spp have been reported to exhibit a remarkable spectrum of beneficial biological activity like antioxidant, anticancer, antibacterial, antifungal, antiparasitic, anti-viral and anti-proliferation activities[24]. Archaeological discoveries in Chile indicate that man’s use of macroalgae goes back at least 14 000 y to the Neolithic period. The coastal people have been harvesting the seaweeds for domestic purposes for a long time. This has been one of the practices of the traditional life in such countries as Japan (13 000 BC), China (2700 BC), Egypt (1550 BC), and India (300 BC), in which marine algae were traditionally utilized as folk remedies for various illnesses.
In the Mediterranean, seaweeds were used in the ancient Roman and Greek periods like medicine, animal fodder, and dye. According to the pre- Christian period in particular red algae was applied to treat parasitic infections[25]. However, only wild seaweed were accessible in this period, and this was used only for food purposes until the middle age. In the Asian countries such as China, Korea and Japan, seaweeds were crucial for traditional medicine, cuisine for more than six centuries.
Natural polysaccharides isolated from seaweed have attracted more attention during the recent decades due to a set of desirable properties-biocompatibility, biodegradability, hydrophilicity, non-toxicity, and protective capacity, which makes them a very interesting object for the pharmaceutical and cosmetic industries. Agar, which was initially identified in Japan in the 17th century, is usually obtained from the Gracilaria species[26]. In China, the Gracilaria is grown to be used as a food ingredient as well as a binding agent[27]. These species are edible and consumed in such places as Japan, Southeast Asia, Hawaii, and Caribbean.
Agar isolated from Gracilaria is a food grade agent used as a common gelling and stabilizing material in sugar-confection production such as jelly candies, icing and pies. In the Western cuisine, agar is commonly used as a weight-loss agent and as a vegan replacement for gelatin. Its use in Japan is in the soups, snacks and other culinary dishes [28].
Great beyond these traditional uses, polysaccharide extract from Gracilaria species has been found to have a variety of therapeutic activities such as anticancer[29], anti-inflammatory, anti-diabetic, and immunostimulatory effects. Some of the factors that affect the degree of these bioactivities include molecular weight, monosaccharide composition, degree of sulphation, and chain length. These in turn are influenced by taxonomy, environmental factor i.e salinity, temperature, light, nutrient availability, biotic interactions like epiphytism and method of extraction and purification[30]. Therefore, optimization of such variables is vital for the optimization of the level of sulphation, purity and biological activity of Gracilaria-derived compounds, the identification of new therapeutic agents.
In this study, K. alvarezii and Gracilaria debilis (G. debilis) were investigated as potential alternative drug treatments for breast cancer. The Molegro Virtual Docker program was utilized to target the BRCA1 gene. The docking score, which measures the phytoconstituent's binding affinity and efficacy against the target proteins, was used to evaluate the activity of the algae.
Materials and Methods
Protein preparation:
The 3D crystal structure of breast cancer target protein BRCA1, namely, the BRCT domain (PDB ID: 4IGK in both its free and complexed with the ATRIP (Ataxia Telangiectasia and Rad3-related protein–Interacting Protein) peptide forms was retrieved from the Protein Data Bank (www.rcsb. org/pdb). In order to gain insight into structural traits and active binding sectors of the protein, CASTP (Computed Atlas of Surface Topography of Proteins) tool was utilized besides PyMOL software in drawing molecular simulations. This method allowed for an overall analysis of the topography of the protein surface and possible interaction of ligands.
In order to yield potential drug-like compounds, Lipinski’s Rule of five was used. Based on this rule, the higher probability of being orally active, the lower the molecular weight (<500 g/mol), less value of log P (<5), no more than ten hydrogen bond acceptors, no more than five hydrogen bond donors, and the fewer rotatable bonds (less than ten). Such criteria provide good pharmacokinetic properties and bioavailability. Only compounds that satisfied these criteria were taken through molecular docking studies to determine their affinity with BRCA1 and BRCA2. This strict process of screening sought to determine prospective phytochemicals that may be useful in the fight against BRCA-linked breast cancer as a potential therapeutic agent.
Ligand preparation:
The main phytoconstituents that are responsible for anticancer activity in K. alvarezii and G. debilis were identified earlier by Gas Chromatography- Mass Spectrometry (GC-MS) analysis carried out by other researchers. The three-dimensional structures of these bioactive compounds were retrieved from PubChem database and prepared for molecular docking simulation.
Molecular import and preparation:
The molecular structure files were parsed to pick out the relevant components such as ligands, co factors, water molecules and proteins and were automatically prepared for docking. Molegro Virtual Docker (MVD) was used to obtain information about chemical bonds, aromatic structures, charge assignment tasks, and the inclusion of explicit hydrogen atoms. Using the integrated cavity detection facility, it was possible to determine the promising areas for the ligand interaction on the surface of the protein and, the search could be limited by those.
Docking:
Molecular docking is the simulation of the interaction of a small molecule, the ligand, and a target protein, the receptor or enzyme. The MVD software was used to carry out docking simulations in this research. The docking scoring function that is used by MVD is derived from the Piecewise Linear Potential (PLP), assessing for the binding affinity with the help of a mixture of electrostatic and hydrogen bonding interactions.
For each ligand, the program determined the best orientation of the ligand in the protein-ligand binding site. Hydrogen bonds were detected, named and taken into the calculation of energy. The computation of ligand energies were obtained by using the MolDock score, which is a linear combination of the essential interaction terms. Before docking, all valence tests and hydrogen atoms addition were done. The algorithm of cavity detection directed the search method based on differential evolution during simulation on the detected binding pockets. All the warnings caused by the errors related to the structure preparation or data (e.g., unknown residues) were checked and fixed. Subsequent to this, a re-ranking procedure was used in ranking the top scorer binding poses.
Analysis:
Pose organizer tool was used in analysing the resulting ligand poses from the docking simulation and this allowed dynamic loading and viewing of the docking results. This feature allowed an efficient search in a wide range of ligand poses. Several parameters of energy and interaction type were assessed at the same time, additional re-ranking and binding affinity were calculated. Hydrogen bonding and electrostatic interactions were revised dynamically through the course of reviewing poses.
The MolDock scores of the selected ligand then compared with a standard drug (Olaparib). The most favourable (for this purpose the lowest score of MolDock) ligands were recognized as the most likely candidates and candidates of future lead compounds for breast cancer treatment[2].
Results and Discussion
MolDock relies on a new heuristic search engine combining cavity prediction and differential evolution, which increases the efficacy and precision of molecular docking. Its scoring function is based on the PLP, which is parameterized on a proper set of refined electrostatic and hydrogen bonding interaction terms. The MolDock score is the principal criteria of how the quality of the ligand-target protein binding can be estimated.
For the determination of efficacy of ligands MolDock score, rerank score, and hydrogen bond interactions were studied. A higher (less negative) value of MolDock score is indicative of weaker affinity of the ligand for the target protein. The outcome of in silico docking experiment with selected ligands are displayed in Table 1 and Table 2 against the human BRCA1 protein.
| Ligand | Ligand name | MolDock score | Rerank score | H Bond |
|---|---|---|---|---|
| 22485713 | Bromoindole-3-acetic acid | -114.54 | -95.032 | -6.514 |
| 2794830 | 6-Bromoindole-3-carbaldehyde | -103.48 | -93.658 | -3.025 |
| 23725625 | Standard Drug (Olaparib) | -101.98 | -73.845 | -0.639 |
Table 1: Ranking of ligands from k. Alvarezii against human BRCA-1 based on moldock score
| Ligand | Ligand Name | MolDock score | Rerank score | H Bond |
|---|---|---|---|---|
| 21672114 | Aplysiatoxin | -139.03 | -108.28 | -8.713 |
| 192847 | Gigartinine | -113.48 | -95.772 | -3.049 |
| 23725625 | Standard Drug (Olaparib) | -101.98 | -73.845 | -0.639 |
Table 2: Ranking of ligands from g. debilis against human BRCA-1 based on moldock score
Based on the docking results from MVD, we can draw several insights about the interaction of the selected ligands with the target proteins.
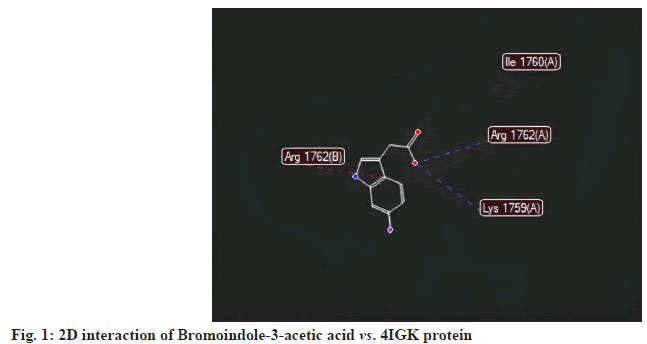
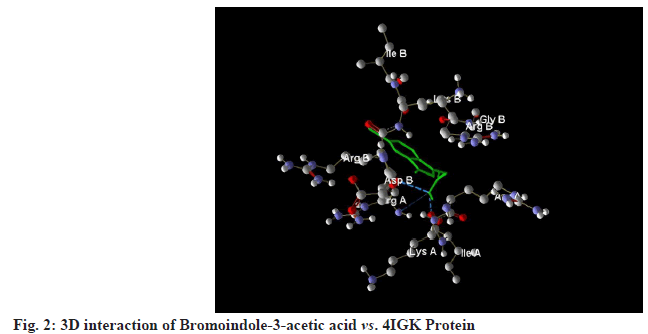
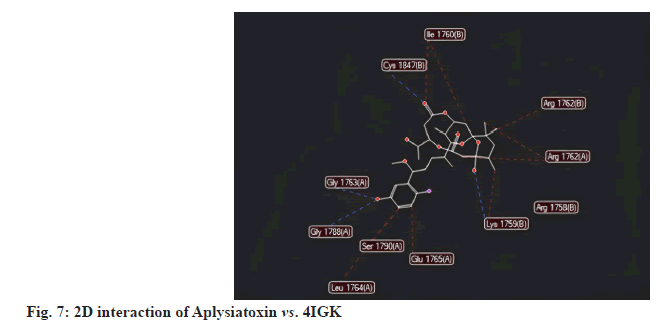
All selected ligands, including the standard, exhibited strong binding affinities. The results indicated that Bromoindole-3-acetic acid achieved a MolDock score of -114.537 against 4IGK, along with a rerank score of -95.032. Its hydrogen bonding value was measured at -6.514. Bromoindole-3-acetic acid demonstrated the highest binding affinity, than 6-Bromoindole- 3-carbaldehyde. A lower (more negative) score suggests a stronger binding interaction, which is further supported by the substantial rerank score of -95.032. The ligand’s significant binding affinity to the protein is likely due to the formation of stable hydrogen bonds, as indicated by the hydrogen bonding score of -6.514 (fig. 1-fig.3).
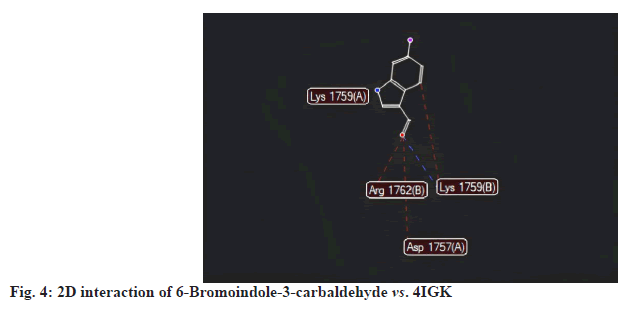
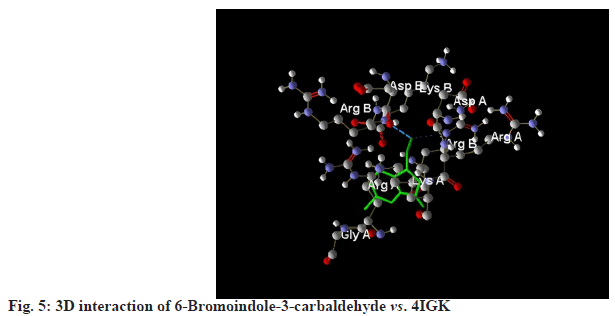
6-Bromoindole-3-carbaldehyde recorded a MolDock score of -103.484 against 4IGK, along with a rerank score of -93.658. The hydrogen bonding value was measured at -3.025. With a MolDock score of -103.484, 6-Bromoindole-3-carbaldehyde demonstrates a strong binding affinity. However, its rerank score of -93.658 suggests that while the initial binding may be robust, the overall stability of its binding conformation is likely less favorable compared to that of Bromoindole-3-acetic acid (fig. 3-fig.6).
Olaparib, a standard treatment for breast cancer, achieved a MolDock score of -101.976 against 4IGK, along with a rerank score of -73.845. Its hydrogen bonding value was found to be -0.639. While Olaparib exhibits excellent MolDock and rerank scores, its hydrogen bonding score of -0.639 is notably lower than those of the selected ligands.
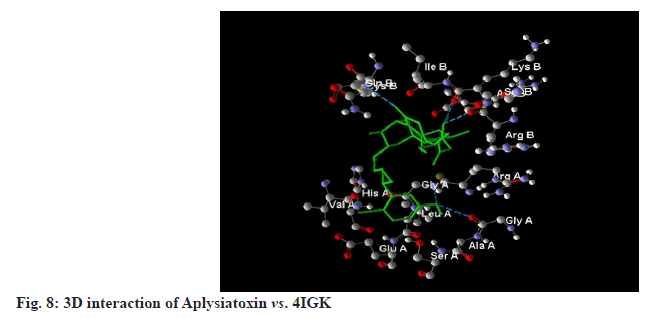
All selected ligands, including the standard, exhibited strong binding affinities. The results indicated that Aplysiatoxin achieved a MolDock score of -139.032 against 4IGK, along with a rerank score of -108.278. Its hydrogen bonding value was measured at -8.713. Aplysiatoxin demonstrated the highest binding affinity, than Gigartinine. A lower (more negative) score suggests a stronger binding interaction, which is further supported by the substantial rerank score of -108.278. The ligand's significant binding affinity to the protein is likely due to the formation of stable hydrogen bonds, as indicated by the hydrogen bonding score of -8.713 (fig.7-fig.9).
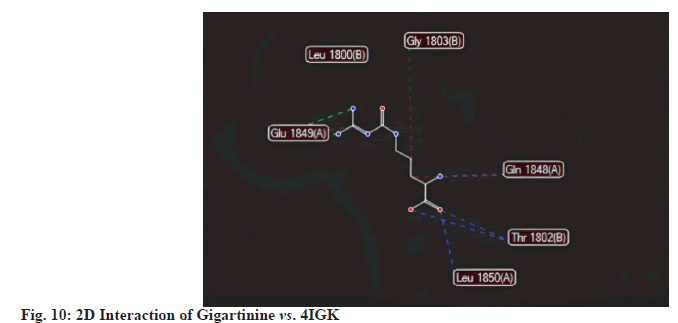
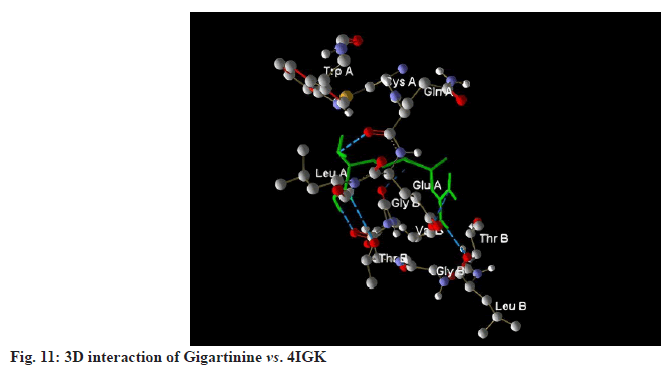
Gigartinine recorded a MolDock score of -113.484 against 4IGK, along with a rerank score of -95.772. The hydrogen bonding value was measured at -3.049. With a MolDock score of -113.484, Gigartinine demonstrates a strong binding affinity. However, its rerank score of -95.772 suggests that while the initial binding may be robust, the overall stability of its binding conformation is likely less favourable compared to that of Aplysiatoxin (fig.10-fig.12).
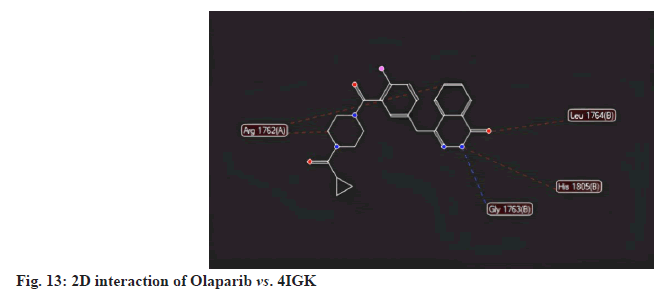
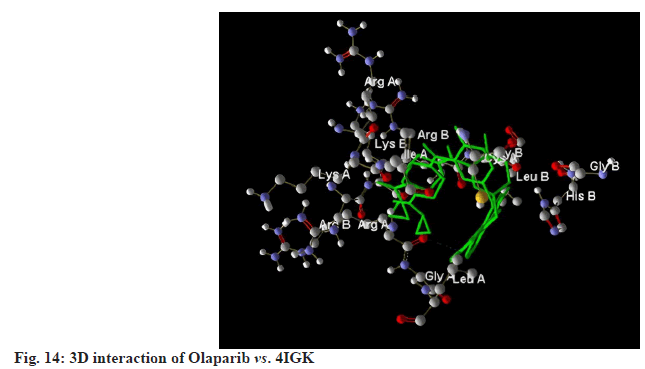
Olaparib, a standard treatment for breast cancer, achieved a MolDock score of -101.976 against 4IGK, along with a rerank score of -73.845. Its hydrogen bonding value was found to be -0.639. While Olaparib exhibits excellent MolDock and rerank scores, its hydrogen bonding score of -0.639 is notably lower than those of the selected ligands ( fig.13-fig.15).
This research identifies the growth prospects for anti-cancer agents of marine origin from the K. alvarezii and G. debilis by in silico docking into the breast cancer target protein BRCA1. Among the tested ligands, Bromoindole-3-acetic acid, K. alvarezii and Aplysiatoxin, G. debilis showed the highest binding affinities, even to a higher extent as compared to the standard drug Olaparib, in terms of MolDock scores and hydrogen bonding interactions. These results highlight a great promise of the marine red algae as sources of bioactive compounds of breast cancer treatment. Some additional in vitro and in vivo studies should be used to confirm these computational predictions and conduct the pharmacokinetics and safety assessments of the most promising candidates.
Acknowledgements
The authors gratefully acknowledge Dhanalakshmi Srinivasan University, Samayapuram, Tiruchirappalli, for providing the computational facilities and institutional support required to carry out the molecular docking studies. We also extend our sincere thanks to Dr. S. Akilandeswari for her continuous guidance, valuable suggestions, and encouragement throughout this work.
Conflict of interests:
The authors declared no conflict of interests.
References
- Corben AD. Pathology of invasive breast disease. Surg Clin. 2013;93(2):363-92.
[Crossref] [Google Scholar] [PubMed]
- American Cancer Society. Global cancer facts and figures 2009. Atlanta: American Cancer Society; 2009.
- Paterson MC. BRCA1 and BRCA2: Molecules and models. Trends Genet; 1998;14(10):396-402.
- Miki Y, Swensen J, Shattuck-Eidens D, Futreal PA, Harshman K, Tavtigian S, et al. A strong candidate for the breast and ovarian cancer susceptibility gene BRCA1. Science 1994;266(5182):66-71.
[Crossref] [Google Scholar] [PubMed]
- Ruffner H, Verma IM. BRCA1 is a cell cycle-regulated nuclear phosphoprotein. Proc Natl Acad Sci 1997;94(14):7138-43.
[Crossref] [Google Scholar] [PubMed]
- Miyake T, Maeda T, Nomura T. BRCT domains of the BRCA1 protein interact with p300 to regulate transcription. Biochem Biophys Res Commun 2000;271(3):1023-6.
- Wilson CA, Payton MN, Elliott GS. BRCA1 nuclear localization signal. J Biol Chem 1999;274(52):37895-900.
- Couch FJ, Weber BL. Mutations and polymorphisms in the familial early?onset breast cancer (BRCA1) gene. Hum Mutat 1996;8(1):8-18.
[Crossref] [Google Scholar] [PubMed]
- Friedenson B. BRCA1 and BRCA2 pathways and the risk of cancers other than breast or ovarian. MedGenMed 2005;7(2):60.
[Google Scholar] [PubMed]
- Wang Y, Cortez D, Yazdi P, Neff N, Elledge SJ, Qin J. BASC, a super complex of BRCA1-associated proteins involved in the recognition and repair of aberrant DNA structures. Genes Devel 2000;14(8):927-39.
[Google Scholar] [PubMed]
- Starita LM, Parvin JD. Substrates of the BRCA1-dependent ubiquitin ligase. Cancer Biol Ther 2006;5(2):137-41.
[Crossref] [Google Scholar] [PubMed]
- Thakur MH, Phadke SR. BRCA1 and BRCA2: genetics and clinical implications. Indian J Hum Genet 2005;11(1):1-10.
- Ford D, Easton DF, Stratton M, Narod S, Goldgar D, Devilee P, et al. Genetic heterogeneity and penetrance analysis of the BRCA1 and BRCA2 genes in breast cancer families. Am J Hum Gen 1998;62(3):676-89.
[Crossref] [Google Scholar] [PubMed]
- Coelho SM, Heesch S, Grimsley N, Moreau H, Cock JM. Genomics of marine algae. In: Introduction to Marine Genomics; 2010;1:179–211.
- Hayashi L, Santos AA, Faria GS, Nunes BG, Souza MS, Fonseca AL, et al. Kappaphycus alvarezii (Rhodophyta, Areschougiaceae) cultivated in subtropical waters in Southern Brazil. J Appl Phycol 2011;23(3):337-43.
- Bixler HJ. Recent developments in manufacturing and marketing carrageenan. Hydrobiologia 1996;326(1):35-57.
- Pickering TD, Skelton P, Sulu RJ. Intentional introductions of commercially harvested alien seaweeds. Bot Mar. 2007;50:338-50.
- Ferraces-Casais P, Lage-Yusty MA, Rodríguez-Bernaldo de Quirós A, López-Hernández J. Evaluation of bioactive compounds in fresh edible seaweeds. Food Anal Methods 2012;5(4):828-34.
- Senthil A, Mamatha BS, Vishwanath P, Bhat KK, Ravishankar GA. Studies on development and storage stability of instant spice adjunct mix from seaweed (Eucheuma). J Food Sci Technol 2011;48(6):712-7.
- Fayaz M, Namitha KK, Murthy KC, Swamy MM, Sarada R, Khanam S, et al. Chemical composition, iron bioavailability, and antioxidant activity of Kappaphycus alvarezzi (Doty). J Agric Food Chem 2005;53(3):792-7.
[Crossref] [Google Scholar] [PubMed]
- Khotimchenko YS. Antitumor properties of nonstarch polysaccharides: Fucoidans and chitosans. Russian J Marine Biol 2010;36(5):321-30.
- Yuan H, Song J, Li X, Li N, Liu S. Enhanced immunostimulatory and antitumor activity of different derivatives of κ-carrageenan oligosaccharides from Kappaphycus striatum. J Appl Phycol 2011;23(1):59-65.
- Liao X, Yang L, Chen M, Yu J, Zhang S, Ju Y. The hypoglycemic effect of a polysaccharide (GLP) from Gracilaria lemaneiformis and its degradation products in diabetic mice. Food Funct 2015;6(8):2542-9.
[Crossref] [Google Scholar] [PubMed]
- Choudhary B, Chauhan OP, Mishra A. Edible seaweeds: A potential novel source of bioactive metabolites and nutraceuticals with human health benefits. Front Marine Sci 2021;8:740054.
- Jeong SC, Jeong YT, Lee SM, Kim JH. Immune-modulating activities of polysaccharides extracted from brown algae Hizikia fusiforme. Biosci Biotechnol Biochem 2015;79(8):1362-5.
[Crossref] [Google Scholar] [PubMed]
- Ahmed ABA, Adel M, Talati A, Kumar MS, Abdulrahim K, Abdulhameed MM. In: Venkatesan J, Anil S, Kim S-K, editors. Seaweed polysaccharides and their production and applications. Seaweed Polysaccharides. Amsterdam: Elsevier; 2017. p. 369-82.
- Padmesh S, Singh A. Agars: Properties and applications. In: Polysaccharides. Hoboken (NJ): John Wiley & Sons; 2021. p. 75-93.
- Rasyid A, Ardiansyah A, Pangestuti R. Nutrient composition of dried seaweed Gracilaria gracilis. ILMU Kelaut Indones J Mar Sci 2019;24(1).
- Torres P, Santos JP, Chow F, dos Santos DYAC. Traditional uses, bioactivity potential, and chemical diversity of the genus Gracilaria (Gracilariales, Rhodophyta): A comprehensive review. Algal Res 2019;37:288-306.
- Rosemary T, Arulkumar A, Paramasivam S, Mondragon-Portocarrero A, Miranda JM. Biochemical, micronutrient and physicochemical properties of the dried red seaweeds Gracilaria edulis and Gracilaria corticata. Molecules 2019;24(12):2225.
[Crossref] [Google Scholar] [PubMed]